Abstract
Background
Besides Crohn's disease (CD), there are a variety of other causes that can also lead to ulcerations in the terminal ileum. The purpose of this study was to identify useful diagnostic features for CD when evaluating terminal ileum biopsies in patients with endoscopic finding of ulcers.
Methods
Five hundred and seventy-one patients with endoscopic finding of ulcers were included in this retrospective study. Five main histological features were analysed, which were crypt irregularity, mucosal thickening, villous stromal widening (including villous atrophy), granulomas, and pseudopyloric gland metaplasia. Clinical and pathological features were determined by uni- and multivariable logistic regression. Then another independent cohort of 99 patients was established for verifying this nomogram.
Results
The crypt irregularity, mucosal thickening, and villous stromal widening were combined to be considered as one new variable named mucosal architectural change which was an independent variable in diagnosing CD. We found that mucosal architectural change, age <40 years, the presence of granulomas, and the presence of pseudopyloric gland metaplasia were independent factors for the pathological diagnosis of CD. Then nomogram was developed, with receiver operating characteristic (ROC) curve (area under the ROC curve [AUC] = 0.927) in training sets, and ROC curve (AUC = 0.913) in validation sets.
Conclusions
We found mucosal architectural change is very helpful in distinguishing CD from non-CD patients. In the context of small biopsy which may lack full scope of changes, the model developed by combining these key features is valuable in predicting a diagnosis of CD, especially in younger patients (age <40 years).
Introduction
Crohn's disease (CD), 1 of 2 subtypes of inflammatory bowel disease along with ulcerative colitis (UC), is an idiopathic chronic inflammatory disease of the gastrointestinal tract, the symptoms of which develop with recurrence and remission. All parts of the gastrointestinal tract are affected in CD, but most commonly affecting the distal small bowel and colon.1,2
Ileo-colonoscopy with multiple biopsy specimens is the preferred method for the diagnosis of CD.3–5 Ileoscopy and biopsy can be performed in at least 85% of colonoscopy procedures and increase the diagnostic yield.6–9 The diagnosis of CD in the ileal periphery by ileoscopy and biopsy is superior to that of radiological techniques, including magnetic resonance imaging and computed tomography.10–12 The presence of ulcers in the terminal ileum, such as aphthous ulcers, longitudinal or fissuring ulcers, is common and characteristic endoscopic feature in CD.
The pathological features of CD in terminal ileum biopsy samples include chronic inflammation (increased lymphocytes and plasma cells in lamina propria), active inflammation (neutrophils in lamina propria and epithelium, cryptitis, and crypt abscess), epithelial injuries (erosion and ulceration), crypt irregularity (patchy crypt distortion), non-necrotizing granulomas (unrelated to crypt damage), pseudopyloric gland metaplasia, and abnormal villous structures.13,14 However, none of the above features is unique in CD. Although the presence of granulomas is the most important feature, granulomas can only be found in less than 50% patients in the terminal ileum biopsy.15,16 With that being said, it is still very challenging to make a diagnosis of CD based on mucosal biopsy tissues.
The main purpose of this study was to select a suitable and practical model for the diagnosis of CD by evaluating the combination of histological and clinical characteristics of patients with terminal ileum ulcers to improve the yield and accuracy of diagnosing CD.
Material and Methods
Case Selection
The human ethics committee of Sir Run Run Shaw hospital affiliated to Zhejiang university approved the study (No. 2022-0169). Five hundred and seventy-one cases with endoscopic findings of ulceration at the terminal ileum were identified in ileo-colonoscopy reports between January 2005 and December 2020. At least 3 mucosal biopsy specimens were taken from each patient. The final diagnosis of CD was combined with clinical, endoscopic, radiological, and histological examinations. 13
Microscopic Analysis
The biopsy pathology reports and slides of all the patients were retrieved. All the slides were reviewed for the following histological features: Crypt irregularity, mucosal thickening, villous stromal widening (including villous atrophy), granulomas, and pseudopyloric gland metaplasia. Some variables are defined below:
Crypt Irregularity
Crypt irregularity was referred to in more than 10% of crypts showing abnormalities. The characteristics of crypt irregularity were irregular crypts (non-parallel crypts, variable diameters, or cystic expansion crypts), crypt loss, and crypt branching. The presence of more than 2 branches in well-oriented biopsy specimens was considered abnormal.3,17,18
Mucosal Thickening
Mucosal thickness was measured as the distance from the villus depression to the uppermost part of the muscularis mucosae (Figure 1A). If no mucosal muscle was observed or the mucosal muscle was not clear, it was considered a missing value.
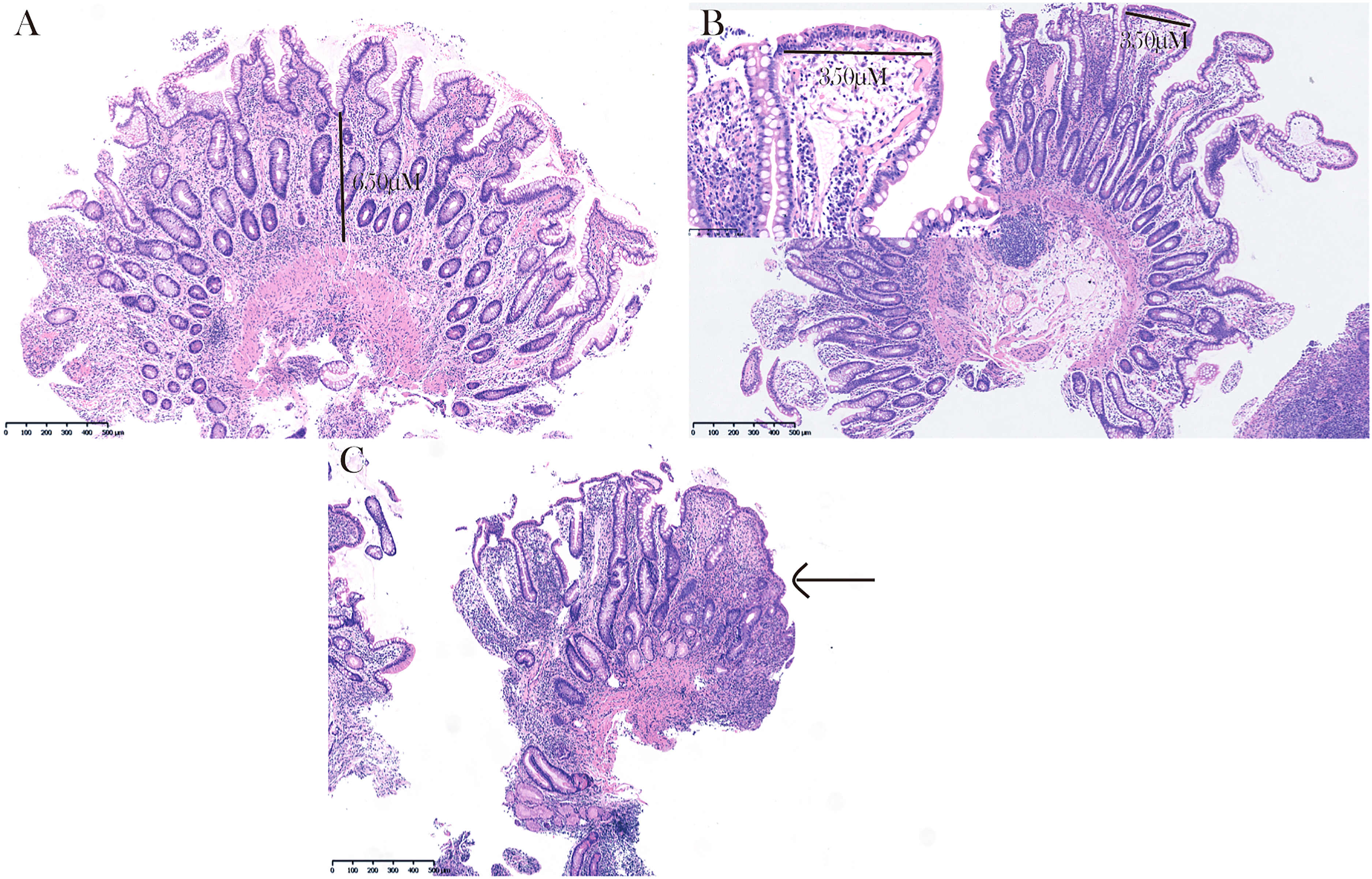
Histological features. (A), the mucosal thickness. (B and C), villous changes, including (B) villous stromal widening and (C) villous atrophy.
Villus Stromal Widening
Ileal villi were typically shorter and less straight than the proximal small intestinal villi. 19 To better evaluate villous width, we proposed the concept of villous stromal width, which was referred to the distance between the basement membrane beneath the overlying epithelial cells on both sides of the villus. In this study, the largest villous stromal width was measured in villus-intact biopsy tissue (Figure 1B). Villous atrophy was considered a special phenomenon of villous stromal widening (Figure 1C). Simple villous shortening without villous stromal widening was not included in this study. The width of the villus just above the lymphoid follicles was not calculated.
Nomogram Construction and Validation
The patients were randomly split into the training and validation cohorts at a ratio of 4:1. Receiver operating characteristic (ROC) curves were established and the areas under the ROC curves (AUC) were calculated to evaluate the significance of mucosal thickness and villous stromal width in the diagnosis of CD on the training cohort. According to their sensitivities and specificities, the optimal cut-off points for each curve were determined, both of which were of equal importance. Positive predictive values (PPVs) and negative predictive values (NPVs) were also reported.
The relationship between CD as the dependent variable and the diagnostic factor as the independent variable was analysed using binary logistic regression. To avoid multicollinearity, highly correlated risk factors were combined. Then, the clinical and pathological features were determined by multivariable logistic regression, and a nomogram was built based on these results.
Model validation included 2 parts: internal and external validation. First, calibration was performed for the nomogram and the number for bootstrap repetitions was 1000 to internally validate the nomogram. Discriminant ability was determined by analysing the area under the ROC curve. Second, external authentication was studied in a validation cohort.
Statistical Analysis
Data were analysed by SPSS (version 22.0; IBM Corporation) and R software (Version 4.1.1, http://www.r-project.org) for Windows. In all cases, statistical significance was set at P < .05.
Results
Patient Characteristics and Histopathological Findings
All 571 cases were followed up; some of them were reviewed using the latest electronic medical records, and the patients in some were contacted by telephone. Thirty-two patients were not contacted. Next, the pathologist provided a microscopic description of the samples. Among them, there were 48 specimens without the muscularis mucosae, and 31 specimens in which villous stromal width could not be measured. Finally, 460 cases were included in this study with a training set (n = 361) and a validation set (n = 99).
The small intestine biopsy in CD usually showed various changes in the mucosa and infiltration of inflammatory cells. The mucosal changes included abnormal crypts (distortion, widening, and shortening), mucosal thickening, pseudopyloric gland metaplasia, lymphatic expansion, granulomas, and villous widening. The entire mucosa was infiltrated by mixed inflammatory cells, including lymphocytes, plasma cells, neutrophils, and eosinophils. There were also many lymphoid follicles. The mucosal changes in the small intestine of CD were usually diffuse, which meant that almost all crypts were abnormal on 1 piece of biopsy tissue. In the non-CD group, the diseases included UC, intestinal tuberculosis, Henoch-Schönlein purpura, and drug-induced colitis, etc (Table 1).
Differential Diagnoses of Ulcer in the Terminal Ileum.

Definition of Mucosal Architectural Change
We compared the intergroup differences between the CD group and the non-CD group in the training set. Among 361 cases, 186 patients were confirmed with CD and 175 patients without CD (Table 2).
Demographics and Clinicopathological Characteristics in Crohn's disease (CD) and non-CD Patients.
For mucosal thickness measurement, a cut-off value of 500 μm showed a sensitivity of 77.4% (95% confidence interval [CI] 70.7-83.2), specificity of 87.4% (95% CI 81.6-92.0), PPV of 86.7% (95% CI 80.4-91.3), NPV of 78.5% (95% CI 71.9-83.9), and AUC of 88.4% (Figure 2A). Therefore, thickened mucosa was defined when the mucosal thickness was more than 500 μm.
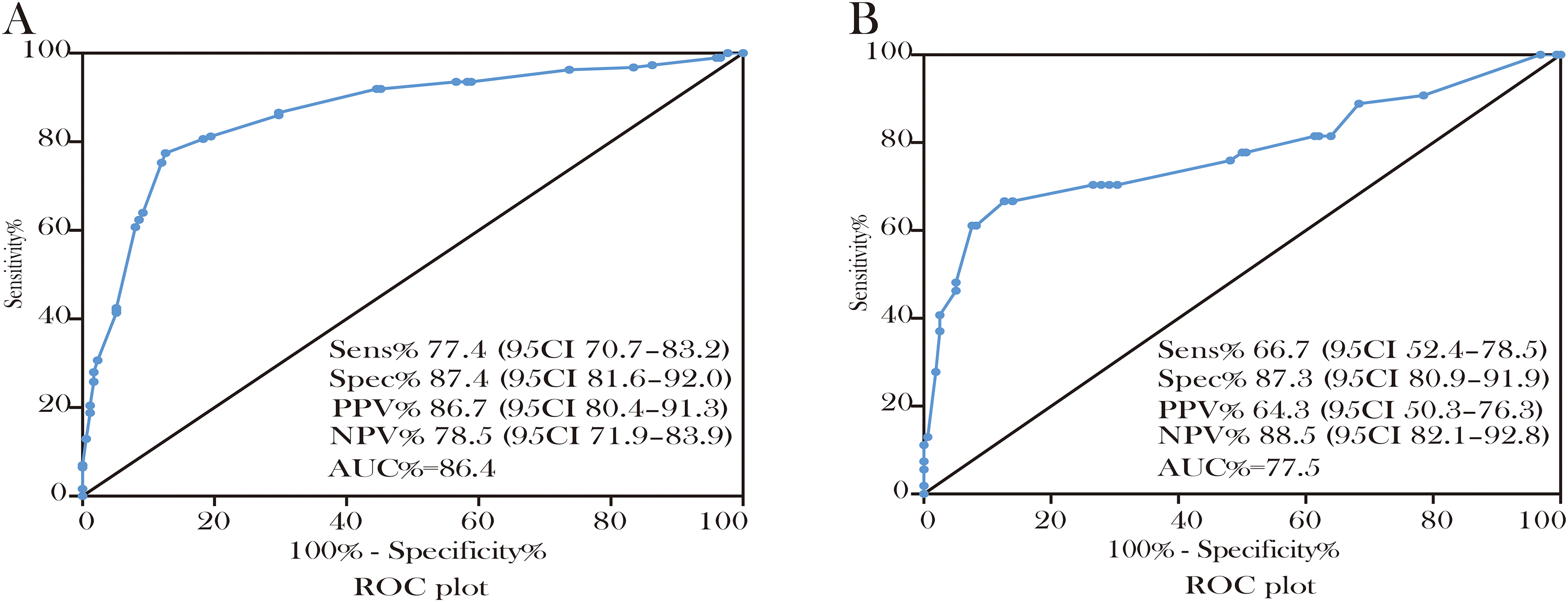
Definition of mucosal architectural change. (A and B), ROC curves for the best cut-off values of mucosal thickness and largest villus stromal width. Sensitivity, specificity, PPV, and NPV with their 95% CI and AUC were reported. Abbreviations: ROC, receiver operating characteristic curve; AUC, area under the ROC curve; PPV, positive predictive value; NPV, negative predictive value; CI, confidence interval.
For the measurement of the largest villous stromal width, we found that a cut-off value of 275 μm showed a sensitivity of 66.7% (95% CI 52.4-78.5), specificity of 87.3% (95% CI 80.9-91.9), PPV of 64.3% (95% CI 50.3-76.3), NPV of 88.5% (95% CI 82.1-92.8), and AUC of 77.5% (Figure 2B). The villous with the largest villous stromal width of more than 275 μm was considered as a widened villus.
The correlations among crypt irregularity, mucosal thickening, and villous stromal widening were examined using Spearman's rank correlation coefficient, and they each showed a significant positive correlation (Spearman's coefficient > 0.7, P < .001). They were combined to obtain a new variable named mucosal architectural change.
Mucosal architectural change was defined as having the following 3 characteristics: Crypt irregularity, mucosal thickening, and villous stromal widening. In univariate logistic regression, the mucosal architectural change was an independent variable. The sensitivity, specificity, PPV, and NPV for the presence of CD were 74.2%, 91.4%, 90.2%, and 76.9%. It showed even higher specificity (98.9%) among patients under the age of 40.
Univariate and Multivariate Logistic Regression of the Histological Variables
The following indicators were selected as the diagnostic variables of CD: Sex, age, mucosal architectural change, granulomas, and pseudopyloric gland metaplasia. The results of the univariate logistic regression analysis of each biopsy feature association with CD indicated that all the variables showed statistical significance.
The above variables were analyzed using multivariate logistic regression. In the binary logistic regression, we found that mucosal architectural change (odds ratio [OR] 24.670; 95% CI 11.197-54.358; P < .001), age <40 years (OR 8.531; 95% CI 4.060-17.925; P < .001), the presence of granulomas (OR 12.846; 95% CI 3.983-41.429; P < .001), and the presence of pseudopyloric gland metaplasia (OR 6.813; 95% CI 1.200-38.664; P = .03) were independent diagnostic factors for CD (Table 3).
Univariate and Multivariate Logistic Regression for Histological Variables.

Abbreviations: OR, odds ratio; CI, confidence interval.
Nomogram Development and Model Evaluation
A diagnostic model was developed by applying 4 key features identified in the multivariate logistic regression analysis, which was diagnostic index = Exp (3.206*X1 + 2.144*X2 + 2.553*X3 + 1.919*X4 – 6.988) / (1 + Exp (3.206*X1 + 2.144*X2 + 2.553*X3 + 1.919*X4 – 6.988)) [X1 = mucosal architectural change (1: Yes, 0: No), X2 = age (1: < 40, 0 ≥ 40); X3 = granulomas (1: Yes, 0: No); X4 = pseudopyloric gland metaplasia (1: Yes, 0: No)].
To make the matrix easier to use in clinical practice, a nomogram model incorporating mucosal architectural change, age, granulomas, and pseudopyloric gland metaplasia was developed (Figure 3A). To estimate the predictive accuracy of the nomogram model, a ROC curve was drawn, validating the discriminative capacity with an AUC of 0.927 (Figure 3B). For the validation group, the AUC of the nomogram model was 0.913 (Figure 3C). The calibration curve generated by 1000 repetitions of bootstrap sample corrections was shown and the curve showed that the predicted probability of this model was highly consistent with the actual probability in both the training and validation cohorts (Figure 3D and E).
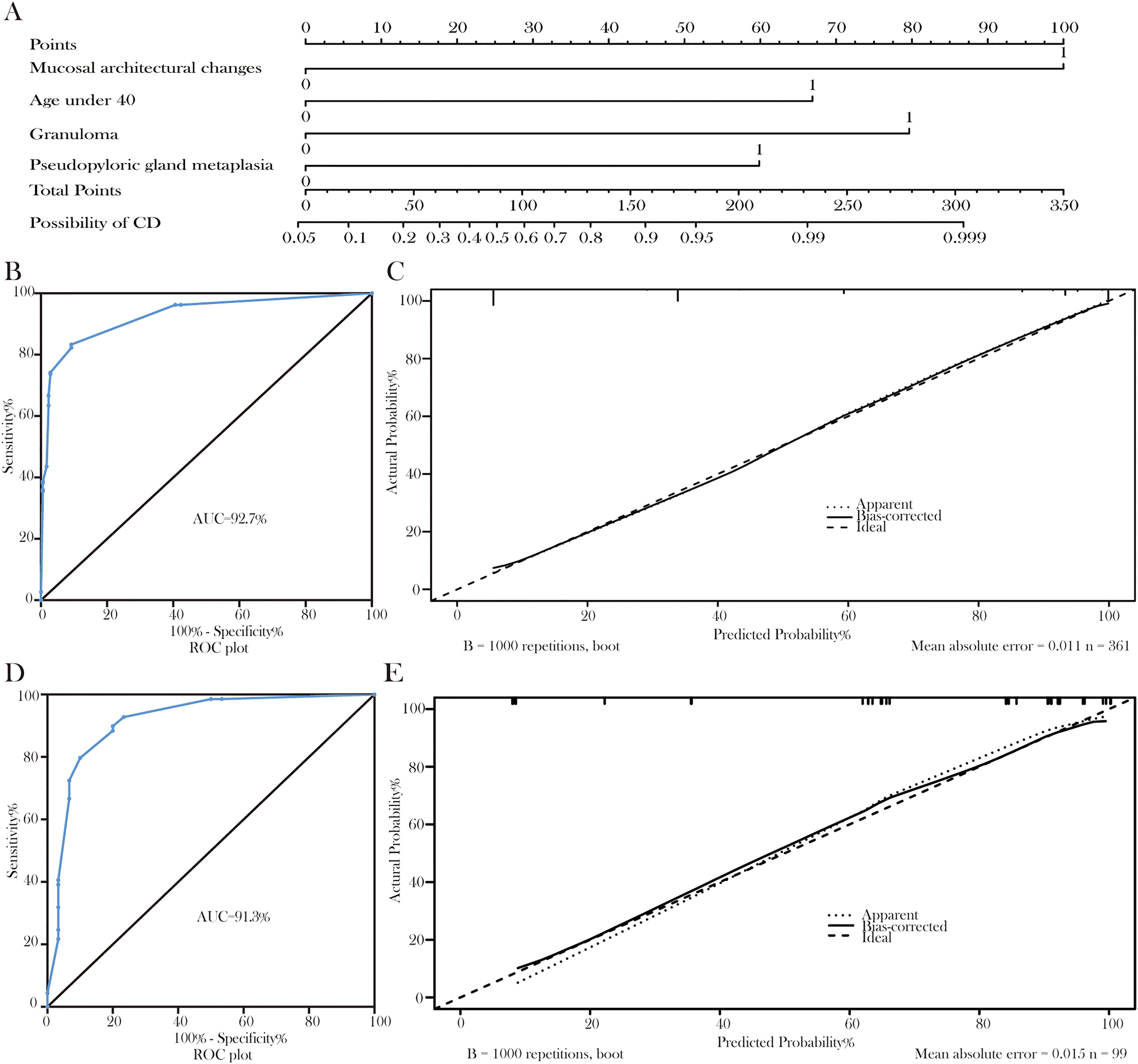
Internal and external validation of the nomogram model. (A) Nomogram for predicting CD based on mucosal architectural change and other parameters. (B) The ROC curve and (C) the calibration curve shown in the training set. (D) The ROC curve and (E) the calibration curve shown in the validation set. Abbreviations: ROC, receiver operating characteristic curve; CD, Crohn's disease.
Discussion
The pathological diagnosis of small bowel CD is usually straightforward on resection specimens, based on characteristic gross findings and microscopic features. However, on biopsy samples, most of these diagnostic features are absent or are not easily identified because of superficial nature, small sample size, and artifacts of the mucosal biopsy. The main histological patterns of CD on biopsy can also be confused with other disorders such as nonspecific active chronic ileitis, granulomatous inflammation, and especially ileal ulceration. Terminal ileum ulcers are often seen endoscopically, but the ulcer itself is not etiologic specific. Although CD is the commonest cause, there are many other conditions that cause terminal ileum ulcerations such as NSAIDs-induced ulcers, intestinal tuberculosis, ischemic/mechanical injury, eosinophilic enteritis, or others. 20 Courville et al 21 found that isolated asymptomatic ileitis does not progress to overt CD on long-term follow-up despite features of chronicity in ileal biopsies. Therefore, it is very important to recognize other useful features in diagnosing CD with or without granulomas. In this study, we quantitatively analysed mucosal changes in the terminal ileum, and we found that significant mucosal architectural change is an independent feature in CD, and in conjunction with other features, it can help to establish the diagnosis of CD and to differentiate CD from other causes of terminal ileum ulcers.
We found that in CD patients with ulcerations at the terminal ileum, mucosal thickening was often seen in the biopsy specimens, with most of the biopsy specimens having a maximum mucosal thickness in excess of 500 µM (approximately a high-power microscopic field diameter). The maximal villous stromal width in the terminal ileum biopsy specimen in CD was often widened to more than 275 µM (approximately half of the high-power microscopic field). We proposed the term “mucosal architectural change” to describe the chronic changes in the terminal ileum with crypt irregularity, mucosal thickening, and villous stromal widening. Being an independent variable, its sensitivity, specificity, PPV, and NPV for the presence of CD were 74.2%, 91.4%, 90.2%, and 76.9%. It showed even higher specificity (98.9%) among patients under the age of 40. The point of mucosal architectural change could also be intuitively seen as the highest point in the nomogram model. In conclusion, mucosal architectural change in the terminal ileum is a compelling feature in CD which may be a more dominant factor than granulomas when examining biopsy specimens, since the presence of granulomas can only be identified in a small fraction of CD patients in terminal ileum biopsies.
In fact, the mucosal architectural change is one of the most important factors in determining chronicity in chronic colitis either due to UC or CD. The significance of this feature in the terminal ileum biopsy is less emphasized in the textbook, literatures, and daily practice. This is probably because the evaluation of mucosal architectural change in the terminal ileum is relatively subjective and there are many factors that can compromise the accuracy, such as the presence of lymphoid follicles/aggregates, the small tissue, and biopsy artifacts. Most pathologists rely on the finding of pseudopyloric gland metaplasia to determine the chronicity in the terminal ileum, and again, like the finding of granulomas in the terminal ileum, the yield is very low on biopsy samples. The mucosal architectural change described in this study includes increased lamina propria inflammation, villous and crypt irregularity, and overall mucosal change. These features should be able to be evaluated in all terminal ileum biopsies. The cut-off values we determined in this study will also help pathologists to more reliably evaluate the mucosal architectural change. If either or both other 2 key features, non-necrotizing granulomas, and pseudopyloric gland metaplasia, are also identified on the biopsy, the overall pathologic findings are strongly supportive of a diagnosis of CD.
No cases were identified in the non-CD group that simultaneously fulfilled these 4 features. We analysed non-CD cases with mucosal architectural change under 40 years old, especially intestinal tuberculosis. Intestinal tuberculosis can present with very similar histological changes mimicking CD, and the differential diagnosis between the 2 diseases can be difficult. Intestinal tuberculosis is characterized by large, confluent granulomas often with caseation.22,23 With help of the model developed in this study, and in conjunction with clinical, endoscopic, and radiologic findings, we could confidently differentiate intestinal tuberculosis from CD.
This study has some limitations. We only examined the specimens with endoscopic finding of terminal ileum ulcers. CD patients with no terminal ileum ulcers but may present with other features such as strictures were not included in this histopathologic study. In addition, we found that endoscopically unremarkable terminal ileum in both CD and non-CD patients were also unremarkable microscopically in most cases. This may represent minimal disease activity, nonspecific reaction, or patients being in remission. Therefore, the values of our finding of mucosal architectural change and the model in CD patients with no terminal ileum ulcers are uncertain, and future study can address this issue. Another limitation is that we did not compare the histologic findings in colonic mucosal biopsies in these patients. The correlation of findings in the terminal ileum with the changes in the colon biopsies was not investigated. The main reason is that the chronic colitis in both CD and UC on the colonic mucosal biopsy is essentially indistinguishable, even with the presence of granulomas.
Conclusion
Diagnosing CD based on the terminal ileum mucosal biopsy is still challenging. Although the presence of non-necrotizing granulomas is important, it is not entirely specific to CD and it can only be identified in a small percentage of the cases. On the other hand, we carefully described and defined the mucosal architectural change of the terminal ileum in CD in this study. The findings emphasize the importance of recognizing mucosal architectural change as a reliable feature to confirm the chronicity of the terminal ileum in patients with CD. The model developed based on this finding and other key features can predict CD in patients with terminal ileum ulcers, especially in younger patients (age <40 years).
Footnotes
Authors’ Contributions
T.Z., L.Z., and Z.J. contributed to the study concept and design, and drafted the manuscript. T.Z., W.T., Y.W., Y.P., Y.F., J.X., and Q.C. performed procedures, and collected the data. W.T., Y.W., Y.P., Y.F., J.X., Q.C., and Z.J. analysed and interpreted the data. All authors read and approved the final paper.
Ethical Approval
This study complies with the Declaration of Helsinki. The study was approved by the Ethics Committee of Sir Run Run Shaw hospital affiliated to Zhejiang university (No. 2022-0169).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 81800459)
