Abstract
Aims. Immunoscore, based on the evaluation of CD3+ and CD8+ densities in the center of the tumor and its invasive margin, is currently considered as a potential prognostic factor, particularly in colorectal carcinomas. In the current study, we aimed to assess the prognostic value of immunoscore in colorectal cancer stage I to IV, through a survival study. Methods and Results. It was a descriptive and retrospective study involving 104 cases of colorectal cancer. Data were collected over 3 years (2014-2016). An immunohistochemical study (anti-CD3, anti-CD8) by the tissue microarray technique was carried out in the areas of “hot spot” in the tumor center and invasive margin. A percentage was assigned to each marker and within each region. Then, the density was classified as “low” or “high,” according to a cutoff fixed at the median of percentages. immunoscore was calculated by the method described by Galon et al. The prognostic value of the immunoscore was assessed through a survival study. The mean age of patients was 61.6 years. immunoscore was low in 60.6% (n = 63). Our study had shown that low immunoscore significantly deteriorates survival and a high immunoscore enhances survival significantly (P < .001). We found a correlation between immunoscore and T stage (P = .026). A multivariate showed that the predictive factors for survival were immunoscore (P = .001) and age (P = .035). Conclusions. Our study highlights the potential role of immunoscore as a prognostic factor in colorectal cancer. Its reproducibility and reliability allow its introduction into daily practice for better therapeutic management.
Keywords
Introduction
Colorectal carcinoma is a major public health problem. Its annual incidence is increasing representing the third most occurring cancer in the world and the second cause of death by cancer in 2020. 1
Colorectal cancer is a heterogeneous disease with different suggested prognostic factors. 2 The prognostic value of pTNM tumor staging classification of colorectal cancer has been known for decades. However, it is currently accepted that the clinical course can vary considerably between patients within the same histological stage pTNM.3,4 This has directed research toward new prognostic factors, in particular, those related to the tumor microenvironment, and more particularly, to tumor-infiltrating lymphocytes (TILs).
In the literature, the infiltration of the tumor by lymphocytes is predictive of cancer progression and would, therefore, represent a potential prognostic factor. 2 TILs would play a role in inhibiting tumor growth and would be associated with a better prognosis. 5
In this context, a new test called immunoscore, based on the immunohistochemical evaluation of total T lymphocytes (CD3+) and cytotoxic T lymphocytes (CD8+) densities in the center of the tumor and its invasive margin has been proposed. 6 Studies have shown that this immune test is an independent prognostic factor, correlated with better survival and lower risk of recurrence. 7 The prognostic value of immunoscore was validated in 2018 during an international consortium. 8 Thus, immunoscore seems to have a therapeutic impact, particularly for stage II and III colorectal cancer. 8
In Tunisia, the evaluation of immunoscore is not yet carried out and a few studies on the prognostic significance of immunoscore were conducted until now. The objective of our study was to evaluate the prognostic value of immunoscore in a Tunisian series of colorectal cancer.
Methods
One-hundred and four patients diagnosed with colorectal cancer at the Pathology department of Habib Thameur Hospital in Tunisia during a 3-year period (2014-2016) were included in this trial.
Inclusion Criteria
Our study included all patients with histologically proven colorectal carcinoma stage I to IV, who underwent surgical resection of the tumor during the study period (2014-2016) at Habib Thameur Hospital in Tunisia.
Exclusion Criteria
We did not include in this study patients who received neoadjuvant therapy that could influence the immune response in the tumor microenvironment.
Management of Patients
Preoperative assessment included a complete medical history, a physical examination, a colonoscopy with biopsies, and computed tomographic scans. Gender, age, tumor location, tumor size, gross appearance, lymphovascular, or perineural invasion, histological classification according to the 2019 WHO classification, TNM stage according to the eighth edition of the American Joint Committee on Cancer/International Union Against Cancer and type of surgery were recorded.
Selection of “Hot Spot” Areas
For each case of colorectal cancer, the “hot spot” areas of lymphocyte infiltration at the tumor center and at the invasive margin were selected on H&E slides. Tumor center was defined as the central region containing the tumor stroma and cancer cell nests. Invasive margin corresponded to the invasive front. Only lymphocytes were counted. Granulocytes, macrophages, mast cells, and plasma cells were not considered. Areas of necrosis and fibrosis have been avoided.
Immunohistochemical Staining
An immunohistochemical study was carried out on the tissue microarray slides, obtained from the recipient blocks, allowing the analysis on a single slide, of several samples. The study was carried out using the complex Avidin-streptavidin technique using the BOND-MAX automaton (Leica Biosystems). Anti-CD3 and anti-CD8 antibodies (Leica Biosystems) were used for the study of T cells.
Evaluation of T Lymphocytes Densities
A semiquantitative evaluation of T cells densities in the “hot spot” area on a surface of 0.95 mm2 was carried out, following these procedures:
Measurement of an area of 0.95 mm2 which corresponds to a field at magnification 200×. For each region (tumor center and invasive margin) and for each marker (anti-CD3 and anti-CD8), a photo where the lymphocyte infiltrate was maximal at magnification 200× was taken. A percentage of the lymphocyte infiltration was assigned to each photo (Figure 1). The median of the infiltration percentages was calculated and retained as a cutoff for the determination of densities (Table 1).
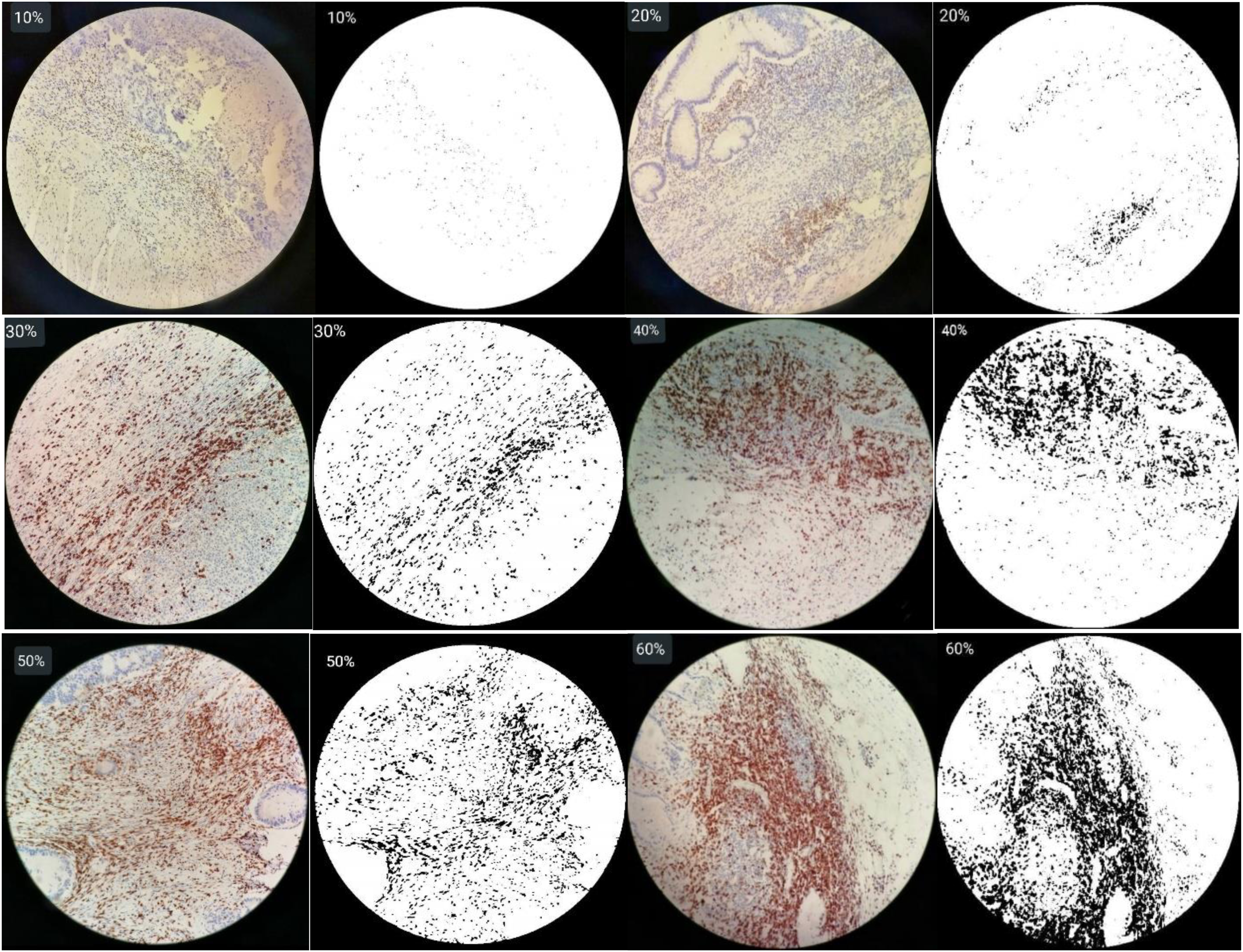
Semiquantitative evaluation of CD3+ densities in immunohistochemical tissue sections obtained at 200× magnification: for each photo, a percentage was assigned reflecting the density of T cells.
Percentage of CD3+ and CD8+ Lymphocyte Densities in CT and IM and Their Median.

The density was considered “low” when it was lower than the median of the percentages. It was considered “high” when it was above this median.
Computation of Immunoscore
We followed a method published by Galon et al.
6
This method has been validated in a task force in which 22 worldwide experts have participated. Indeed, for each marker (CD3 and CD8) and in each region studied (tumor center and invasive margin), a score of 0 was assigned when the density was “low” and a score of 1 when the density was “high.” The immunoscore was obtained by the addition of these scores to give a minimum score of 0 and a maximum score of 4 (Figure 2):
I0: low density of CD3+ and CD8+ in the tumor center and the invasive margin. I1: high density of one of the markers in a single region (tumor center or invasive margin). I2: corresponding to a score of 2. I3: corresponding to a score of 3. I4: high density of the 2 markers (CD3+ and CD8+) in the 2 regions (tumor center and invasive margin).
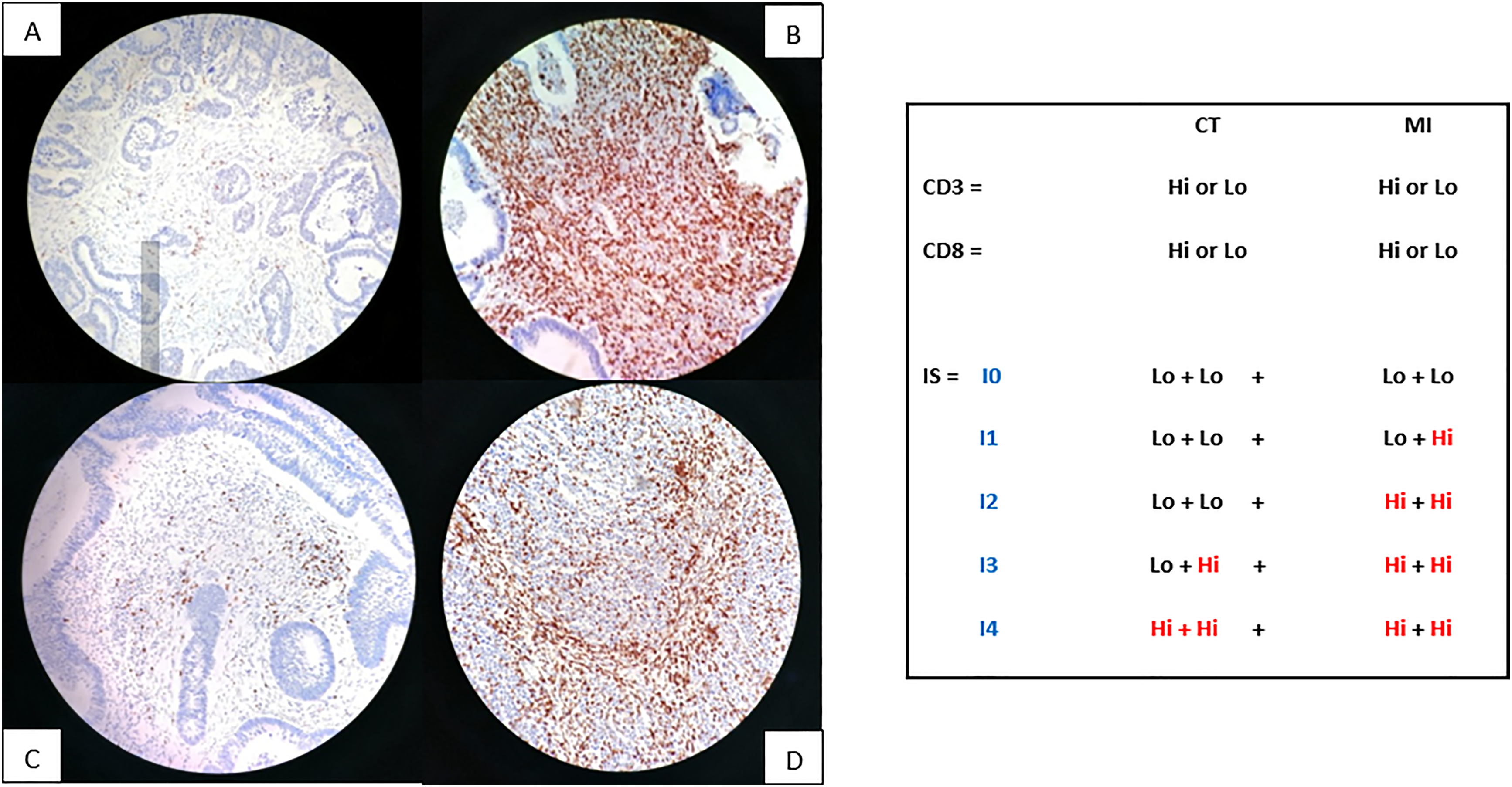
Immunoscore calculation model (200× magnification): (A) Low density of CD3, (B) High density of CD3, (C) Low density of CD8, (D) High density of CD8.
Statistical Analysis
We carried out a descriptive and analytical study. Data collection was stopped on 04/10/2021. Overall survival and predictive factors of survival were analyzed. Univariate analysis aimed to determine dependent prognosis factors. Qualitative variables were compared using the χ2 test or Fisher exact test. Quantitative variables were compared by the independent Student t test. Survival was analyzed by the Kaplan-Meier method and included postoperative deaths. The log-rank test was used to compare survival curves. Multivariate analysis aimed to determine independent prognosis factors. It was performed by linear regression by Cox proportional hazards stepwise procedure, including nonredundant variables chosen by univariate analysis. All statistical analyses were performed using SPSS version 18.00. A P value .05 was considered statistically significant.
Results
A total of 104 patients were included in the study. The clinicopathological features are detailed in Table 2. The average age was 61.6 years. A male predominance was noted (sex ratio = 1.36). The location of the tumor was colonic in 92.3% of cases (n = 96). The tumor size varied from 1.5 cm to 13 cm with an average of 5.52 cm. Histologically, adenocarcinoma not otherwise specified was the most represented histological type (84.6%; n = 88). The grade was low in 95.5% (n = 84). Colorectal cancers were mainly classified as pT3 and pT4 (88.4%; n = 92). Vascular emboli and perineural invasion were present in 49% (n = 51) and 46% (n = 32) of cases, respectively. Lymph node metastasis was revealed in 43.3% (n = 45) of cases. Visceral metastasis was noted in 12.5% (n = 13) of cases. The pTNM stage was mostly II or III (75.9%), and IV in 12.5% of cases.
Demographic and Clinicopathological Features of 104 Primary Colorectal Carcinomas.

Abbreviation: NOS, not otherwise specified.
Of the 104 patients, 16 patients were lost to follow-up. We were only able to judge the evolution in 88 patients. The average follow-up was 40.29 months. The 1-year, 3-year, and 5-year survival rates of colorectal cancer patients were 86.3%, 63.9%, and 51.2%, respectively.
Immunoscore was I0 in 19.2% (n = 20), I1 in 15.4% (n = 16), I2 in 26% (n = 27), I3 in 16.3% (n = 17), and I4 in 23.1% (n = 24). Two groups were identified: those with a low immunoscore (≤2: I0-I2) and those with high immunoscore (>2: I3 and I4). A predominance of low immunoscore (60.6%, n = 63) was noted.
Univariate analysis showed a strong association between the different groups of immunoscore (from I0 to I4) and the overall survival (OS) (P = .001). Similarly, low immunoscore (I0-I2) was correlated with shorter survival, and high immunoscore (I3 and I4) was correlated with longer survival (P < .001). Survival curves illustrating the association between immunoscore and OS are shown in Figure 3.
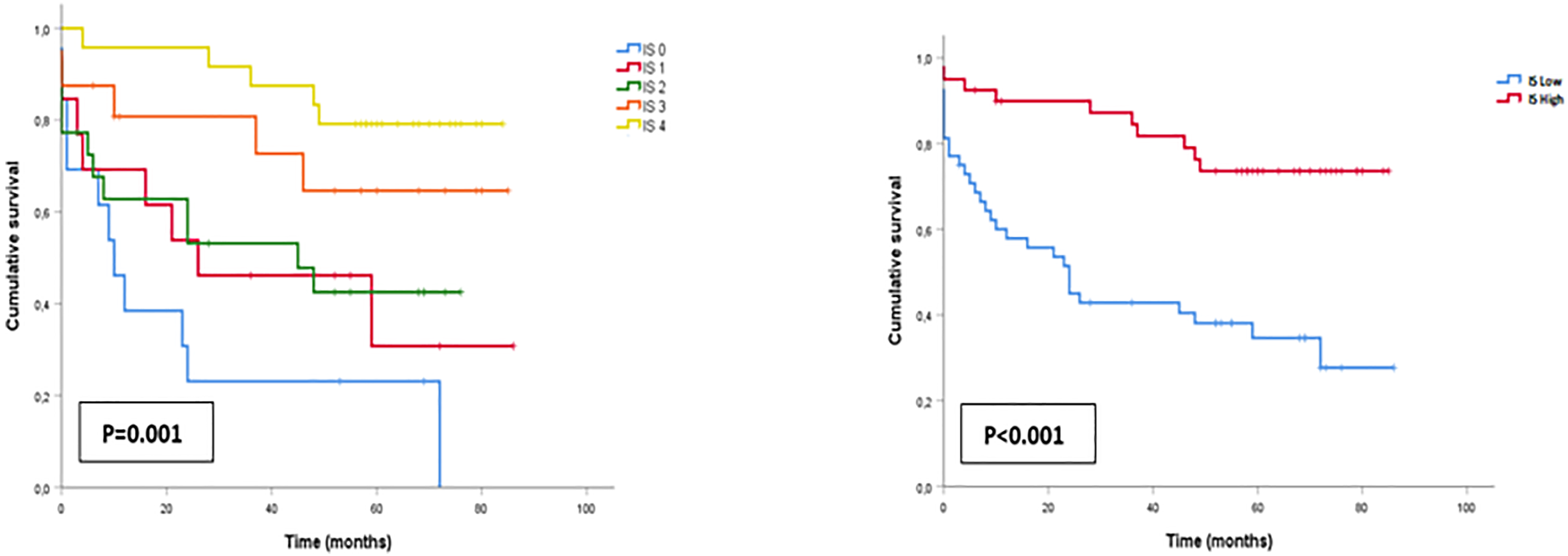
The correlation of immunoscore with survival.
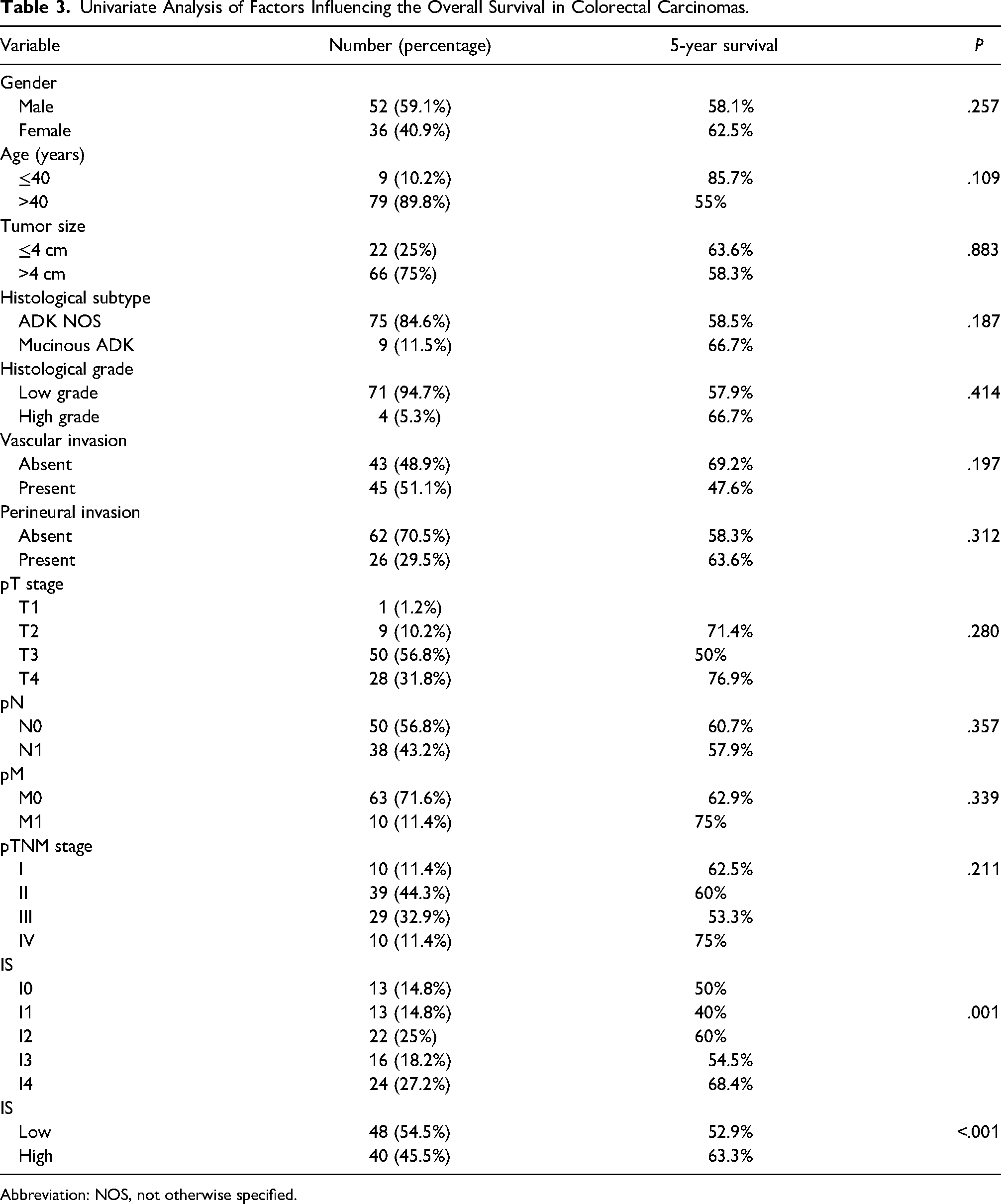
The other prognostic factors: age, tumor size, histologic grade, pT stage, pM, pTNM stage, vascular invasion, and perineural invasion tended to reduce survival without significant difference (Table 3).
Univariate Analysis of Factors Influencing the Overall Survival in Colorectal Carcinomas.
Abbreviation: NOS, not otherwise specified.
Multivariate Cox proportional hazards models included variables associated with survival in univariate analysis, age, degree of parietal invasion, presence of vascular emboli, stage, and immunoscore showed that immunoscore was the strongest independent prognostic factor (P = .001), follow-up by age (P = .035) (Table 4).
Multivariate Analysis of Overall Survival in Colorectal Carcinomas.

A significant association was found between advanced pT stage and low immunoscore (P = .026). On the other hand, immunoscore was not correlated with age, tumor size, tumor grade, vascular emboli, perineural invasion, metastasis, and pTNM stage (Table 5).
Association Between Immunoscore and Other Prognostic Factors.
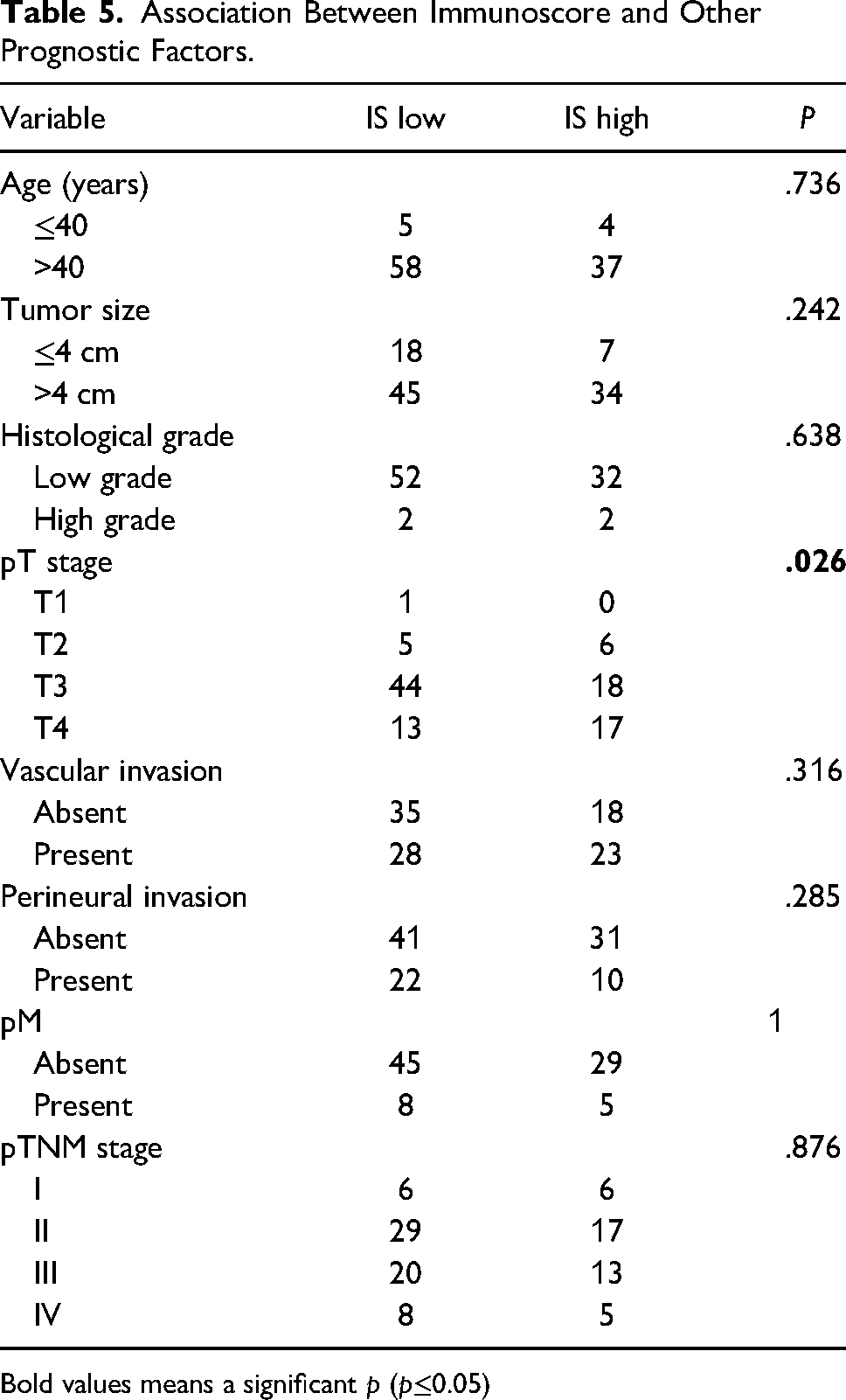
Bold values means a significant p (p≤0.05)
Discussion
We calculated the immunoscore in 104 patients with colorectal cancer following the recommendations published by Galon et al 6 but we have replaced the automated quantification method to determine the densities of T cells lymphocytes with a semiquantitative method. We found that the immune response varied considerably between patients; Indeed, 19.2% of patients had an immunoscore I0, 15.4% had an immunoscore I1, 26% had an immunoscore I2, 16.3% had an immunoscore I3, and 23.1% had an immunoscore I4.
Two groups of patients were individualized: those with a low immunoscore (≤2: I0-I2) and those with high immunoscore (>2: I3 and I4). A predominance of the group with a low immunoscore (60.6%, n = 63) compared to the other group (39.4%, n = 41) was revealed.
The univariate analysis showed that there was a significant correlation between the different immunoscore groups and 5-year survival (P = .001). Thus, a low immunoscore was correlated with short survival, and a high immunoscore was correlated with longer survival (P < .0001). The immunoscore was correlated with the degree of parietal invasion (P = .026).
For decades, the pTNM stage was thought to be the best prognostic factor in colorectal cancer. However, the clinical outcome can vary in patients with the same stage. The pTNM stage focuses on tumor cells and does not consider the host immune response. Recent studies support the role of immune cells in tumor progression, particularly, lymphocytes infiltrating tumors are demonstrated to have a protective role against tumor cells. 5
Of all immune cells involved in the tumor microenvironment, CD3, CD8, and CD45RO lymphocytes had the best prognostic impact.8,9 In the present study, CD3 and CD8 were chosen as markers because of the stability of these antigens and the good quality of staining. CD45RO was excluded because of background staining and loss of antigenicity in stored sections as proven in other studies. 10 Besides, the prognostic value of immunoscore in different patient groups was better in a combined analysis of 2 regions: tumor center and invasive margin compared to a single region analysis. 11 So, immunoscore is a test based on the evaluation of densities of both lymphocytes CD3 and CD8 in 2 specific regions: tumor center and invasive margin. 12
The determination of immunoscore is automated in most studies using software dedicated to immunoscore. In Tunisia, we did not have software to enumerate lymphocytes in tumors. A semiquantitative study was carried out and validated with significant results. To the best of our knowledge, this is the first Tunisian study that confirmed the prognostic value of immunoscore using a semiquantitative method. Therefore, it represents an alternative tool for the quantification of immunoscore, particularly, in countries that do not have the software. However, this semiquantitative method remains less precise than the automated method.
Based on the analysis of a cohort of 104 patients with colorectal cancer, we demonstrated the prognostic value of immunoscore on OS: high immunoscore was associated with longer OS, while low immunoscore was associated with shorter OS. These findings support the conclusions of many previous studies implicating immune cells in colorectal cancer. The majority have confirmed an association between immunoscore and OS or disease-free survival (DFS).6–8,12–19
Pagès et al 8 have proved that the prognostic value of immunoscore was superior to the pTNM staging system. They proposed the introduction of immunoscore as a new component of the classification of cancers designated TNM-Immune (TNM-I).
Immunoscore has also a therapeutic role in colorectal cancer. On the one hand, patients with colorectal cancer stage II, having a low immunoscore, will be justified for adjuvant chemotherapy which would be useless for the group with a high immunoscore. 20 On the other hand, patients with colorectal cancer stage III and a high immunoscore will be justified for shorter adjuvant chemotherapy (3 months) compared to the group with a low immunoscore (6 months).20,21 Thus, the immunoscore allows personalized therapeutic management of patients with colorectal cancer stage II and III.
Conclusion
Immunoscore is a simple, rapid, and reproducible immune test with a strong prognostic value and therapeutic impact. The introduction of this parameter in daily practice becomes mandatory.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
