Abstract
Introduction
There are scant data on renal cell carcinoma (RCC) from relatively younger patients in South America using contemporary classification.
Methods
Fifty-nine consecutively treated patients with RCC (≤40 years old) were assessed from the National Institute of Neoplastic Diseases in Peru from 2008 to 2020 (34 males; 25 females), age range of 13 to 40 years.
Results
Most common presenting symptoms were flank pain (n = 40), hematuria (n = 19), and weight loss (n = 12). Associated conditions included 4 patients with proven or presumed tuberous sclerosis and 1 patient with von Hippel Lindau syndrome, all with clear cell RCC. Tumor histopathology was clear cell RCC in 32 of 59 (54%), chromophobe RCC in 6 of 59 (10%), and 5 of 59 (8%) each of papillary RCC and MiT family translocation-associated RCC. Four of 59 (7%) were FH-deficient RCC and 2 of 59 (3%) remained unclassified. The remaining tumors were isolated examples of clear cell papillary renal cell tumor, eosinophilic solid and cystic RCC (ESC RCC), RCC with fibromyomatous stroma, sarcomatoid RCC, and sarcomatoid clear cell RCC. Of the 4 FH-deficient RCCs, none had the classic morphology. The 5 MiT family translocation RCCs had variable morphology. There were 41 tumors without recurrence or metastases, 3 tumors with local recurrence only, 8 tumors with metastases only, and 7 tumors with both local recurrence and metastases.
Conclusions
The current study demonstrates the importance of special studies in accurately classifying RCC in younger individuals. The distribution of RCC subtypes in younger individuals is similar between 2 representative large institutions of the United States and Peru.
Keywords
Introduction
Renal cell carcinoma (RCC) accounts for less than 5% of all pediatric renal neoplasms. 1 It is the most common malignancy in the second decade with a median age of 12.9 years and an equal distribution between males and females. 2 The majority of RCC studies in young adults and children have analyzed patients from North America and Europe. There are scant data on RCC from relatively younger patients in South America using contemporary classification.
Methods
With the approval of the institutional review board, 59 consecutively treated patients with RCC in patients aged ≤40 years of age were assessed from National Institute of Neoplastic Diseases in Peru from 2008 to 2020. The National Institute of Neoplastic Diseases is a major referral center for cancer in Peru. In 2018, it treated 256 new renal cell cancers. This compares to 2714 new renal cancers seen in Peru in 2020, approximately 12% of Peru's kidney cancers.
Where to draw an age cut-off to define young patients with RCC is controversial. We chose ≤40 years of age because it is significantly younger than the median age of occurrence of RCC (55 years). Moreover, while our files contained only 13 patients aged ≤30 years of age, patients in the 31 to 40 year age range ultimately were shown to have a very similar distribution of pathological classification.
A histological review was performed on all tumors. In tumors, where the hematoxylin and eosin (H&E) stained sections showed the classical morphology of clear cell RCC, chromophobe RCC, papillary RCC without features suggestive of fumarate hydratase (FH) deficiency, or clear cell papillary renal cell tumor no additional special studies were performed. In all the remaining tumors where the diagnosis was uncertain based on the H&E morphology, additional studies were routinely performed including immunohistochemistry for FH, succinate dehydrogenase B (SDHB), cathepsin K, carbonic anhydrase IX (CA9), and keratin 20 in the one tumor with features suggestive of eosinophilic solid and cystic RCC (ESC RCC). Tumors that remained unclassified had fluorescent in situ hybridization (FISH) performed for the rearrangement of TFE3 and TFEB.
Immunohistochemistry (IHC) was performed on the Benchmark XT autostainer (Ventana Medical Systems Inc., Tucson, AZ, USA) for FH (J-1 Cruz Biotechnology, Santa Cruz, CA, USA; 1:200 dilution), Cathepsin K (clone 3F9, dilution 1:2000, Abcam), CA9 (polyclonal rabbit, dilution 1:1000, Abcam), and keratin 20 (ks20.8; Dako, Carpinteria, CA, USA; pre-diluted). IHC for SDHB (catalog no. M7195; Abcam, Cambridge, MA, USA; 1:100 dilution) was performed on the Leica Biosystems autostainer. For FH and SDHB, a complete absence of labeling of neoplastic cells in the presence of intact labeling of normal tissue was considered a loss of expression. For keratin 20, any labeling of neoplastic cells was considered as a positive result. TFE3 and TFEB FISH were performed as previously reported.3,4
Results
The patient population consisted of 34 males and 25 females. The age range was 13 to 40 years. The breakdown in years of age was: 10 to 20 (n = 5); 21 to 30 (n = 8); 31 to 35 (n = 16); and 36 to 40 (n = 30). The 5 patients with TFE3 or TFEB translocated associated RCC were 15, 34, 35, 35, and 40 years of age, respectively. The most common presenting symptom was flank pain in 40 patients, followed by hematuria and weight loss in 19 and 12 patients, respectively. Three patients presented with all 3 symptoms. Only 11 (17%) patients lacked any presenting symptoms.
Six patients had a partial nephrectomy, with the remaining patients undergoing radical nephrectomy. All tumors were unifocal with the exception of 1 patient with bilateral single tumors. A 13-year-old patient had proven tuberous sclerosis with multiple angiomyolipomas and ESC RCC. One patient had a history of von Hippel Lindau syndrome with clear cell renal cell carcinoma (RCC). There were 3 additional patients with presumed VHL that also had hemangioblastomas in the cerebellum (n = 2) and eye (n = 1). Although there were 2 patients with uterine leiomyomas, none of them were in patients with FH-deficient RCC.
Upon workup at the National Institute of Neoplastic Diseases Hospital, 15/59 tumors were re-classified in terms of their diagnosis. In some tumors, reclassification resulted after performing FISH for TFE3/TFEB or after immunohistochemistry with antibodies that were not available in the original institution. The final histological classification is seen in Table 1. Overall, the most common tumor was clear cell RCC in 32 of 59 (54%) patients, followed by chromophobe RCC in 6 of 59 (10%), and 5 of 59 (8.5%) each of papillary RCC and translocation-associated RCC. Tumors diagnosed as clear cell RCC, papillary RCC, and chromophobe RCC had the typical histology for these entities and are not illustrated. Four of 59 (7%) tumors were FH-deficient RCC and 2 of 59 (3%) remained unclassified. Isolated examples of other tumors are listed in Table 1.
Histological classification of renal cell carcinoma in Peruvian patients ≤40 years of age.
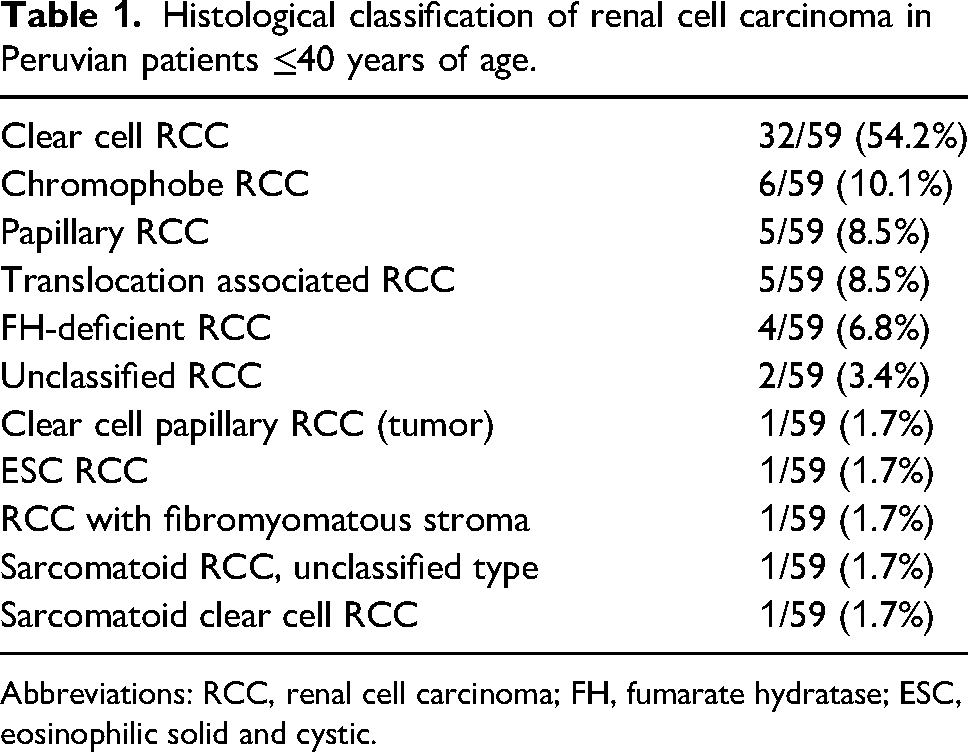
Abbreviations: RCC, renal cell carcinoma; FH, fumarate hydratase; ESC, eosinophilic solid and cystic.
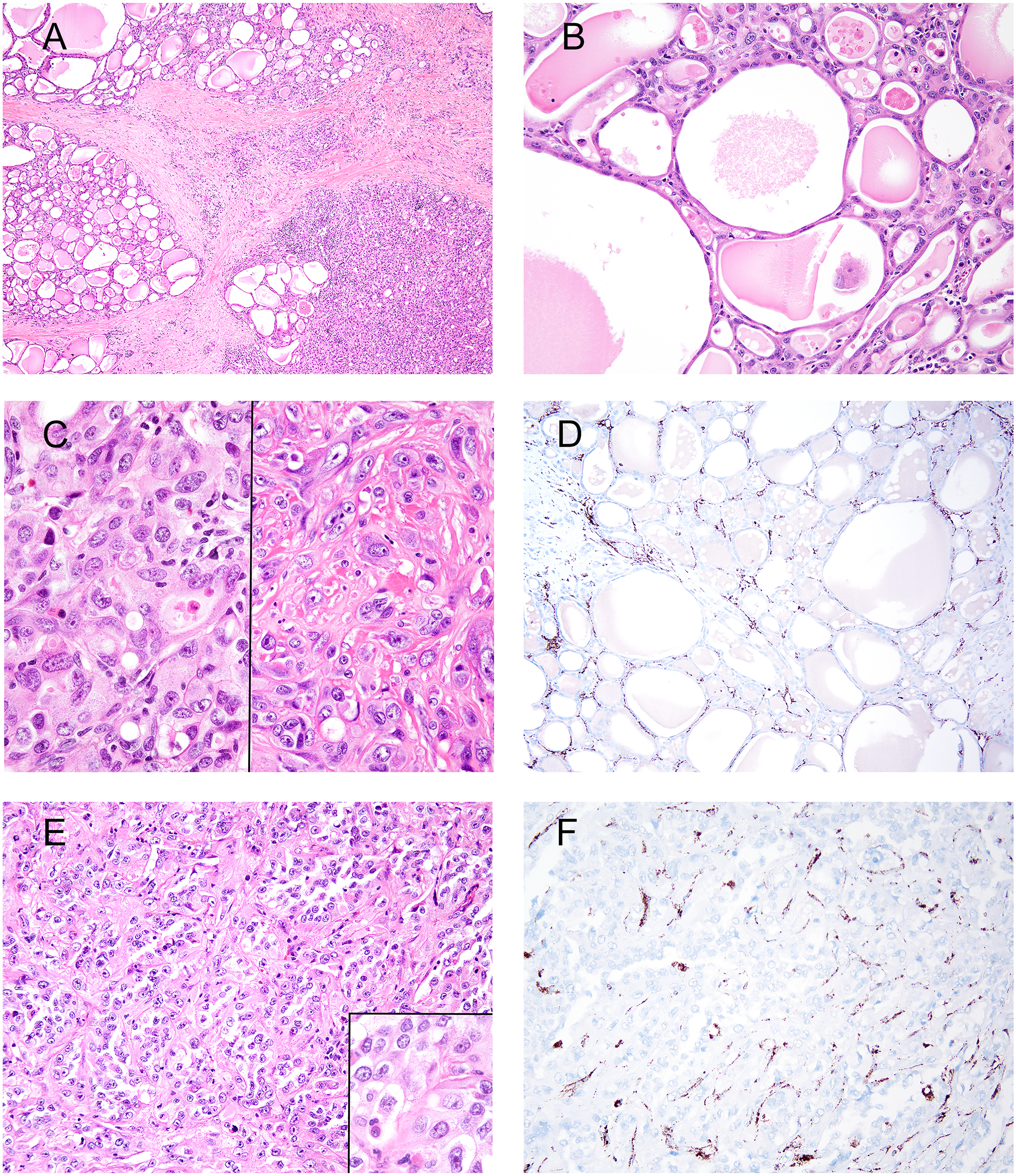
Of the 4 FH-deficient RCCs, none had the classic morphology with diffuse large prominent nucleoli surrounded by halos (Figures 1 and 2). The 5 MiT family translocation RCCs had variable morphology (Figures 3 and 4). Of the 5 translocation-associated RCCs, 2 had the classic appearance of admixed clear cell and papillary features typical of TFE3 rearranged translocation-associated RCCs. One tumor more closely resembled TFEB rearranged RCC with calcified hyaline material and a biphasic appearance, yet showed a TFE3 rearrangement. The remaining 2 tumors were not particularly suggestive of translocation-associated RCCs, 1 TFE3 and the other TFEB rearranged. The RCC with fibromyomatous stroma had the characteristic morphology of this entity, resembling clear cell papillary renal cell tumor, yet with the presence of broad bands of fibromyomatous stroma separating the tumor into variably sized nodules (Figure 5). The one ESC RCC in this study was solid without cysts (Figure 5).
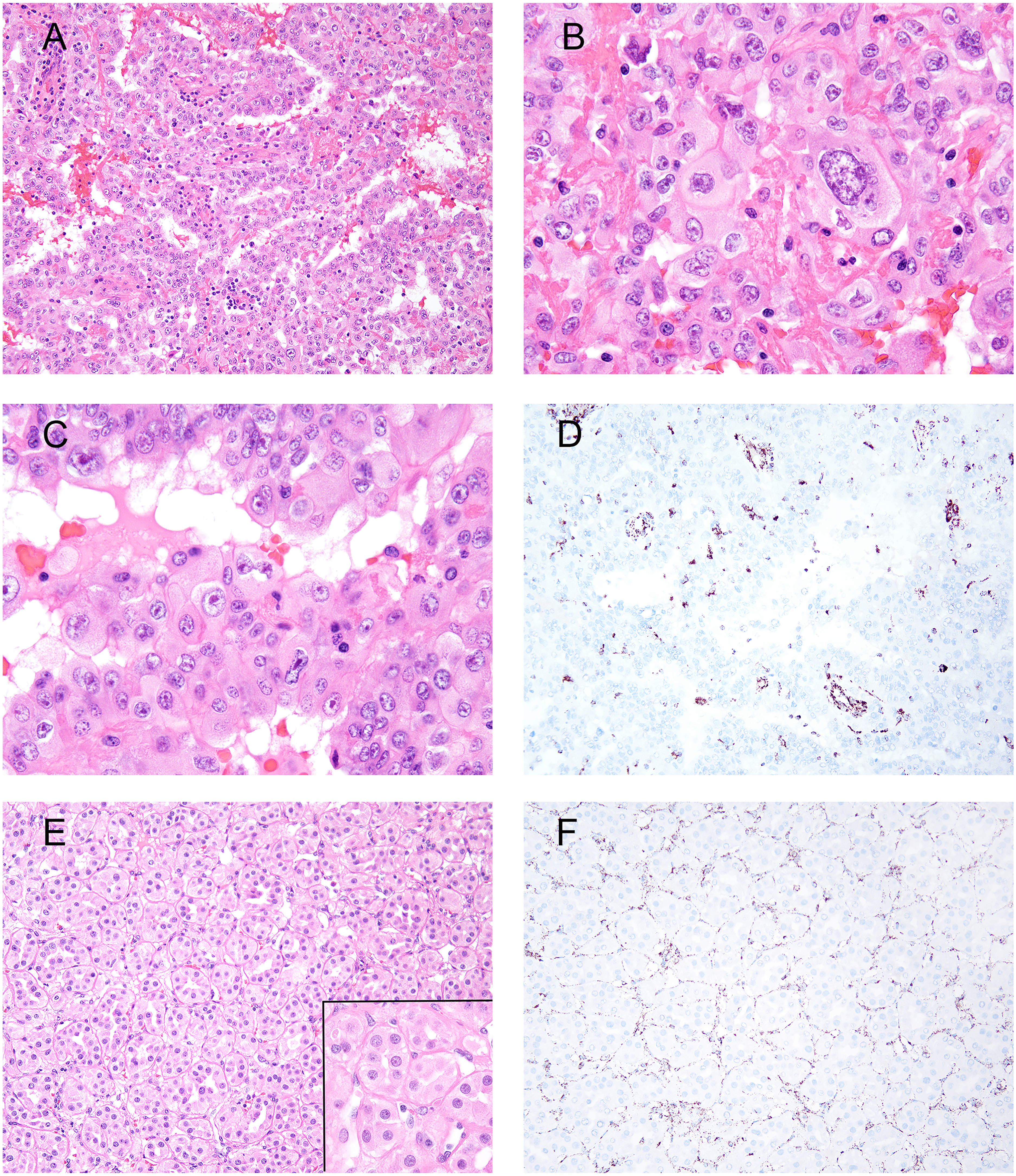
FH-deficient RCC. A to D represent one tumor and E to F another tumor. (A) Tubulopapillary architecture and abundant eosinophilic cytoplasm and patchy interstitial chronic inflammation. (B) Diffuse architecture with cells having moderate to marked nuclear pleomorphism and variably prominent nucleoli. (C) Occasional nuclei having large prominent eosinophilic nucleoli with peri-nucleolar halos, which were only focally present. (D) Loss of FH. (E) Small uniform nests and tubules separated by thin vasculature. Cells had eosinophilic cytoplasm with round nuclei, lacking prominent nucleoli (inset). (F) Loss of FH.
FH-deficient RCC. A to D represent one tumor and E to F another tumor. (A) Lobules of tubules with eosinophilic secretions separated by fibrous tissue. (B) Tubules lined by atrophic cells with abundant eosinophilic cytoplasm. (C) Solid areas with variable-sized nuclei (left panel) lacking prominent nucleoli. Other areas (right panel) had individual cells in fibrotic stroma, some with more prominent nucleoli, but not the large eosinophilic nucleoli characteristic of FH-deficient RCC. Although there was slight spindling of tumor cells, no definitive sarcomatoid features were present. (D) Loss of FH. (E) Small poorly-formed nests with abundant cytoplasm. Nuclei were moderately pleomorphic with occasional prominent nucleoli, yet without peri-nucleolar halos (inset). (F) Loss of FH.
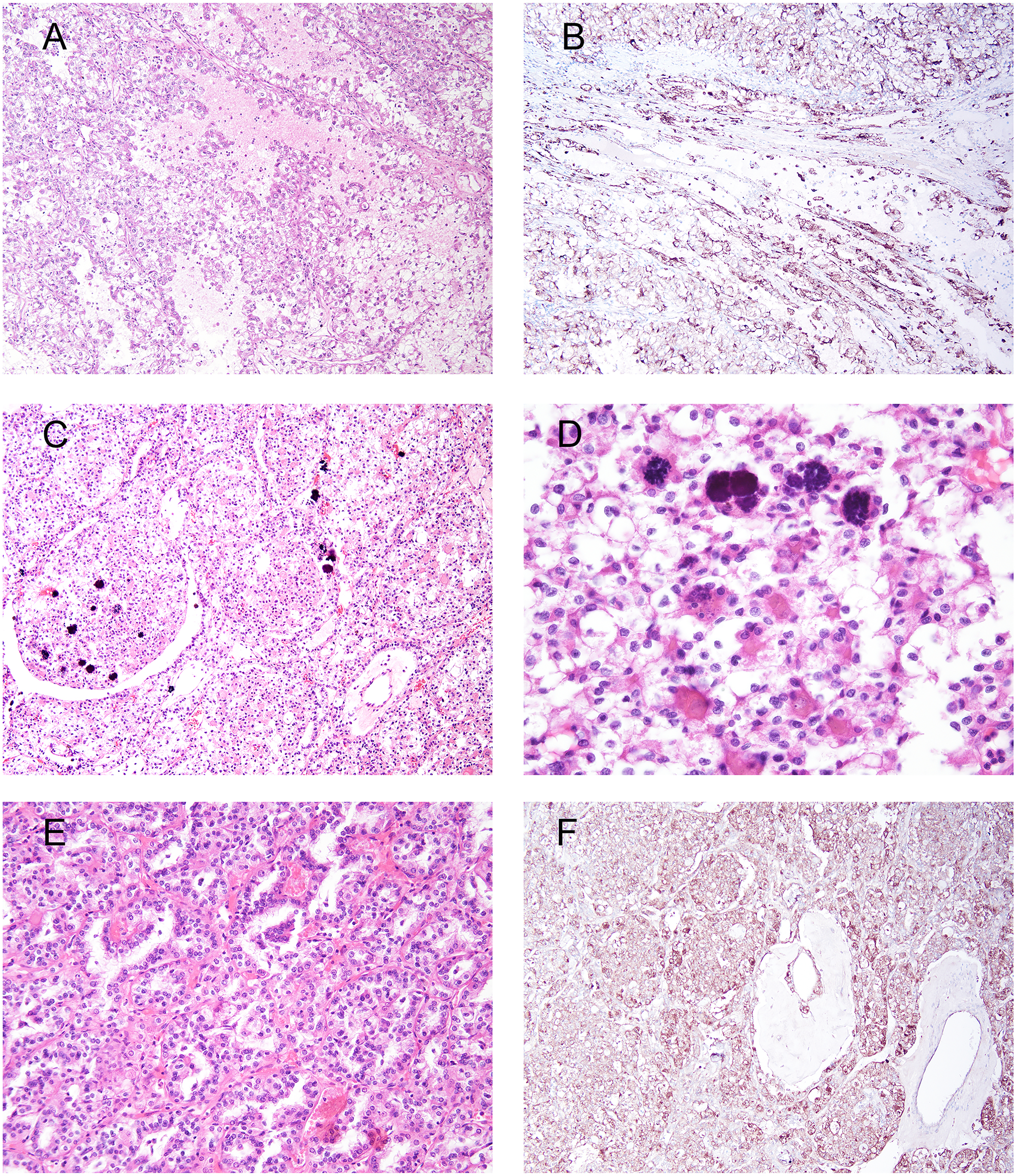
MiT family translocation associated RCC. A to B represent one tumor and C to F another tumor. (A) FISH proven TFE3 rearrangement composed of tubulopapillary architecture lined by clear cells. Tumor was positive for cathepsin K and negative for CA9. (B) Diffuse cathepsin K. (C) FISH proven TFE3 rearrangement composed of large nests of cells with a mixture of cells with eosinophilic and clear cytoplasm and scattered calcifications. (D) Calcified hyaline stromal nodules and relatively low-grade nuclei. (E) Other areas displaying tubulopapillary architecture. (F) Diffuse cathepsin K. CA9 was negative (not shown).
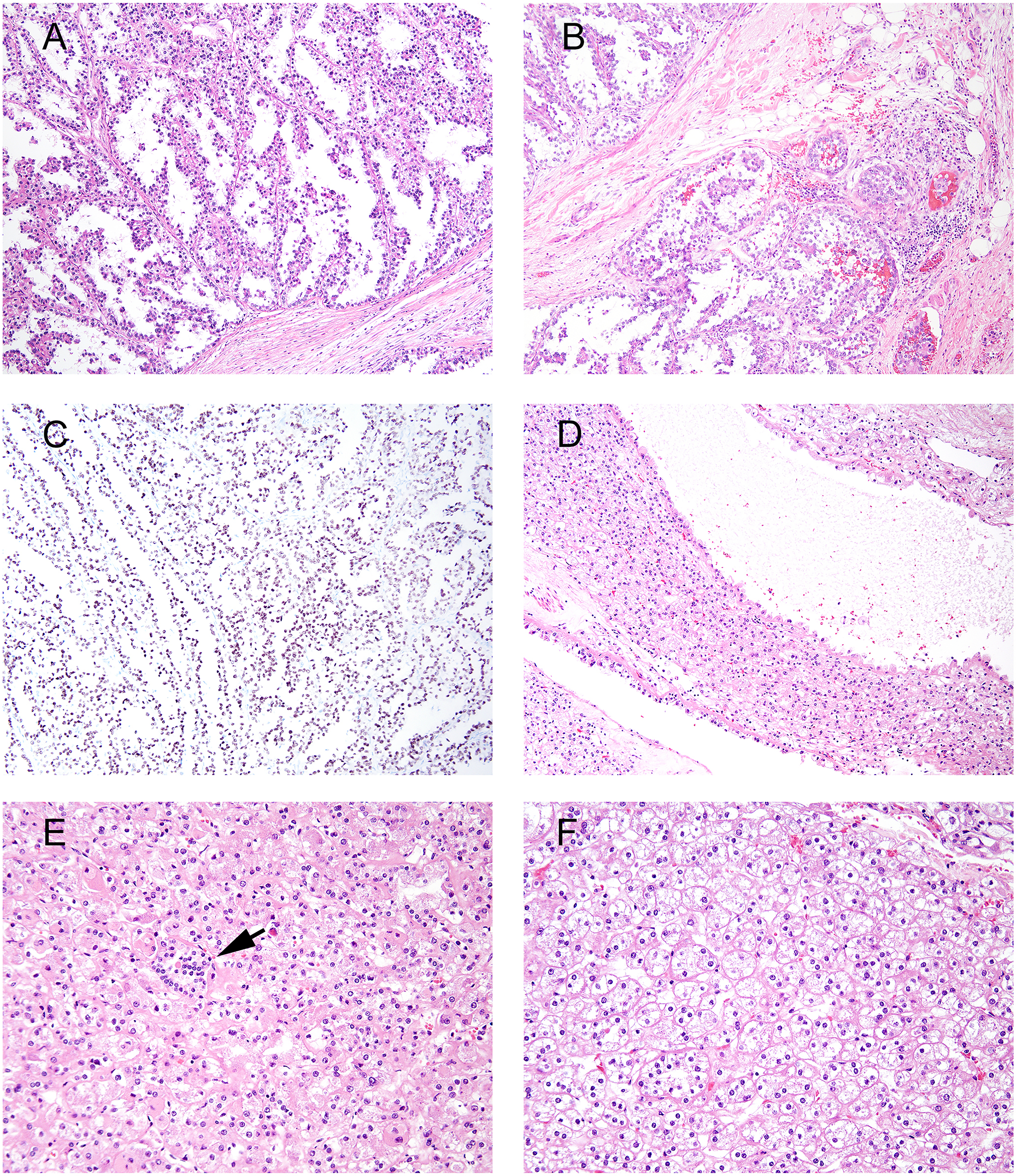
MiT family translocation associated RCC. A to C represent one tumor and D-F another tumor. (A) FISH proven TFE3 rearrangement composed of tubulopapillary architecture lined by clear cells. Tumor was positive for cathepsin K and negative for CA9. (B) Tumor extending into renal hilar adipose tissue. (C) Diffuse staining for TFE3 (performed at the outside institution). Tumor was negative for cathepsin K and CA9 (not shown). (D) FISH proven TFEB rearrangement composed of solid and cystic tumor. (E) Solid areas and ill-defined nests with cells having eosinophilic cytoplasm. A small cluster of cells with scant cytoplasm is present suggestive of a biphasic tumor population (arrow). (F) Nests and tubules composed of low nucleolar grade cells with lightly eosinophilic cytoplasm.
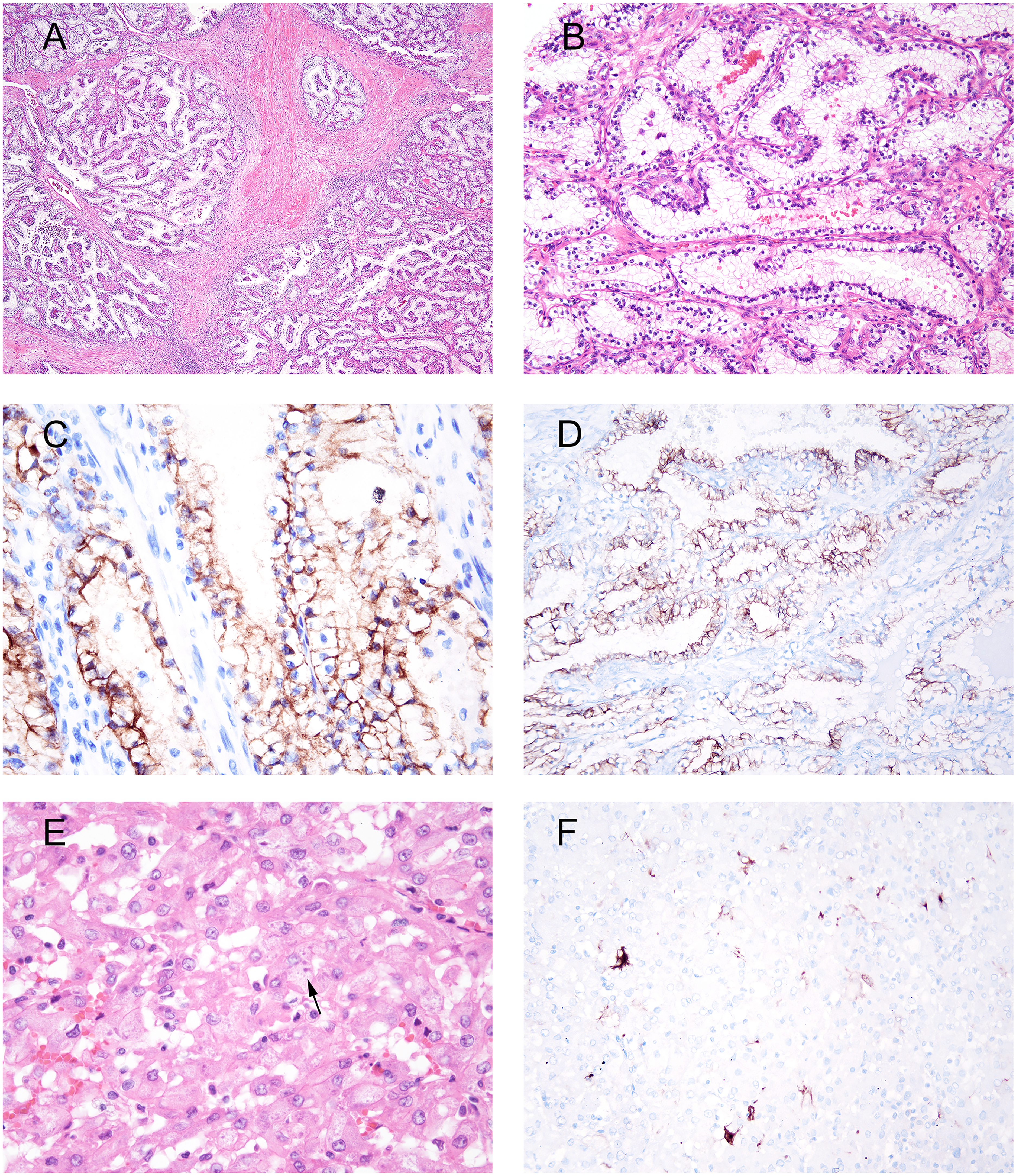
RCC with fibromyomatous stroma and eosinophilic solid and cystic RCC (ESC RCC). A to C represent one tumor and E to F another tumor. (A) RCC with fibromyomatous stroma. (B) Tubulopapillary architecture lined by cells with clear cytoplasm, areas of reverse polarity, and low-grade cytology. (C) CA9 shows predominantly circumferential labeling. (D) Diffuse keratin 7 immunoreactivity. (E) ESC RCC with solid pattern with occasional cells having cytoplasmic amphophilic stippling (arrow). (F) Patchy staining for keratin 20.
It was difficult comparing the 13 patients aged ≤30 years of age to the older cohort of 31 to 40 years old due to the small number patients in the former younger cohort (Table 2). Nonetheless, in both groups clear cell RCC was the predominant variant seen in 26 of 46 (56%) and 6 of 13 (46%) of patients 31 to 40 years old and 1 to 30 years old, respectively. Of note, MiT family translocation-associated RCC was also similar between the 2 age groups, 4 of 46 (9%) and 1 of 13 (8%) of patients 31 to 40 years old and 1 of 30 years old, respectively. The mean and median tumor size for all tumors were 9.2 and 9.0 cm, respectively, with a range from 2 to 22 cm.
Histological classification of renal cell carcinoma in Peruvian patients stratified by age subgroups.
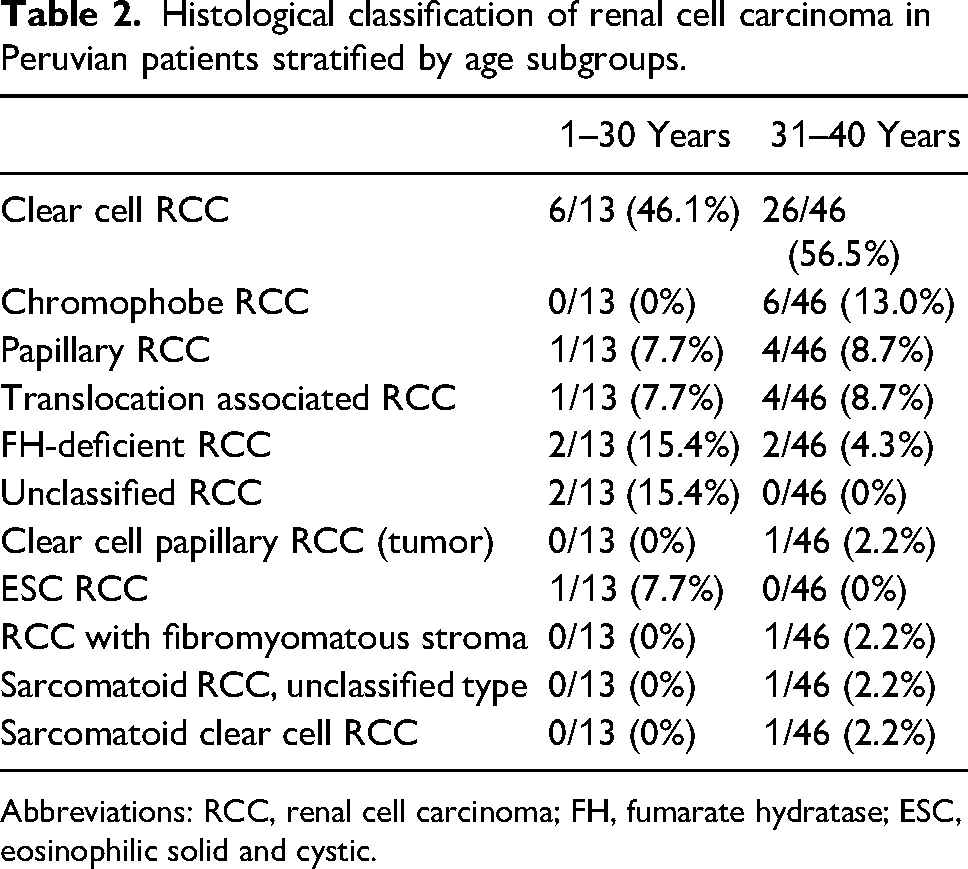
Abbreviations: RCC, renal cell carcinoma; FH, fumarate hydratase; ESC, eosinophilic solid and cystic.
There were 41 tumors without recurrence or metastases with follow-ups <1 year in 13, 1 to 2 years in 9, 2 to 3 years in 4, 3 to 4 years in 3, 4 to 5 years in 1, and ≥5 years in 11. Three tumors locally recurred only. Eight tumors were associated with only metastases, and 7 tumors had both local recurrence and metastases. Follow-up data was available for 9 of 15 patients with metastatic disease and 8 of 14 (57%) were reported dead in less than 3 years. Of the 15 patients with metastatic disease, 14 had pT3a or pT4 disease at presentation. Seven of 8 of the patients with pN1 disease had metastatic disease to a distant site. Lungs were the most common metastatic site. Metastases were seen in Grades 3 to 4 clear cell RCC (n = 6), Grade 3 papillary RCC (n = 3), and single tumors each of TFE3 translocation associated RCC, RCC with fibromyomatous stroma, FH deficient RCC, ESC RCC, sarcomatoid RCC, and unclassified RCC.
Discussion
Whereas the majority of studies on RCC in young adults and children have analyzed patients from North America and Europe, very little is found in the literature about RCC from younger patients in South America. The literature on younger patients with RCC is difficult to analyze and compare to the current series for several reasons. Several of the series have had relatively few tumors, which is understanding given the rarity of RCC in younger individuals. Another confounding factor has been variable inclusion age-based criteria for “young patients” with RCC. Some of the cut-offs have been <18 years old, 5 ≤23 years old, 6 ≤25 years old, 7 and ≤35 years old. 2 In the current study, we used ≤40 years of age to expand the number in the series, with the recognition that we could do subset analyses for younger cohorts and compare them to older patients (ie, 31-40 years old). As noted in the Results, the breakdown in RCC subtypes was for the most part similar between patients 1 to 30 and 31 to 40 years of age. The one exception that could not be explained by the smaller numbers of patients in the younger-aged cohort was the absence of chromophobe RCC in patients 1 to 30 years old compared to a 13.0% incidence in the 31 to 40 year age group. Another potential difference is that there were 2 sarcomatoid carcinomas in patients 31 to 40 years old, yet none in the younger patients. One potential rationale for narrowing down the age group for “young patients” with RCC is to focus on tumors with a predilection for younger individuals. However, in the current study, the incidence of MiT family translocation-associated RCC was the same in patients 31 to 40 years old, compared to those aged 1 to 30 years. Based on our data, it therefore seems reasonable evaluating RCC in individuals ≤40 years of age as a population of “young” patients with RCC.
It is difficult comparing data in our study from Peru to studies from North America or Europe for various reasons. Van Der Baek performed a review in 2020 of children with RCC including only studies with ≥10 patients aged ≤25 years at diagnosis. 8 Seven of these studies used the WHO 2004 classification system, with the remaining 9 studies reporting pathology that predated the use of the WHO 2004 classification.
One of the 7 studies and the largest contemporary study on young patients with RCC is from the Children's Oncology Group (COG) protocol AREN0532 including 212 RCCs from patients <30 years old from the United States, Canada, and Australia. 9 This protocol is primarily a study on the treatment for very low and standard risk favorable histology Wilms tumor. In this study, 41.5% were MiT family translocation-associated RCC, 16.5% papillary RCC, 12.3% renal medullary RCC, 6.6% chromophobe RCC, 4.2% tuberous sclerosis-associated RCC, 3.8% anaplastic lymphoma kinase (ALK)-rearranged RCC, and only 3.3% clear cell RCC. This study suffers from selection bias as straightforward tumors in young patients, such as clear cell RCC, might not be sent to COG for a second opinion compared to tumors with more unusual morphology. In addition, only tumors with “a histologic appearance consistent with the diagnosis of MiT-RCC” were studied by IHC, FISH, or NGS to confirm the diagnosis.
One of the 7 studies included in the van der Beek review by Kim et al was a retrospective review without any pathologist as an author. 10 The remaining 5 studies were predominantly pathology focused yet are still difficult to compare to the current study for 2 reasons. First, their use of the 2004 WHO classification predated the acceptance of more currently accepted RCCs that were evaluated for in our study, including SDHB and FH-deficient RCCs, RCC with fibromyomatous stroma, and ESC RCC.
Secondly, all 5 of these studies used only TFE3 immunohistochemistry to confirm the diagnosis of MiT family translocation-associated RCC. The incidences of MiT family translocation-associated RCCs in these studies were 19 of 46 (41.3%), 11 8 of 11 (72.7%), 12 6 of 13 (46.2%), 6 11 of 26 (42.3%), 13 and 7 of 13 (53.8%), 14 respectively. In all of these studies, MiT family translocation-associated RCCs were more common than clear cell RCC. However, in many laboratories TFE3 immunohistochemistry lacks specificity for MiT family translocation-associated RCC due to both technical and interpretation issues. In a large study performed in conjunction with the Mayo Clinic and PhenoPath laboratories, 2 methods of TFE3 immunohistochemistry were compared analyzing 98 tumors without known TFE3 rearrangements. 15 The overall specificity of TFE3 immunohistochemistry for TFE3-rearranged neoplasms was 57% (56/98) at Laboratory A and 95% (93/98) at Laboratory B. The overall sensitivity of TFE3 immunohistochemistry for 27 proven TFE3 rearranged tumors was 85% (23/27) at Laboratory A and 70% (19/27) at Laboratory B. In a survey of genitourinary pathologists, strong (3+) or diffuse (>75% of tumor cells) nuclear TFE3 immunohistochemistry expression was considered diagnostic by only 13 of 46 (28%) and 12 of 47 (26%) participants, respectively 16 though it was found to be a reliable surrogate in another recent study. 17 Problems with TFE3 immunohistochemistry cited were the low specificity, unreliable staining performance, and background staining. There are also different criteria as to what constitutes a positive TFE3 immunohistochemistry stain. In contrast to the more stringent criteria noted above in the genitourinary pathologist survey, other authors have considered TFE3-positive tumors with as little as 5–10% staining of any intensity.18,19 It has been shown that use of automated stainers which are common to most laboratories decreases the specificity of TFE3 immunohistochemistry relative to the originally validated overnight incubation technique.20,21 TFE3 immunohistochemistry is often best used as a supplement to TFE3 break apart FISH, as it is particularly helpful in identifying TFE3 rearranged tumors resulting from subtle chromosome inversions, such as the RBM10::TFE3 fusion. 22
Another large international study not summarized in the van der Beek review studied pediatric patients (0 to 18 years), diagnosed with histologically proven RCC registered in the International Society of Paedriatic Oncology (SIOP) (1993-2001), 2001 (2001–2018) and UK-IMPORT (2012–2019) databases. 7 These clinical trials and studies were primarily focused on Wilms’ tumor patients but allowed prospective registration of pediatric RCCs. Histology was, where feasible, classified according to the WHO 2004 classification. In 65 of 122 (53.3%) patients, MiT-RCC status was assessed, predominantly by TFE3-immunohistochemistry. The diagnoses were MIT-RCC in 36 of 64 (56.3%), papillary RCC in 19 of 65 (29.7%), clear cell RCC in 4 of 64 (6.3%), and other RCC subtypes in 5 of 64 (7.8%). One patient's histological subtype was not reported. This study, similar to the COG study cited above, suffers from selection bias as straightforward tumors in young patients, such as clear cell RCC, might not be sent to these oncology groups for a second opinion compared to tumors with more unusual morphology. Another difference in this study compared to ours is the lack of analysis by FISH for TFE3 and TFEB rearrangements, as well as using the WHO 2004 classification system which would not include newer variants of RCC.
The most recent work was a French–Italian series of 93 RCCs collected from 1990 to 2019 in patients aged less than 18 years was reclassified according to the 2016 World Health Organization (WHO) classification and the latest literature. 23 The median age at diagnosis was 11 years (range = 9 months-17 years). The distribution of cases were MITF-translocation RCC (52%), papillary RCC, high grade (11%), papillary RCC, type 1 (10%), clear cell RCC (4%), chromophobe RCC (3%), ALK translocation RCC 2%), and 1% each of collecting duct carcinoma, medullary RCC, SDHB-deficient RCC, FH-deficient RCC. The remaining 14% were unclassified. FISH analyses were performed on selected areas of whole sections of all cases. Immunohistochemical expression of TFE3 was present in all TFE3 rearranged cases and in 15 of 52 (28%) of TFE3 not rearranged RCCs (sensitivity 100%, specificity 72% for TFE3::TRCC). The higher proportion of cases with MITF-translocation RCC compared to our current series can be attributed to the much younger age of the French–Italian series.
The only comparable study to ours is the one published by Abdulfatah in 2020 from the University of Michigan, analyzing 68 RCCs in patients 30 years or younger seen over a 33-year interval. 2 This study reevaluated the tumors using the WHO 2016 classification system including work-up for SDHB-deficient RCC, FH-deficient RCC, and RCC with fibromyomatous stroma. FISH for TFE3 and/or TFEB gene rearrangements was performed only from tumors that were morphologically suspicious for MiT family translocation RCC. This work-up for MiT family translocation-associated RCC differed from the current study, where all tumors that remained unclassified based on a combination of H&E morphology and immunohistochemistry had FISH to assess for TFE3 or TFEB rearrangements, as some MiT family translocation-associated RCCs do not have histological features suggestive of the entity. In the University of Michigan study, 53% were clear cell RCC (our study 54%), 13% MiT family translocation associated RCC (our study 9%), 8% papillary RCC (our study 9%), 4% chromophobe RCC compared to our study (our study 10%). One difference between our study of Peruvian patients and the one from U. Michigan is that we had a lower percentage of unclassified RCC (3%) than U. Michigan (12%). A possible explanation is that we performed immunohistochemistry for FH on all unclassified tumors even if the morphology was not suggestive of an FH-deficient RCC, leading to the diagnosis of FH-deficient RCC in our 4 tumors which lacked the diffuse large prominent eosinophilic nucleoli surrounded by halos typical of this variant. We currently perform immunohistochemistry for 2SC in tumors with highly suggestive morphology of FH-deficient RCC and retained FH immunohistochemistry to identify the uncommon tumor with dysfunctional FH protein. As none of the tumors in the current study had such morphology, immunohistochemistry for 2SC was not performed in the current series. In contrast, in the U. Michigan study it states: “The renal tumours were placed into their appropriate diagnostic categories using appropriate immunohistochemical stains as and when necessary.” This difference would also account for our 5.1% incidence of FH-deficient RCC compared to no such tumors seen in the U. Michigan study. With the exception of renal medullary carcinoma which would not be expected to be seen in the Peruvian population who lack sickle cell trait (1% of young patients in the U. Michigan study), the distribution of RCC in both the Peruvian and U.S. population appear to be relatively similar.
Although rare, RCC in young adults typically spans from adolescence and teenagers into early adulthood. In contrast to adults where most RCCs are currently incidentally detected by imaging studies performed for various reasons, the most common presenting symptoms in young patients are hematuria, abdominal/flank pain and abdominal/flank palpable mass. 8 In our study we found that 83% of the patients presented with symptoms such as hematuria, flank pain or weight loss. Another notable differences between adult and younger adult RCC include less advance stage of presentation with less aggressive behavior. 5 Reported factors that impact survival include age, tumor size, lymph node status, MiT-Rcc and pathologic stage. 8 An exception is with MiT family translocation-associated RCC, where young patients present at higher stage with early spread to lymph nodes, yet still a favorable clinical outcome. Overall tumor stage is the most important prognostic factor. 5 Of the 15 patients with metastatic disease, 14 had pT3a or pT4 disease at presentation. Metastases were seen in tumors where more aggressive behavior would be expected such as in Grades 3 to 4 clear cell RCC (n = 6), Grade 3 papillary RCC (n = 3), and sarcomatoid RCC. Other tumors with distant metastases were seen with RCC variants less uniformly associated with an adverse prognosis such as single examples each of TFE3 translocation-associated RCC, RCC with fibromyomatous stroma, FH-deficient RCC, ESC RCC, and unclassified RCC.
The current study demonstrates the importance of special studies in accurately classifying RCC in younger individuals. Fifteen of the 59 tumors were reclassified. Ten of 59 (16.9%) required special studies to arrive at the diagnosis of FH-deficient RCC, ESC RCC, and MiT family translocation-associated RCC. Subsequent to this study, we also currently utilize a newer immunohistochemical marker GPNMB which identifies TSC1/2, MTOR-associated, and translocation-associated neoplasms. 24 This study also highlighted the presence of more recently described entities that were not recognized in older studies including FH-deficient RCC, ESC RCC, and RCC with fibromyomatous stroma. 25 It also appears that with the exception of certain RCC variants that are specific to certain ethnic populations, the distribution of RCC subtypes in younger individuals is similar between 2 representative large institutions of the United States and Peru. Additional studies are needed from other South American countries to further address their distribution of RCC subtypes. Some countries in South American may have unique populations, such as a higher proportion of those with an African background in Brazil with an increased prevalence of sickle cell trait and renal medullary carcinoma. 26 A potential weakness of the current study is that we analyzed only one large hospital in Peru. However, National Institute of Neoplastic Diseases Peru's major national referral center for cancer. The only way to broaden the comparison would be to compare national databases, which as the current study demonstrated would likely not be as accurate given the specialized expertise and studies needed to correctly classify RCC in the younger aged population.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
