Abstract
Renal epithelioid angiomyolipoma (EAML) (epithelioid PEComa of the kidney), is a rare subtype of renal angiomyolipoma with the potential for aggressive behavior and a known diagnostically challenging entity. We present a renal EAML with unusual papillary architecture and tumor cells with abundant eosinophilic cytoplasm and cherry-red nucleoli with perinucleolar halos, strongly mimicking a fumarate hydratase (FH) deficient renal cell carcinoma (RCC). We herein report our findings and discuss the morphologic, immunohistochemical, and molecular pitfalls to consider in the differential of EAML, including with FH-deficient RCC and more recently described entities: TFEB-amplified RCC and other renal tumors with alterations in TSC1/2. Novel findings in this tumor include papillary morphology and a novel telomerase reverse transcriptase promoter rearrangement, which has not been previously reported in EAML.
Introduction
Renal epithelioid angiomyolipoma (EAML) (epithelioid PEComa of the kidney) is a rare subtype of renal angiomyolipoma (AML) with the potential for aggressive behavior. EAML is defined by the World Health Organization as a renal AML consisting of greater than 80% epithelioid cells. Two morphologic patterns have been described: (1) carcinoma-like pattern composed of nests of atypical large eosinophilic cells with prominent nucleoli and nuclear inclusions, resembling ganglion cells; and (2) epithelioid and plump spindled cells in diffuse sheets.1,2 We recently encountered a renal EAML with unusual papillary architecture, mimicking renal cell carcinoma (RCC), and we report its clinical, morphologic, and molecular features here.
Case Report
A 56-year-old Asian female with 2 years of abdominal bloating underwent left radical nephrectomy for a left kidney mass. Gross examination revealed a 12.5 × 10.4 × 8 cm inferior pole circumscribed mass with red-brown-orange lobulated cut surfaces, with renal vein extension. The mass was approximately 80% hemorrhagic and necrotic. On morphology, there was a prominent papillary component, comprising approximately 30% of total viable tumor area. The papillae were lined by epithelioid cells containing abundant eosinophilic cytoplasm and round nuclei with conspicuous eosinophilic macronucleoli and peri-nucleolar halos. The remaining viable tumor showed an epithelioid and plump spindled cell pattern (approximately 50%) and carcinoma-like pattern (approximately 20%), compatible with that previously described for EAML. No usual AML areas were seen. Mitotic figures were up to 4 per 10 high-powered fields. On immunohistochemistry (IHC), tumor cells were positive for melan-A, HMB45, and scattered patchy positive for SMA; negative for pan-keratin (AE1/AE3 and CAM5.2), K7 (CK7), PAX8, CD10, CA9, TFE3, ALK; fumarate hydratase (FH), SDHB, and SMARCB1 (INI1) were retained (Figures 1 and 2).
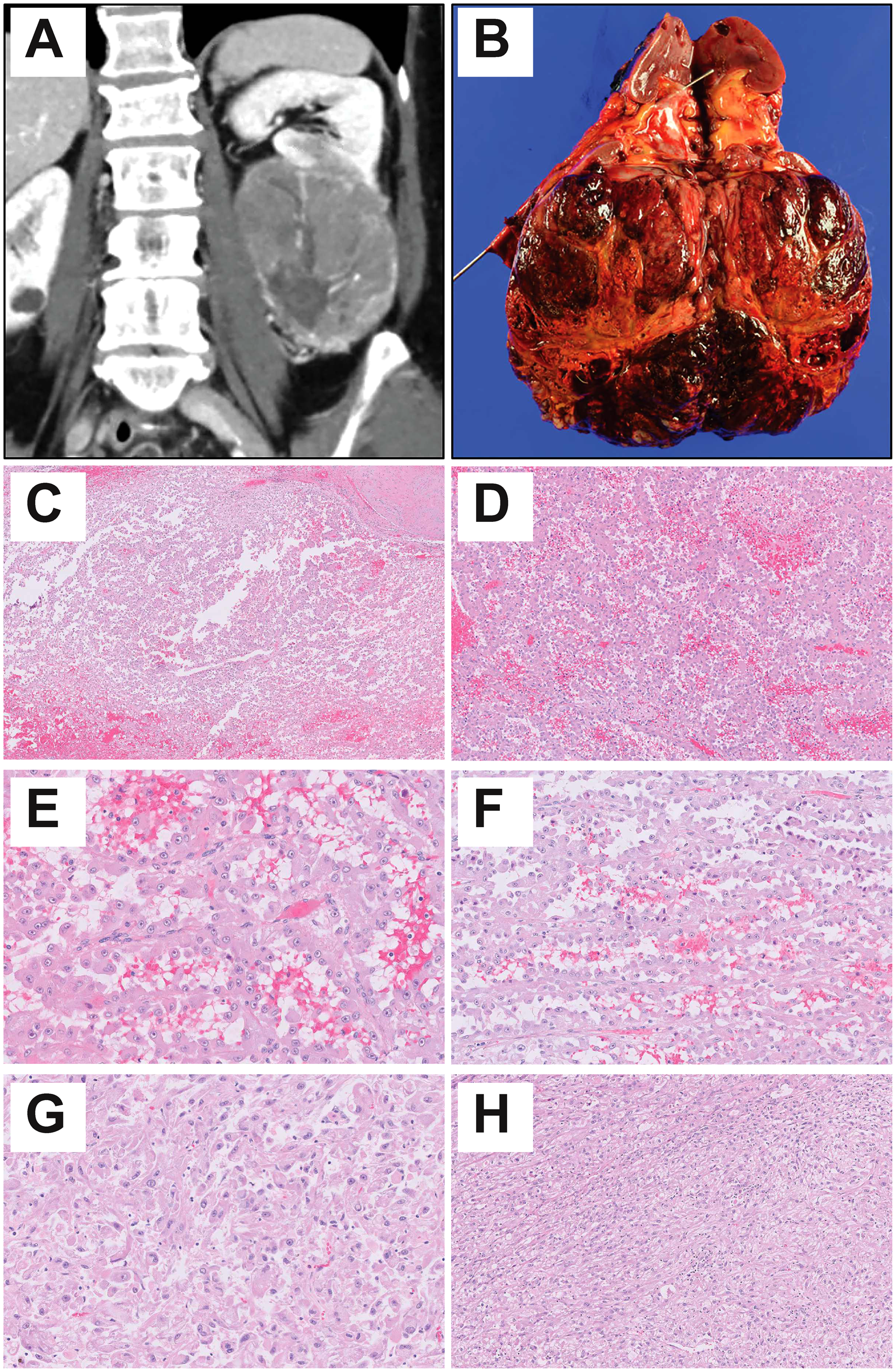
Epithelioid angiomyolipoma with papillary architecture imaging, gross and histologic features. (A) Computer tomography scan. (B) Gross photo. (C-F) Representative papillary morphology. (G) Representative carcinoma-like morphology. (H) Representative epithelioid and plump spindled morphology.
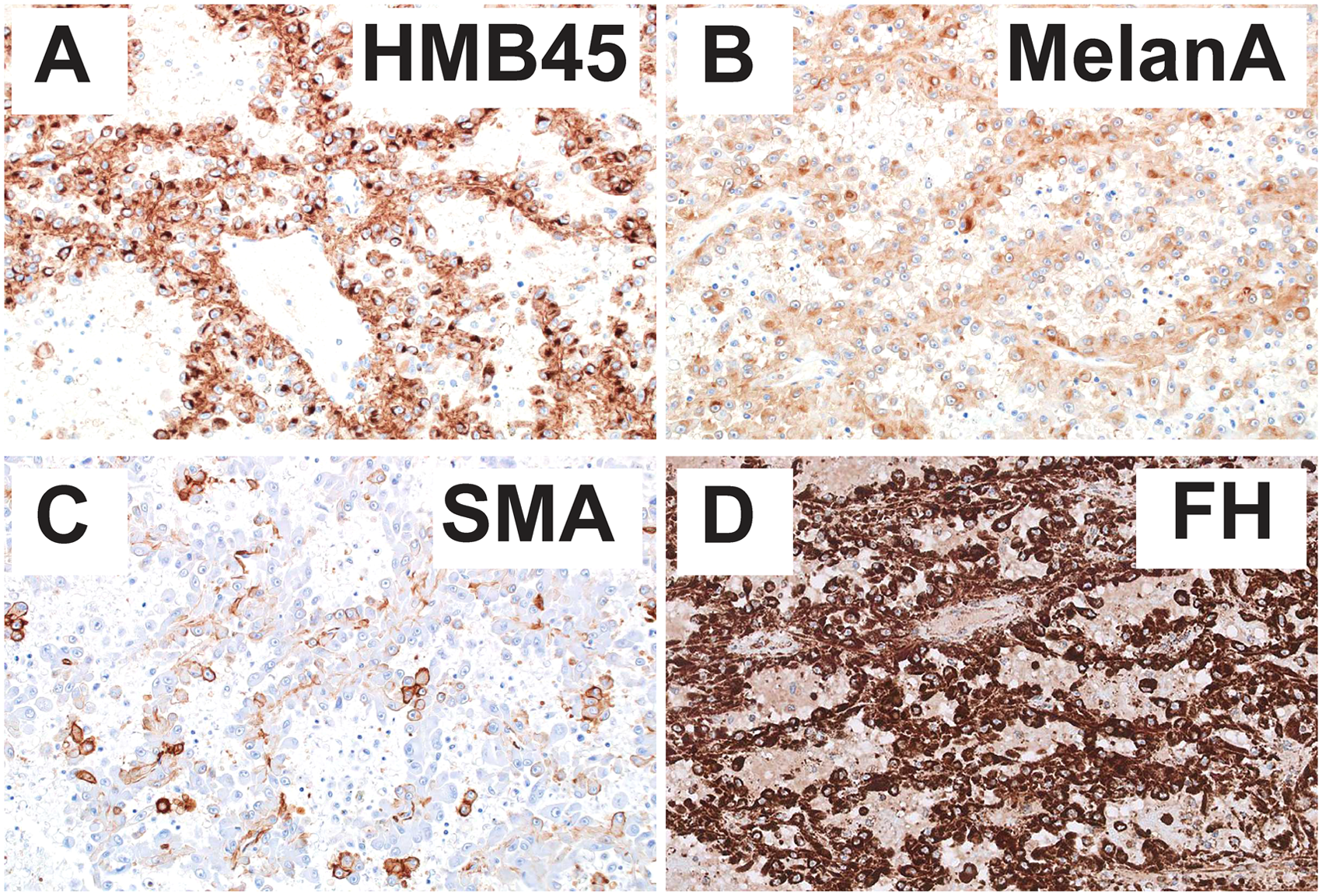
Immunohistochemical findings in epithelioid angiomyolipoma with papillary architecture. (A) HMB45, (B) melan-A, (C) SMA, and (D) fumarate hydratase (FH).
Molecular analysis utilizing the UCSF500 Cancer Gene Test (UCSF Genomic Sequencing Services Lab), a clinically validated test that uses capture-based next-generation sequencing to analyze the coding regions of 529 cancer genes, as well as select introns and noncoding sequence of 73 genes (https://genomics.ucsf.edu/content/ucsf-500-cancer-gene-panel-test-ucsf500-uc500, version 3), revealed 2 pathogenic inactivating frameshift mutations in TSC2 (p.I357fs and p.G1577fs) and telomerase reverse transcriptase (TERT) promoter structural rearrangement. The combined morphologic, IHC, and molecular findings supported a diagnosis of EAML.
Multiple subsequent surveillance scans showed possible but stable local disease recurrence and no evidence of regional spread or distant metastasis at 6 months post-nephrectomy.
Discussion
This article highlights several underrecognized morphologic, IHC, and molecular pitfalls in EAML. First, we describe an unusual papillary architecture that very closely mimics the classic morphology described for FH deficient RCC: abundant eosinophilic cytoplasm and eosinophilic macronucleoli with perinucleolar halos. A diagnosis of FH-deficient RCC requires demonstration of FH loss by IHC (ie, FH loss and/or 2SC expression) and/or molecular ancillary studies. It is important to exclude FH-deficient RCC, as these tumors are often aggressive with high mortality, associated with hereditary leiomyomatosis and RCC syndrome and necessitate genetic counseling and screening for the patient and their family members.3,4
Secondly, the morphology and IHC profile also raised consideration for MITF-altered RCC. In particular, the recently described TFEB-amplified RCC demonstrates high-grade morphology with oncocytic and papillary features. Like other MITF-altered RCC, TFEB-amplified RCC can share IHC overlap with EAML, including the potential for underexpression of pan-CK and increased expression of melanocytic markers.5,6 SMA and PAX8 stains can be most helpful in distinguishing EAML from RCC, though overreliance on a few IHC stains can be treacherous; therefore, further molecular testing to support EAML was performed on the presented specimen.
Furthermore, we highlight the importance of histologic–molecular correlation when using NGS ancillary testing for the subtyping of kidney tumors. Notably, there have been increasing tumor types in the recent kidney neoplasia literature with alterations seen in the tuberous sclerosis gene family (TSC1/TSC2). Most relevant to this differential is the eosinophilic and vacuolated tumor (EVT), also referenced as eosinophilic and vacuolated RCC. 7 In the EAML described here, correlation with the immunoprofile supports EAML over EVT.
Finally, 2 studies have identified clinicopathologic criteria for malignancy which utilize percent atypical epithelioid cells, quantification of mitotic figures, atypical mitotic figures, necrosis, tumor size, carcinoma-like pattern, and extrarenal extension,1,8 both of which would classify the presented EAML as high risk. The genetics of malignant EAML is largely unknown, though a recent study identified alterations in TP53, ATRX, RB1, APC, and NF1 as enriched in a series of metastatic relative to nonmetastatic EAML. 9 In our tumor specimen, only an additional TERT promoter structural rearrangement was identified. Although the significance of this TERT mutation is unclear in EAML, it is known that TERT promoter mutations occur in many human cancers and are associated with poor cancer-specific survival.10, amongst others
In conclusion, the EAML presented here uniquely highlights several morphologic, IHC, and molecular pitfalls to consider in the differential of EAML. Novel findings in this tumor include papillary morphology and a novel TERT promoter rearrangement, which has not been previously reported in EAML.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
