Abstract
Objective
When the clinical presentation is related to the metastatic mass and a radiologically solitary tumor focus is detected, especially in cases where clinical information is not taken into account or is insufficient, if a possible metastatic neoplasia is not kept in mind then it is possible to evaluate the tumor as a primary breast neoplasm. In this study, it is aimed to present our cases of non-hematopoietic metastatic neoplasms and to evaluate the clinicopathological features that may aid in distinguishing metastatic from primary neoplasms.
Material and Methods
This study includes cases diagnosed with metastatic non-hematopoietic breast neoplasm in breast resection materials in our center, between the years 2010–2023. All cases were analyzed retrospectively by evaluating clinicopathological features.
Results
Of the 15 subjects included in the study, 11 (73%) were female and 4 (27%) were male. The mean age of the patients were 46.9 ranged from 22 to 63 years. The most frequent metastatic malignancy was carcinoma (60%), followed by melanoma (33%) and sarcoma (7%). Of the 9 patients with metastatic carcinoma, the primary tumor originated from the lungs in 4, from gastrointestinal system in 2, female genital tract in 2, and kidney in 1 patient. Sarcoma diagnosis was given in a single patient and the histology was a leiomyosarcoma originating from kidney.
Conclusion
A careful histomorphological and immunohistochemical evaluation and a detailed examination of the clinicoradiological data are critical to establish the right course in patient management, treatment plan and to correctly predict the prognosis.
Introduction
Breast cancer, constituting 11.6% of all cancers in both genders, is the second most common malignancy worldwide. 1 More than 98% of breast malignant neoplasms are primary tumors originating from breast parenchyma. 1.2–2% are metastatic neoplasms either originating from non-mammary solid organs or the systemic involvement of hematological malignancies. The rate of metastatic breast neoplasia is up to 6.6% in autopsy series. 2
In neoplasms metastasizing to the breast, the primary tumor focus is most frequently detected in the contralateral breast. 3 If malignancies originating from breast are excluded, the most frequently metastatic neoplasms are malignant melanoma and lung cancer. Metastases of sarcomas and carcinomas originating from the female genital tract (ovary, endometrium, cervix), gastrointestinal tract (stomach, colon, rectum), urinary tract (prostate, bladder), head and neck region, and thyroid gland have also been reported. Rhabdomyosarcoma, medulloblastoma, neuroblastoma, and primitive neuroectodermal tumor/ewing sarcoma are tumors that can metastasize to the breast, particularly in children and young adults.4–6
Despite their rarity, it is essential to distinguish metastatic breast neoplasms from primary neoplasms to ensure correct patient management, avoid unnecessary surgical interventions, and predict prognosis. In this study, it is aimed to present our cases of non-hematopoietic metastatic neoplasms and to evaluate the clinicopathological features that may aid in distinguishing metastatic from primary neoplasms.
Materials and Method
Patients diagnosed with metastatic non-hematopoietic breast neoplasm from breast surgical materials (tru-cut biopsy, lumpectomy, segmental mastectomy, modified radical mastectomy, and breast-conserving surgical material) evaluated in our center, between the years 2010 and 2023 were included in our study. Cases with slides unavailable or unsuitable for re-evaluation were excluded from the study. With available hematoxylin-eosin (H&E) and immunohistochemical stained slides, a retrospective study group consisting of 15 cases was determined.
Demographic information (age, gender), diagnosis of the primary and metastatic tumor, primary tumor localization, clinical presentation, radiological preliminary diagnosis, survival status, and follow-up periods of the cases were obtained from the hospital database and pathology reports. H&E stained and immunohistochemically stained slides with the thickness of 4–5 µm were re-evaluated in terms of histopathological diagnosis.
Ethics approval, dated 16 May 2023 and numbered 2023–11/5, was obtained from the local Clinical Research Ethics Committee.
Results
The general characteristics of 15 patients with metastatic non-hematopoietic breast neoplasm included in the study, are summarized in Table 1. 11 (73%) patients were female and 4 (27%) were male. The ratio of female to male was 2.75. The mean age of the patients was 46.9 ranging from 22 to 63 years.
Clinicopathological features of the cases (n = 15)
The presenting symptom was palpable mass in 13 (87%) and pain in 1 (7%) case. 13 patients (87%) had a clinical history of a primary tumor. Two patients (13%), with no history of primary malignancy, were admitted to the clinic for the first time with complaints related to breast metastasis. The tumor was located in the right breast in 10 cases (67%), left breast in 4 cases (27%), and bilaterally in 1 case (7%). Preliminary radiological diagnoses were as follows: 6 (40%) primary breast neoplasm, 6 (40%) metastatic neoplasia, and 2 (13%) fibroadenoma. The most frequent metastatic malignancy was carcinoma (60%), followed by melanoma (33%) (Figure 1) and sarcoma (7%). Of the 9 patients with metastatic carcinoma, the primary tumor originated from the lungs in 4, from gastrointestinal system in 2, female genital tract in 2, and kidney in 1 patient. When the histopathological diagnoses of primary tumors were evaluated, 3 of the lung tumors were small cell carcinoma and 1 was adenocarcinoma (Figure 2), all tumors originating from the gastrointestinal tract were adenocarcinoma (Figure 3), 1 tumors originating from the female genital tract was serous carcinoma and the other one was adenocarcinoma, and the kidney tumor was diagnosed as renal cell carcinoma (Figure 4). Sarcoma diagnosis was given in a single patient and the histology was a leiomyosarcoma originating from kidney (Figure 5).
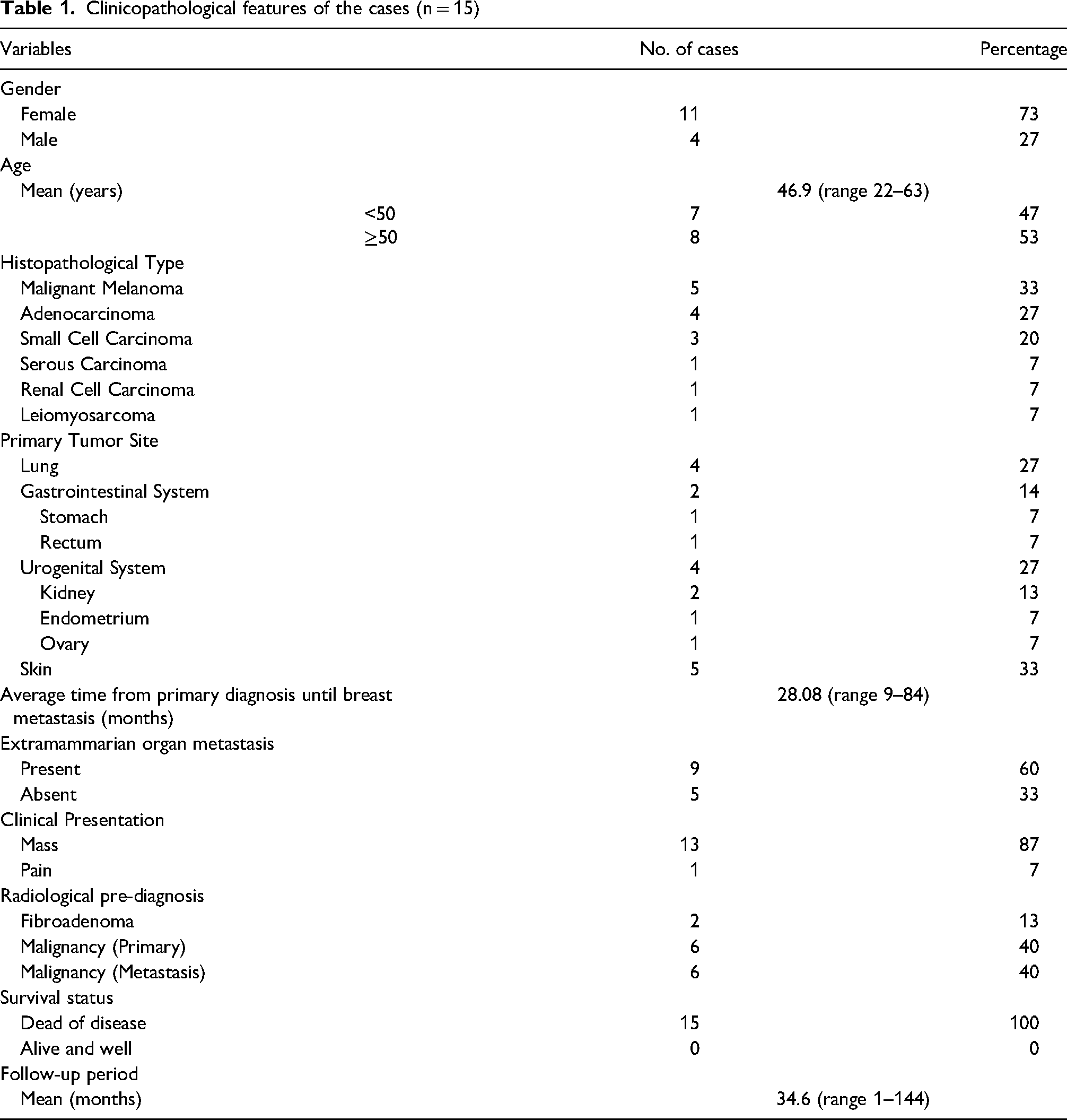
Metastatic malignant melanoma. Tumor cell islands with wide eosinophilic cytoplasm, prominent nucleoli, and foci of necrosis (A, H&E×200). Intracytoplasmic (brown) pigment accumulation (B, H&E×400). Strong cytoplasmic staining in tumor cells by S100 immunohistochemistry (C, ×100). MART1 immunohistochemical staining (D, ×100). HMB-45 immunohistochemical staining (E, ×100).
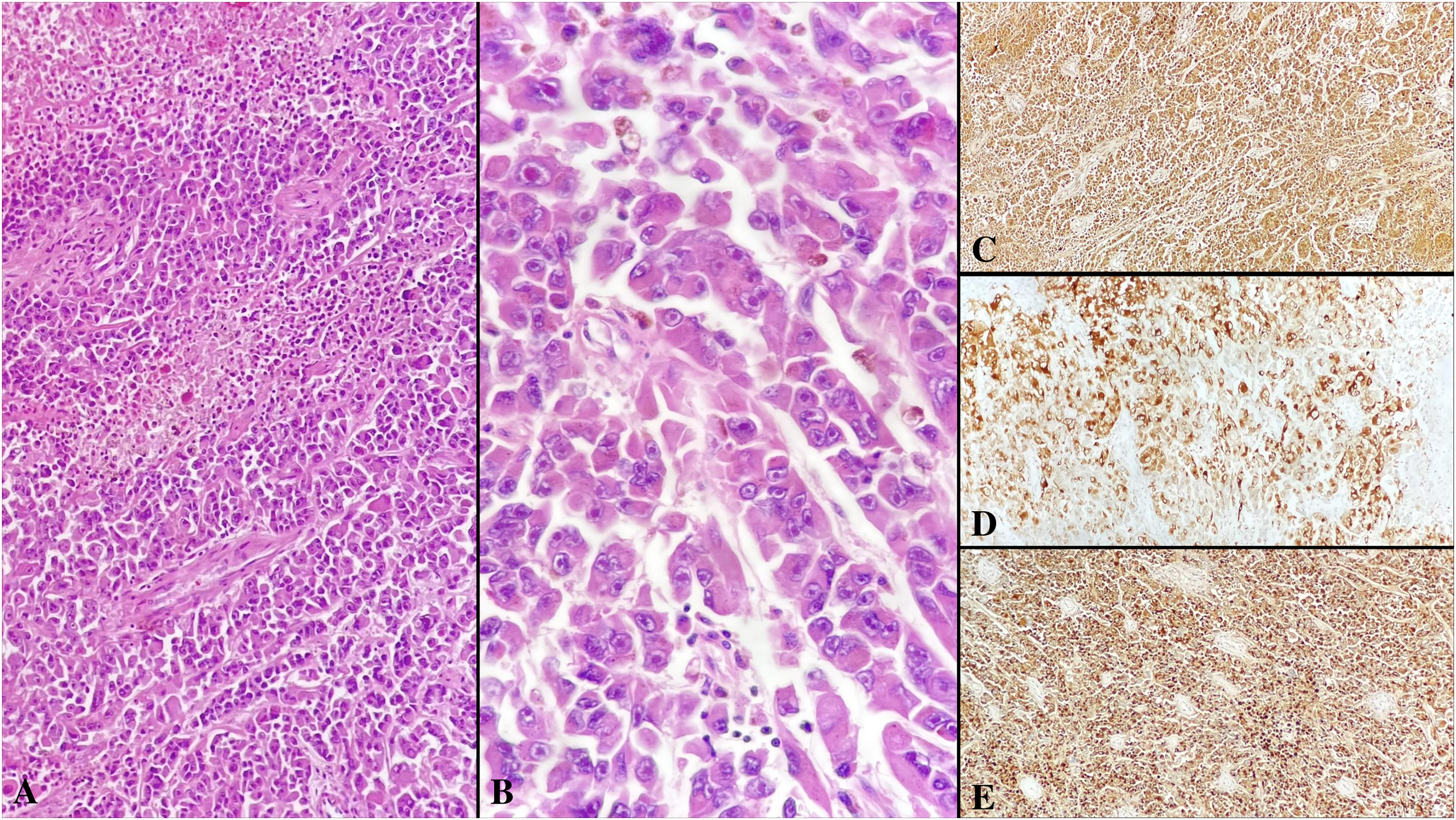
Metastatic lung adenocarcinoma. Next to normal breast ducts, tumor cells showing prominent cytological atypia, occasional intracytoplasmic vacuoles, eosinophilic cytoplasm, and glandular growth pattern (A and B, H&E×400). Tumor cells show strong staining with TTF1, while normal breast ducts do not show immunoreactivity. (C, ×100). Tumor cells show strong staining with Napsin, while normal breast ducts do not show immunoreactivity. (D, ×100). Normal breast ducts are stained with GATA3 and no immunoreactivity is seen in the tumor (E×100).
Metastatic rectal adenocarcinoma. Discohesive tumor cells with large eosinophilic cytoplasm, intracytoplasmic mucin vacuoles next to normal breast ducts (A, H&E×400, B, H&E×200). CDX2 immunoreactivity observed in tumor cells (×200).
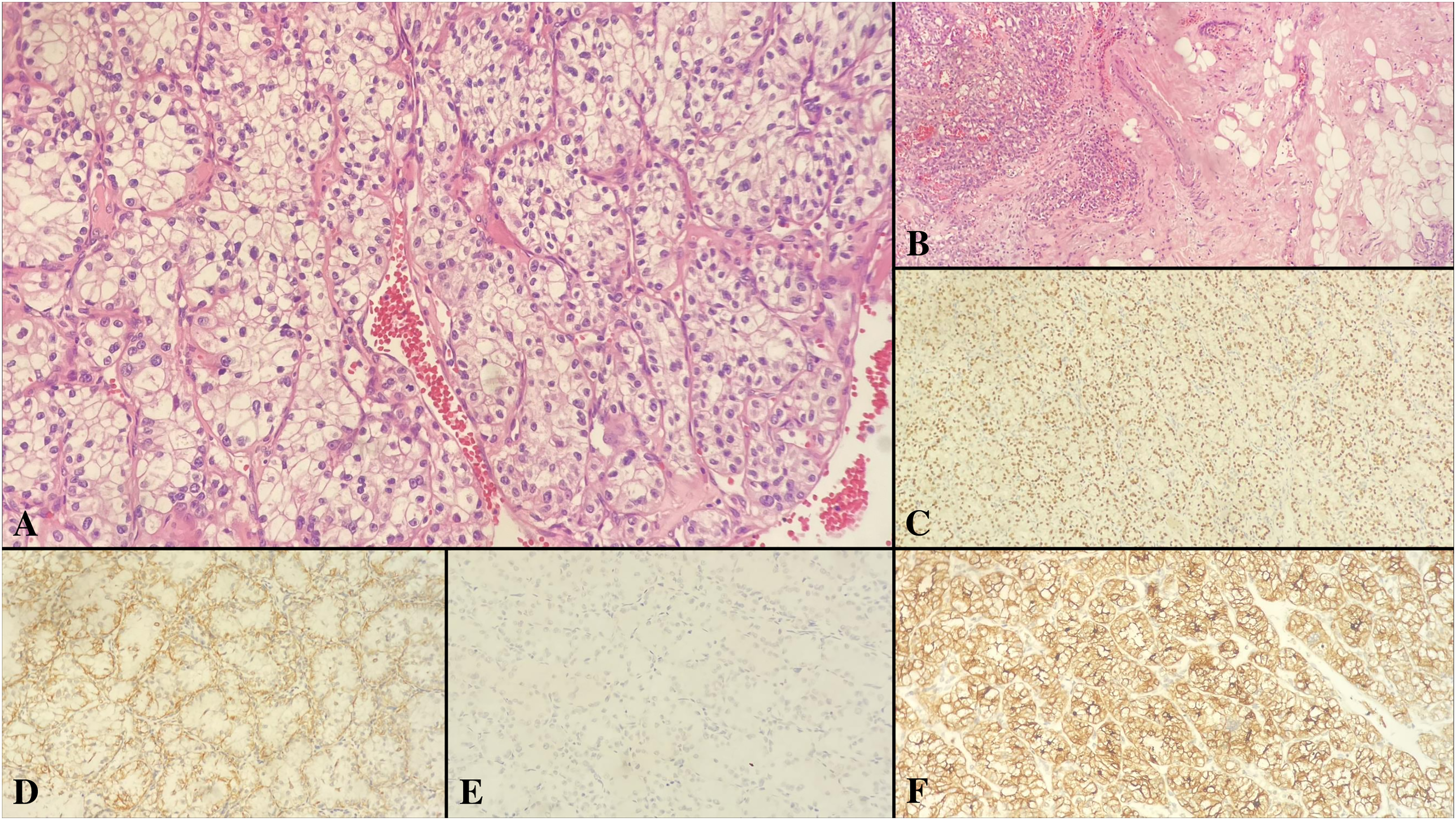
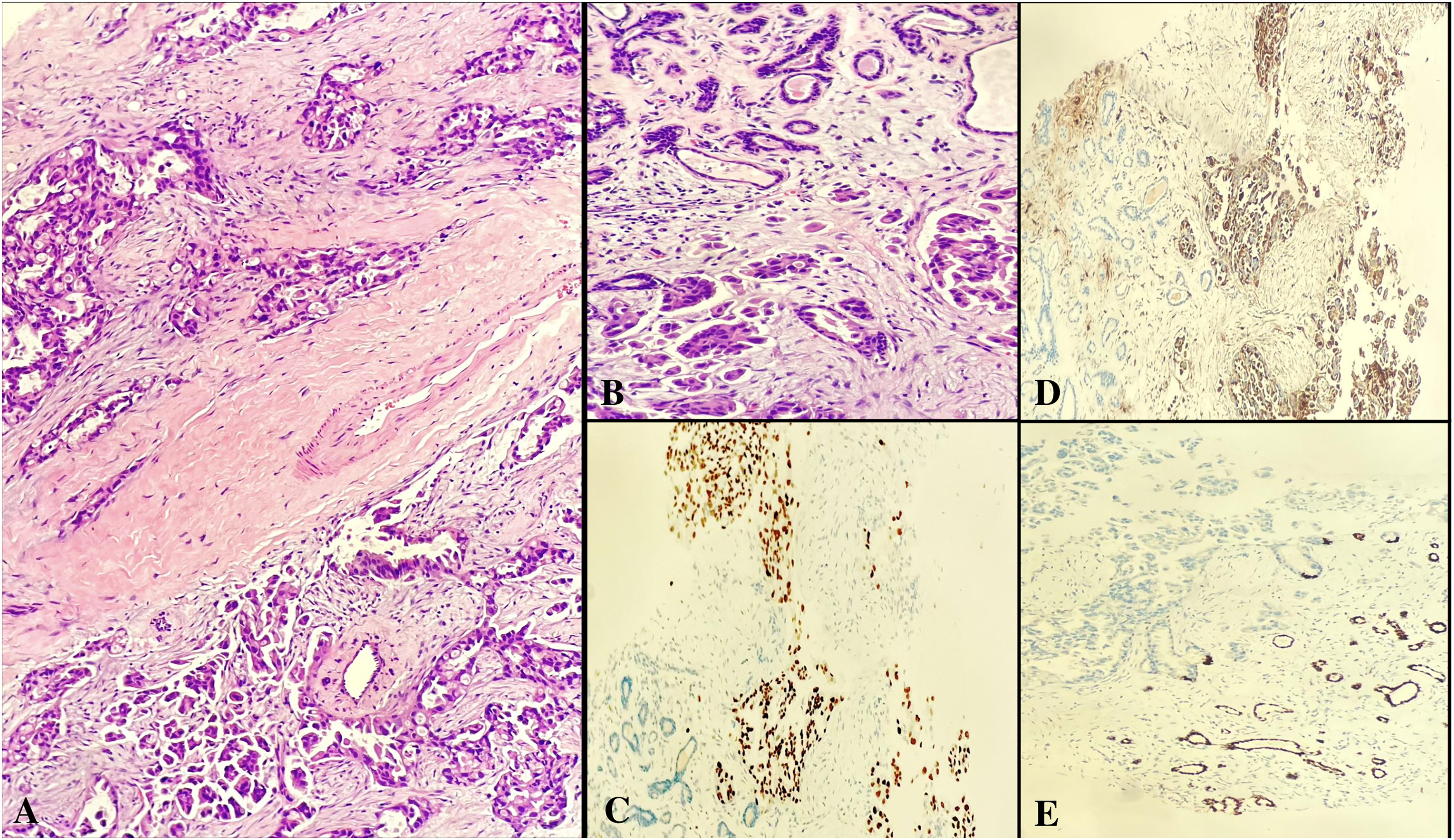
Metastatic renal cell carcinoma. Nests of tumor cells showing clear cytoplasm, prominent nucleoli, well-defined cell borders include thin walled small vascular structures in its stroma (A, H&E×400). Relationship between tumor and normal breast ducts (B, H&E×100). PAX8 immunoreactivity in tumor cells (C, ×200).CD10 immunoreactivity in tumor cells (D, ×200). Negativity in tumor cells with Keratin 7 immunostaining (E, ×200). Vimentin immunoreactivity in tumor cells (F, ×200).
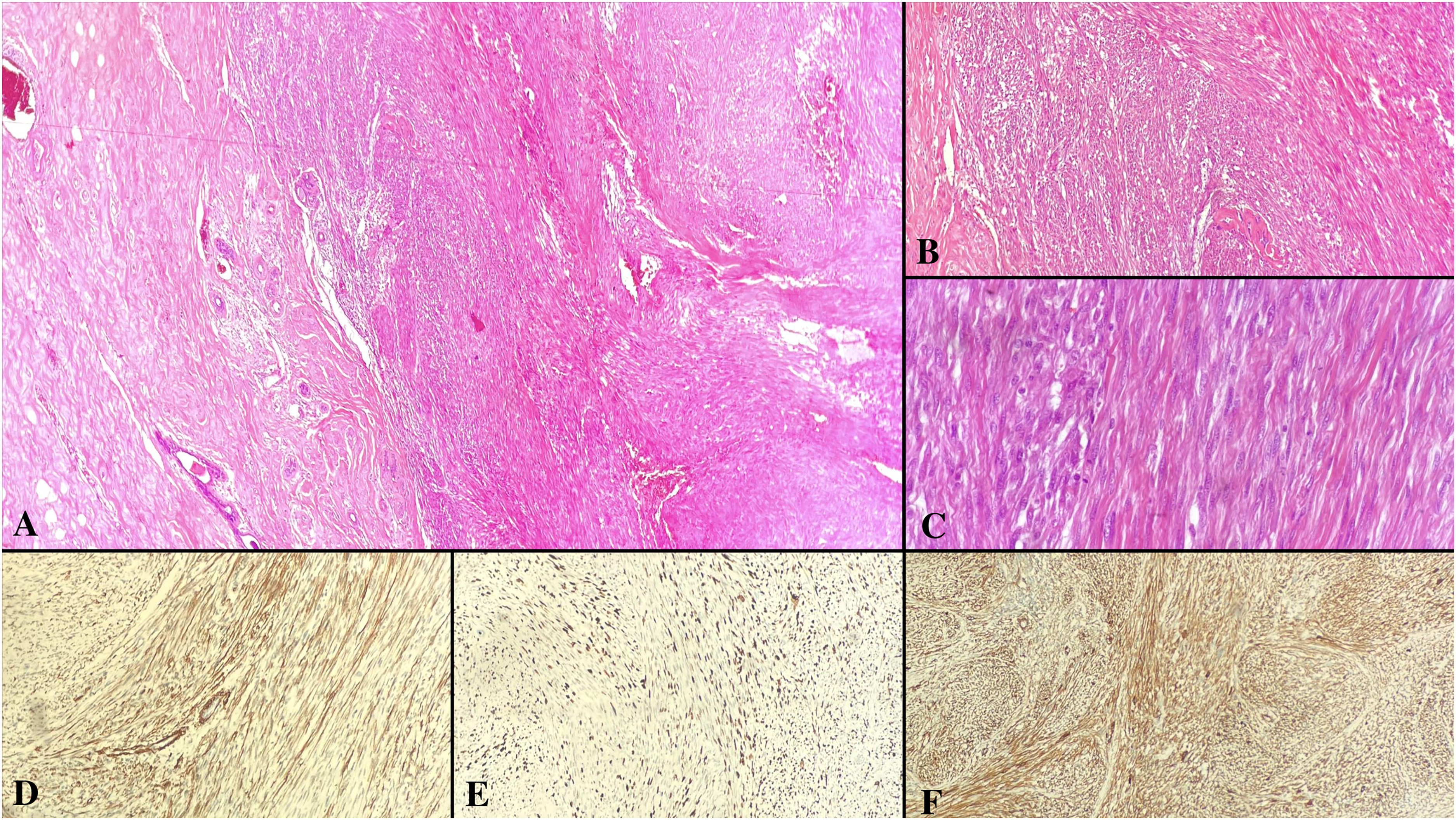
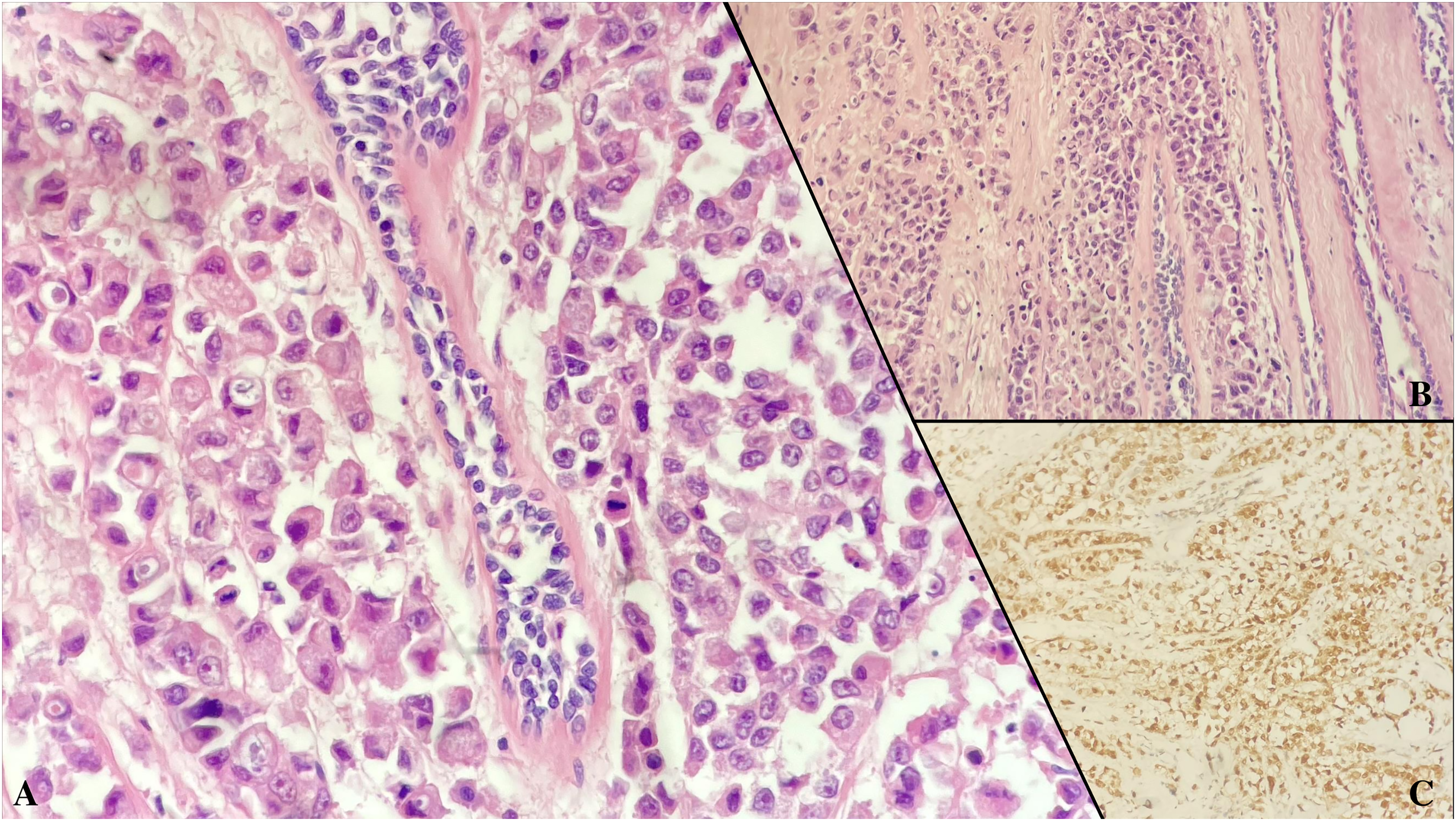
Metastatic leiomyosarcoma. Next to normal breast ducts, fascicular structures formed by tumor cells with spindle-shape, eosinophilic cytoplasm, centrally localized nuclei and oval-shaped ends (A, H&E×200, B, H&E×100). Close-up micrograph of tumor cells with increased mitotic figures (C, H&E×400). SMA immunoreactivity in tumor cells (D, ×200). High ki-67 proliferation index in tumor (E, ×200). Caldesmon immunoreactivity in tumor cells (F, ×200).
Metastases were detected in organs other than breast in 9 (60%) cases. The only metastatic site was breast in 5 (33%) cases. The mean time from primary diagnosis to the detection of breast metastasis was 28.08 months (range: 9–84 months).
The mean follow-up period was 34.6 months, varying between 1 and 144 months. All cases died of disease during the follow-up period.
Discussion
An important part of routine pathology practice is the evaluation of breast resection materials, considering its incidence. Metastatic neoplasms may exhibit histomorphology similar to primary breast neoplasms. Therefore, it is very difficult to make an accurate diagnosis, especially in cases where clinical information is not taken into account or is insufficient. 7 As with primary breast neoplasms, metastatic neoplasms are also detected more frequently in women. 8 The tumor site involved is often the upper outer quadrant of the left breast. Tumor foci are generally unifocal, but in 10% of the cases multifocal involvement, in 5% diffuse involvement and in 25% bilateral involvement is observed.6,9 In our study, the tumor was detected more frequently in the right breast, unlike other reported series.
The most frequent clinical presentation is palpable mass, similar to primary breast neoplasms. A solitary lesions often suggests a primary breast neoplasm and multiple lesions should be investigated for a possible metastatic tumor. Even in cases presenting with a palpable mass in the breast without a previous history of malignancy, involvement in organs other than breast is frequently detected as the result of a systemic investigation. Orange peel sign, skin or nipple retraction or nipple discharge are not expected in metastatic neoplasms. However, skin edema and redness due to extensive lymphovascular invasion are mutual symptoms of metastatic and primary tumors. In that instance, a possible inflammatory breast carcinoma should be included in the differential diagnosis and careful examination should be obtained.1–10
Although ipsilateral axillary lymph node involvement is a warning sign for primary breast neoplasms, it should be known that the ipsilateral axilla may be involved in approximately 15% of metastatic neoplasms. 9 In their series consisting of 85 cases, DeLair et al found the rate of cases showing the first presentation with a mass in the breast or axilla as 11%. 11 In our series, 86.6% of the cases presented with the complaint of a mass. All tumors were unifocal and bilateral involvement was observed in 1 case.
Radiologically about 20% of tumors are categorized as probable benign, while more than 50% are evaluated as suspicious for malignancy. In ultrasonographic evaluation, tumors are often detected as well-circumscribed, round-oval shaped, single or multiple, hypoechoic masses showing variable lobulation. Contrary to primary breast neoplasms showing spiculation, their smooth circumference is not distinctive, but an important feature. Although the presence of microcalcification in the mammographic evaluation of various metastatic malignancies (hepatocellular carcinoma, carcinomas of gastric or ovarian origin) is reported in the literature, it is not a very common finding.12,13 In our study, radiological preliminary diagnoses were malignant in 80% and benign neoplasia in 13.3% of the cases.
Although macroscopic findings vary from case to case, the tumors are generally in form of a solid mass that can be separated from the surrounding breast parenchyma with a smooth contour, similar to the radiological findings. 6 Lesions are usually superficially located and not detected in the deep plane or between muscle fibers. 14 Microscopically, metastatic breast tumors are frequently observed in the periductal and perilobular areas, separated clearly from the surrounding breast parenchyma, show multiple foci, infiltrate subcutaneous adipose tissue, does not show desmoplastic reaction, elastosis or an in situ carcinoma component, and exhibit widespread lymphatic invasion. 15 The absence of a ductal carcinoma in situ is considered important for diagnosing metastasis. However, coincidental carcinoma in situ and metastatic tumor, and carcinoma in situ foci colonizing the ductal-lobular unit which create the impression of carcinoma have been reported. 16
The possibility of metastasis comes to mind more frequently in tumors with histomorphological features different from those expected in primary breast tumors. Differentiating tumors resembling primary breast tumors can be more challenging, especially in cases where clinical information is insufficient. Some examples of these tumors are invasive lobular carcinoma, neuroendocrine neoplasms, mucinous carcinoma, or squamous cell carcinoma with signet ring cell features. Similarly, widespread dermal lymphatic invasion in a tumor metastasizing to the breast via the lymphatic route can be challenging when differentiating from a inflammatory breast carcinoma.17,18 Khalifeh et al presented a case of metastatic peritoneal serous carcinoma in punch biopsy material, which was interpreted as inflammatory breast carcinoma due to diffuse tumor embolism in dermal lymphatics. 19
Among the non-hematopoietic metastatic breast neoplasia series in the literature, one of the most comprehensive is the series of Zhou et al In their study of 238 cases, 77.7% were diagnosed correctly at the first evaluation. The most common metastatic tumors were as follows: malignant melanoma (42%), serous carcinoma (15%), neuroendocrine neoplasia (13%). 20 In our study, carcinoma was the most frequent and malignant melanoma was the second most common tumor group. The skin is the most common primary organ that metastasized.
Classical histomorphological features such as the presence of melanin pigment, nuclear pseudoinclusions, prominent nucleoli, eccentric nuclei and discohesive cells provide diagnostic clues for malignant melanoma. In their series consisting of 20 cases, Bacchi et al detected prominent melanin pigment in only 2 cases. The rate of cases diagnosed as metastatic malignant melanoma in the first evaluation was 15%. 21 Zhou et al mentioned that when the clinical history is available to the pathologist, the correct diagnosis rate is 100% and this rate drops to 58% in cases where the clinical history is not known. 20
Serous carcinoma is one of the tumors that commonly metastasize to the breast and is often misdiagnosed if the clinical history is unknown. Growth pattern similar to micropapillary breast carcinoma, mimic inflammatory breast carcinoma clinically, and showing hormone positivity are some of the reasons for misdiagnosis. 22 Although serous carcinoma case in our series exhibited a micropapillary growth pattern and hormone positivity, it was correctly diagnosed in the first evaluation with the help of history of previous malignancy.
Similar to neuroendocrine neoplasms of other systems, neuroendocrine neoplasms of the breast include well-circumscribed tumor islands, uniform nuclei, salt-and-pepper chromatin pattern and do not show specific histomorphological features supporting breast primary. Therefore, they are unlikely to be diagnosed correctly unless immunohistochemical evaluation is performed. Exceptionally, there are two specific types of breast carcinoma in which neuroendocrine marker expression can be detected and diagnosed histomorphologically. These tumors, which show neuroendocrine marker expression but are not included in the neuroendocrine neoplasia category, are solid papillary carcinoma and mucinous carcinoma type B. Primary breast neuroendocrine tumors are hormone receptor-positive, unlike metastatic tumors that are generally triple-negative. However, for definitive diagnosis, immunohistochemical markers specific to the primary (GATA3, TTF1 (especially SPT24 clone), CDX2, CEA, PAX8, SATB2, ISL1, PDX1, ARX, PSAP, PAX6) should be studied.1,23–25
In the histomorphological evaluation of other neoplasms metastasizing to the breast, salt-pepper chromatin pattern, scant cytoplasm, increased mitosis and apoptosis, presence of diffuse necrosis and crush artifact indicate small cell carcinoma, prominent cytological atypia and clear cytoplasm suggest lung adenocarcinoma, presence of glands including luminal necrosis suggest adenocarcinoma of the gastrointestinal system, presence of focal squamous metaplasia and spindle cells in addition to glandular structures indicate endometrioid carcinoma and tumor cells with clear cytoplasm suggest renal cell carcinoma. 26
Histomorphological features of metastatic tumors may be similar to primary tumors thus, one of the most important evaluation steps for correct diagnosis is immunohistochemical study. Metastatic breast carcinomas generally show a triple negative immunexpression profile. However, ovarian, endometrial, and gastric carcinomas can express estrogen hormone receptor (ER). In metastatic breast neoplasms showing ER expression, focal positivity is a diagnostic finding. In suspected cases, an immunohistochemistry panel consisting of keratin 7 (KRT7), keratin 20 (KRT20), Mammaglobin 1 (SCGB2A2), GATA3 (HDR), NY-BR-1 (ANKRD30A), and GCDFP15 (PIP) helps the diagnosis. GATA3, which is positive in 72–94% of breast carcinomas is also expressed in urothelial carcinomas. It is generally negative in other carcinomas. Mammaglobin and GCDFP15 are markers with sensitivity ranging from 10% to 80% for breast primary. They can stain the tumor focally or may give negative results in small samples containing limited tumor cells. In addition, Mammaglobin can be expressed in salivary gland, sweat gland, ovarian and endometrial carcinomas, while GCDFP15 can be expressed in lung carcinomas in addition to these. The fact that these markers are expressed in non-mammary malignancies and the rate of staining decreasing up to 10% in triple negative breast carcinomas (TNBC), has led to the search for a more sensitive marker in determining breast primary. As a result, TRPS1 emerged as a highly sensitive marker in invasive lobular carcinoma and metaplastic carcinoma, especially in TNBC. Another marker reported to have higher sensitivity than GATA3 for TNBC is SOX10.24–29 In their study which included 100 invasive breast carcinomas, Cimino-Mathews et al found nuclear SOX10 immunoreactivity in 40% of tumors. This rate increased to 46% in metaplastic carcinoma, 69% in basal-like carcinoma, and 77% in unclassified triple negative carcinoma. 30 Keratin 7 is positive in breast, lung, and ovarian carcinomas, while keratin 20 is positive in tumors originating from the gastrointestinal tract, especially colorectal carcinomas. In the immunohistochemical evaluation of neoplasms metastasizing to the breast, TTF1 and Napsin staining was suggestive for lung adenocarcinoma, SOX10, Melan-A and HMB45 for melanoma, CDX2 for colorectal adenocarcinoma, WT1, CA125, and PAX8 staining for serous papillary carcinoma, RCC and CD10 staining for renal carcinoma. In neuroendocrine neoplasms, GATA3 and ER positivity support the breast primary, while TTF1 support lung, CDX2 suggest small intestine and ovary, SATB2 support colon, ISL1 indicate duodenum while ISL1 and ARX indicate pancreas, PSAP suggest genitourinary system, calcitonin and CEA support thyroid origin.24–26,28,31 Even if all resources are available during clinical, radiological and histopathological evaluation, it is not possible to make a definitive diagnosis in some conditions, especially in small biopsy materials. Various artifacts occurring during follow-up or drying processes, crushing artifacts, and small tumor size are among these conditions. In such cases, which prevent a definitive diagnosis, the report should include “invasive carcinoma/adenocarcinoma.” Combining the histomorphological features with the clinical and radiological features of the case, an interpretation of a possible origin of the tumor should be stated. However, if this is not possible, it should be stated that a neoplasia metastasizing to the breast might be the case and systemic screening should be recommended. Metastatic breast neoplasms can cause diagnostic difficulties, especially in cases where the clinical history is unknown. This may prolong the duration of the pathological evaluation, increase costs by causing additional immunohistochemical, histochemical or molecular studies, or cause the pathology report to include a descriptive diagnosis rather than a definitive diagnosis, especially in tru-cut biopsy materials. Clinicians experiencing similar problems can also increase costs, prolong treatment duration, and worsen clinical outcome. 32 A multidisciplinary team including surgeons, radiation and medical oncologists, radiologists, pathologists, nurses, and other healthcare personnel, can minimize these problems in patient management. Multidisciplinary meetings can be in the form of a team meeting in a suitable room, discussing the patient management process, or can be held in the form of video conference.33,34 If multidisciplinary meetings cannot be held for various reasons, all the information obtained from the patient after detailed anamnesis, laboratory, or radiological evaluation and all decisions taken by all physicians who evaluated the patient should be clearly recorded in files or hospital electronic databases that can be available to other physicians for further evaluations.
Patient management, correct treatment plan and predicting the prognosis basically depends on the correct diagnosis. Therefore, histopathological evaluation should be based on a careful histomorphological and algorithmic immunohistochemical evaluation, taking the clinicoradiological data into account.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethics approval, dated 16 May 2023 and numbered 2023-11/5, was obtained from the local Clinical Research Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials
