Abstract
Introduction. Recently, an increased risk of celiac disease or eosinophilic esophagitis has been postulated among patients with either of these disorders, prompting some to suggest a common underlying mechanism, whereas others maintain that their co-existence is coincidental. Methods. We compared clinical and pathological features of 29 patients meeting criteria for both celiac disease and eosinophilic esophagitis to 26 celiac disease and 26 eosinophilic esophagitis controls to determine whether any distinguished study patients from controls. Results. Eight (28%) study patients presented with symptoms of both celiac disease and eosinophilic esophagitis, whereas 14 (48%) had celiac disease symptoms only and 5 had (17%) esophageal symptoms only. Study patients had similar autoimmune and atopic conditions seen in both control groups. Histological severity of disease, including Marsh II-III duodenal histology (study specimens: 87%; controls: 89%), mean peak esophageal eosinophil counts (study specimens: 55/400x field; controls: 80/400X field, P = .1), and presence of eosinophil microabscesses, scale crust, and subepithelial fibrosis were also similar to controls. Gluten-free diet resolved celiac disease-related symptoms (19 of 20, 95%) and histology (10 of 12, 83%), but not esophageal symptoms or eosinophilia in most study patients. Conclusion. Patients with concomitant celiac disease and eosinophilic esophagitis lack distinguishing features compared to controls with celiac disease or eosinophilic esophagitis alone. The occurrence of both disorders is likely coincidental in most cases.
Introduction
Celiac disease and eosinophilic esophagitis are usually considered distinct disorders of the upper gastrointestinal tract. Celiac disease is an aberrant Th1 cell-mediated response to dietary gluten in genetically susceptible individuals. Patients with celiac disease usually harbor HLADQ2 and DQ8 alleles and may have other autoimmune disorders, such as type I diabetes mellitus and autoimmune thyroid disease.1, 2 Celiac disease shows a predilection for females and can develop at any age. It has a worldwide prevalence of approximately 0.7% to 1% and is more common in the Northern hemisphere. The diagnosis is based on detection of immunoglobulin A anti-tissue transglutaminase (TTG) and/or anti-endomysial antibodies, and characteristic histology in duodenal biopsy samples. 3
Eosinophilic esophagitis is a Th2-mediated allergic reaction to a variety of inhaled or ingested antigens. Cytokines released from activated T cells recruit eosinophils to the esophageal mucosa. The disorder shows a predilection for young males; patients often have other atopic conditions, such as asthma or eczema. 4 The genetic basis is incompletely characterized, but a single nucleotide polymorphism in CCL26, which encodes eotaxin-3 is associated with disease susceptibility. 5 Expression of several candidate genes (TSLP, CAPN14, EMSY, LLRC32, STAT6, ANKRD27) have been associated with disease development. 6 Eosinophilic esophagitis is diagnosed in patients with at least 15 eosinophils per high power field in esophageal mucosa in whom other causes of esophageal eosinophilia have been excluded. 4
Several recent studies note an increased prevalence of eosinophilic esophagitis and celiac disease among patients diagnosed with either disorder, particularly in the pediatric age group. By some estimates, approximately 3% to 10% of children with celiac disease have concomitant eosinophilic esophagitis.7–11 Risk of eosinophilic esophagitis in children with celiac disease was increased by 50-fold, whereas risk of celiac disease in children with eosinophilic esophagitis was increased by 75-fold in 1 Canadian study. 12 Two studies that included adults reported a 16-fold increase in eosinophilic esophagitis and 26% higher odds of eosinophilic esophagitis in patients with celiac disease, respectively.13,14 Other large cohorts fail to support any association.15,16 We have also noted an increase in biopsy series in our practices that show features of both of these disorders. In this study, we correlated clinical and pathological features of a multi-institutional cohort of patients with both celiac disease and eosinophilic esophagitis in order to determine whether any clinical or pathological features distinguish these patients from those with isolated celiac disease or eosinophilic esophagitis.
Materials and Methods
This study was approved by the institutional review boards of the participating institutions.
Case Selection
Study patients: We retrospectively searched the pathology databases of 5 tertiary care centers over a 12-year period for cases meeting the following criteria: (1) both duodenal and esophageal biopsies were performed, (2) duodenal biopsies showed at least a Marsh I lesion as defined below (see Histological Review section), (3) at least 1 esophageal biopsy specimen contained ≥15 eosinophils in a 400x field, (4) 1 or more serological assays for celiac disease (anti-TTG, deamidated gliadin peptide, or endomysial antibodies) were positive. Patients without biopsy samples from either site, with slides unavailable for review, with history of other enteropathy or eosinophil-rich disorder, or lacking serological studies were excluded. Patients with Helicobacter pylori-associated gastritis were excluded as this might have accounted for clinical presentation; however, H. pylori-negative chronic inactive gastritis was not an exclusion criterion.
Controls: Consecutive unselected control cases were culled from the archives of 1 participating site (Montefiore Medical Center). All controls cases had biopsy samples taken from both the duodenum and esophagus. Celiac disease controls showed at least a Marsh I lesion, at least 1 positive celiac disease serological assay, and normal esophageal histology. They did not report esophageal symptoms. Eosinophilic esophagitis controls had at least 1 esophageal biopsy specimen containing ≥15 eosinophils in a 400x field and normal duodenal histology. These patients did not have diarrheal symptoms.
Clinical Data
Clinical data were extracted from the electronic medical records to include patient age and sex, history of atopic or autoimmune disorders, presenting symptoms, serological studies, type of treatment, and treatment response. For the purposes of the study, diarrhea, abdominal pain, and bloating were considered symptoms of celiac disease, whereas dysphagia, odynophagia, regurgitation, chest pain, and heart burn were considered esophageal symptoms.
Histological Review
Slides from all patients were reviewed by pathologists with subspecialty training in gastrointestinal pathology at the participating sites. Esophageal biopsy specimens were evaluated for peak eosinophil count in a 400x field, distribution of eosinophils in the mucosa (basally oriented, evenly distributed, or superficially oriented), distribution of eosinophils in the esophagus (distal, middle, and proximal), and presence or absence of eosinophil microabscesses (clusters of at least 4 intact eosinophils), eosinophil scale crust, and subepithelial fibrosis when subepithelial tissue was sampled. The mean peak eosinophil count was defined as the cohort's average derived from the single highest eosinophil count for each patient in all specimens from that patient. Duodenal biopsies were evaluated for peak eosinophil count in a 400x field, and were graded according to the Marsh classification as follows: 0: normal; I: normal architecture with increased intraepithelial lymphocytes; II: normal villi with hyperplastic crypts and increased intraepithelial lymphocytes; III: blunted villi with hyperplastic crypts and increased intraepithelial lymphocytes; and IV: blunted villi with atrophic crypts and increased intraepithelial lymphocytes. 17 Increased intraepithelial lymphocytes were defined as >25 lymphocytes/100 enterocytes. 18
Statistical Analysis
Fisher's exact test was used to compare categorical variables. The student's t test was used to compare means. A P value <.05 was considered significant.
Results
Clinical Features of Study Patients and Controls
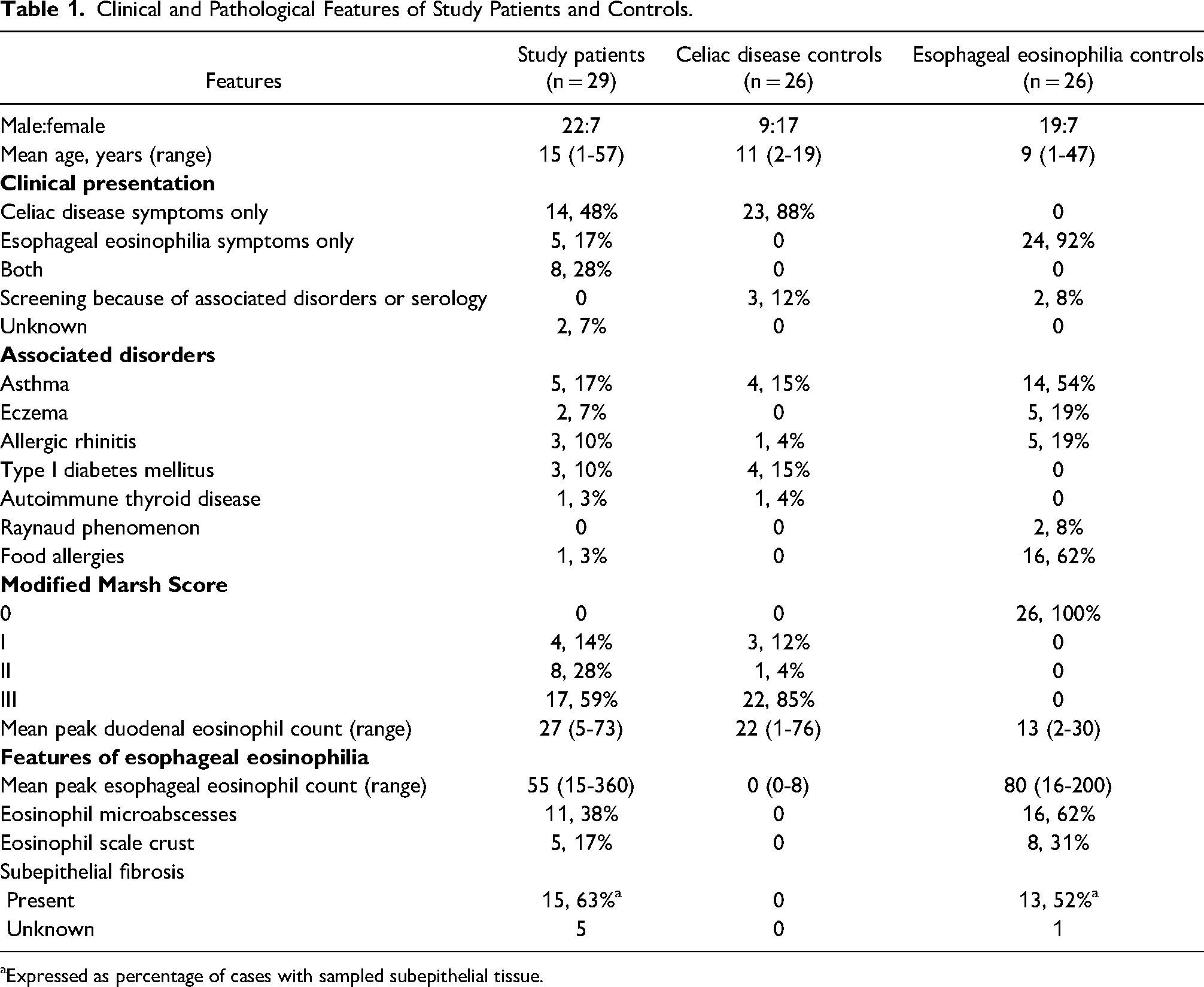
Clinical and histological features of the study patients and controls are summarized in Table 1. All but 1 of the study patients were diagnosed with celiac disease and eosinophilic esophagitis during the same upper endoscopic procedure. The study group included 22 males and 7 females with a mean age of 15 years. Twenty-three patients were ≤18 years old and the remaining 6 ranged from 20 to 57 years old. Five patients had history of 1 or more atopic disorders including asthma (n = 5), eczema (n = 2), or allergic rhinitis (n = 3). One patient had food allergies. Four patients had history of autoimmune disorders, including thyroid disease (n = 1) and type 1 diabetes mellitus (n = 3). One patient had asthma, allergic rhinitis, and type I diabetes mellitus. Twenty-two (76%) patients total experienced symptoms of celiac disease. Thirteen patients total (45%) experienced esophageal symptoms. Fourteen patients (48%) reported only celiac disease-associated symptoms and 5 (17%) reported only esophageal symptoms. Eight patients (28%) experienced symptoms of both disorders.
Clinical and Pathological Features of Study Patients and Controls.
aExpressed as percentage of cases with sampled subepithelial tissue.
Celiac disease controls included 9 males and 17 females with a mean age of 11 years. Twenty-three patients were ≤18 years old and the other 3 were 19 years old. Twenty-three (88%) patients had celiac disease-related symptoms, 1 had positive autoantibodies upon screening in the setting of type 1 diabetes mellitus, another had positive celiac disease antibody screening in the setting of Grave's disease, and another was a child evaluated for failure to thrive. Three additional patients had type I diabetes mellitus, and 4 had asthma, 2 of whom also had type I diabetes mellitus. One had allergic rhinitis.
Eosinophilic esophagitis controls included 19 males and 7 females with a mean age of 9 years. Twenty-four patients were ≤18 years old and the other 2 were 39 and 47. Twenty-four (92%) had esophageal symptoms and the other 2 underwent endoscopy for poor weight gain (n = 1) and melena (n = 1). Twenty-one patients had history of 1 or more atopic disorders, including asthma (n = 14), allergic rhinitis (n = 5), eczema (n = 5), and food allergies (n = 16). Two patients in this group had a history of Raynaud's disease.
The male:female ratio was similar in the study group and eosinophilic esophagitis controls (P = 1.00), but there were significantly more male study patients compared to celiac disease patients (P = .03). Study patients were not significantly older than either the celiac disease (P = .07) or eosinophilic esophagitis (P = .05) group. The type and distribution of underlying atopic and autoimmune disorders in study patients was more comparable to celiac disease controls than to eosinophilic esophagitis controls due to more frequent occurrence of atopic conditions in controls.
Histological Features of Study Specimens and Controls
Most (17 of 29, 59%) study patients presented with Marsh III histology and only 4 (14%) had a Marsh I lesion. Similarly, most celiac disease controls (22, 85%) had Marsh III histology and only 3 (12%) had a Marsh I lesion. The proportion of Marsh III lesions was significantly different (P = .04) due to occurrence of Marsh II histology in 8 (28%) study patients (Figure 1A). The mean peak eosinophil counts in duodenal mucosa were significantly higher in study specimens compared to eosinophilic esophagitis controls (P = .0008), but not compared to celiac disease controls (P = .3) in keeping with previously described increase in duodenal mucosal eosinophils in celiac disease (Figure 1B). 19 The mean peak esophageal eosinophil counts were higher in eosinophilic esophagitis controls (80/400x field, range: 16-200) compared to study specimens (55/400x field, range 15-360) (Figure 1C) but the difference was not significant (P = .1). Eight study patients had sampling of the distal esophagus only. Of the remainder, 19 had involvement of multiple levels of the esophagus, 1 involved the mid-esophagus only, and the site was unspecified in 1 case. Seven study patients had eosinophil counts near the established threshold for eosinophilic esophagitis (range: 15-20 per high power field); all but 3 of these had documented eosinophilia at the mid-esophagus or at multiple levels of the esophagus. All control specimens had involvement of multiple levels of the esophagus. Eosinophils were either luminally oriented or distributed throughout the mucosa in study (2 [7%], 26 [90%], respectively) and control (6 [23%], 20 [77%], respectively) specimens with basal distribution noted in 1 study specimen (3%). No statistically significant differences were found with regard to eosinophil microabscesses, scale crust, or subepithelial fibrosis (Table 1).
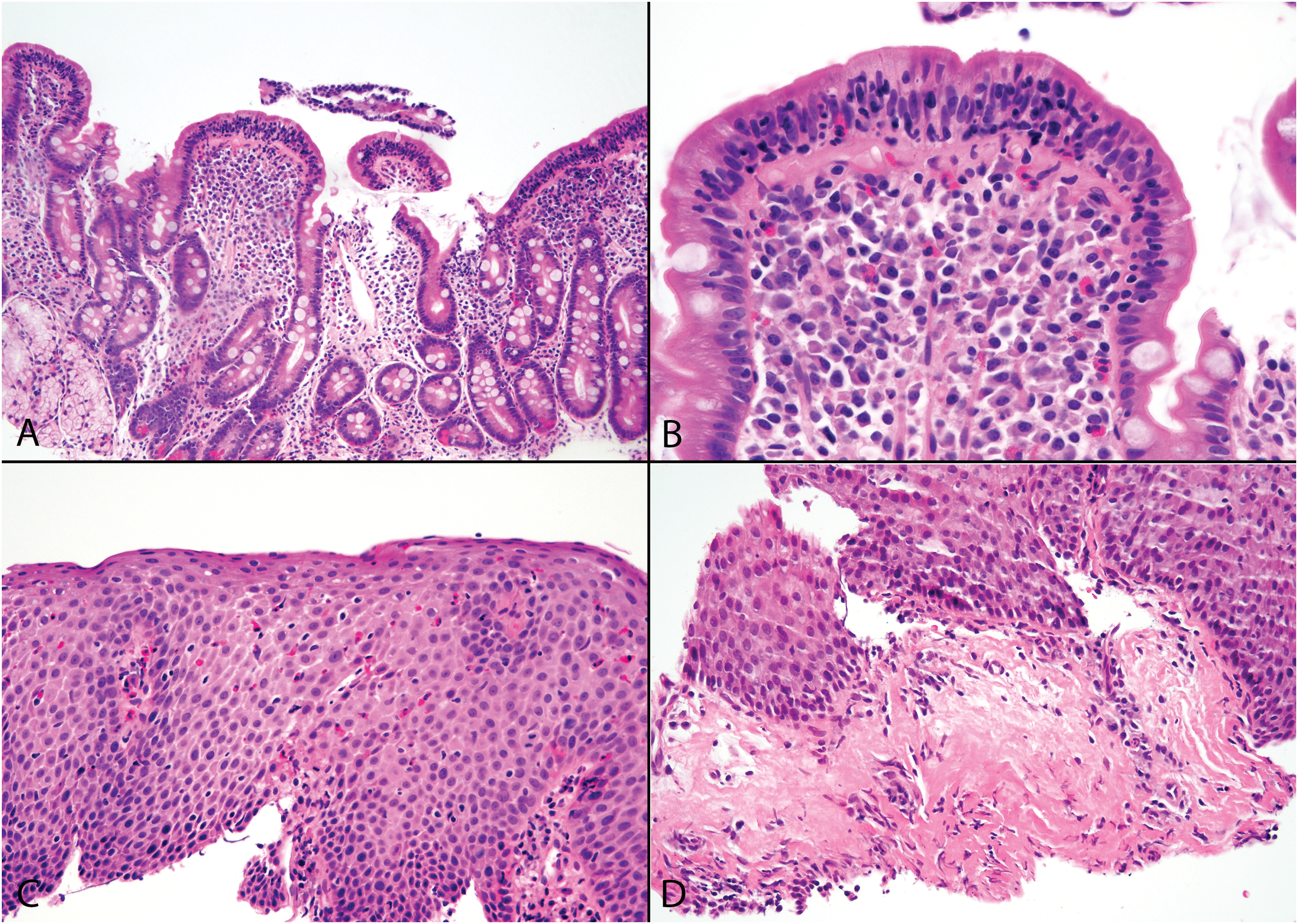
A study specimen displays partial villous blunting and crypt hyperplasia in the duodenum (A). Intraepithelial lymphocytes are increased and the lamina propria is expanded by mixed inflammation, including scattered eosinophils (B). The esophageal mucosa features numerous luminally oriented eosinophils (C). A comparable study specimen from a symptomatic patient showed subepithelial fibrosis (D) (hematoxylin and eosin; original magnifications: A 100X; B, D 400X; C 200X).
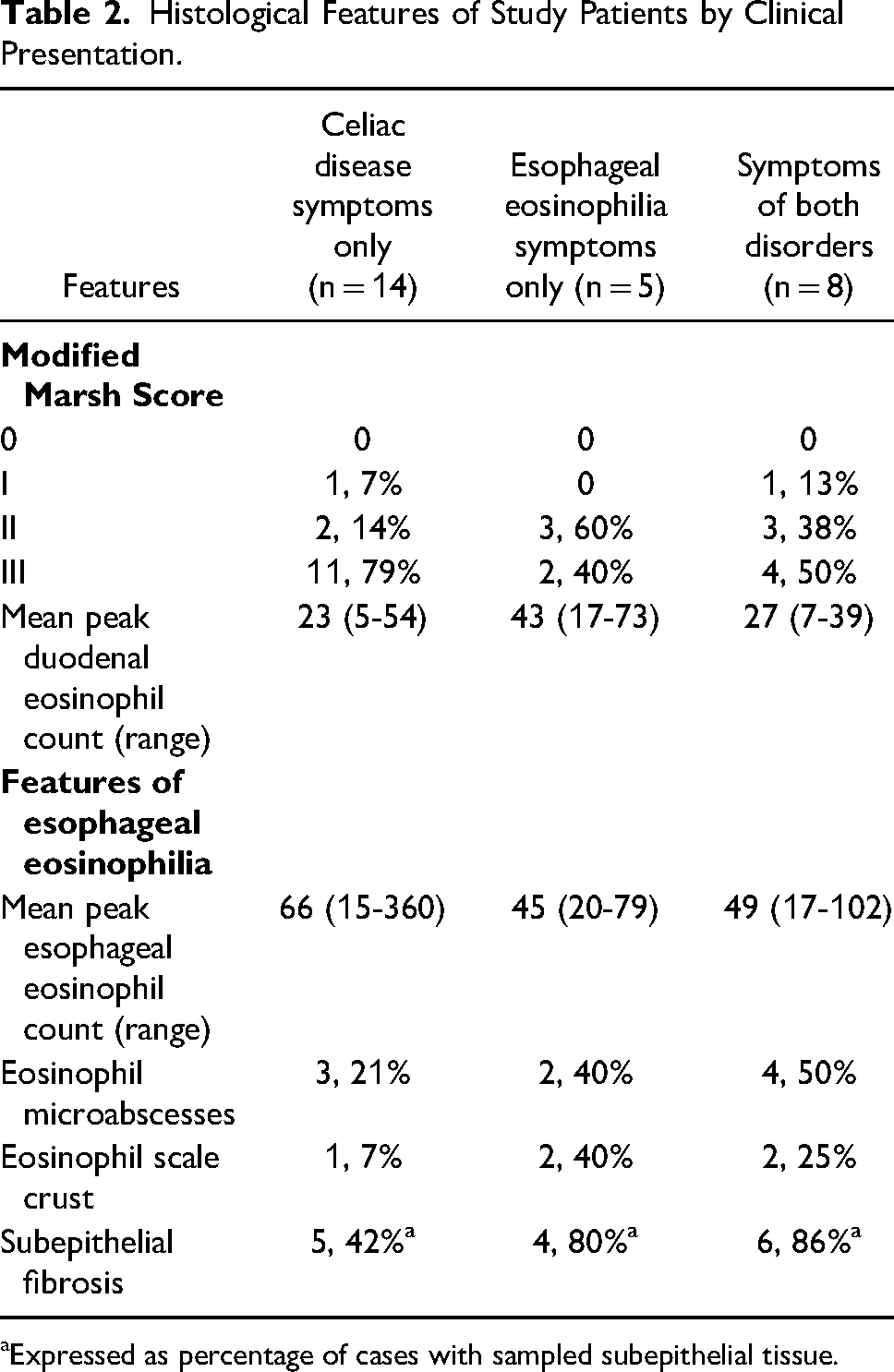
A comparison of histological features of the study patients by clinical presentation is presented in Table 2. Patients with symptoms of celiac disease alone, eosinophilic esophagitis alone, or both disorders had high proportions of Marsh II or III lesions (93%, 100%, 88%, respectively), but the patient numbers were too low for meaningful statistical comparison. Similarly, mean peak eosinophils counts were well above the minimum threshold for a diagnosis of eosinophilic esophagitis in all 3 of the aforementioned groups (66/400x field, 45/400x field, and 49/400x field, respectively). Subepithelial fibrosis (Figure 1D), eosinophil microabscesses, and eosinophil scale crust were more common in patients with esophageal symptoms compared to those with only celiac disease symptoms.
Histological Features of Study Patients by Clinical Presentation.
aExpressed as percentage of cases with sampled subepithelial tissue.
Pediatric Subgroup Analysis
Since most study patients were in the pediatric age group, we separately analyzed major clinical and histological features after exclusion of the 6 adult patients. Of 23 pediatric patients, 4 had history of atopic disorders and 4 had autoimmune diseases. Eighteen presented with celiac disease-associated symptoms, 9 with eosinophilic esophagitis-associated symptoms and 6 had symptoms of both disorders. This group included 3 patients with Marsh I, 5 with Marsh II, and 15 with Marsh III histology. The mean peak eosinophil count in this group was 63 per high power field.
Clinical and Histological Follow Up
Clinical follow-up information was available for 22 patients; 13 of these patients also had follow-up biopsies (Table 3). All were prescribed a gluten-free diet, but treatment for eosinophilic esophagitis was variable. Of 9 patients treated with gluten-free diet alone, all reported improvement in celiac disease-related symptoms. Only 4 originally reported esophageal symptoms, and 2 experienced improvement in eosinophilic esophagitis symptoms. Four patients treated with gluten-free diet alone had follow-up endoscopic biopsies; 3 patients had normalization of duodenal histology, but persistent eosinophilic esophagitis and 1 had histological resolution of both disorders.
Clinical Follow Up of Study Patients.

aNo duodenal biopsy was available for review.
Ten patients were treated with gluten-free diet and proton pump inhibitors. Four had improvement in both celiac disease and esophageal symptoms and 4 who presented with celiac disease symptoms only reported improvement. One initially reported only esophageal symptoms and also experienced symptomatic relief. The last patient in this group did not adhere to gluten-free diet and symptoms persisted; information regarding response to proton pump inhibitors was not available. Seven follow-up endoscopic procedures were performed in this group. Of these, 3 patients had improved (n = 1) or normal (n = 2) duodenal histology, but persistent eosinophilic esophagitis, 2 had resolution of both histological abnormalities, and 2 had persistent or more severe disease at both sites. Of the latter 2, 1 was a patient who reported nonadherence to gluten-free diet.
Three patients received inhaled steroids in addition to gluten-free diet, 2 of whom initially reported only celiac disease-associated symptoms and experienced improvement and the other of whom had improvement in both types of symptoms. Two had follow-up biopsies, 1 showed normal duodenal histology, but persistent eosinophilic esophagitis and the other had improved esophageal findings, but no duodenal biopsy was available.
Discussion
Since Th1-mediated and Th2-mediated disorders can share environmental triggers, some authors postulate that wheat may stimulate both celiac disease and eosinophilic esophagitis in some patients. 20 For example, gluten exposure may initiate an immune response in the small intestine that increases overall intestinal permeability exposing esophageal mucosa to allergens. 8 Their co-occurrence may also be coincidental given increasing worldwide incidences of both celiac disease and eosinophilic esophagitis.21–23 We report the largest series to date of patients with both celiac disease and eosinophilic esophagitis, including correlation of pretreatment and post-treatment histology with clinical features. Study patients did not have distinguishing clinical or histological features compared to controls. All but 2 required separate interventions for each disorder. Our results support the latter theory, namely that most cases are likely coincidental.
Overall, 76% of patients presented with celiac disease-related symptoms, and 45% of patients reported esophageal symptoms, including 14 (48%) who only had symptoms related to celiac disease and 5 (17%) who only had esophageal symptoms. Others have also noted that most patients have celiac disease-related symptoms whether or not esophageal symptoms are present.7–9 Only 1 series described 6 children diagnosed with eosinophilic esophagitis who were incidentally found to have concomitant celiac disease. 10 Our cohort included both scenarios, although incidental eosinophilic esophagitis was more common. We hypothesize that cases wherein patients reported only 1 type of symptom came to clinical attention after development of the first disorder and in a subclinical phase of the second; however, this does not necessarily imply a causal link between the 2. In our series gluten-free diet alone did not address esophageal symptoms, with the exception of 2 cases. Histologically, esophageal eosinophilia also persisted in 75% of patients treated with gluten-free diet alone. Most other groups have also found that eosinophilic esophagitis does not respond to dietary gluten elimination.8,9,13
To our knowledge, this is the first detailed histological analysis and comparison with both celiac disease and eosinophilic esophagitis control groups. We did not find histological features in either the esophagus or duodenum that consistently identify patients who might have combined celiac disease and eosinophilic esophagitis. Mean peak eosinophil count (55/400x field) and frequency of Marsh II-III histological lesions (87%) in study specimens were similar to controls and among groups of study patients, regardless of clinical presentation. Four prior series with histological data, totaling 25 cases, reported that all patients had Marsh II or III histology.10–12,15 Similar to our findings, previously reported peak eosinophil counts have ranged from 35 to 52/400x field without substantial variation in those with and without esophageal symptoms.9,10,15 In our study, eosinophil microabscesses, scale crust, and subepithelial fibrosis were more common among study patients with esophageal symptoms, not surprisingly.
Some weaknesses in this study are inherent in its retrospective nature. Data on treatment and response were not available for all patients, and not all underwent follow-up biopsy procedures. Although all slides were reviewed by pathologists with subspecialty training in gastrointestinal pathology, we did not perform a central review of the specimens. This being a multi-institutional study, we are not able to determine the denominator of all celiac disease and eosinophilic esophagitis patients evaluated in all of these institutions in order to determine the incidence of co-occurence of these diseases. Finally, our inclusion criteria required that all study patients and controls had biopsies of both the duodenum and esophagus; this increases the likelihood that some control cases had symptoms or subtle abnormalities at either site that were not documented in clinical notes. As such, potential differences between patients with celiac disease and eosinophilic esophagitis and those with only one of these disorders may be underrepresented.
Clinical and pathological correlation reveals that combined celiac disease and eosinophilic esophagitis is likely coincidental in most instances. These patients are a heterogeneous group who may present with symptoms usually associated with 1 or both of these disorders. Histological features in the esophagus and duodenum did not differ substantially from controls. Rare patients who respond to gluten-free diet alone may result from inter-related Th1 and Th2 responses to wheat, but specific therapy directed at celiac disease and eosinophilic esophagitis is necessary in most, implying independent mechanisms of disease development.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
