Abstract
In some instances, the central scar of renal oncocytoma can demonstrate entrapped cells with unusual morphology and aberrant immunoprofile creating potential diagnostic confusion. Herein, 100 renal oncocytomas containing scars with embedded epithelial cells were identified from 6 institutions, including nephrectomies (64% partial, 36% radical) of similar laterality (left = 51%) and sex distribution (male = 56%), with patient ages ranging from 38 to 86 years (mean = 64.3years) and tumor sizes ranging from 2 to 16 cm (mean = 5.3 cm). Immunohistochemistry was performed on all tumors for KRT7, KIT, vimentin, and CA9 with staining intensity and extensity separately analyzed. Of 4 architectural patterns of cells within the scar, 60% showed tubular pattern. Of 4 cytologies within the scar, flat/elongated (49%) and cuboidal cells (40%) predominated. Within the scar, 62% showed eosinophilic cytoplasm, with 38% showing both cleared and eosinophilic cytoplasm; notably, 79% showed higher grade nuclei than typical oncocytes. A subset of scar cells showed mucinous-like basophilic secretions (19%). Compared to background renal oncocytoma, tumor cells within the scar were more often positive for vimentin, KRT7, and CA9 and more frequently negativity for KIT. Specifically, of the notable “aberrant” immunoprofiles, 79% showed KRT7 positivity/KIT negativity/vimentin positive, 84% showed vimentin positivity/CA9 positivity, and 78% showed KIT negativity/vimentin positivity/CA9 positivity. While encountering scars within renal oncocytomas is not uncommon, what is not well appreciated is the unique morphology and immunohistochemistry of tumor cells within the scar. Comparing tumor morphology and immunoprofile of the scar to the background oncocytoma is helpful to avoid interpretative confusion.
Introduction
Of all the various diagnostic problems that genitourinary pathologists encounter with renal oncocytic neoplasia, separating renal oncocytomas from their malignant epithelial counterparts remains both a frequent and often cumbersome occurrence. Although morphology and immunohistochemistry are often sufficient for accurate diagnostic distinction, problem scenarios do occur. Specifically, we have encountered scenarios where the central scars of renal oncocytoma (reported in up to 54% in one series 1 ) demonstrate unusual morphology and with an immunoprofile not in keeping with typical renal oncocytoma. This can be especially problematic in renal mass biopsy assessment. While this situation is occasionally alluded to,2,3 delving into the literature reveals few if any formal studies targeting this phenomenon. After encountering such an index renal mass core biopsy in which the scar showed discrete potentially problematic morphologic and immunophenotypic features from the background tumor (Figure 1), herein, the aim of this study was to investigate the morphological and immunohistochemical features of renal oncocytoma scars from a large multi-institutional series.
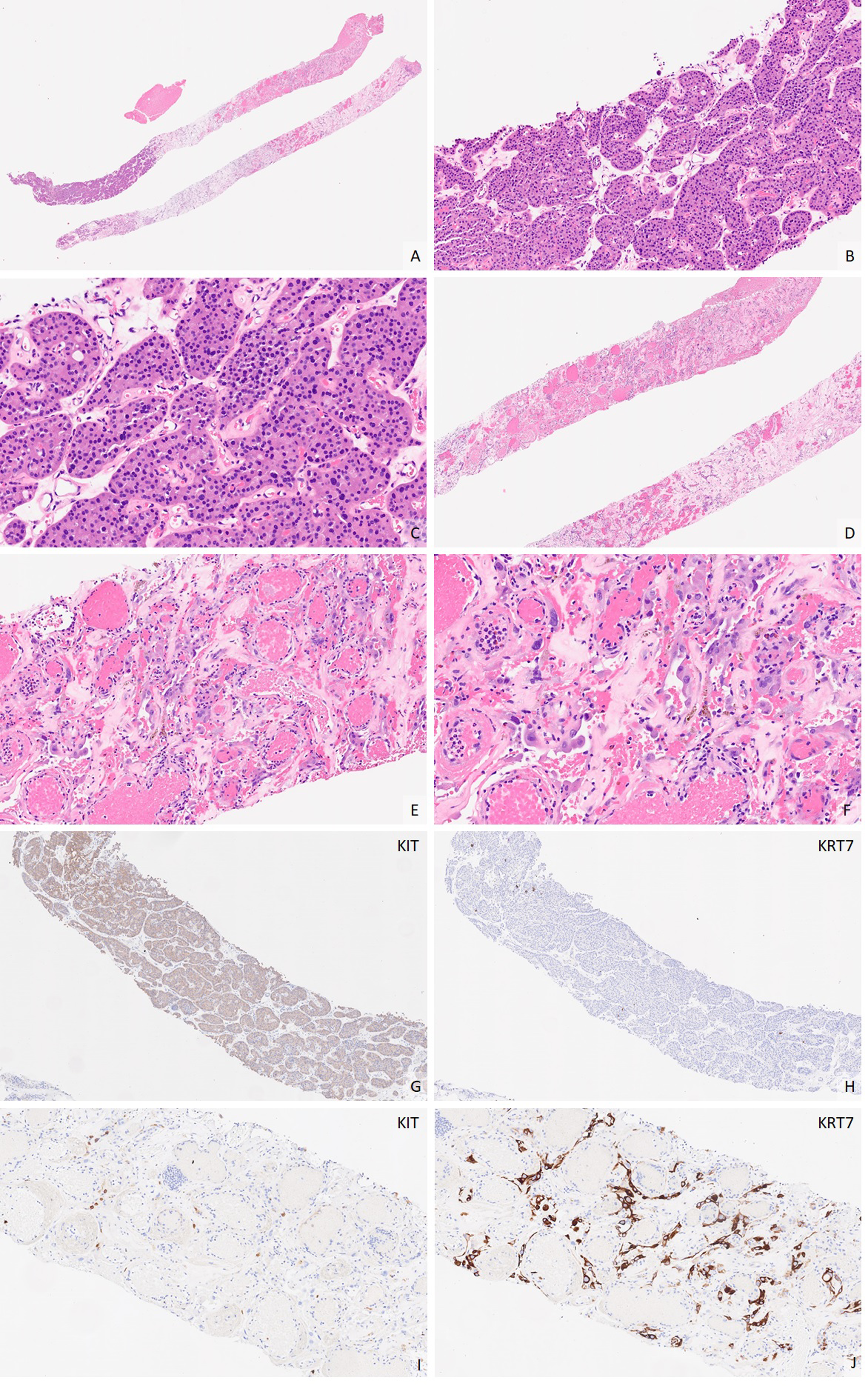
Low-power photomicrograph of a renal oncocytoma core biopsy (A, 10×) demonstrates notable demarcation between clusters of tumor cells with bland eosinophilic cells (B, 40×; C, 200×) and scarred tumor region (D, 20×), the latter showing distinct cytologic features including hobnail cuboidal cells with marked nuclear degenerative atypia (E, 100×; F, 200×). Expected diffuse KIT (G, 40×) and rare and scattered KRT7 expression (H, 40×) are present in non-scarred tumor; however scarred tumor regions frequently exhibit aberrant immunophenotype, such as a decreased or lack of KIT expression (I, 100×) and aberrant KRT7 (J, 100×) expression. KRT7, keratin 7.
Methods and Materials
A retrospective search was undertaken at 6 institutions to identify potential study specimens. A total of 400 nephrectomies were reviewed for renal oncocytomas containing gross central scar or microscopic central scars (encompassing at least one 4× objective low power field) with admixed epithelial cellular elements.
Diagnoses for all patients were confirmed by at least one genitourinary pathologist. Clinicopathologic features were recorded on all specimens. Detailed morphologic features within the scar were recorded including predominant architectural pattern, predominant cytology, cytoplasmic quality, nuclear characteristics, and luminal contents. Similarly, an immunohistochemical assessment (performed on one formalin-fixed paraffin-embedded whole slide section) of the epithelial cell component within the scar compared to background tumor was performed on one representative section per specimen for KRT7 (Leica clone RN7), KIT (Leica clone YR145), vimentin (Leica clone SRL33), and CA9 (Leica clone TH22). Staining extent (percentage; negative = none or less than 5%) and intensity (semi-quantitatively scored as 0-3+) were separately evaluated, with
Results
From 400 nephrectomies of renal oncocytoma, 100 tumors (25%) were identified that showed gross central scar (specimens only noted the presence of scar, not linear measurement) or microscopic central scars with admixed epithelial cellular elements. Specimens included almost partial nephrectomies (64%) and radical nephrectomies (36%) of similar laterality (left = 51%), with slight male predominance (male = 56%). Patient ages ranged from 38 to 86 years (mean = 64.3 years), with tumor sizes ranging from 2 to 16 cm (mean = 5.3 cm). No tumor had a prior biopsy. Significant mitotic activity, tumor cell necrosis, or papillary formation were not found in any tumor. Five renal oncocytomas showed vascular invasion, and 12 tumors showed adipose invasion (11 perinephric fat, 1 sinus fat); 1 tumor showed both vascular and perinephric adipose tissue invasion. Of 4 architectural patterns of the cells within the scar (tubular, trabecular/solid, microcystic/, and angiomatoid), tubular pattern was most common (60%). Of 4 cytologies within the scar (flat/elongated, cuboidal, hobnail, vacuolated/signet ring-like), flat/elongated and cuboidal were the predominant morphologies noted (49% and 40%, respectively). Within the scar, the predominant cytoplasm within the admixed epithelial cells was more often eosinophilic (62%; vs both cleared and eosinophilic in 38%). The cells showed “higher grade” nuclei with prominent nucleoli compared to typical oncocytes (79%; vs oncocyte-like nucleus in 21%), and infrequently showed mucinous-like basophilic secretions (19%). Table 1 depicts detailed morphologic features of study specimens. Figure 2 depicts prototypic histology of study tumors.
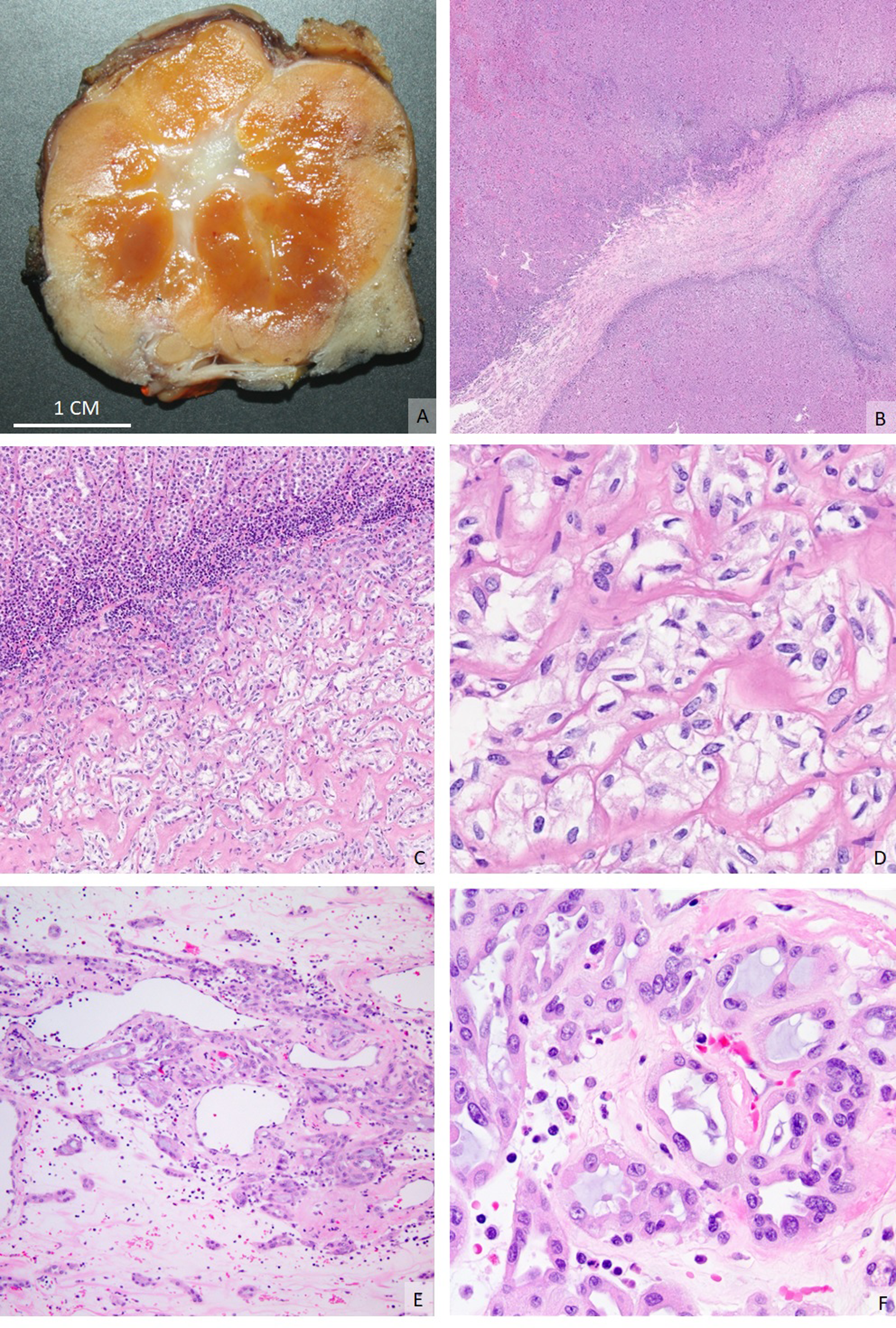
Partial nephrectomy of oncocytoma exhibiting grossly-identifiable central stellate scar (A), readily appreciable at low power magnification (B, 40×), showing a transition of typical oncocytoma cells with round nuclei and eosinophilic cytoplasm (C, 40×) to flattened/cuboidal tumor cells with striking cytoplasmic clearing embedded in the scar (D, 200×). Especially in a biopsy setting consisting of mostly scar elements of tumor, there is significant morphologic overlap with clear cell renal cell carcinoma. Another case of entrapped cells within an oncocytoma scar showing an unusual microcystic pattern of cuboidal eosinophilic cells with mucinous-like basophilic secretions (E, 40×; F, 200×).
Morphologic Features of Renal Oncocytoma Scars.

Immunohistochemically, tumor cells within the scar compared to background renal oncocytoma were often positive for vimentin, showed more frequent positivity/stronger positivity for KRT7, showed more frequent negativity/reduced positivity for KIT, and showed frequent positivity for CA9 (Table 2). Specifically, of the notable “aberrant” immunoprofiles (
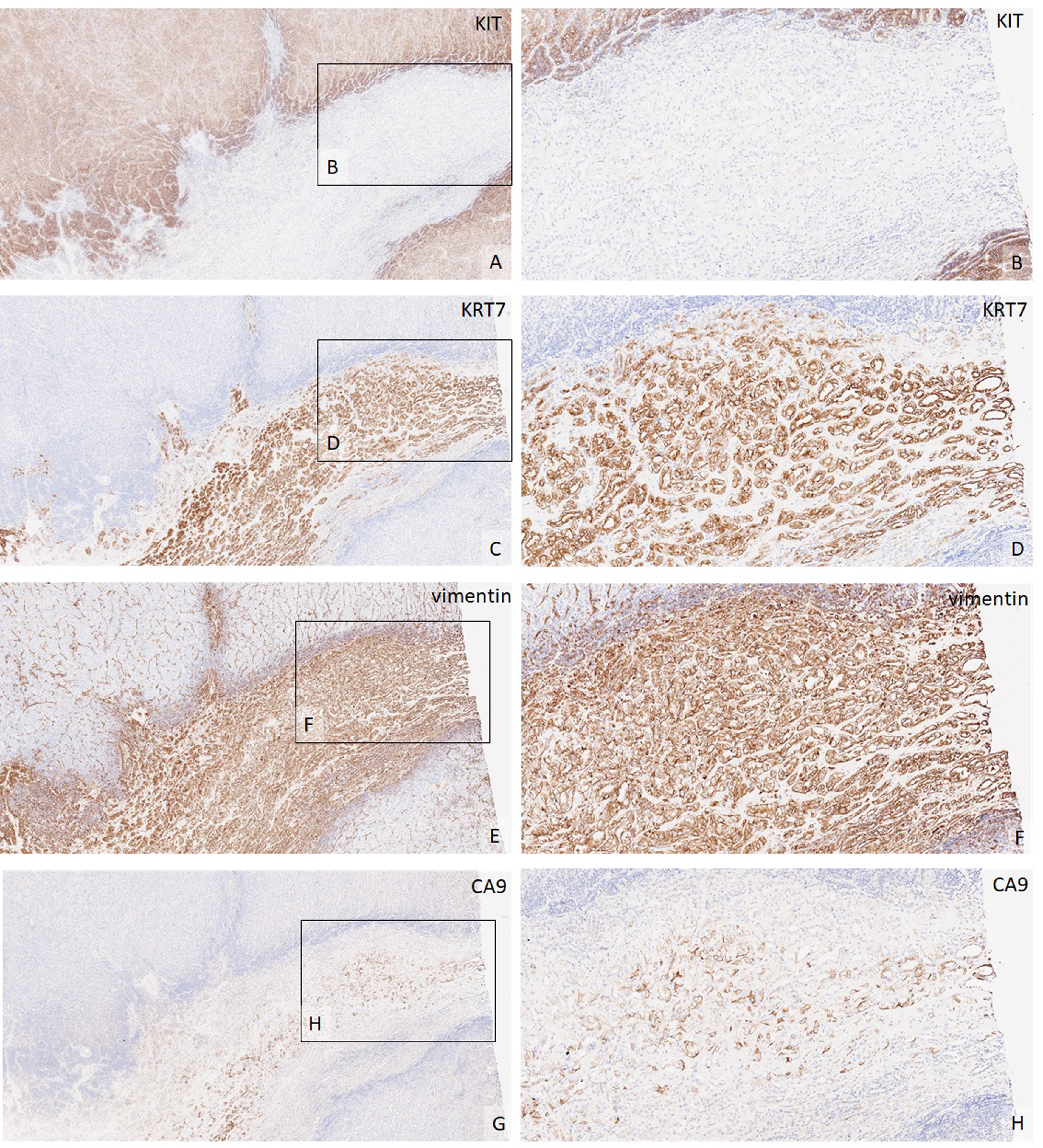
Paired immunophenotype of tumor cells from oncocytoma in figure 2 showing notable variation in staining between scarred and non-scarred regions. Tumor cells in the scarred region lack KIT (A, 40×; B, 100×), and demonstrate diffuse expression for KRT7 (C, 40×; D, 100×), diffuse expression for vimentin (E, 40×; F, 100×), and moderate expression for CA9 (G, 40×; H, 100×). CA9, carbonic anhydrase 9; KRT7, keratin 7.
Comparative Immunohisochemical Assessment of Tumor cells Within Renal Oncocytoma Scar Versus Background Tumor.
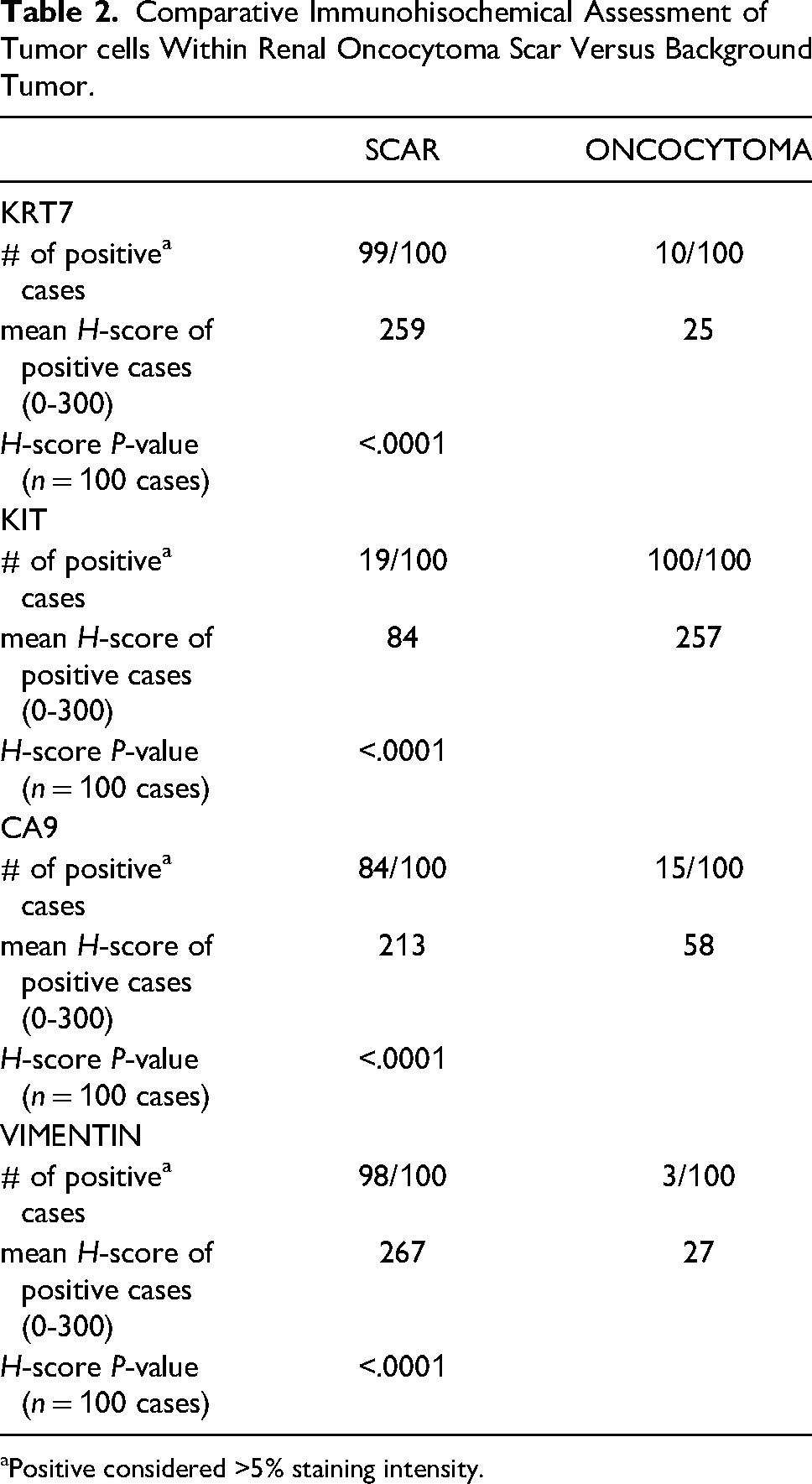
Positive considered >5% staining intensity.
Discussion
The identification of prominent central scars within renal oncocytoma is not a novel finding, with some of the important early renal oncocytoma studies reporting an incidence of 33% to 54%.1,4 This is overall similar to the 25% we encountered in our retrospective review (our incidence is slightly lower than that previously reported as we only included tumors that contained entrapped epithelial cellular elements, not just central acellular scar). However, formal diagnostic assessment of renal oncocytoma scars has mainly been restricted to the radiology literature,5,6 with few studies reviewing any potential immunohistochemical differences in the scar versus background tumor (focused mainly on vimentin7,8), and none, to our knowledge, performing a detailed morphologic comparative assessment. While in some instances this may not present as a diagnostic conundrum, given the lack of adequate pathology literature on this topic, we found it important to recognize potential diagnostic issues, especially when scars are prominent and/or in renal mass biopsy scenarios in which only the scar is sampled. After we encountered an index core biopsy showing a striking example of renal oncocytoma with clear-cut areas of traditional pattern oncocytoma as well as oncocytoma scar (the latter showing discrete morphology and immunoprofile compared to background oncocytoma elements; Figure 1), the current study was undertaken on resection specimens to further expand this potentially diagnostic issue.
In addition to lacking significant mitotic activity, tumor cell necrosis, or true papillary formation, renal oncocytomas should lack significant areas of clear cells.9–11 In our series, while over one-third (38%) of the renal oncocytoma scars showed cytoplasmic clearing, it is important to note that these foci were typically less than one low power field (4× objective) and showed an abrupt change in cytoplasmic tinctorial clearing (Figure 2). In the absence of a collision tumor (uncommon 12 ) or tumor-to-tumor metastasis (rare 13 ) as a putative explanation for this unusual clear cell finding, appreciating the background/typical renal oncocytoma morphology which should be apparent on a resection specimen should help arrive at the correct diagnosis. However, diagnostic hazards abound when these clear cell foci of renal scars (which often show “atypical” non-oncocyte-like nuclei in up to 79% of tumors) are exclusively sampled during renal mass biopsy.
In our experience, although encountering mucin-like cytoplasmic elements in a renal oncocytoma is uncommon (Figure 2) and would make other oncocytic tumors such as urothelial carcinoma with glandular differentiation or metastasis a diagnostic consideration, it has been rarely reported in renal oncocytoma. 14 Nonetheless, finding mucinous-like secretions in cuboidal cells of microcystic architecture with cleared/eosinophilic cytoplasm showing atypical nuclei (Table 1) could present a diagnostic pitfall if the fact that these findings are mainly restricted to the scar is not appreciated (one wonders if putative reported “collision” oncocytoma with mucinous tubular and spindle cell carcinoma 12 could in fact represent the aforementioned phenomenon described in our study).
With KRT7 the single most utilized diagnostic marker (94%) among urologic pathologists in the assessment of renal oncocytic neoplasms,
11
it is notable that in our series almost all (99%) tumors showed statistically significant positive reactivity, including many with moderate staining extent/intensity (mean
Often vilified as an futile diagnostic immunostain in general surgical pathology, vimentin remains a helpful marker in renal epithelial tumor assessment,2,8,10,15–18,23,24 utilized as the third most used diagnostic stain (65%) among urologic pathologists in oncocytic renal neoplasia. 11 Particularly in the renal mass biopsy setting of oncocytic renal neoplasms,17,18 vimentin positivity (observed within oncocytoma scars in 98% of our tumors) among tumors in the differential diagnosis may raise the possibility of either papillary renal cell carcinoma or clear cell renal cell carcinoma. Vimentin positivity compounded with KRT7 positivity/KIT negativity (seen in 79% of our study tumors), would seemingly make renal oncocytoma even less likely a diagnosis. Importantly, in reviewing the literature focused on potential immunohistochemical differences in the scar versus background oncocytoma, we found only 2 studies notably depicting vimentin positivity in up to 73% of oncocytomas restricted mainly to cells either within or bordering the scar, findings similar our study showing statistically significant positivity compared to the background tumor (Table 2). However, these 2 previously-published studies have not received recognition as potential pitfalls of incorporating vimentin among diagnostic immunohistochemistry of renal oncocytomas.2,8,10,15–24,26
Compared to older immunostains, CA9 is a more novel helpful addition to the growing list of available diagnostic markers in the assessment of renal oncocytic neoplasms. Of the few large-series studies that have investigated CA9 staining in renal oncocytomas, they have shown utility with expected negative staining.17,26 In our series, while all (100%) of our background renal oncocytomas were also negative for CA9, a majority (84%) of the cellular component in renal oncocytoma scars showed positive staining for CA9, notably with moderate positivity (mean
Although the exact etiology of scars within renal oncocytomas remains unknown and whose presence may not necessarily correlate with tumor size (in our series scars were found in tumors as small as 2 cm; lack of available on scars’ gross size measurement precluded formal scar to tumor volume comparison), we hypothesize it is a degenerative and perhaps hypoxic phenomenon (based on expression of CA9, upregulated in hypoxic environments via the hypoxia-inducible factor 1 pathology, 27 in a subset of our tumors) which might explain some of the unique morphologic and immunohistochemical features observed in our study. While the striking KRT7 positivity found in renal oncocytoma scars could raise the possibility that these cells represent entrapped non-neoplastic tubules (KRT7 positivity within distal tubule/collecting duct has been previously described20,21), based on the graduated transition in morphology from typical oncocytoma to aforementioned scar cell morphology with co-expression of KIT in many tumors, in our opinion, the scar cells are indeed neoplastic.
In summary, while we and others have shown that encountering scars within renal oncocytomas is not uncommon, what is not appreciated is the unique morphologic and immunophenotypic features of the tumor cells within the scar. On a resection specimen, comparing tumor morphology and immunoprofile of the scar to the background oncocytoma is helpful to avoid interpretative confusion. However, as renal mass biopsy is increasingly being used in the work-up of smaller (≤4 cm) tumors including some that may be amenable to active surveillance, 28 it may be prudent to communicate with our clinicians to determine which portion of the renal mass was biopsied to avoid diagnostic pitfalls where background traditional oncocytoma elements may not be available.
Footnotes
Authors’ Contribution
ARS and AP established the study design, and ARS wrote the original draft of the manuscript. LMNC, MA, FJQ, and MGM contributed study specimens and contributed to editorial changes; MA also assisted with figures and figure legends.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Retrospective study not interfering with diagnosis and patient management.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
