Abstract
Lymphoplasmacytic lymphoma often needs to be differentiated from other B-cell lymphomas with plasmacytic differentiation, especially marginal zone cell lymphoma. Molecular detection of MYD88 p.L265P hotspot mutation supports the diagnosis of lymphoplasmacytic lymphoma since it is seen in about 90% of such lymphoma, which is much higher than other B-cell lymphomas. MYD88 p.L265P is a gain-of-function mutation that enhances the activity of the NF-κB signaling pathway and therefore drives lymphomagenesis. Other mutations in MYD88 are rarely reported. This study aims to report an unusual MYD88 in-frame deletion in an aggressive lymphoplasmacytic neoplasm. This is an IgM-positive, CD5- and CD10-negative mature B-cell lymphoma with prominent plasmacytic differentiation and aggressive features. The clinical and pathologic findings were most consistent with lymphoplasmacytic lymphoma. Next-generation sequencing identified an unusual MYD88 in-frame deletion in the absence of the hotpot p.L265P mutation. Other concurrent pathogenic mutations also include truncating mutations of TRAF3, which is a negative regulator of the NF-κB signaling pathway, and a missense mutation of TP53. Karyotype analysis showed complex karyotypes, including chromosome 6q deletion. By searching literature and online cancer databases, we identified only 8 other mature B-cell lymphomas with MYD88 in-frame deletions, but none of them was diagnosed with lymphoplasmacytic lymphoma. Recognizing such in-frame deletions is necessary to help understand the mutational spectrum of MYD88 in B-cell lymphomas. It remains to be further investigated whether such MYD88 in-frame deletions are also overrepresented in lymphoplasmacytic lymphoma among other B-cell lymphomas.
Introduction
Lymphoplasmacytic lymphoma is a small B-cell neoplasm genetically characterized by a myeloid differentiating factor 88 (MYD88) mutations. The hotspot mutation MYD88 p.L265P is seen in over 90% of lymphoplasmacytic lymphoma patients, although this mutation can be seen in other B-cell lymphomas with lower frequencies. 1 MYD88 is an adaptor protein of Toll-like receptors and transduce signaling of the NF-κB pathway. Constitutive activation of this pro-inflammatory pathway promotes cell survival and proliferation in lymphoma. MYD88 encodes 296 amino acids which can be grouped into an N-terminal death domain (DD), a short connecting linker, and a highly conserved C-terminal Toll/Interleukin-1 receptor (TIR) domain. 2 Upon Toll-like receptor activation, MYD88 is recruited to Toll-like receptor on cell membrane through TIR–TIR domain interactions with other adaptor proteins. MYD88 can also oligomerize through its TIR domain to form a higher-order complex leading to clustering of its DD domains. 3 The clustered DD domains result in the recruitment of downstream Interleukin-1 receptor-associated kinases and the propagation of the NF-κB signaling pathway.
The oncogenic mutation MYD88 p.L265P is located in the TIR domain, which can cause augmented oligomerization of MYD88, thus enhancing activation of the NF-κB signaling. 4 MYD88 in-frame deletions in the TIR domain are very rare and, to the best of our knowledge, have only been reported in 8 other B-cell lymphomas by searching literature and online cancer databases. Here, we report an aggressive lymphoplasmacytic neoplasm with an unusual in-frame MYD88 deletion in the TIR domain in the absence of the hotspot p.L265P mutation. The findings of this report can help to expand the mutational spectrum of MYD88 in B-cell lymphomas
Clinical and Pathologic Presentation
A 54-year-old man with no significant medical history presented with B symptoms, diffuse abdominopelvic and axillary lymphadenopathy, hepatosplenomegaly, hyperviscosity, elevated uric acid (12.6 mg/dL), and acute renal failure. Laboratory tests showed anemia (hemoglobin: 8.1 g/dL), leukocytosis (14.5 × 109/L), IgM paraprotein (7720 mg/dL), and free lambda light chain (655 mg/dL). A blood smear revealed rouleaux formation, granulocytic left shift, and atypical lymphoid cells. Excisional biopsy of the axillary lymph node and bone marrow biopsy was performed for further diagnosis.
Excisional biopsy of an axillary lymph node revealed an enlarged lymph node with total architectural effacement by a nodular proliferation of lymphoid cells (Figure 1A). Cells at the periphery of the nodules showed small lymphocytes and prominent plasmacytic differentiation with conspicuous Dutcher bodies (Figure 1B). In comparison, cells within the nodules were small-to-medium sized with relatively regular nuclear contours and punctate small nucleoli. Monocytoid cells were not seen. In addition, scattered large cells with more prominent nucleoli were present within the nodules, although sheets of large cells were absent (Figure 1C). Frequent mitotic figures and apoptotic bodies were also seen. CD138 immunostaining demonstrated the prominent plasmacytic differentiation at the periphery of the nodules (Figure 1D). The lymphoma cells were highlighted by CD20 (Figure 1E), while CD3 only stained background T-lymphocytes. The lymphoma cells were also positive for BCL2 but were negative for CD5, CD10, CyclinD1, CD30, HHV8, and EBER. Only very few follicular dendritic cell meshworks were highlighted by CD21 (Figure 1F) and CD23. The lymphoma cells expressed IgM and restricted lambda light chain. Ki-67 proliferation index was 40–50% (Figure 1G) with MYC expression in roughly 40% of the lymphoma cells (Figure 1H). On bone marrow biopsy, prominent paratrabecular lymphoid aggregates were present in the background of hypercellular bone marrow (Figure 2A). Similar to the lymph node, a heterogeneous population of small lymphocytes, plasmacytoid lymphocytes, plasma cells, and scattered large cells were appreciated in the bone marrow (Figure 2B). Staining revealed similar immunophenotypes as seen in the lymph node, including numerous CD20- and CD138-positive cells with scattered large forms (Figure 2C and D). Flow cytometry was performed on the bone marrow sample and demonstrated a CD5- and CD10-negative clonal B cell population with lambda light chain restriction. No abnormal T-cell population was identified.
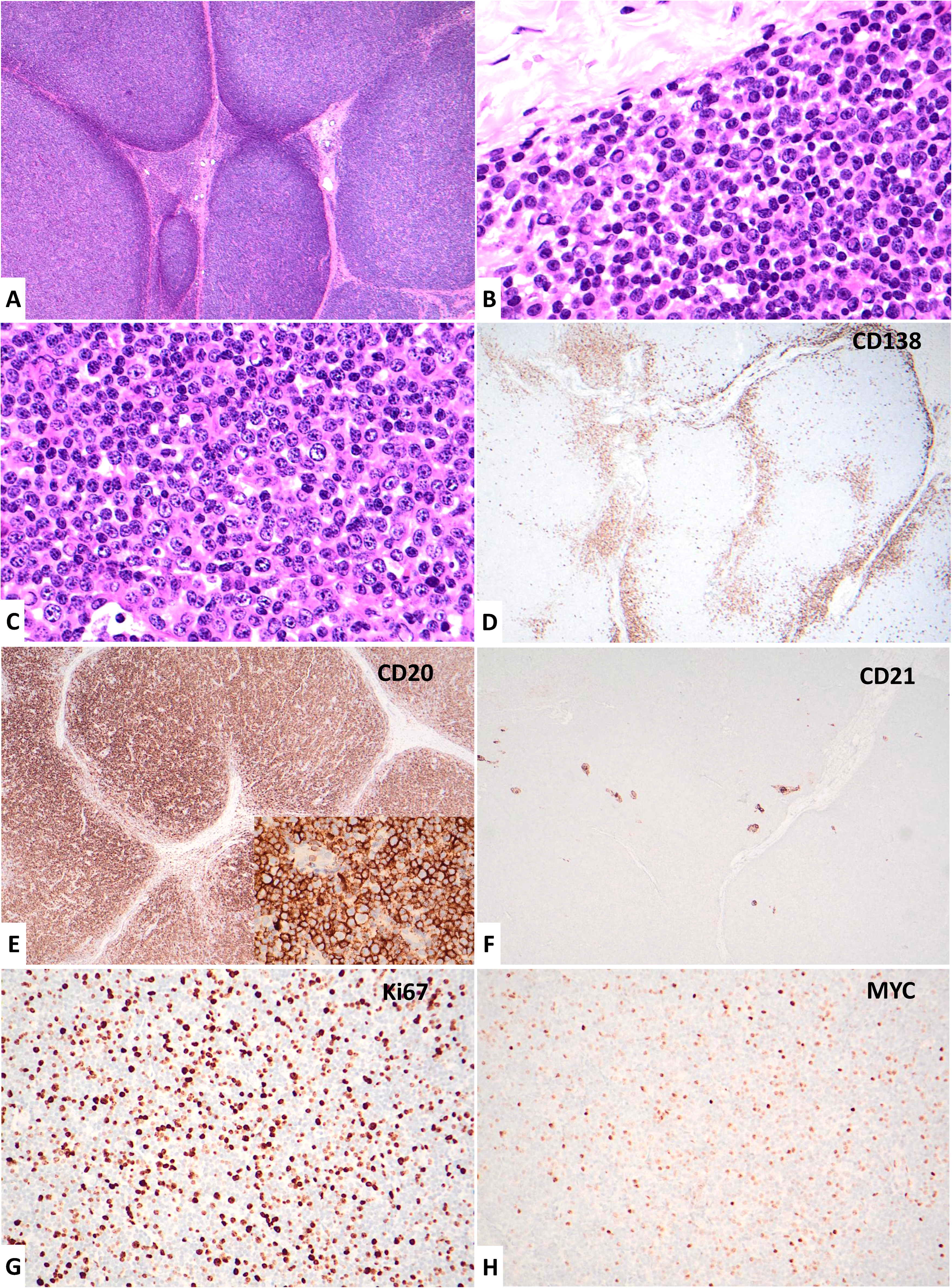
Pathologic presentations of the lymphoma involving an axillary lymph node. (A) Total replacement by nodular lymphoid infiltrate, 4x. (B) Plasmacytic differentiation with abundant Dutcher bodies at the periphery of lymphoid nodules, 60x. (C) Small-to-intermediate lymphoma cells within the nodules, with scattered large cells and mitotic figures, 60x. (D) CD138 immunohistochemistry highlights plasma cells at peripheries of lymphoid nodules, 4x. (E) CD20 immunohistochemistry highlights numerous B-lymphocytes within the lymphoid nodules, 4x. High power image shows large lymphoma cells highlighted by CD20 (inset), 60x. (F) CD21 immunohistochemistry shows disruption of lymph node architecture with minimally preserved follicular dendritic cell meshwork, 4x. (G) Ki67 immunohistochemistry shows a high proliferation index (40–50%), 20x. (H) MYC immunohistochemistry shows relatively high expression (about 40%) by lymphoma cells, 20x.
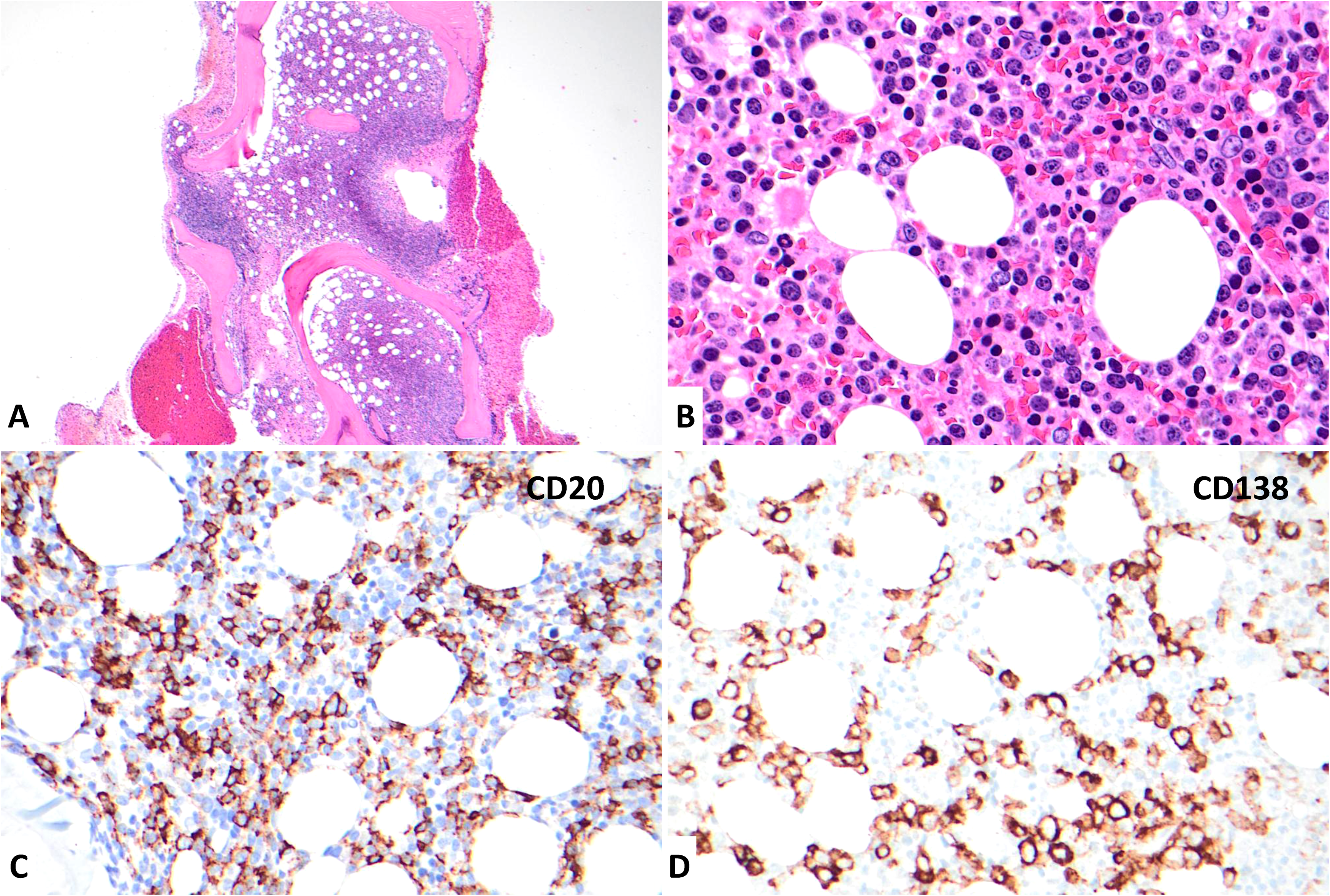
Pathologic presentations of the lymphoma involving bone marrow. (A) Hypercellular bone marrow with paratrabecular lymphoid aggregates, 2x. (B) Lymphoid aggregates reveal plasma cells and plasmacytoid lymphocytes with scattered large forms, 60x. (C) CD20 immunohistochemistry highlights many neoplastic B lymphocytes including some large cells, 40x. (D) CD138 immunohistochemistry highlights numerous plasmacytic cells, 40x.
Next-generation sequencing (NGS) of a targeted 30-gene lymphoid panel performed on the axillary lymph node identified the following pathogenic variants and corresponding variant allele frequencies (VAF):
MYD88 p.C287_C293delCDYTNPC (NM_002468 c.859_879del21) VAF: 18.1% (Figure 3) TRAF3 p.E313Pfs*9 (NM_003300 c.937_940delGAAT) VAF: 34.8% TP53 p.R248 W (NM_000546 c.742C > T) VAF: 25%
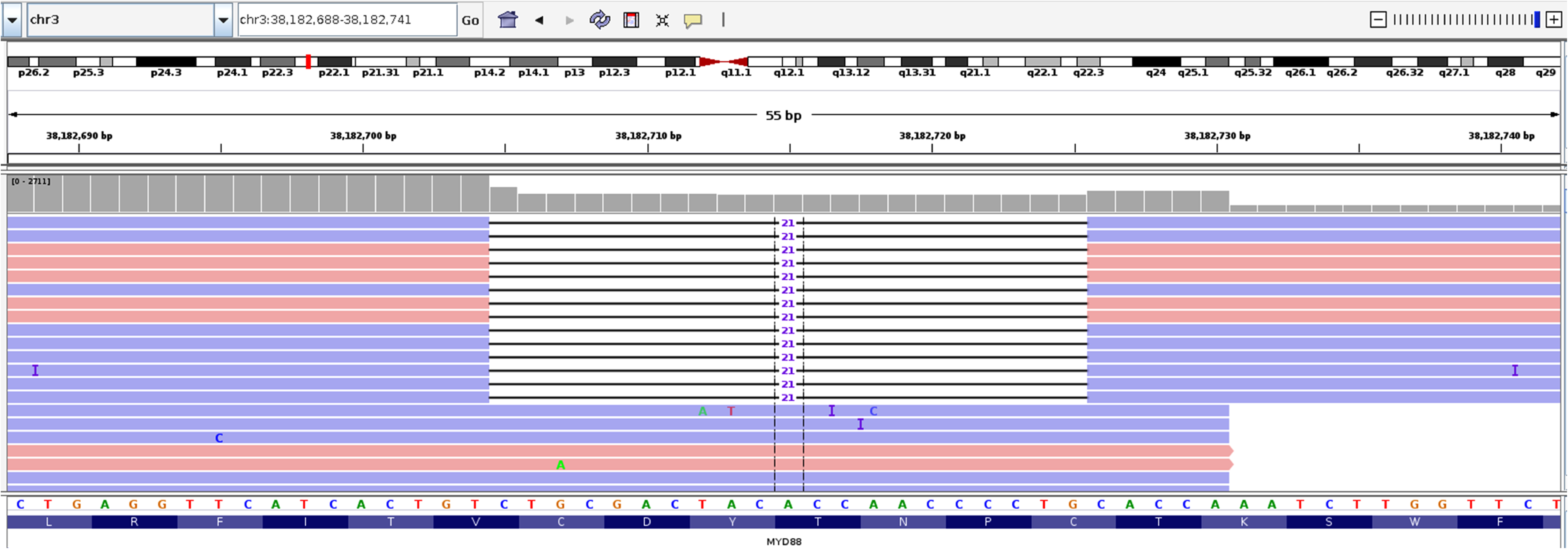
In-frame deletion of MYD88. MYD88 p.C287_C293delCDYTNPC (NM_002468 c.859_879del21) shown in Integrative Genomics Viewer (IGV). 5
Chromosomal analysis revealed a complex karyotype: 47,XY,del(1)(q42),+3,del(4)(q25),dup(5)(q12q31),del(6)(q21),del(13)(q12q22),del(14)(q31)[5]/46,XY[15]
The read depth at MYD88 p.L265 was 2711 for this sample, and there were 5 alternative reads for T > C which caused MYD88 L265P. The low VAF (0.1844%) for this variant was very similar to other cases on the same run and was interpreted as background noise/sequencing error. No other MYD88 variants were detected by our pipeline.
The disease was difficult to classify; however, a diagnosis of lymphoplasmacytic lymphoma was supported by the immunophenotype, prominent plasmacytic differentiation, IgM paraprotein, extensive bone marrow involvement with paratrabecular distribution, and chromosome 6q deletion. The presence of bone marrow involvement and IgM paraprotein also satisfied the criteria for diagnosing Waldenstrom macroglobulinemia. Both clinical and pathologic findings, including acute kidney injury, elevated uric acid level, B symptoms, presence of scattered large cells, increased Ki67 fraction, TP53 mutation, and complex karyotype, suggested aggressive features. Thus, a transforming lymphoplasmacytic lymphoma should be considered. It is noteworthy that unusual features for lymphoplasmacytic lymphoma were also present in this lymphoma, including diffuse lymphadenopathy with total effacement of lymph node architecture and peripheral blood involvement, which may possibly be explained by its aggressive behavior.
The patient was treated with zanubrutinib, CyBord (bortezomib, cyclophosphamide, and dexamethasone) and plasma exchange (6 procedures in total). After a follow-up of 6 months, the patient still showed persistent disease with elevated IgM paraprotein (810 mg/dL) and free lambda light chain (332 mg/dL).
Discussion
By reviewing literature and online cancer databases, we were only able to identify 8 additional MYD88 in-frame deletions reported in B-cell lymphomas (Table 1). Based on a limited number of cases, extranodal marginal zone lymphoma appears to be the most common type of B-cell lymphoma harboring this unusual type of mutation (4 out of 9). Importantly, all these mutations, including the one reported here, were located within the C-terminal TIR domain, which is the same genomic region harboring the hotspot p.L265P missense mutation. Although each mutation is not exactly the same, they all have a common sequence of amino acids being deleted, which is p.D288_C293delDYTNPC. The mutational spectrum spans 3 amino acids both upstream and downstream of this commonly deleted sequence.
Summary of Reported MYD88 In-Frame Deletions in b-cell lymphomas.

Abbreviations: LPL, lymphoplasmacytic lymphoma; EMZL, extranodal marginal zone lymphoma; DLBCL, diffuse large B-cell lymphoma; CLL/SLL, chronic lymphocytic leukemia/small lymphocytic lymphoma; PCNSL, primary central nervous system lymphoma; N/A, Not available; VAF, variant allele frequency.
Structural characterization of the MYD88 TIR domain identified 3 highly conserved motifs, namely boxes 1, 2, and 3. Box 3 is near the C-terminal end which is crucial in regulating Toll-like receptor signaling by participating in MYD88 oligomerization. 9 The in-frame deletion reported here is in close proximity to box 3 (2 codons upstream) and may thus dysregulate the Toll-like receptor signaling by interfering in the function of the box 3 region. The close similarities of these MYD88 in-frame deletions further support their common function in driving lymphomagenesis. Two previously reported lymphomas were from the same patient, who initially had chronic lymphocytic leukemia/small lymphocytic lymphoma and later developed diffuse large B-cell lymphoma. 8 For this patient, the VAF was much higher in the transformed diffuse large B-cell lymphoma (19%) than in the chronic lymphocytic leukemia/small lymphocytic lymphoma (2%), suggesting a possible gain-of-function of the MYD88 in-frame deletion. However, due to the rarity of this type of mutation, there is currently no annotation of its biological function in cancer database. 8 Further study is needed to understand if such in-frame deletions have similar gain-of-function effect in promoting Toll-like receptor signaling as the hotspot p.L265P missense mutation.
In our patient, a truncating mutation of TRAF3 was also identified with a VAF nearly double that of the VAF for the MYD88 in-frame deletion. TRAF3 is a negative regulator of noncanonical NF-κB signaling pathway, and its biallelic inactivation is seen in about 5% of patients with Waldenstrom macroglobulinemia. 10 TRAF3 is located at chromosome 14q32.32, which is close to a deleted region in the karyotype analysis (del(14)(q31)). Therefore, it is possible that bi-allelic inactivation of TRAF3 in this lymphoma was accomplished by loss of heterozygosity. Alternatively, the MYD88 mutation may have occurred as a subclone of the lymphoma with TRAF3 inactivating mutation. Together, TRAF3 inactivation and the MYD88 in-frame deletion with possible gain-of-function could work together to drive lymphomagenesis by activating the NF-κB signaling pathway via 2 distinct mechanisms. In addition, TP53 alterations are observed in 5% to 11% of Waldenstrom macroglobulinemia patients, often coexist with MYD88 mutations, and pose a worse prognosis for patients.11,12 The aggressive disease reported here could possibly be contributed by the concurrent TP53 mutation.
In summary, we report an unusual in-frame deletion of MYD88 for the first time in an aggressive lymphoplasmacytic neoplasm. Due to clinical and histopathologic features, lymphoplasmacytic lymphoma is favored to be the most likely diagnosis. The observed deletion is within the MYD88 TIR domain and is downstream of the hotspot p.L265P mutation. It is important to recognize the full mutational spectrum of MYD88 beyond the hotspot mutation. MYD88 in-frame deletions in the TIR domain may represent a different mechanism to activate NF-κB signaling. It requires further study to understand whether such MYD88 in-frame deletions are also overrepresented in lymphoplasmacytic lymphoma among other B-cell lymphomas.
Footnotes
Author Contributions
Jennifer Yoest designed the study, collected the data, and revised the manuscript. Howard Meyerson and Kwadwo Oduro collected data and revised the manuscript. Shashirekha Shetty collected the data. Mingfei Yan collected the data and wrote the manuscript.
Ethnic Approval
This study is approved by University Hospitals Cleveland Medical Center IRB STUDY20191252.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
