Abstract
Xanthogranulomatous ureteritis is a very rare process characterized by the presence of foamy histiocytes in a background of chronic active inflammation affecting the ureteral wall. Herein, we describe a case of a 64-year-old man with bladder cancer affecting the left posterolateral wall of the bladder. Radiologically, there was a suspicion of multifocal involvement of the ureteral wall. The patient underwent a radical cystectomy with bilateral pelvic lymphadenectomy and a laparoscopic left nephroureterectomy. Histopathologic examination of the radical cystectomy revealed an invasive high-grade urothelial carcinoma. The wall of the left ureter was replaced by abundant foamy histocytes and a mixed inflammatory infiltrate with lymphocytes and plasma cells consistent with xanthogranulomatous ureteritis. In this report, we highlight the importance of awareness of this benign process when observing a ureteral mass in cancer patients.
Introduction
Xanthogranulomatous pyelonephritis is a well-known and well-characterized histological entity that typically is associated with urinary tract infections by gram-negative bacteria or, less commonly, by Staphylococcus aureus, group B Streptococcus, Candida, Klebsiella, or Bacteroides.1,2 In contrast, xanthogranulomatous ureteritis is a very rare process that affects the ureteral wall. It is characterized by the infiltration of foamy histiocytes in a background of chronic inflammation (lymphocytes, giant cells, and plasma cells) and, occasionally, acute inflammation (neutrophils). 3 To our knowledge, only five cases of xanthogranulomatous ureteritis have been published to date.3‐7 Here, we describe a new case and conduct a review of the literature on xanthogranulomatous ureteritis.
Case Report
A 64-year-old man with no personal or family medical history of interest presented with nocturia, frequency, urinary urgency, and hematuria for a year. Abdominal ultrasound revealed left ureterohydronephrosis and signs of renal cortical atrophy. In addition, an 8 × 7 × 4 cm mass in the left posterolateral wall of the bladder and lymphadenopathy in the left iliac chain were observed.
Computed tomography (CT) scan revealed that the bladder mass infiltrated the seminal vesicles and prostate. Left uretero-hydronephrosis and renal atrophy were also seen. In addition, solid areas were identified in the wall of the left ureter suggestive of probable multifocal tumor involvement.
Given these findings and due to the large size of the lesion, a transurethral resection of the bladder (TURB) with partial resection of the lesion was performed. The TURB biopsy was reported as a high-grade urothelial cell carcinoma infiltrating the muscularis propria. Therefore, the patient received six cycles of neoadjuvant chemotherapy with carboplatin plus gemcitabine and subsequent maintenance treatment with avelumab (five doses). Subsequently, a new CT scan was performed, which revealed that the retroperitoneal lymphadenopathy did not show any changes. Thus, a radical cystectomy with bilateral pelvic lymphadenectomy and a laparoscopic left nephroureterectomy was performed. During surgery, a segment of the left ureter that was sent for an intraoperative study by frozen sections showed no presence of invasive carcinoma.
In the radical cystoprostatectomy specimen, a 3 × 2.5 cm tumor mass was identified in the bladder wall. The histologic diagnosis was invasive high-grade urothelial carcinoma originating in the left posterolateral wall of the bladder that invaded the perivesical soft tissue and the prostatic stroma (pT4a).
In the left nephroureterectomy specimen, the ureter measured 15 cm in length by 1.4 cm in diameter and was dilated. In serial sections, 4 cm from the renal pelvis, thickening of the wall and a yellowish pasty material was observed (Figure 1). Microscopically, the mucosa, submucosa, and muscular propria of the left ureter were replaced by abundant foamy histocytes causing proximal dilatation and total obliteration of the lumen (Figure 2A to D). Foamy histiocytes showed clear, pale, and slightly granular cytoplasm with small, round nuclei without nucleoli. In addition, a mixed inflammatory infiltrate of lymphocytes and plasma cells and hemorrhagic areas were observed. No multinucleated giant cells, emperipolesis, phlebosclerosis, or any type of fibrosis were observed. Immunohistochemically, the foamy cells were positive for CD68 (Figure 3A) and negative for keratin AE1/AE3 (Figure 3B), thus confirming the histiocytic nature of the cells. Other negative immunostains included immunoglobulin (Ig)G, IgG4, Octamer binding protein 2 (OCT2), CyclinD1, CD1a, and S100. The laboratory tests did not show relevant abnormalities, and the serum level of IgG4 was not studied.
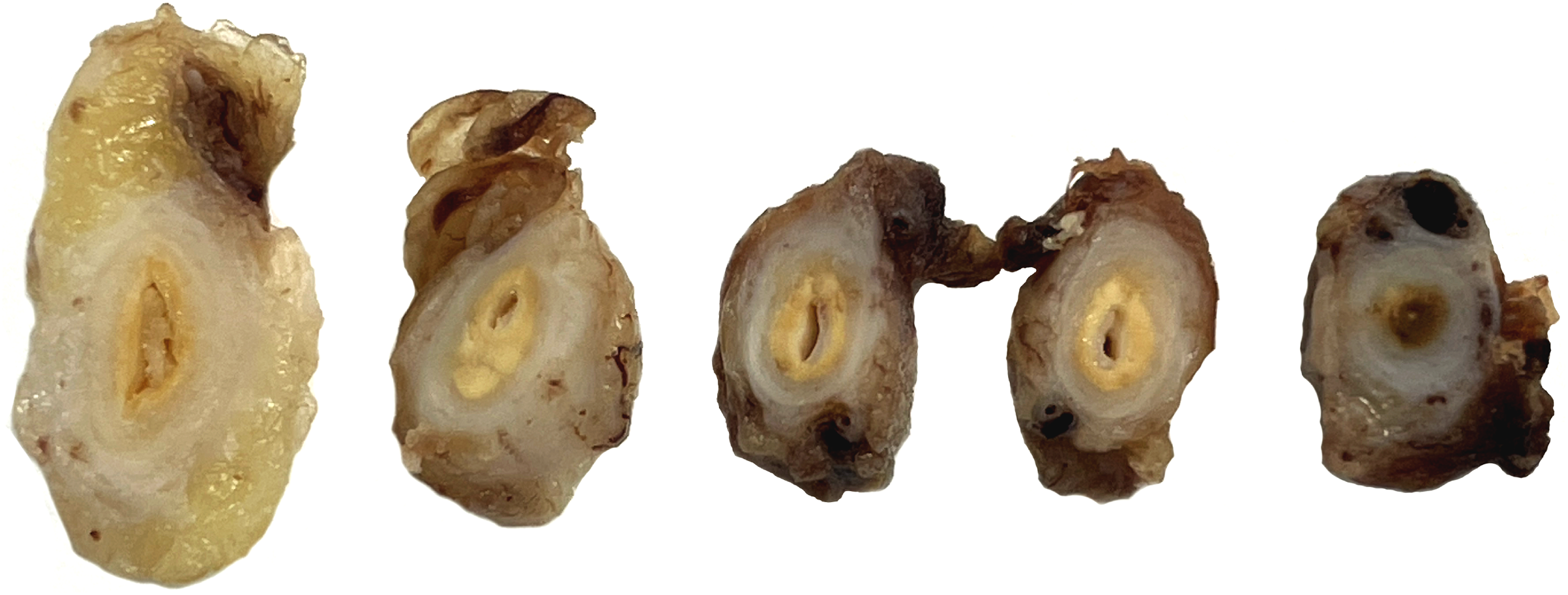
Macroscopic image of serial sections of the ureter showing thickening of its wall with a pasty material. Some areas of dilation and others of occlusion of the lumen are observed.
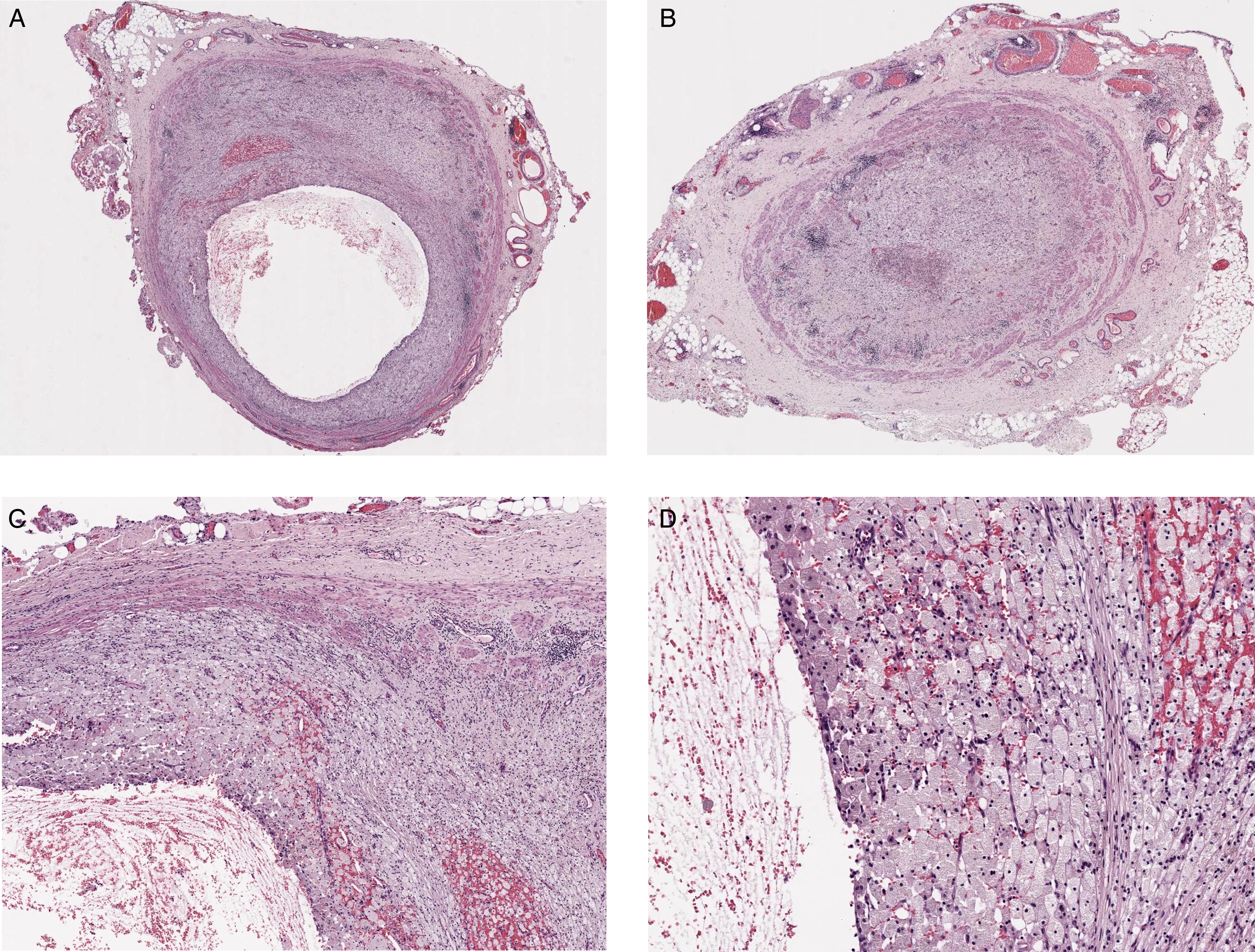
Microscopic images of xanthogranulomatous ureteritis. A, areas of dilated lumen with abundant foamy histiocytes in the ureteral wall (hematoxylin and eosin; 10x magnification). B, areas where the ureteral lumen was occluded due to the abundant presence of foamy histiocytes (hematoxylin and eosin; 10x magnification). C-D, Higher magnification images showing the presence of predominantly foamy histiocytes with accompanying lymphoplasmacytic inflammation (hematoxylin and eosin; C, 40x; D, 100x magnification).
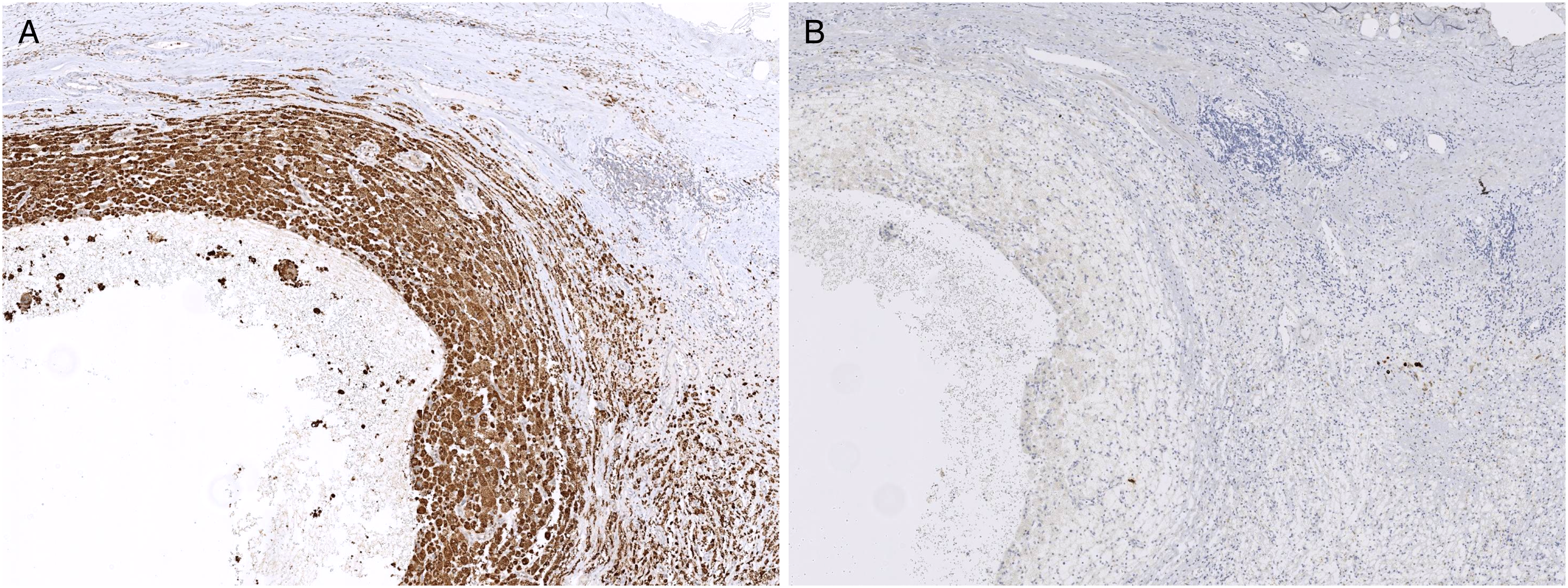
The immunohistochemical study confirming the histiocytic nature of the cells. A, CD68 positive (40x magnification). B, keratin AE1/AE3 negative (40x magnification).
Discussion
Xanthogranulomatous ureteritis is a very rare process in comparison with xantogranulomatous pyelonephritis. It is an atypical inflammatory response characterized by the presence of foamy histiocytes occupying the lumen and infiltrating the ureteral wall. 3 Few cases have been reported in the literature, and its etiology is not well defined (Table 1).3‐7 In one case, 5 the ureteral involvement was observed as an extension of xanthogranulomatous pyelonephritis, while in two other cases, the ureteral involvement was not contiguous.4,7
Reported Cases of Xanthogranulomatous Ureteritis.
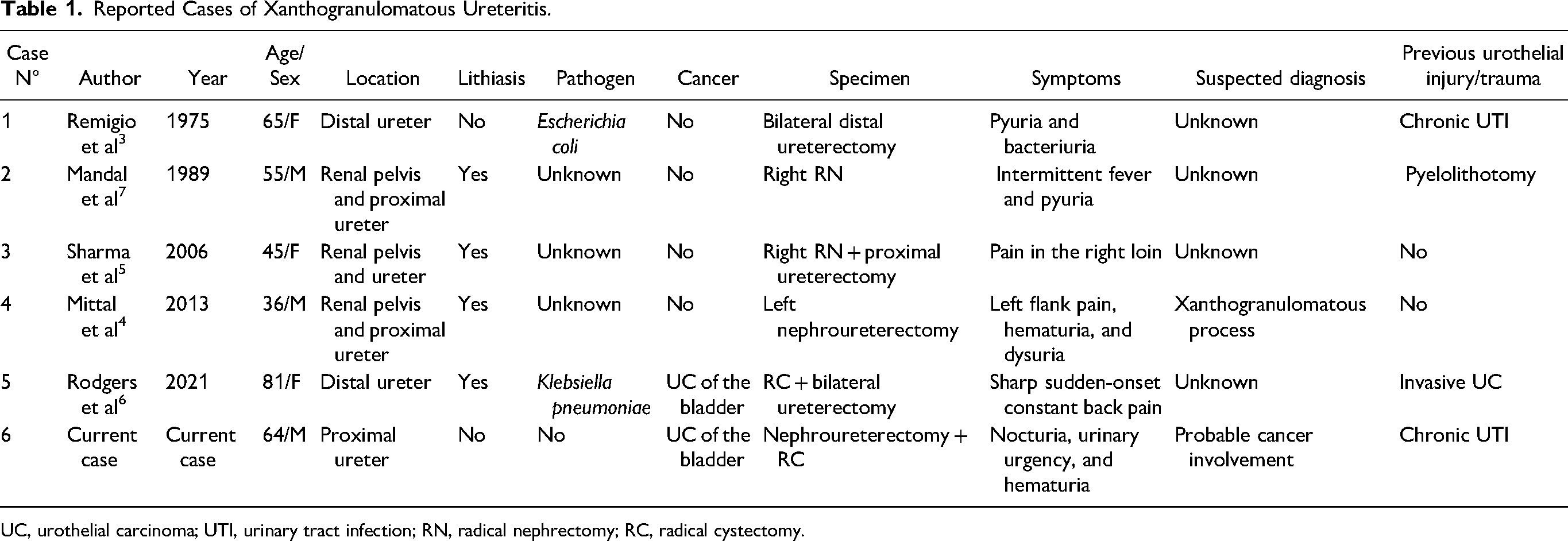
UC, urothelial carcinoma; UTI, urinary tract infection; RN, radical nephrectomy; RC, radical cystectomy.
In patients with cancer, especially bladder cancer, this process could be a clinical and radiological diagnostic challenge due to its mass effect on the ureter; this could lead to a differential diagnosis with tumor infiltration. Microscopically, it is characterized by the presence of abundant foamy histiocytes in a background of chronic active inflammation (lymphocytes, plasma cells, neutrophils, and eosinophils). Although the presence of foamy histiocytes is the main feature of other xanthogranulomatous processes, such as xanthogranulomatous cholecystitis or xanthogranulomatous pyelonephritis, it could be challenging when present in very uncommon locations like the ureter, especially in patients with bladder cancer. It can also be a diagnostic challenge as it could be confused microscopically with some carcinomas such as clear cell renal cell carcinoma.
Other differential diagnoses included non-Langerhans cell histiocytosis such malakoplakia, IgG4-mediated disease, ALK-mediated histiocytosis, and Rosai-Dorfman disease.8,9 However, the presence of a predominant foamy histiocytes infiltrate, that is CD68 positive with keratin AE1/AE3, IgG, IgG4, OCT2, CyclinD1, CD1a, and S100 negative and has no other histologic features that could cause us to consider these other diagnoses, could help us in the differential diagnosis and confirmation of xanthogranulomatous ureteritis.
In conclusion, xanthogranulomatous ureteritis is another benign process that is important to consider when observing a ureteral mass, especially in patients with bladder cancer. It is characterized by abundant foamy histiocytes in a background of chronic active inflammation. In some instances, it can be necessary to use immunohistochemistry (CD68 and keratins) for a diagnostic differential with infiltration by carcinoma.
Footnotes
Acknowledgements
L.M.N.C, A.V.C., A.V.M., contributed to the design and implementation of the research and to the analysis of the results. L.M.N.C wrote the manuscript. L.M.N.C, A.V.C., A.V.M., approved the final manuscript. We thank Lauren Tucker for her collaboration with English.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
