Abstract
Background
Malignant ovarian germ cell tumors represent small percentage of malignant ovarian neoplasms but they affect significantly young age group.
Aim of the study
To investigate the immunohistochemical expression of p16 tumor suppressor protein in malignant ovarian germ cell tumors.
Materials and Methods
Twenty-two malignant ovarian germ cell tumors (five dysgerminoma, eight immature teratoma, and nine yolk sac tumors), twenty mature cystic teratoma tumors and twenty normal ovarian tissue were immunohistochemically stained with p16 monoclonal antibody. Ki67 immunohistochemical staining was done for malignant ovarian germ cell tumors to assess proliferation.
Results
We found that p16 tumor suppressor protein is overexpressed in all malignant ovarian germ cell tumors in both nuclear and cytoplasmic locations compared to control and to mature cystic teratoma (p-value <0.001). Cytoplasmic p16 expression was significantly correlated to Ki67 proliferation index in malignant ovarian germ cell tumors (p-value = 0.033, r = 0.445).
Conclusion
Overexpression of p16 in malignant ovarian germ cell tumors denotes that dysfunction of the cyclin dependent kinase pathway is involved in tumorigenesis of malignant ovarian germ cell tumors.
Keywords
Introduction
Malignant ovarian germ cell tumors comprise about 5% of all malignant ovarian neoplasms. 1 They typically present in adolescence. 2 Although germ cell tumors of the ovary are rare and accounting for only 1% of ovarian malignancies, most pediatric tumors of the ovary are of germ cell origin. 3 The most frequently presenting symptoms are abdominal pain and abdominal distension by pelvic or abdominal mass. 4 The treatment is either surgery alone or combination of surgery, adjuvant chemotherapy and radiotherapy in advanced tumors.5,6 Malignant ovarian germ cell tumors are believed to be derived from pluripotent primordial germ cell. 5 According to World Health Organization (WHO) classification of ovarian tumors, malignant ovarian germ cell tumors include immature teratomas, dysgerminomas, yolk sac tumors, embryonal carcinomas, choriocarcinomas, malignant struma ovarii and teratomas with malignant transformation. 7 Owing to rarity of these tumors, targeted therapeutic options for chemotherapy-resistant patients are not well investigated.
Dysregulation of cyclin dependent kinase (CDK) pathway is involved in pathogenesis of many tumors and kinase inhibitors are recently US FDA approved as targeted therapy for many neoplasms. 8 P16 protein is one of the major members of cyclin dependent kinase inhibitors from (INK4) family. 9 It is also named inhibitor of cyclin kinase 4a (INK4a or P16INK4a), major tumor suppressor-1 (MTS-1) and cyclin-dependent kinases inhibitor 2A (CDKN2A). 10 P16 protein is encoded by CDKN2A on chromosome 9 (9p21.3) within INK4/ARF locus. 11 Its function is arresting the cell cycle in the G1-to-S phase transition via inhibition of cyclin D1-CDK4/6 complex formation and CDK4/6-mediated phosphorylation of the retinoblastoma (Rb) protein. Phosphorylation of Rb leads to dissociation of E2F-RB complex and progression of the cell cycle. Thus, p16 maintains Rb family members in a hypophosphorylated state to prevent proliferation, so it is known as a tumor suppressor protein. 12 P16 protein is overexpressed with aging and in response to injurious agents as ionizing radiation or DNA damaging agents resulting in cellular senescence. 13 Abnormal expression of tumor suppressor proteins leads to unrestricted proliferation of cells and generation of a neoplasm.14,15 The expression of p16 protein is deranged in many tumors either by inactivation or overexpression. 11 Overexpression of p16 is notably present in human papilloma virus (HPV)-related endocervical neoplasms,16,17 HPV-related head and neck carcinoma, and high-grade squamous intraepithelial lesions of the vulvovaginal and anogenital region.9,17 However, it is also expressed in some HPV-independent tumors as high-grade serous carcinomas of the ovary, 18 colorectal carcinomas, 19 and breast cancer. 20 Inactivation of p16 protein occurs in many tumors by different genetic and epigenetic mechanisms.21–23 Loss of p16 expression was reported to be associated with poor prognosis in nasopharyngeal carcinomas 22 and in melanomas. 24 Although p16 downregulation leads to cancer progression, its overexpression is also associated with poor prognosis in different tumors. 25
Materials and Methods
The study protocol was approved by the Institutional Review Board at Faculty of Medicine, Assiut University (IRB No.17100871), and Ethical approval for this study was obtained from the committee of medical ethics, Faculty of Medicine, Assiut University. Formalin-fixed, paraffin-embedded tissue blocks from 62 ovarian specimens were collected from pathology archives. These specimens include group A: 22 malignant ovarian germ cell tumors representing all malignant ovarian germ cell tumors from January 2010 to January 2021; of them, five were dysgerminomas, eight immature teratomas, and nine yolk sac tumors, group B include 20 mature cystic teratomas as benign group and group C include 20 apparent normal ovarian tissue specimens obtained from specimens of total abdominal hysterectomy and salpingo-oophorectomy for non-ovarian, non-malignant causes such as multiple fibroid uterus and adenomyosis as a control group. The initial diagnosis of each malignant ovarian germ cell tumor was re-evaluated according to the WHO classification of ovarian tumors, 2020.
All available hematoxylin and eosin stained slides were examined by two independent pathologists by routine light microscopy and the most representative one or two tissue blocks were selected for immunohistochemical staining.
Immunohistochemistry
Sections from cervical non-keratinized squamous cell carcinoma with block p16 expression were used as positive controls. Sections from tonsil are used as positive control for Ki67. Negative controls were obtained by omitting the primary antibody.
Immunohistochemical evaluation
For p16 IHC staining, the percentage of p16 positive cells and the location of positive signals (nuclear or cytoplasmic) were visually estimated for neoplastic components of all lesions. German semi-quantitative scoring system was used to evaluate p16 expression, every section was given a score according to the staining intensity in both cytoplasmic and nuclear components (0 = no staining, 1 = weak, 2 = moderate, 3 = strong) and the extent of stained cells (0 = 0%, 1 = 1%–10%, 2 = 11%–50%, 3 = 51%–80%, 4 = 81%–100%). The final score was determined by multiplying the intensity scores with the extent of positivity scores of stained cells, with the minimum score of 0 and a maximum score of 12. 26
For Ki67 IHC staining for malignant ovarian germ cell tumors, the labeling index for Ki67 antigen was estimated by average score method in which at least three independent areas that had prevalent Ki67 staining per section were selected. All slides were visually estimated, and proportions of positive cells were scored at 10% intervals. 27
Statistical analysis
Data were analyzed using IBM-SPSS version 24. Numerical data were presented as mean and standard deviation, while categorical data were presented as number and percentage; independent sample Kruskal–Wallis was used to compare between groups. Correlation analysis was used (Spearman ranked correlation). A p-value < 0.05 was considered significant.
Results
The study investigated 22 malignant ovarian germ cell tumors, 20 mature cystic teratoma & 20 specimens of normal ovaries as a control group. The mean age for malignant ovarian germ cell tumors was 16.5 years old (range 12−23 years) and for mature cystic teratomas was 31.5 years old (range 14−50 years). Malignant ovarian germ cell tumors affect significantly younger age group than mature cystic teratomas (p value 0.001).
Group A is composed of 22 malignant ovarian germ cell tumors: five dysgerminomas (22.7%), eight immature teratomas (36.4%) (Four of them were grade 2 and four were grade 3), and nine yolk sac tumors (40.9%). According FIGO staging system for ovarian cancer, 28 more than half of malignant ovarian germ cell tumors in our study were presented at stage I (54.5%), six tumors were presented at stage II (27.3%) and four tumors at stage III (18.2%). (Table 1)
Distribution of the Studied Malignant Group According to FIGO Staging.

Chi-square test.
*p-value < 0.05 was considered significant.
Immunohistochemical Expression of p16
In control group (Figure 1), p16 was not expressed in epithelial component and rarely expressed in few stromal cells (mean IHC cytoplasmic score is 0.41 ± 1.06 and mean IHC nuclear score is 0.18 ± 0.73). In benign mature cystic teratomas (Figure 1), strong nuclear p16 expression was seen in basal layer of stratified squamous epithelium, while weak cytoplasmic staining was noted in rest of stratified squamous epithelium layers, sebaceous glands, and hair follicles. Also, moderate cytoplasmic staining was noted in pyramidal cells in mature brain tissue found in mature cystic teratomas (mean IHC cytoplasm score is 3.44 ± 1.54 and mean IHC nuclear is 2.72 ± 1.57).
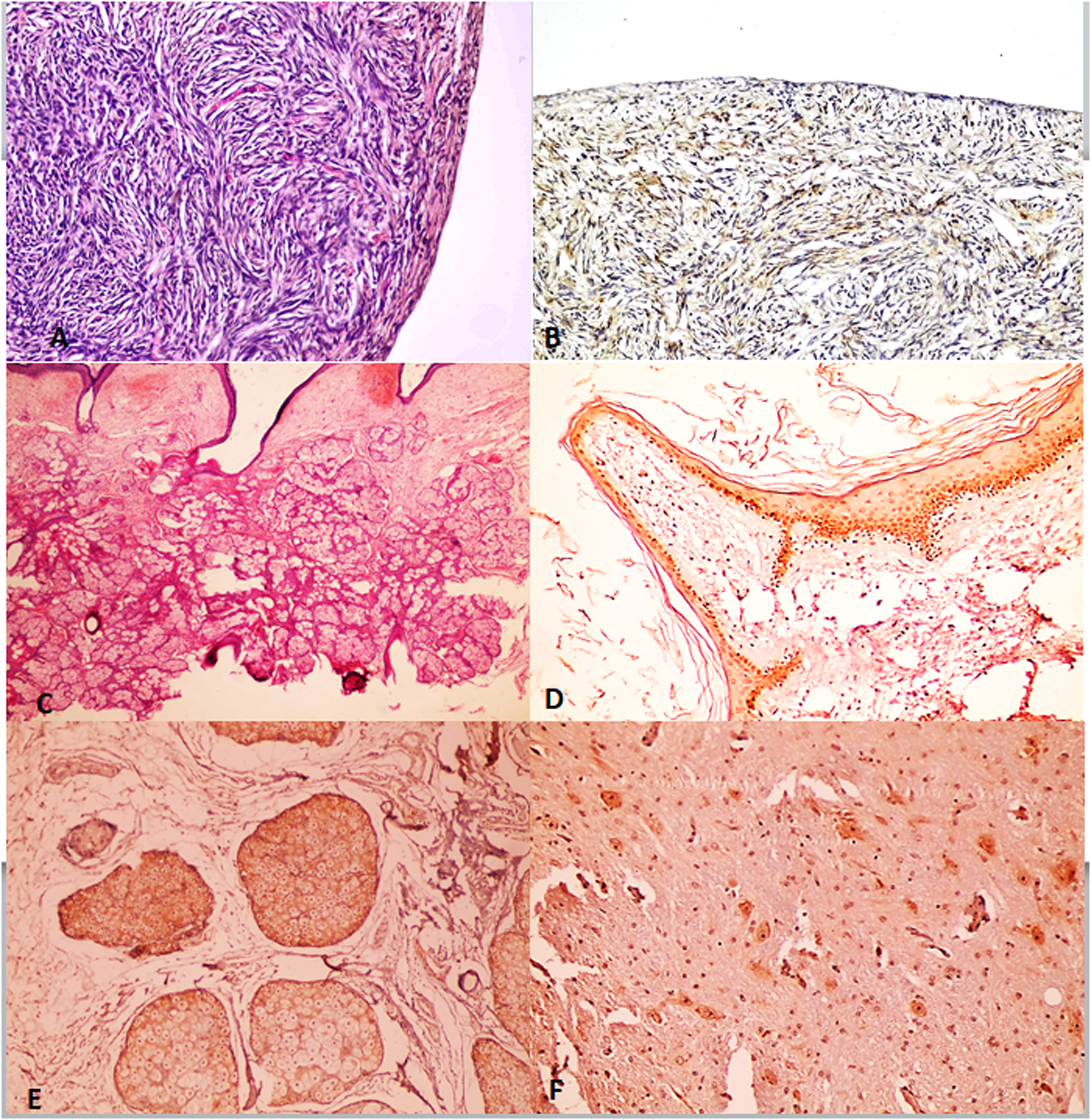
A: Normal ovary, H & E stain, x200. B: Normal ovary, p16 IHC stain, no immunoreactivity in epithelial cells and rare stromal cells reactivity, x200. C: MCT, H&E section, x200. D: MCT, P16 IHC at x200 power that showed moderate nuclear staining in basal layer of stratified squamous epithelium. E: MCT, P16 IHC stain showed weak cytoplasmic intensity limited to sebaceous glands and sweat glands, x200
In malignant ovarian germ cell tumors (Figures 2 to 4), significant strong p16 cytoplasmic and nuclear staining was present (mean IHC cytoplasmic score is 7.23 ± 0.92 and mean IHC nuclear score is 6.14 ± 1.32) compared to control group (p value < 0.001) and also to benign mature cystic teratomas (p value < 0.001) (Table 2). There was no statistically significant difference in either cytoplasmic or nuclear p16 expression in different subtypes of malignant ovarian germ cell tumors under study (p value = 0.641 and 0.207, respectively) (Table 3). Also, there was no statistically significant difference between different grades of immature teratomas (p value = 1.00 and 0.533, respectively) or different FIGO stages of malignant ovarian germ cell tumors (p value = 0.262 and 0.540, respectively).
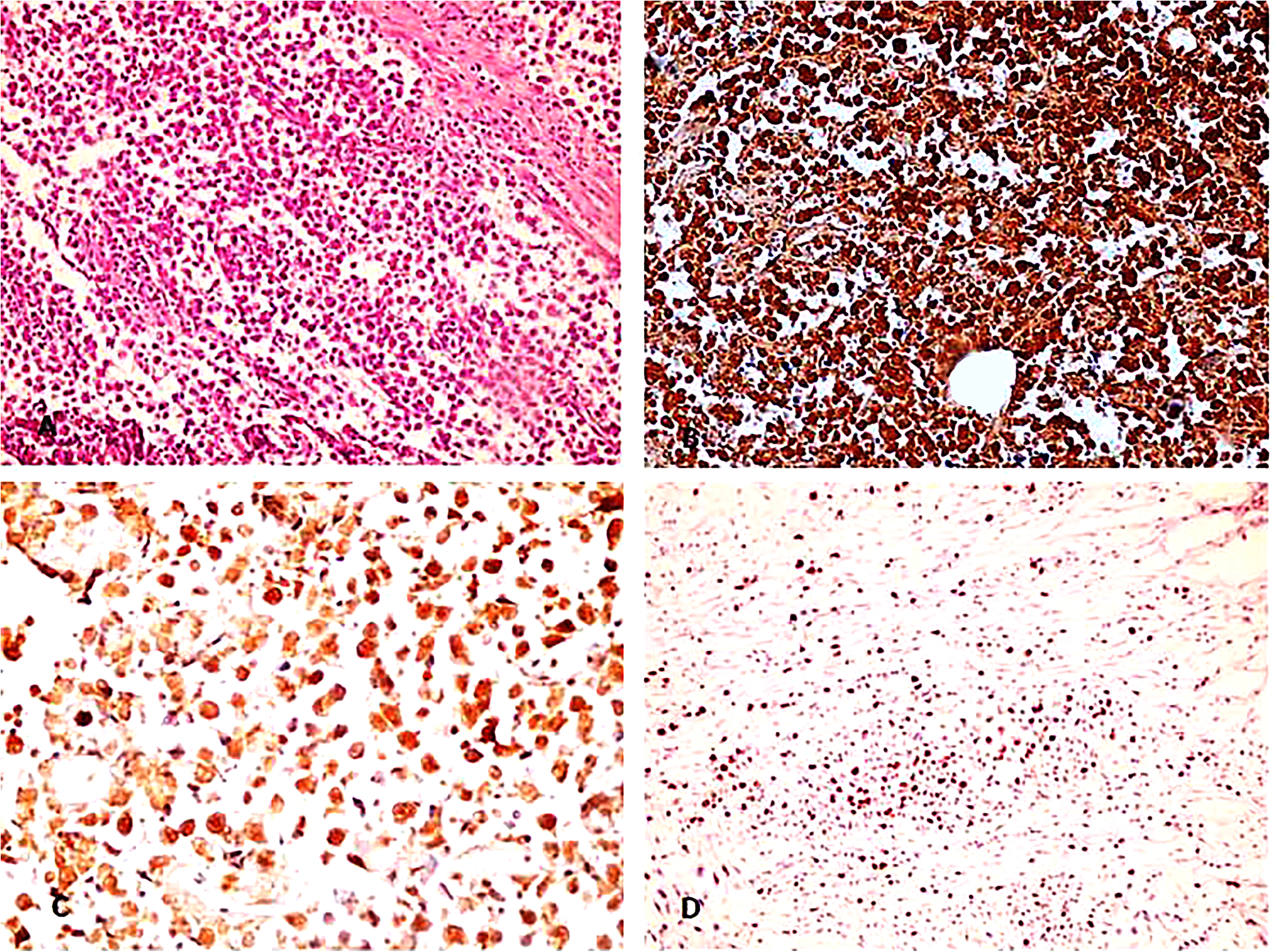
Dysgerminoma, A: H&E section, x200. B: p16 IHC stain at x200 power. C: p16 IHC stain, x400 power. Both B and C showed strong diffuse positivity in both nuclear & cytoplasmic components. D: Ki67 IHC stain, x200.
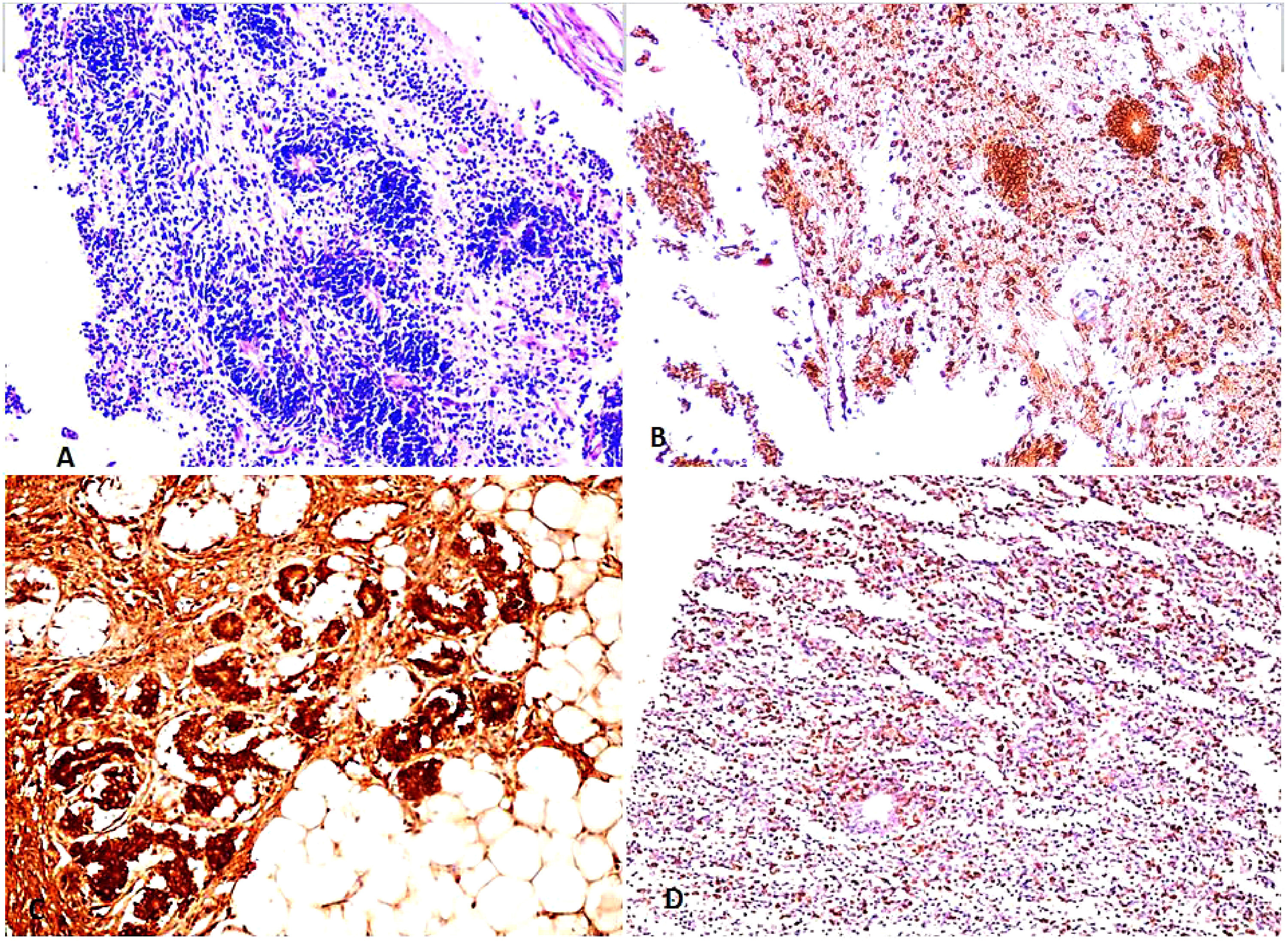
Immature teratoma. A: H&E section, x200. B: p16 IHC stain, x200. C: P16 IHC at x400 power. Both B and C showed strong diffuse positivity in both nuclear & cytoplasmic components. D: Ki67 IHC, x200.
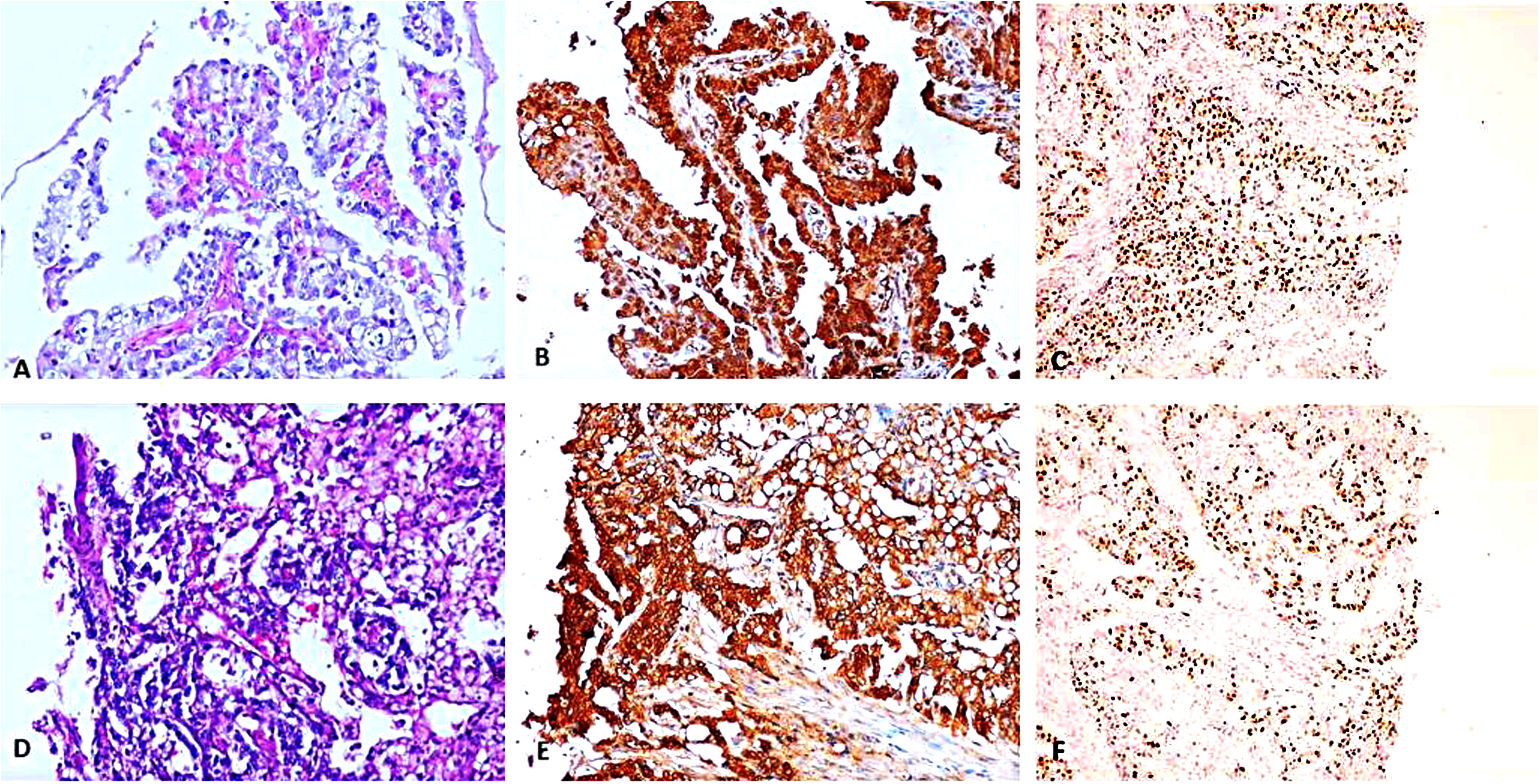
Yolk sac tumor. A: H&E section showing papillary pattern, x200. B: p16 IHC stain, showed strong diffuse positivity in both nuclear & cytoplasmic components, x200. C: Ki67 IHC stain, x200. D: H&E section showing microcystic pattern, x200. E: p16 IHC stain, showed strong diffuse positivity in both nuclear & cytoplasmic components, x200. C: Ki67 IHC stain, x200.
p16 IHC Score Among the Studied Groups.
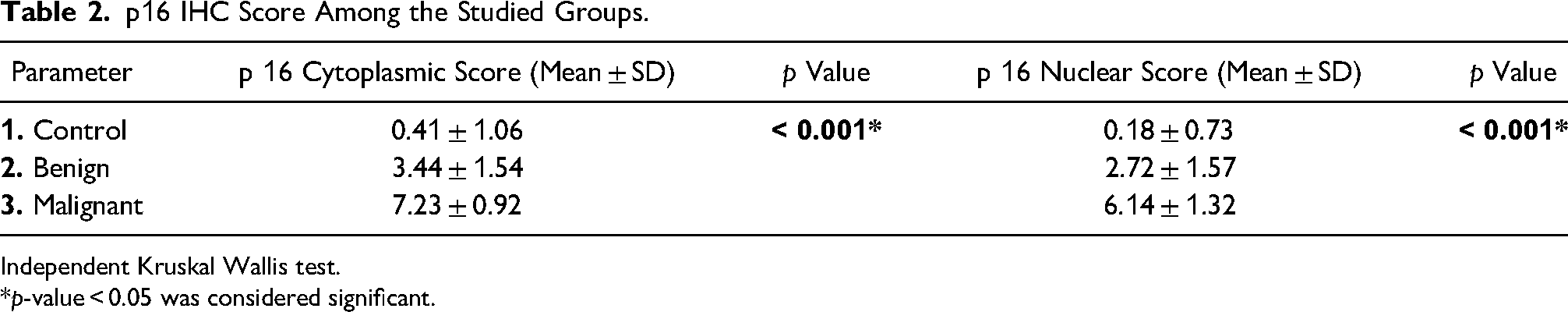
Independent Kruskal Wallis test.
*p-value < 0.05 was considered significant.
p16 IHC Expression in Different Types of MOGCTs.

Independent Kruskal Wallis test.
Data were expressed as mean ± SD.
Ki 67 Expression in MOGCTs
By analyzing Ki67 labeling index (LI) expression in studied malignant ovarian germ cell tumors, we found that the highest Ki67 LI was present in yolk sac tumors but with no statistically significant difference (p value = 0.216) (Table 4). There was no statistically significant difference between Ki67 LI in different grades of immature teratomas (p value = 0.702) or with different FIGO stages (p value = 0.474). The Ki67 LI showed statistical significant correlation with p16 cytoplasmic expression (p value = 0.033, r = 0.445) (Figure 5), but not with p16 nuclear score (p value = 0.926).
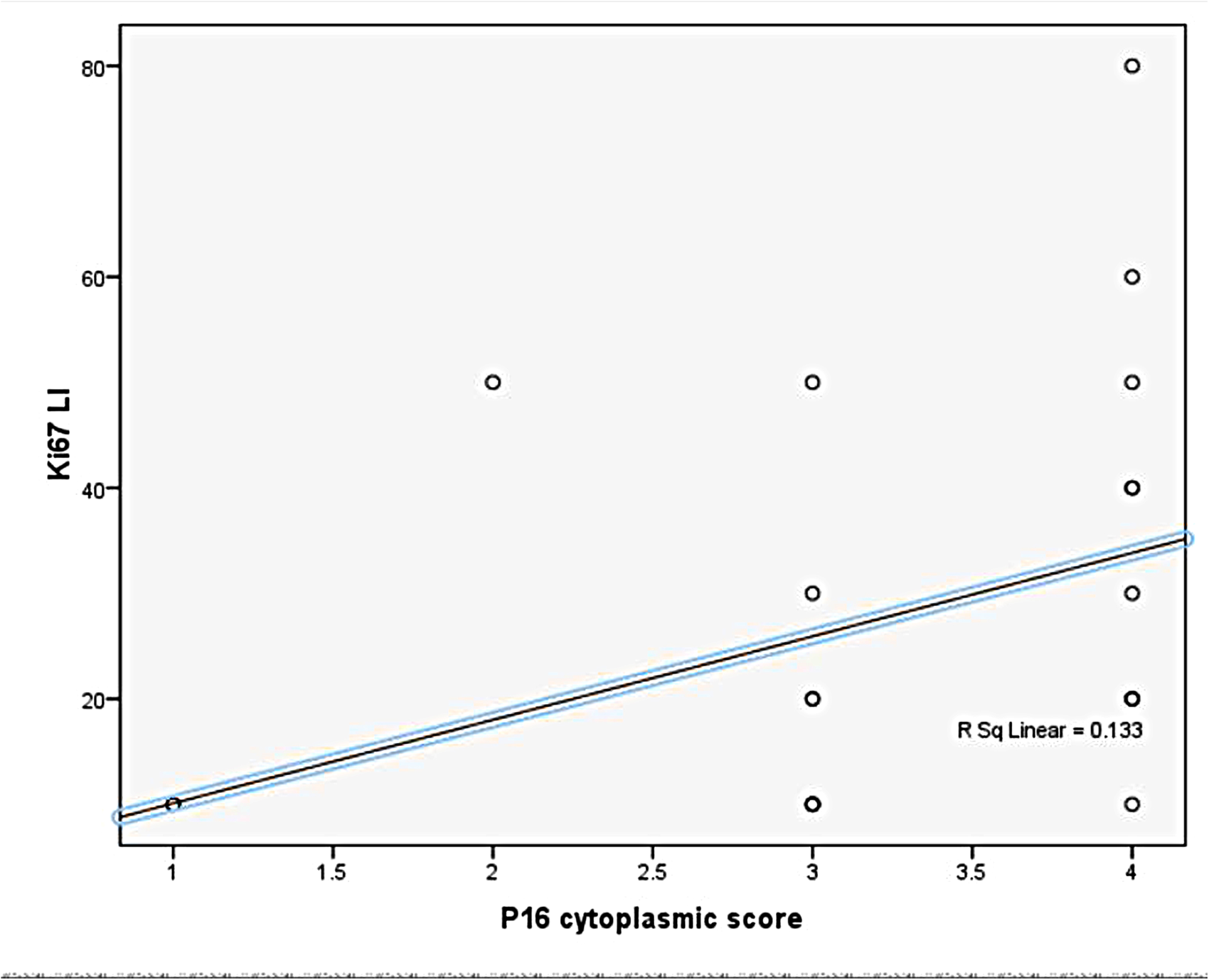
Correlation between level of ki-67 li and p16 cytoplasmic IHC score in MOGCT.
Ki-67 Labeling Index (li) in MOGCTs.

Independent Kruskal Wallis test.
Data were expressed as mean ±SD.
Discussion
Ovarian germ cell tumors are not uncommon, they constitute 5%−20% of all ovarian neoplasms 29 and about 60% of ovarian tumors in first two decades of life. 30 Most of them are benign mature cystic teratomas, while the malignant ovarian germ cell tumors constitute about 5% of all ovarian germ cell tumors. 1 In this study, we investigated 22 malignant ovarian germ cell tumors with mean age 16.5 years old, 20 mature cystic teratomas with mean age 31.5 years old, and 20 normal ovarian tissue as a control. The mean age of malignant ovarian germ cell tumors in this study matched with the universal incidence.30,31 Also, the mean age of mature cystic teratomas agreed with other studies that reported that mean age of mature cystic teratoma is in the fourth decade of life.32,33 In the present study, more than half of malignant ovarian germ cell tumors were presented at stage I. It was reported that malignant ovarian germ cell tumors especially dysgerminomas usually have a good prognosis with good response to chemotherapy, as they are usually detected in stage I, 34 and unilateral salpingo-oophorectomy with preservation of contralateral ovary is the best surgical management. 35 Chen et al. 36 also reported that majority of yolk sac tumors are presented in stage I and 70% of immature teratoma cases were diagnosed at stage IA. Although malignant ovarian germ cell tumors are rare, they usually present at early stage and are curable with optimal therapy. 37
This study investigated the expression of p16 protein as a tumor suppressor protein in malignant ovarian germ cell tumors. P16 protein arrests cell cycle at G1-S transition by binding to CDK4/6 preventing phosphorylation of Rb, so maintains association between Rb and E2F1 transcription factor preventing transcription of target genes responsible for G1-S transition. P16 expression is regulated by RB1 as phosphorylated Rb results in increased p16 expression. 13 In normal ovarian tissues, p16 was not expressed in surface epithelium of normal ovaries and was rarely expressed in few stromal cells. These findings agreed with other studies that reported that epithelial and stromal cells of the non-neoplastic ovary showed little or no p16 expression. 38 Absent or low p16 expression in normal ovarian tissue is attributed to the low p16 expression in healthy cells.39,40 However, Surowiak et al. 41 found strong nuclear p16 expression in surface epithelium of healthy ovarian tissue in comparison with the studied malignant ovarian tumors. This discrepancy might be attributed to the fact that physiological aging and stresses that trigger cellular senescence might increase the expression of p16 protein. 42
In mature cystic teratomas, strong nuclear p16 expression was observed in basal layer of stratified squamous epithelium of studied benign mature cystic teratoma, while weak cytoplasmic staining was noted in rest of stratified squamous epithelium layers, sebaceous glands, and hair follicles. Also, moderate cytoplasmic staining was noted in pyramidal cells in mature brain tissue found in studied mature cystic teratomas. Limited reports were published about p16 expression among mature cystic teratomas. Multiple clinical trials showed the potential effect of CDK4/6 inhibitors in treatment of unresectable cases of mature teratoma in critical sites as intracranial teratomas.43,44 Thus, these findings might suggest that the accumulation of mutant forms of p16 might have a role in mature cystic teratomas tumorigenesis. On the contrary to our results, Zhu et al. 45 reported negative p16 expression in teratomatous skin of ovarian mature cystic teratoma and concluded the low risk of malignant transformation of mature cystic teratoma. However, normal genital squamous epithelium usually showed absent or patchy p16 expression without continuous basal expression.11,46,47 Abnormal p16 expression in squamous epithelium is defined by strong and diffuse nuclear or more typically nuclear and cytoplasmic expression in all epithelial cells in basal and parabasal cells with upward extension involving at least one-third of epithelial thickening; the so called block positivity and this pattern is considered a surrogate marker for HPV infection. 46 Further studies are required to declare the role of p16 in mature cystic teratomas.
In the present study, strong and diffuse p16 cytoplasmic and nuclear staining was detected in all malignant ovarian germ cell tumors under study with significant difference compared to normal ovaries, and also to benign mature cystic teratomas. These findings agreed with Kawauchi et al. 48 study who reported that more than two-thirds of the studied malignant ovarian germ cell tumors showed diffuse p16 expression. Tumors with elevated levels of p16 protein were referred to as RB1 deficient tumors as p16 overexpression denotes dysregulation of p16/RB1 pathway. Inactivation of RB1 in these tumors occurs either as a primary event as in HPV-positive cervical cancer and HPV-positive head and neck cancer or occurs secondary to induction of p16 by oncogenic stresses. 42 However, although p16 is overexpressed, the neoplastic process cannot be completely inhibited and continuous tumor growth occurs possibly due to check point defect at G1-S phase. Thus, accumulation of p16 protein which is normally inhibited by hypophosphorylated RB1 gene 13 suggests that an aberrant dysfunction in the RB1 pathway is implicated in the pathogenesis of malignant ovarian germ cell tumors. It was also claimed that overexpression of p16 protein whether in presence or absence of RB1 function is a mechanism of resistance to CDK 4/6 inhibitors. 49 On the other hand, overexpression of p16 protein in cancer might also be attributed to other functions of p16 protein such as angiogenesis, invasion, and apoptosis. 9
In the present study, we found significant positive correlation between cytoplasmic (but not nuclear) p16 staining in malignant ovarian germ cell tumors and Ki67 labeling index which points to a defect in the function of the accumulated p16 protein in inhibition of tumor proliferation. Although some authors regard cytoplasmic p16 staining as non-specific and should be interpreted as negative,50,51 Evangelou et al. 52 provided electron microscopic evidence that p16 cytoplasmic overexpression in small cell lung carcinoma is specific and not an artifact as they detected cytoplasmic immunoreactivity near the rough endoplasmic reticulum. Romagosa et al. 9 supposed that p16 has different functions according to its subcellular location, so nuclear p16 controls proliferation while cytoplasmic accumulation of p16 occurs through different mechanisms in different tumors. Aberrant p16 cytoplasmic expression was associated with a subset of breast cancer with high proliferation rate. 53 In colorectal carcinoma, cytoplasmic expression of p16 and CDK4 was reported to have a role in early malignant transformation but not in tumor progression. 54 Arifin et al. 55 reported that cytoplasmic overexpression of p16 in high-grade astrocytomas is unfavorable prognostic factor and supposed that sequestration of p16 in the cytoplasm is an alternative mechanism for p16 inactivation. Thus, the significant correlation found in this study between cytoplasmic p16 expression and Ki67 LI studies suggests that dysregulation of RB1/P16 pathway is involved in tumorigenesis of malignant ovarian germ cell tumor and that overexpression of p16 protein is a mechanism for its inactivation and that these tumors might be resistant to cyclin dependent kinase inhibitor therapy. Further studies are needed to confirm these preliminary results.
To summarize, in this study we documented the overexpression of p16 protein in malignant ovarian germ cell tumors and the correlation between the cytoplasmic p16 expression and tumor proliferation. This points to dysregulation of cyclin dependent kinase pathway in malignant ovarian germ cell tumors and suggesting that p16 cytoplasmic expression is a mechanism for its inactivation.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by research grant office, Faculty of Medicine, Assiut University, Egypt
Declaration of Conflicting Interests
The author declares that the manuscript has not been published in any journal or other citable form. There was no conflict of interest. This manuscript has been approved by all co-authors.
Trial Registration
Not applicable.
Ethical Approval
Not applicable.
Informed Consent
Not applicable.
