Abstract
EBV-positive inflammatory follicular dendritic cell sarcoma (EBV+ inflammatory FDCS) is a rare neoplasm almost exclusively located in the spleen or liver. It is characterized by a proliferation of EBV-positive spindle-shaped cells bearing follicular dendritic cell markers, associated with an abundant lymphoplasmacytic infiltrate. EBV+ inflammatory FDCS is often asymptomatic or responsible for mild symptoms. It usually displays an indolent course and its prognosis is excellent after tumor removal, although relapsing and metastatic forms exist. Herein, we describe an aggressive form of splenic EBV+ inflammatory FDCS in a 79-year-old woman presenting with abdominal pain, deterioration of general health status, major inflammatory syndrome, and symptomatic hypercalcemia. A splenectomy was performed leading to a rapid improvement in her clinical condition and normalization of laboratory abnormalities. Unfortunately, her symptoms and laboratory abnormalities reappeared 4 months later. Computed tomography showed a mass in the splenectomy site and multiple liver and peritoneal nodules. Further analyses were performed on tumor tissue and showed positive phospho-ERK staining of tumoral cells indicating activation of MAPK pathway. Inactivating mutations were found on CDKN2A and NF1 genes. Subsequently, the patient's condition deteriorated rapidly. Since interleukin-6 levels were dramatically increased, tocilizumab was used but only had a transient effect on the patient's symptoms and inflammatory syndrome. Antitumor agent gemcitabine was initiated but her clinical condition continued to deteriorate and the patient died 2 weeks later. The management of aggressive forms of EBV+ inflammatory FDCS remains challenging. However, since these tumors seem to display genetic alterations, better characterization could lead to molecular targeted therapies.
Keywords
Introduction
EBV-positive inflammatory follicular dendritic cell sarcoma (EBV+ inflammatory FDCS) is a rare neoplasm considered a subtype of follicular dendritic cell sarcoma (FDCS).1,2 FDCS usually occurs in adults with a median age between 50 and 60 years. The tumor is almost exclusively located in the spleen or liver and usually displays an indolent course, although systemic symptoms can be present. EBV+ inflammatory FDCS is characterized histologically by a proliferation of spindle-shaped cells positive for at least 1 follicular dendritic cell (FDC) marker, mainly CD21, CD23, and CD35. 1 It is usually accepted that both EBV+ inflammatory FDCS and conventional FDCS arise from FDC, a subset of stromal cells essential for antigen presentation to B cells within secondary lymphoid organs.1,3 EBV+ inflammatory FDCS differs from conventional FDCS by the presence of abundant lymphoplasmacytic infiltrate and consistent positivity of spindle cells for EBV. Regarding genetic alterations in EBV+ inflammatory FDCS, the data are scarce in the literature. Treatment is based on surgical excision and leads to complete remission in most cases. Although rare, relapses can occur after surgical removal and their management is challenging given the paucity of literature on this subject. 1 Herein, we report a case of splenic EBV+ inflammatory FDCS with major systemic manifestations, relapsing after surgical removal. We also provide molecular analysis of this rare tumor.
Case Presentation
A 79-year-old woman presented to our hospital with a 6-month history of worsening abdominal pain, asthenia, and weight loss. Her past medical history only included breast cancer, in remission for more than 10 years. Upon initial presentation, the patient was afebrile and, apart from mild tenderness in the upper left abdomen, her physical examination was unremarkable. Laboratory tests showed marked inflammatory syndrome, anemia, neutrophilia, monocytosis, eosinophilia and mild hypercalcemia (Figure 1). Hypercalcemia was associated with normal phosphorus, low parathyroid hormone, normal 25-OH vitamin D with increased 1,25-OH vitamin D, and indetectable parathyroid hormone-related peptide levels. Whole-body computed tomography (CT) was performed and showed a 70-mm splenic mass (Figure 2A) without any other radiological abnormality. Infectious diseases, especially tuberculosis and invasive mycoses, were excluded after an extensive workup. Bone marrow aspiration and biopsy did not show any evidence of hematological malignancy. Fluorodeoxyglucose positron emission tomography (18FDG PET/CT) revealed intense uptake at the site of the splenic lesion (maximum standardized uptake value of 21); no other abdominal or distant hypermetabolic lesion we found (Figure 2B). The patient underwent a splenic biopsy that showed a proliferation of EBV-positive cells of indeterminate nature. Since the patient's condition was worsening and no diagnosis could be made, a splenectomy was finally performed.
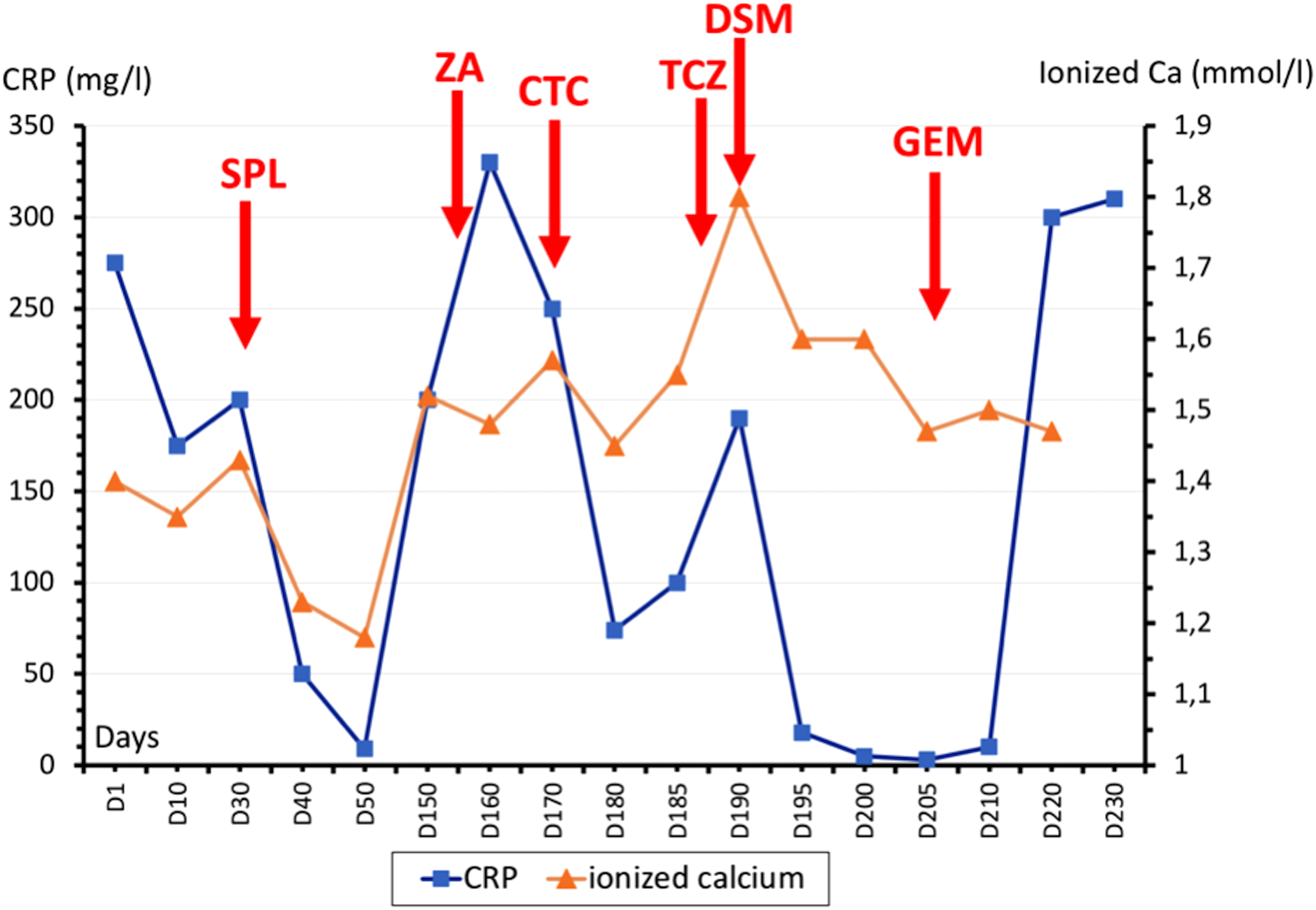
Evolution of CRP and blood calcium according to therapeutic regimens.
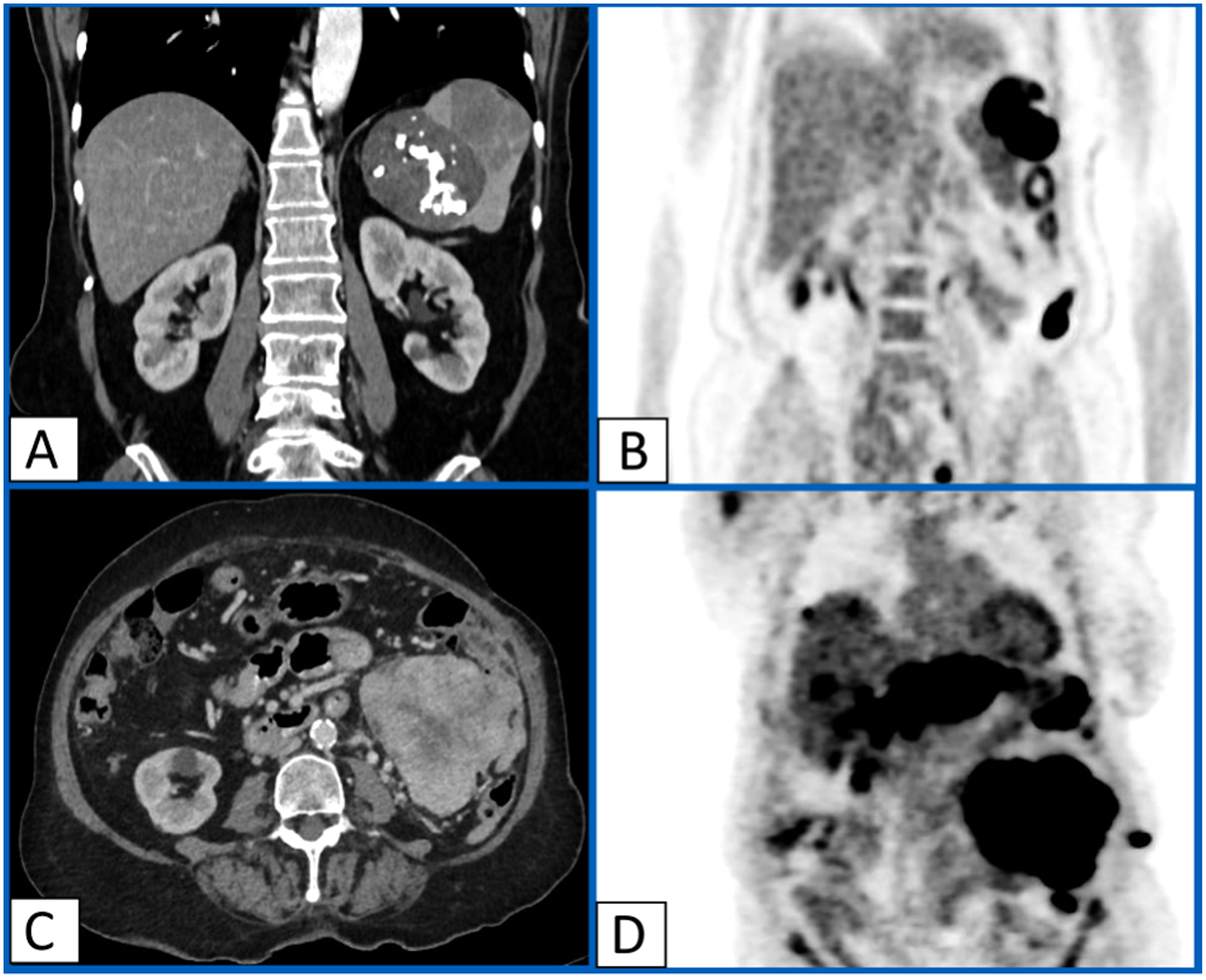
CT and 18FDG PET/CT imaging of the tumor. Panels A and B: CT at initial presentation demonstrated a 70-mm splenic mass (A) with intense uptake (SUVmax of 21) on 18FDG PET/CT (B). Panels C and D: CT at relapse showed 85 × 95 mm mass in the splenectomy site associated with diffuse peritoneal and liver nodules (C). These lesions were all intensely hypermetabolic on 18FDG PET/CT (SUVmax of 14 to 23) (D).
Histological analysis of splenic tissue revealed a proliferation of spindle-shaped cells with small cytoplasmic amount, large nuclei, and vesicular chromatin (Figure 3), surrounded by an extensive inflammatory infiltrate of lymphocytes, plasma cells, neutrophils, and histiocytes, and large areas of necrosis with rupture of the splenic capsule were noted. No increase of IgG4-positive plasma cells or granuloma lesions were found. Tumor cells were focally positive for smooth muscle actin and had heterogenous expression of CD45 but were negative for CD30, CD21, CD23, PS100, CD3, CD5, CD79a, CD138, desmin, ALK, and HHV8. Tumor cells showed positivity for EBER but not inflammatory cells (Figure 3). Bacterial and fungal tissue cultures were negative. Overall, a diagnosis of EBV+ inflammatory FDCS was retained. In the days following the splenectomy, her general condition and laboratory features rapidly improved, including inflammatory parameters, hyperleukocytosis, and hypercalcemia.
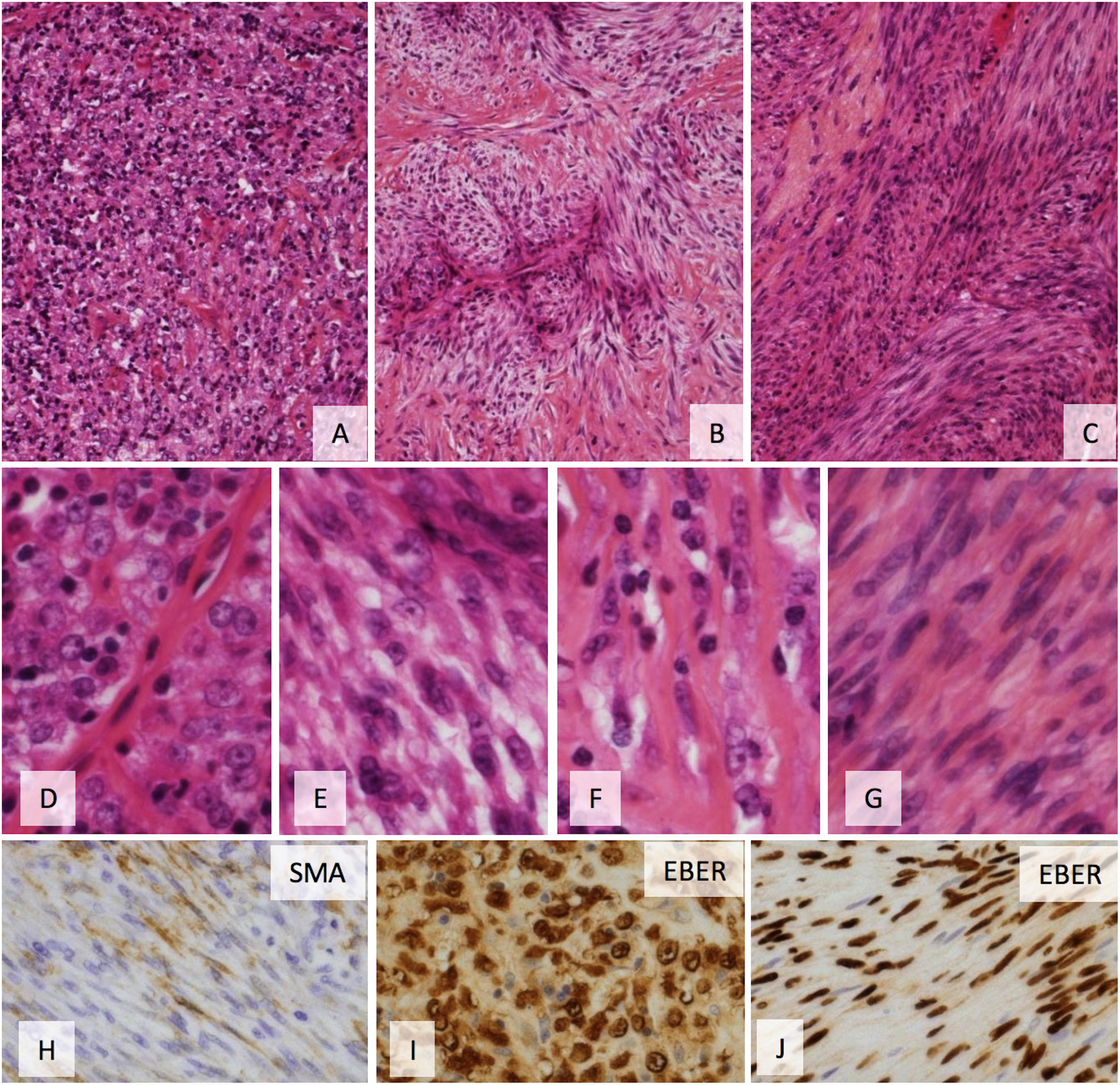
Pathological findings. EBV-positive inflammatory follicular dendritic cell sarcoma (EBV+ inflammatory FDCS). Different patterns are remarkable at low magnification (obj × 10): diffuse tumoral proliferation in an important inflammatory background (A), short fascicles joining fibrotic areas (B) and obvious fascicles. Tumor cells present a large morphological spectrum from round mono- or binucleated cells to spindle-shaped (D to G). Tumor cells are focally positive for smooth muscle actin (H) and diffusely positive for EBER in situ hybridization both in round (I) and spindle cells (J).
However, the patient was readmitted 4 months later because of symptoms recurrence and a new abdominal mass in the upper left quadrant. Laboratory tests showed the same abnormalities as before splenectomy (Figure 1). Abdominal CT showed an 85 × 95 mm mass in the splenectomy site associated with diffuse peritoneal and liver nodules. Lesions were all intensely hypermetabolic on 18FDG PET/CT (Figure 2). Biopsy of the abdominal mass revealed abnormalities identical to those previously observed in the spleen, thus leading to the diagnosis of EBV+ inflammatory FDCS recurrence. Immunohistochemistry analysis was performed and showed negative PDL-1 staining and positive phospho-ERK staining. Next-generation sequencing analysis of 50 genes frequently implicated in cancer (Ion AmpliSeq Cancer Hotspot Panel v2) revealed a mutation in the CDKN2A gene (NM_000077.4:c.225_243dup, p.Val82Argfs*44, variant allele frequency [VAF] 20%). Measuring of tumor mutational load (Oncomine Tumor Mutational Load Assay) identified 1.6 mutations per megabase, including an inactivating mutation in the NF1 gene (NM_001042492.2 :c.6852_6855delTTAC, p.Tyr2285Thrfs*5, VAF 14%), encoding for neurofibromin 1. No fusion genes were detected (Archer FusionPlex Lung panel).
In the days following the diagnosis of EBV+ inflammatory FDCS recurrence, the patient presented clinical deterioration with an increase in hypercalcemia and inflammatory parameters. Symptomatic treatment of hypercalcemia with intravenous hydration, glucocorticoids, and zoledronic acid was unsuccessful while denosumab only resulted in a partial decrease in serum calcium. Since interleukin-6 (IL-6) levels were dramatically increased (616 pg/mL; normal value: < 7.5 pg/mL), tocilizumab, an anti-IL-6 receptor monoclonal antibody, was initiated, leading to transient clinical improvement and decrease in inflammatory markers, but without any effect on calcium levels (Figure 1). Antitumor agent gemcitabine was then initiated but had no beneficial effect. It should be noted that tocilizumab and gemcitabine were used off-label and, thus, discussed in multidisciplinary meetings; the patient was informed of this and gave her consent. Unfortunately, her condition continued to deteriorate, and the patient died 2 weeks later, shortly after the results of molecular analyzes.
Discussion
We report a case of splenic EBV+ inflammatory FDCS responsible for severe systemic manifestations and relapsing after surgical removal. The diagnosis was made on the basis of histopathological data. In our patient, histologic features were suggestive of EBV+ inflammatory FDCS. However, spindle cells were positive for smooth muscle actin and did not express CD21 and CD23. Previous studious showed that the expression of FDC markers is inconsistent in EBV+ inflammatory FDCS and that the expression of myofibroblast or fibroblast markers is sometimes observed. 1 Unfortunately, the expression of CD35 could not be assessed. Furthermore, a heterogeneous expression of the pan-leukocyte marker CD45 was found, as reported in other studies. 4
Aggressive presentation with major systemic inflammation, as in our patient, is uncommon in this disease, with most patients being usually asymptomatic or showing mild symptoms.1,5 Our patient also had symptomatic and resistant hypercalcemia, with increased levels of 1,25-OH vitamin D and normal 25-OH vitamin D, suggesting 1-alpha hydroxylase activity within the tumor. However, no granuloma was found, raising the hypothesis of 1-alpha hydroxylase activity by the tumoral cells themselves, as reported in other types of tumors. 6 Our patient's outcome was also remarkable. In a literature review on 50 EBV+ inflammatory FDCS patients, only 5 (10%) patients relapsed after tumor resection; none of them had a splenic localization. 5
The use of chemotherapy was only reported in 2 patients with EBV+ inflammatory FDCS patients and consisted of adjuvant chemotherapy (cyclophosphamide, doxorubicin, vincristine, prednisolone) associated with surgery. One did not develop recurrence after a 7-year follow-up while data was not available for the other.7,8 Since our patient's condition was deteriorating rapidly and blood IL-6 and CRP were dramatically high, we initiated tocilizumab. This led to a dramatic fall in CRP levels and moderate clinical improvement. Unfortunately, this improvement was only temporary in our patient and tocilizumab had no effect on tumor volume. Given its possible efficacy in FDCS 9 and patient frailty, a gemcitabine regimen was decided but had no beneficial effect.
In order to find a therapeutic target, molecular analyzes were performed and detected mutations on CDKN2A and NF1. CKDN2A encodes proteins involved in cell cycle regulation by inhibition of cyclin-dependent kinase (CDK). 10 NF1 encodes for neurofibromin 1, a protein implicated in the inhibition of RAS proteins and hence MAPK pathway. 11 In 2014, Go et al reported BRAF V600E mutations in FDCS and especially in the EBV+ inflammatory subtype with 2 out of 5 patients displaying this mutation. 12 However, in a study including 6 patients with EBV+ inflammatory FDCS, Frigola et al found no mutation in this gene or in other genes involved in the MAPK pathway. 13 This mutation was not detected in our patient either. Later, several mutations implicated in cell cycle regulation (including CDKN2A), NFkB pathway or immune evasion have been identified in FDCS.14,15 More recently, Bruehl et al described 2 cases of EBV+ inflammatory FDCS whose molecular analysis showed variants in several genes but all were of uncertain clinical significance. 2 The presence of genetic alterations in our patient raised the question of targeted therapies. Clinical trials evaluating cyclin-dependent kinase inhibitors in CDKN2A mutated tumors are in progress. 16 Moreover, our patient's tumor exhibited positive phospho-ERK staining indicating activation of MAPK pathway. 11 Together with NF1 mutation, this could support the use of MEK inhibitors.
In conclusion, although usually indolent, aggressive forms of splenic EBV+ inflammatory FDCS with systemic manifestations, metastasis, and recurrence after surgery are possible. These tumors can also be responsible for severe systemic manifestations. Anti-IL-6 therapies may be useful to reduce systemic manifestations but showed only a transient effect. EBV+ inflammatory FDCS seem to frequently display genetic alterations, and even if the significance of these mutations is frequently uncertain, better characterization could lead to molecular targeted therapies.
Footnotes
Acknowledgments
We would like to thank Pr. Jean François Emile (Hôpital Ambroise Paré, Department of Pathology; Boulogne-Billancourt, France) for having performed phospho-ERK staining of tumoral cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Since our patient died before we started writing this case report, we were not able to seek her consent. However, all data, including patient's characteristics and figures, are anonymized; identification of the patient is therefore impossible.
Trial Registration
Not applicable, because this article does not contain any studies with human or animal subjects.
