Abstract
Widespread use of vaccinations worldwide in the coronavirus disease (COVID-19) pandemic has resulted in various side effects. Here, we presented a 27-year-old man with autoimmune-like hepatitis after the first dose of the BNT162b2 (mRNA) COVID-19 vaccine and reviewed previous reports. He presented with sweating, febrile sensations, and general weakness. He did not have any medical histories. Although he was treated with biphenyl dimethyl dicarboxylate and ursodeoxycholic acid, the elevated liver enzyme levels persisted for 2 months. Liver biopsy demonstrated portal inflammation with rosette formation, interface hepatitis, and infiltration of lymphocytes, histiocytes, plasma cells, and eosinophils. Especially, centrilobular edema and necrosis were found. The symptoms and liver enzymes improved with prednisolone treatment. If persistently elevated liver enzymes are found after COVID-19 mRNA vaccination, the possibility of autoimmune-like hepatitis induced by the vaccine should be considered and a careful pathologic evaluation is required.
Introduction
The profound disruption of life, economy, and sociality caused by the coronavirus disease (COVID-19) pandemic has resulted in rapid development and unprecedented approval of various COVID-19 vaccines. mRNA vaccines (BNT162b2 and mRNA-1273) serve as both immunogens and adjuvants. It works by activating the interferon pathway, which raises the issue of inflammatory disorders associated with interferon activation. 1 A recent study reported that antibodies against the spike protein S1 of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have a high affinity against some human tissue proteins, indicating a potential antigenic cross-reactivity between SARS-CoV-2 and human tissue. 2 Because both mRNA and vector vaccines induce the production of protein S, they can trigger autoimmune diseases in patients predisposed to them. mRNA has modified nucleotides to reduce the excessive production of type I interferon, resulting in limited binding to immune sensors. Nevertheless, there are concerns regarding the possibility of triggering various autoimmune conditions.
A wide range of autoimmune diseases have been reported following COVID-19 vaccination, and autoimmune hepatitis (AIH) has been reported in a few studies. 3 Here, we presented the pathologically confirmed AIH in a 27-year-old man after receiving the first dose of the BNT162b2 COVID-19 vaccine and reviewed the previous literatures.
Case Description
A 27-year-old man received the first dose of the mRNA COVID-19 vaccine (Pfizer-BioNTech, BNT162b2). After 2 weeks, he presented with sweating, febrile sensations, and general weakness. He denied any history of chronic alcohol abuse, previous hepatotropic virus infections, familial medical histories, and other comorbidities. He did not have any medical histories and his historical liver function tests were normal. He took no over-the-counter or prescription medications, and no herbal or dietary supplements. His body mass index was 24.22 kg/m² (normal range). Physical examination revealed normal findings.
Aspartate aminotransferase (640 U/L), alanine aminotransferase (1416 U/L), bilirubin (1.52 mg/dL), and alkaline phosphatase (ALP, 39 U/L) levels were all elevated in the initial laboratory tests. Hepatic viral markers were negative for hepatitis A, B, C, and E. His complete blood count results with cell differentiation, electrolyte profiles, and coagulation laboratory findings, including the prothrombin time, were unremarkable. Additional blood tests showed that Epstein-Barr virus, cytomegalovirus, and human immune deficiency virus were all negative. An autoimmune serology test showed an antinuclear antibody (ANA) titer of 1:80 with a speckled pattern. However, anti-mitochondrial, anti-smooth muscle, liver–kidney microsomal, and antineutrophil cytoplasmic antibodies were negative. Immunoglobulin G (IgG) level was slightly increased (2069 mg/dL). Serum ceruloplasmin level was within the normal range. An abdominal ultrasound examination and contrast computed tomography revealed no remarkable findings. Chest radiography, echocardiography, or transthoracic echocardiography had no specific findings. PCR test for COVID-19 were negative. Although he was treated with biphenyl dimethyl dicarboxylate and ursodeoxycholic acid, the elevated liver enzyme levels persisted for 2 months and the cholestatic pattern was identified (Figure 1). Liver biopsy was performed to evaluate AIH. 4
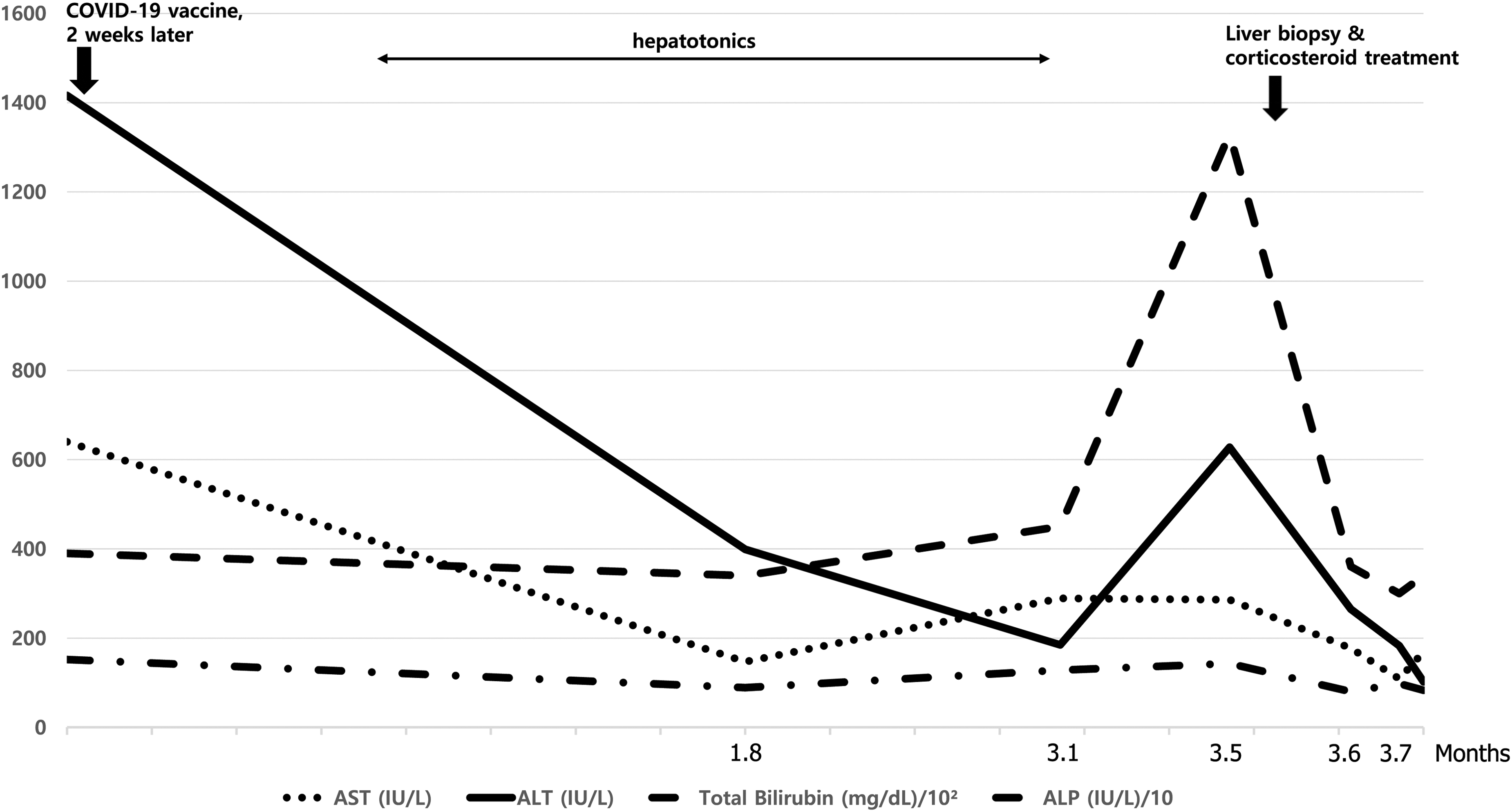
Trends of liver function test. AST, aspartate transaminase; ALT, alanine transaminase.
Liver biopsy demonstrated portal and lobular inflammation (Figure 2). The histology showed moderate inflammation and hepatocellular injury at the interface as well as hepatic rosette formation. Prominent plasma cells with some eosinophils were infiltrated in portal area, and necrosis of hepatocytes was identified in the centrilobular area. Fibrosis or steatosis was not observed. Given these histological findings, AIH versus drug-induced liver injury (including drug-induced AIH) can be considered.
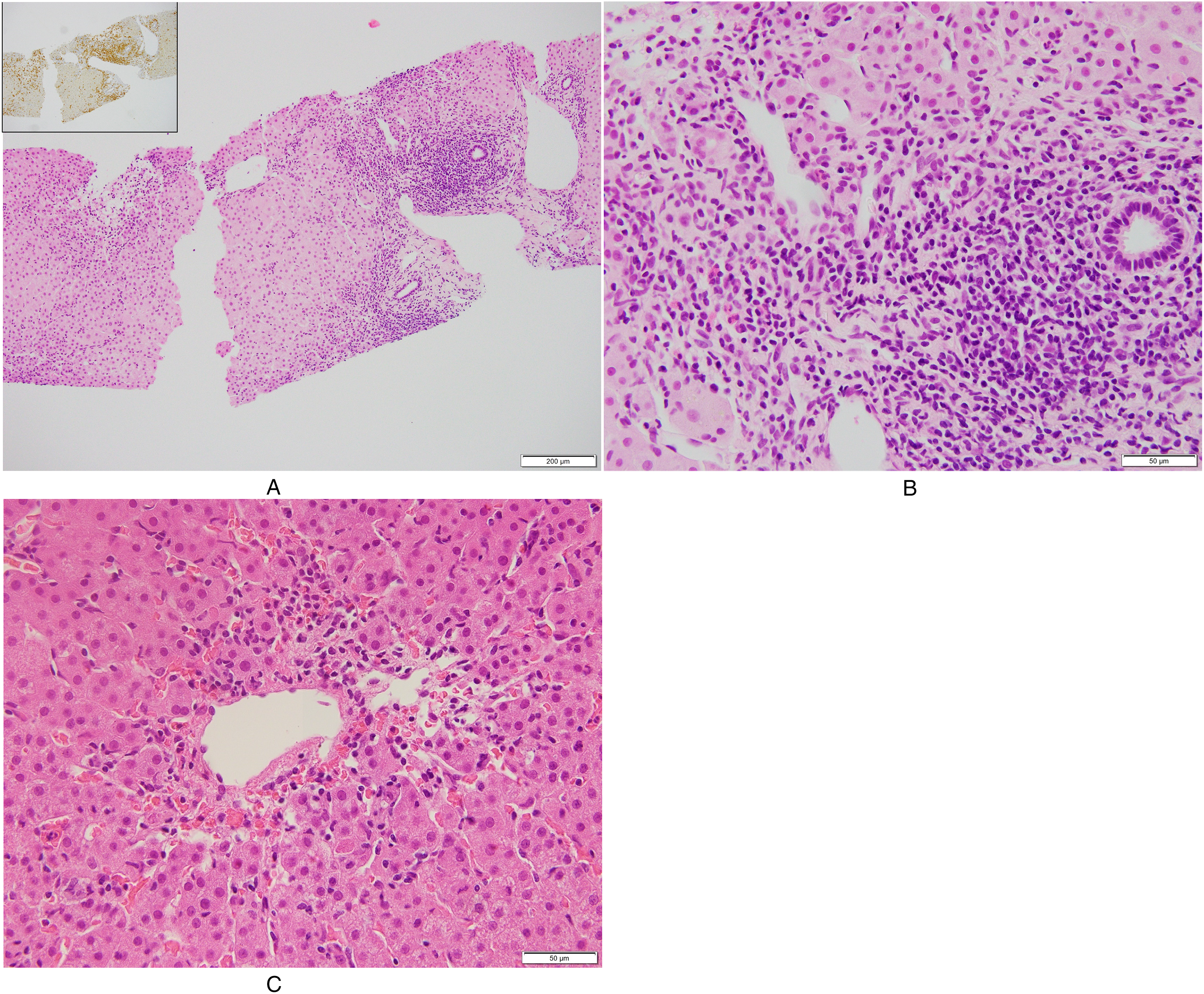
Microscopy shows portal inflammation with interface hepatitis (A, ×100). The infiltrated cells are mostly CD8 T lymphocytes (A, inset, CD8). Prominent plasma cells with some eosinophils were infiltrated in portal area (B, ×400). The centrilobular lesion shows mild edema, necrosis, and infiltrates of lymphocytes and histiocytes (C, ×400).
Prednisolone 30 mg/d was started, and a week later, the patient's liver function test showed a good response and he was discharged (Figure 1). The patient was monitored to taper the prednisolone dose in an outpatient clinic. He has been in good health for 6 months. The institutional review board approved this study (CBNUH IRB, No. 2022-07-005).
Discussion
We present AIH in a 27-year-old Korean man who developed symptoms 2 weeks after receiving the first dose of COVID-19 mRNA vaccine (BNT162b2). The patient's clinicopathological features, including laboratory tests and histology, were consistent with AIH.
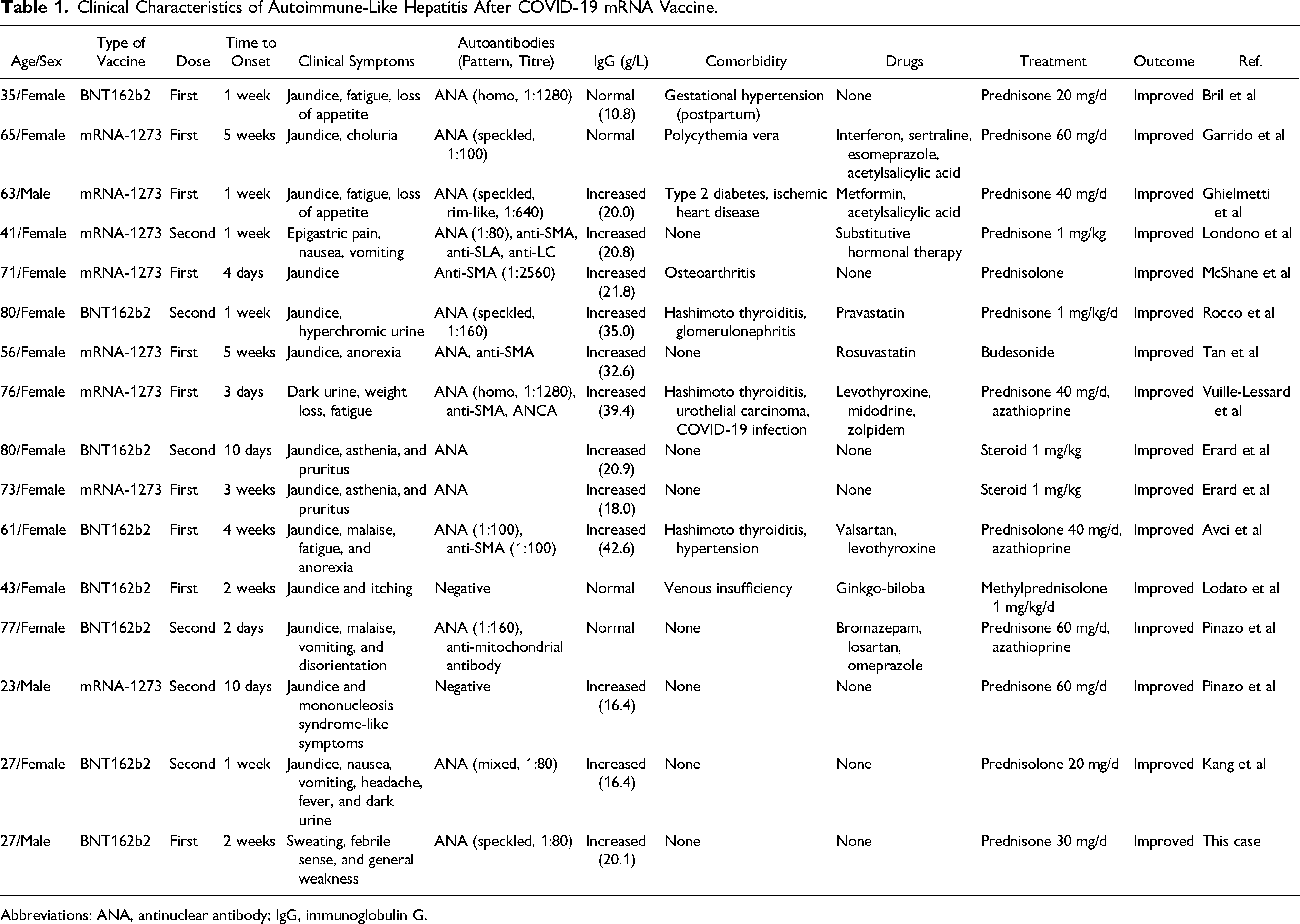
Although the specific link cannot be established, similar patients following COVID-19 mRNA vaccination have been reported (Table 1).5–17 This is suggested by the temporal relationship in the absence of other potential triggers or risk factors for AIH. 18 These reports including ours, support the relationship between COVID-19 mRNA vaccination and AIH. The age of the patients ranged from 23 to 80 years and 5 were young adults (5/16, 31.3%). Female to male ratio was 5.3. Most patients complained of symptoms between 1 and 5 weeks after vaccination and about half of them showed symptoms between 1 and 2 weeks. These latency periods imply that mRNA vaccines stimulate innate immunity. Jaundice was the most common symptom (12/16, 75.0%). However, 2 patients had nonspecific symptoms, including epigastric pain, nausea, vomiting, sweating, febrile sensation, and general weakness.
Clinical Characteristics of Autoimmune-Like Hepatitis After COVID-19 mRNA Vaccine.
Abbreviations: ANA, antinuclear antibody; IgG, immunoglobulin G.
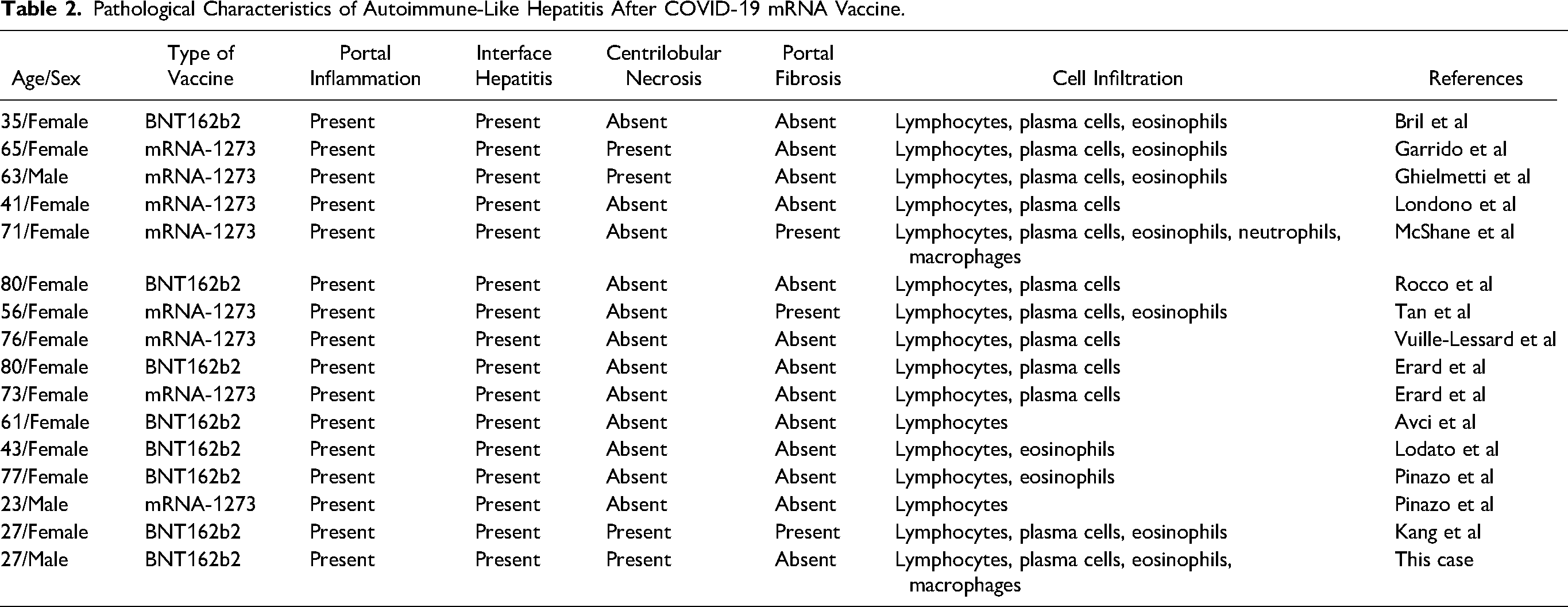
Pathological Characteristics of Autoimmune-Like Hepatitis After COVID-19 mRNA Vaccine.
In our patients, the constantly rising levels of liver enzymes necessitated an autoimmune liver serology test. The autoimmune serology of our patient was remarkable, with a speckled pattern at a titer of 1:80. ANA was positive in almost patients (13/16, 81.3%) after the mRNA COVID-19 vaccine, followed by anti-SMA antibody (5/16, 31.3%). Our patient exhibited low titer ANA (1:80). Labs vary in their standards for positivity. In general, many labs will report any titer above 1:160 as positive. Although the ANA titer was low, the ANA pattern was speckled the result was positive. Of 11 available patients, 3 presented with low ANA titers. Low titers would not fully exclude an AIH-like hepatitis. IgG levels were elevated in 12 patients (12/16, 75.0%) and 3 patients (3/16, 18.7%) had the concomitant autoimmune disease (Hashimoto thyroiditis).
The patients’ drug history varied. Seven patients had no drug history and the remaining 9 patients had a long exposure time to the previous medication. There was no history of administration of a new drug in all patients, which is inconsistent with drug-induced hepatitis. Our patient and other all reported patients were treated with steroids. Although one died of an extrahepatic brain lesion (stroke), all patients successfully responded to steroid treatment. The brain lesion was unrelated to the vaccine and/or steroid treatment.
Histologically, almost all specimens had portal inflammation with interface hepatitis and rosette formation, consistent with the AIH histology (Table 2). Eosinophil infiltration was observed in more than half (9/16, 56.3%) of the patients. Four, including our patient, had a distinct feature, centrilobular necrosis around the central vein in zone 3, which should consider drug-induced liver injury (4/16, 25.0%). This centrilobular necrosis, particularly when accompanied by inflammation, can also be a feature of AIH. Although all patients including our patients showed elevated enzymes (ALP and/or bilirubin) of cholestatic pattern, there was no bile duct injury or cholestatic lesions in all specimens. That is the common and differing features of these patients in comparison to a “bona-fide AIH.” These clinicopathological findings indicated AIH or AIH-like drug-induced liver injury by vaccination. However, whether vaccination is a discloser or a trigger of autoimmune-like hepatitis is unclear.
One patient had a latency period of 2 days after the second vaccination. Despite the very short interval (less than 7 days) between vaccination and symptoms, 3 patients showed histology of chronic inflammation including bridging necrosis and fibrosis. This histology implies bona fide AIH. Given these clinicopathological features, 3 patients have a possibility of unmasking AIH by vaccination. mRNA of the vaccine serves as both immunogen and adjuvant, which could activate the inflammatory conditions associated with interferon activation. 1 Antibodies against the spike protein S1 of SARS-CoV-2 can recognize human tissue proteins and have a potential antigenic cross-reactivity. 2 Because mRNA vaccines induce the production of protein S, they can trigger autoimmune diseases. Although mRNA has modified nucleotides to reduce excessive inflammatory reactions, there are concerns regarding the possibility of triggering various autoimmune conditions.
In conclusion, we present a patient of AIH caused by COVID-19 mRNA vaccination with distinct clinicopathological features. Because of the widespread use of vaccinations worldwide, vaccine-induced AIH has important implications. If persistently elevated liver enzymes are found after COVID-19 mRNA vaccination, the possibility of autoimmune-like hepatitis induced by the vaccine should be considered, and a careful pathologic evaluation is required. This should not discourage vaccination but raises awareness regarding diagnosing and treating as soon as possible because it can lead to liver failure.
Footnotes
Author Contributions
Conceptualization: Kim JH, Woo CG. Data curation: Kim JH, Chae HB, Kim HJ, Woo CG. Formal analysis: Kim JH, Woo CG. Investigation: Kim JH, Woo S, Song MS. Methodology: Kim JH, Woo CG. Software: Woo CG. Validation: Kim JH. Visualization: Kim JH, Kim HJ. Writing - original draft: Kim JH, Woo CG. Writing - review & editing: Chae HB, Woo S, Song MS, Kim HJ.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Ethical Approval
This study adhered to the guidelines established by the Declaration of Helsinki and was approved by the Institutional Review Board of Chungbuk National University Hospital (CBNUH IRB, No. 2022-07-005).
Informed Consent
The patients provided written informed consent for publication.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
