Abstract
Primary effusion lymphoma (PEL) is an aggressive neoplasm often diagnosed in immunosuppressed patients demonstrating peritoneal, pleural, or pericardial effusions. This high-grade lymphoma is strongly associated with human herpesvirus 8 (HHV8) infection and most of the lesions also show the presence of Epstein–Barr virus in tumor cells, which lacks CD20 expression and reveals a plasmablastic morphology and phenotype. The extracavitary or solid variant of PEL is even rarer and usually affects the lymph nodes and is currently considered a clinical manifestation of the classic PEL. In the oral cavity, extracavitary PEL is extremely rare and only a few patients have been previously reported, with no detailed clinicopathological description. The recognition of oral extracavitary PEL is even more important given the occurrence of plasmablastic lymphoma in the oral mucosa, which shares many clinical, microscopic, and phenotypic features with PEL, therefore, demanding from pathologists the search for HHV8, especially in immunosuppressed patients, and an appropriate clinical evaluation. In this report, we aim to describe a very rare extracavitary PEL affecting the palate of a 36-year-old patient and to review the literature regarding the extracavitary presentation of this aggressive lymphoma. This report demonstrates the importance of searching for HHV8 infection in oral lymphomas with plasmablastic features.
Introduction
Primary effusion lymphoma (PEL) is a high-grade neoplasm usually diagnosed in immunosuppressed patients that characteristically affects the body cavity manifesting as pleural, peritoneal, or pericardial lymphomatous effusions. 1 It was originally described in 1989 during the acquired immunodeficiency syndrome (AIDS) epidemic outburst and recognized by the World Health Organization in its third edition published in 2001.2–5 It accounts for approximately 4% to 8.5% of all AIDS-related lymphomas, and for only 0.3% of aggressive lymphomas in human immunodeficiency virus (HIV)-negative patients.4,6,7 In 1995 Cesarman et al 8 demonstrated the occurrence of human herpesvirus 8 (HHV8)/Kaposi's sarcoma herpesvirus deoxyribonucleic acid (DNA) sequence within the tumor cells and the presence of this virus in now considered mandatory for establishing PEL diagnosis. Moreover, Epstein–Barr virus (EBV) is also observed in the majority of the cases and it is believed to play some role in the pathogenesis of this lymphoma subtype. 9
It was later recognized by Chadburn et al 11 that PEL may manifest as solid masses out of body cavities in a varied proportion of the patients, 10 and the occurrence of this extracavitary presentation may follow or precede the occurrence of effusions, although patients exclusively showing solid tumors are well recognized. Extracavitary PEL may affect different anatomic regions and lymph nodes are the most frequently described, followed by the skin and the gastrointestinal tract.4,9
In the oral cavity, the occurrence of an extracavitary PEL represents an even more challenge in diagnosis because of its strong morphological and immunophenotypic resemblance with plasmablastic lymphomas that usually affect this location in immunosuppressed patients, since both lesions are characterized by large atypical plasmablasts, lack of B-cell markers, positivity to plasma cell proteins and association with EBV infection, 12 as originally described by Delecluse et al 13 in 1997. Therefore, oral pathologists must be aware of this rare lymphoma subtype to be able to appropriately recognize this entity. The aim of this manuscript is to report a very rare case of extracavitary PEL affecting the oral cavity.
Case Report
This report follows the Case Report (CARE) guidelines. It refers to a 36-year-old male patient that presented a crateriform ulcerative lesion of approximately 3 cm in diameter in the palate with 5 weeks of duration. The medical history of the patient was positive for Kaposi's sarcoma in the oral cavity, which was indicative of an immunosuppressive state, although the exact reason was unknown. This tumor was originally diagnosed in 1991 based on microscopic findings only and received a descriptive diagnosis of immunoblastic sarcoma, large-cell lymphoma with immunoblastic appearance, and plasmacytic features. It was sent to the immunohistochemical laboratory of our institution some years later to comprise one of the oral pathology studies developed in this center, but no further investigation was carried out thereafter. More recently, as part of our studies with oral lymphomas, this neoplasm was retrieved and a detailed microscopic and immunohistochemical analysis was done.
A new hematoxylin and eosin-stained slide was revised and showed the presence of a lymphoid tumor in the connective tissue and an overlying stratified epithelium showing areas of acanthosis and hyperkeratosis, consistent with the palate mucosa. The neoplastic cells exhibited a diffuse growth pattern cytologically showing a plasmablastic aspect characterized by the presence of large atypical neoplastic cells with displaced nuclei and one or more prominent nucleoli. Some cells had an immunoblastic appearance with a centrally located nucleolus, while few anaplastic tumor cells exhibiting a high degree of pleomorphism were also found. Tingible body macrophages phagocytizing apoptotic bodies/debris were present and gave rise to a starry sky pattern. Atypical mitotic figures were common and some areas of tissue necrosis could also be observed (Figure 1). The main diagnostic hypotheses included a diffuse large B-cell lymphoma, a plasmablastic lymphoma, and an anaplastic large-cell lymphoma, which should have been confirmed with additional laboratorial reactions.
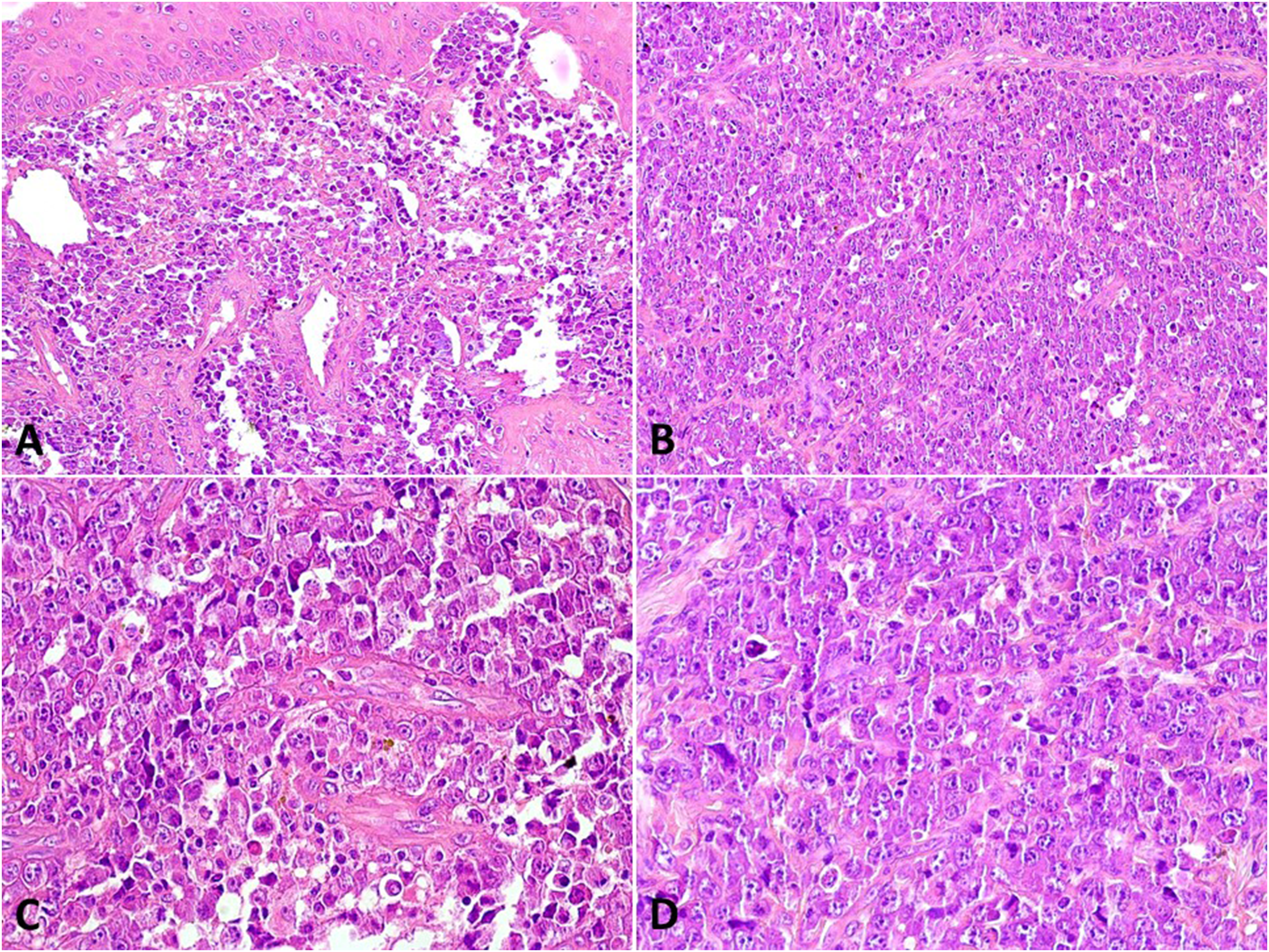
Microscopic features of extracavitary PEL. (A) Diffuse proliferation of neoplastic cells in the connective tissue of the hard palate mucosa. Observe the overlying epithelium of the oral mucosa (H&E: 100×). (B) Tumor cells were diffusely distributed with the presence of tingible body macrophages that led to the so-called starry sky pattern (H&E; 100×). (C) Atypical plasmablasts predominated in the sample (H&E; 200×), but (D) pleomorphic cells and immunoblasts were also found (H&E; 200×). Abbreviations: PEL, primary effusion lymphoma; H&E, hematoxylin and eosin.
A large panel of immunoreactions was done to characterize the tumor cells and we observed a focal staining for leucocyte common antigen and a strong staining for CD30, as well as a diffuse and strong reaction to CD138, MUM1, and epithelial membrane antigen, and a focal staining for plasma cell (VS38c) (Figure 2). Mature B cell markers like CD20 and CD79a were negative, with a strong aberrant expression of CD3. Other reactions against CD10, BCL2, and CD56 were negative, while CD43 was focally and weakly positive. Light chain monoclonality was observed for lambda protein. Proliferative index measured by Ki67 expression achieved approximately 70%. Given the patient's previous diagnosis of Kaposi's sarcoma, CD34 reaction was done, but only normal blood vessels showed reactivity and together with the lack of a spindle cell morphology, excluded the presence of a Kaposi's sarcoma together with the lymphoid neoplasm.
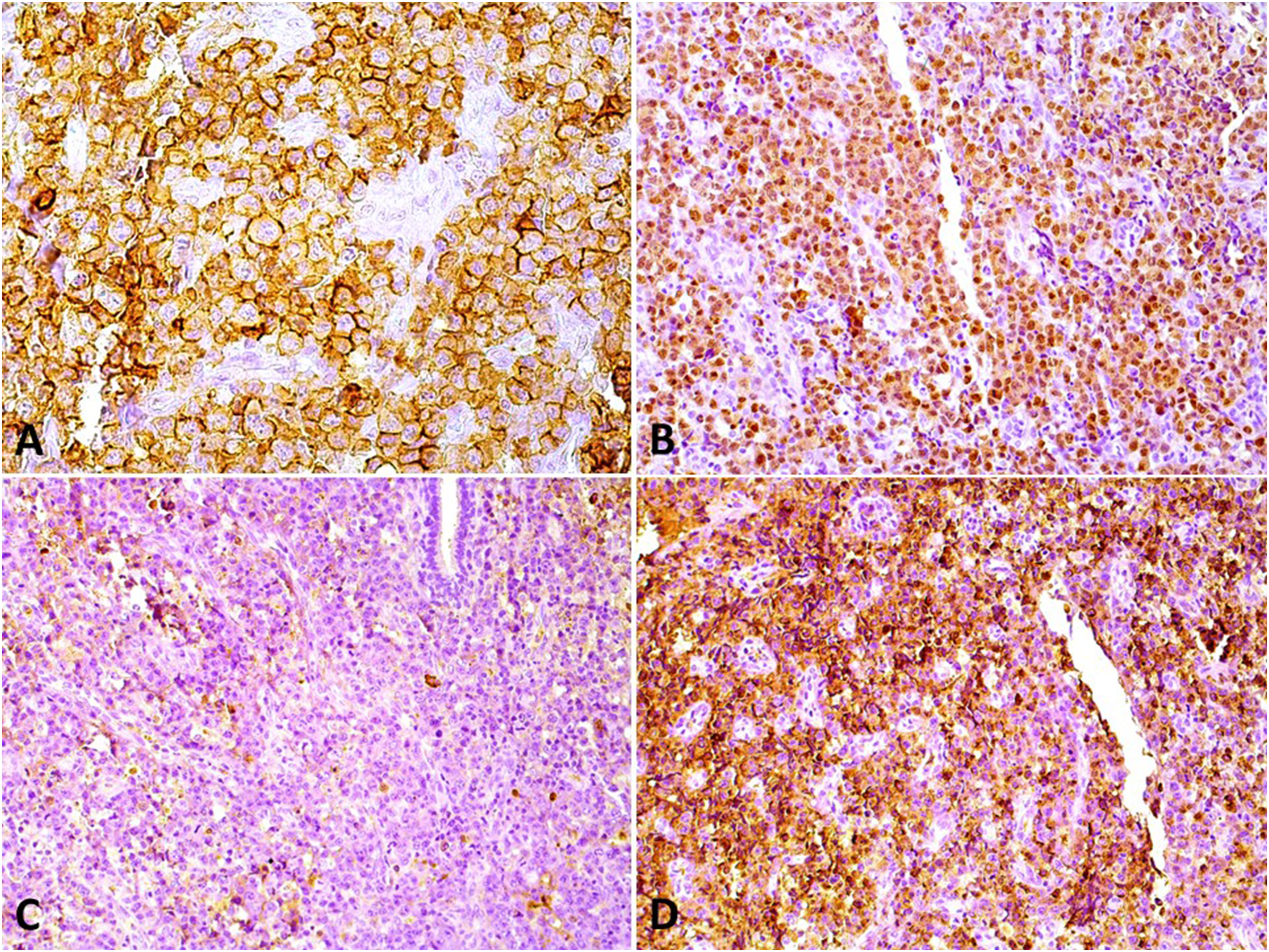
Immunohistochemical findings of the current extracavitary primary effusion lymphoma (PEL) showing the plasma cell phenotype of the neoplasm. (A) A strong and diffuse membrane expression of CD138 (DAB; 200×) and (B) a strong nuclear expression of MUM1 (DAB; 100×). (C) Tumor cells were negative for Kappa light chain (DAB; 100×) and (D) positive for lambda light chain (DAB; 100×).
This plasma cell immunophenotype associated with the plasmablastic morphology of the tumor cells, in an immunosuppressed patient, led to a provisional diagnosis of plasmablastic lymphoma, but reactions to identify EBV and HHV8 viruses were recommended. The immunoreaction to HHV8 using latency-associated nuclear antigen-1 clone was strongly and diffusely positive, while in situ hybridization for EBV (EBV-encoded small RNAs) also showed strong and diffuse positivity in the nuclei of tumor cells (Figure 3). Fluorescence in situ hybridization reactions using break-apart probes to investigate the mutational statuses of BCL2, BCL6, and MYC genes were negative for all of them. Therefore, a final diagnosis of an extracavitary PEL in the palate was done since the occurrence of cavitary effusions is not necessary to determine the diagnosis of the solid variant of this lymphoma. Unfortunately, we do not have information regarding the treatment and the follow-up of this patient.
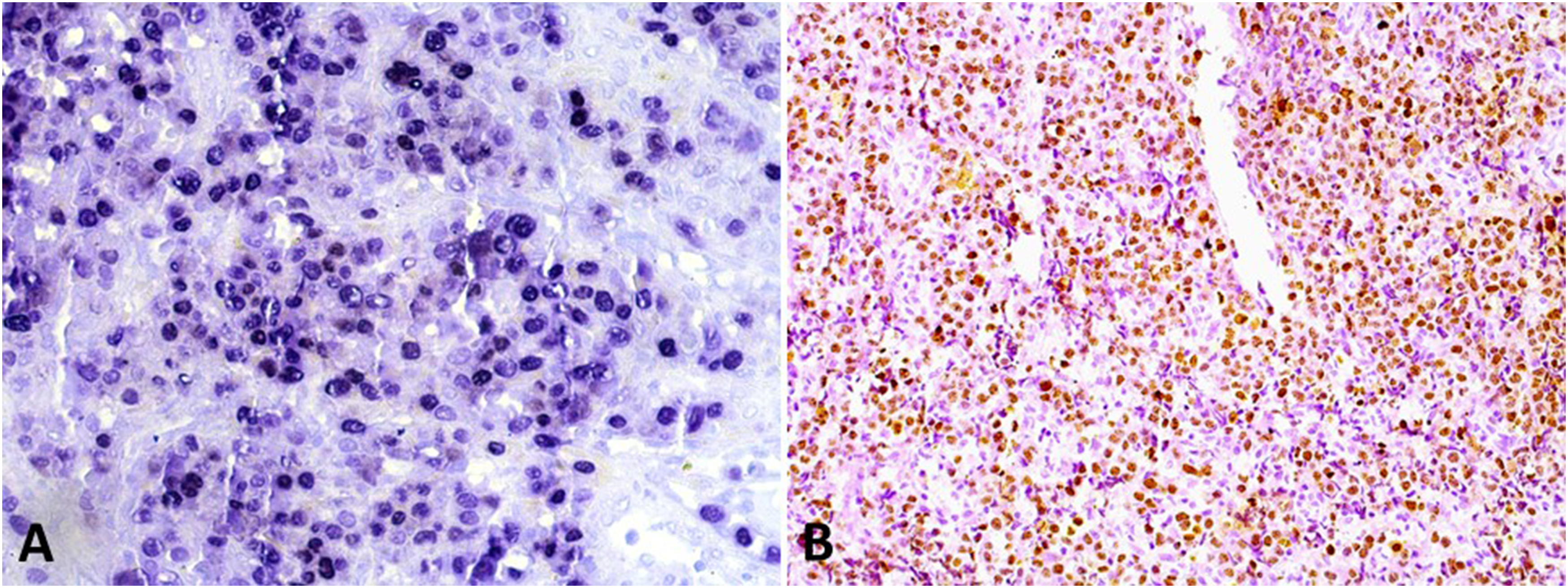
Presence of HHV8 and EBV in the extracavitary PEL. (A) Tumor cells were diffusely positive for EBV (EBER; 200×). (B) The presence of HHV8 was demonstrated in this neoplasm by immunohistochemistry (LANA-1) (DAB; 100×). Abbreviations: PEL, primary effusion lymphoma; HHV8, human herpesvirus 8; EBV, Epstein–Barr virus; LANA-1, latency-associated nuclear antigen-1; EBER, EBV-encoded small RNAs.
This study was approved by the local Ethical Committee and all procedures followed the ethical standards of the Helsinki Declaration of 1975, as revised in 2008.
Discussion
PEL is a very uncommon lymphoma that characteristically manifests as a body cavity effusion in immunosuppressed patients, more often males with a median age ranging from 45 to 60 years old, but that may also present as an extracavitary disease.1,4,6,14 Following a detailed review since the publication of the fourth edition of the World Health Organization guidelines in 2008, 15 we were able to identify 83 patients reported with clinicopathological data available for each case (Table 1). Some series investigating PEL showed that extracavitary manifestation comprises from 20% to 50% of the affected individuals.6,10,14,16
Solid/Extracavitary Primary Effusion Lymphoma Previously Described in Literature (From 2008 to 2022).
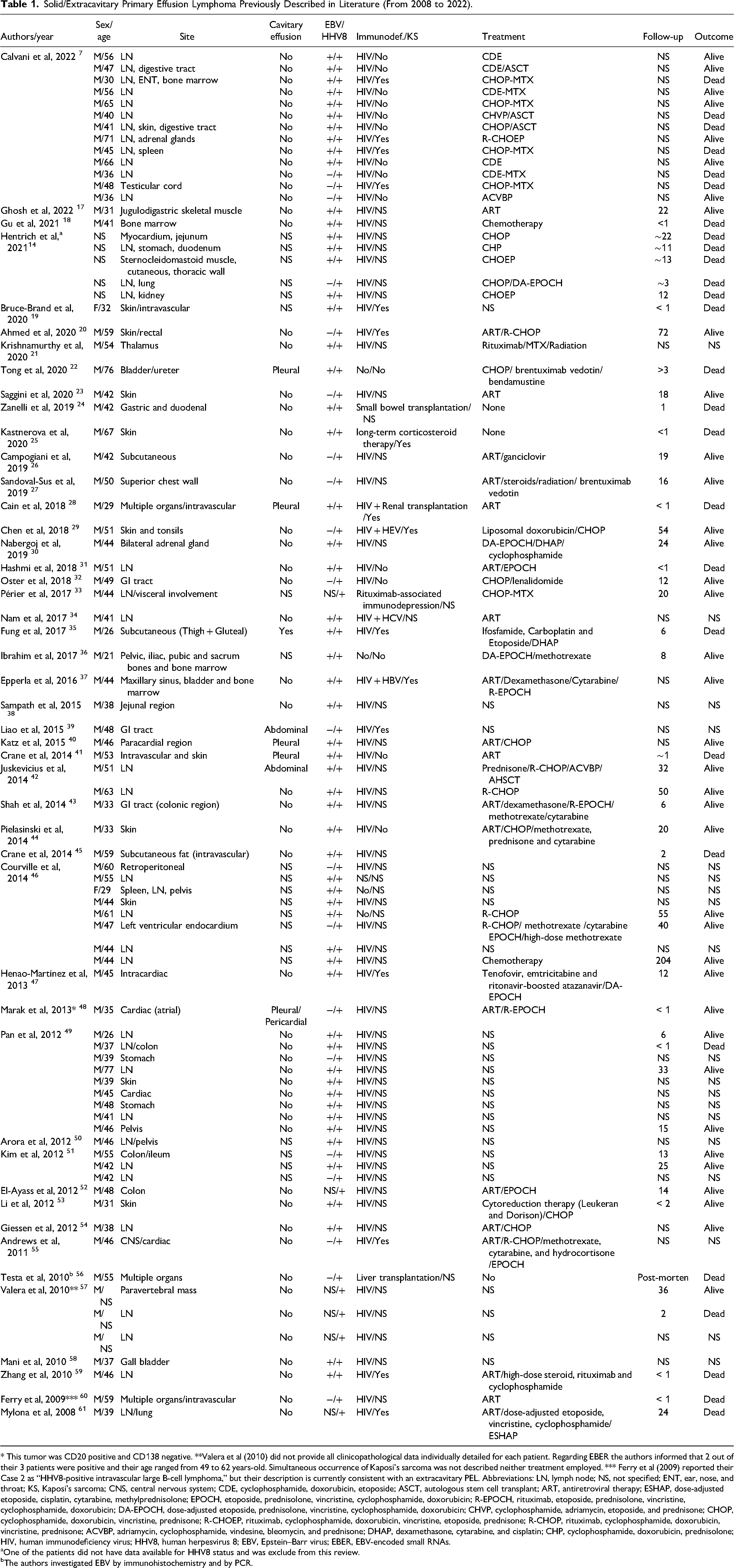
* This tumor was CD20 positive and CD138 negative. **Valera et al (2010) did not provide all clinicopathological data individually detailed for each patient. Regarding EBER the authors informed that 2 out of their 3 patients were positive and their age ranged from 49 to 62 years-old. Simultaneous occurrence of Kaposi's sarcoma was not described neither treatment employed. *** Ferry et al (2009) reported their Case 2 as “HHV8-positive intravascular large B-cell lymphoma,” but their description is currently consistent with an extracavitary PEL. Abbreviations: LN, lymph node; NS, not specified; ENT, ear, nose, and throat; KS, Kaposi's sarcoma; CNS, central nervous system; CDE, cyclophosphamide, doxorubicin, etoposide; ASCT, autologous stem cell transplant; ART, antiretroviral therapy; ESHAP, dose-adjusted etoposide, cisplatin, cytarabine, methylprednisolone; EPOCH, etoposide, prednisolone, vincristine, cyclophosphamide, doxorubicin; R-EPOCH, rituximab, etoposide, prednisolone, vincristine, cyclophosphamide, doxorubicin; DA-EPOCH, dose-adjusted etoposide, prednisolone, vincristine, cyclophosphamide, doxorubicin; CHVP, cyclophosphamide, adriamycin, etoposide, and prednisone; CHOP, cyclophosphamide, doxorubicin, vincristine, prednisone; R-CHOEP, rituximab, cyclophosphamide, doxorubicin, vincristine, etoposide, prednisone; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone; ACVBP, adriamycin, cyclophosphamide, vindesine, bleomycin, and prednisone; DHAP, dexamethasone, cytarabine, and cisplatin; CHP, cyclophosphamide, doxorubicin, prednisolone; HIV, human immunodeficiency virus; HHV8, human herpesvirus 8; EBV, Epstein–Barr virus; EBER, EBV-encoded small RNAs.
One of the patients did not have data available for HHV8 status and was exclude from this review.
The authors investigated EBV by immunohistochemistry and by PCR.
Despite some slight differences in the protein expression patterns, 49 the microscopic, the immunophenotype, and the association with HHV8 and EBV viruses are similar between classic PEL and extracavitary PEL, suggesting that they represent the clinical spectrum of the same entity. This assumption was later supported by genetic and proteomic studies that showed a strong similarity regarding molecular expression profile in both manifestations,62,63 as well as survival rates. 6
Solid PEL is more commonly diagnosed in the lymph nodes, although many other extranodal and extracavitary locations may also be affected, like the skin, the gastrointestinal tract, and other organs. 49 Some reports described an intravascular presentation of the disease, which must be differentiated from intravascular large B-cell lymphoma. 41 Moreover, disseminated solid PEL has also been described affecting 2 or more sites simultaneously, 32 and postmortem reports are also available in literature. 56 In the oral cavity, extracavitary PEL is extremely rare and very poorly documented. Hu et al 16 in their series of 70 PEL included 2 tumors affecting the skin/buccal region and Mate et al 12 described a solid PEL affecting the tongue of a 42-year-old HIV-positive male patient who died 2 weeks after treatment initiation. In 1997 Buske et al 64 described a patient diagnosed as anaplastic large cell lymphoma of the B-cell type positive for HHV8 and EBV (Latent membrane protein 1—LMP1) that affected the hard palate and other sites, while Carbone et al 62 included 2 patients affecting the Waldeyer's ring in their series of 4 PEL, both died after a short follow-up, and Boulanger et al 65 included 1 patient affected by a maxillary sinus in their series of 28 PEL.
Other lymphomas manifesting as cavitary effusions must be distinguished from classic PEL, 66 while the list of differential diagnoses for the solid variant is also complex. Lymphomas characterized by a plasmablastic differentiation represent the most important entities to be distinguished from extracavitary PEL (Table 2). Plasmablastic lymphoma more frequently affects the oral mucosa 67 and was the main differential diagnosis in the present case; however, it does not present HHV8 positivity. Similarly, anaplastic lymphoma kinase (ALK)+ large B-cell lymphoma is also negative for HHV8 and EBV, while anaplastic variant of plasmacytoma/multiple myeloma lacks an association with HHV8, and EBV is only rarely found.16,68 Although diffuse large B cell lymphoma not otherwise specified represents the most common lymphoma subtype in the oral cavity 69 and must be considered in the differential diagnosis, it strongly expresses CD20 protein and other mature B cell markers. Anaplastic large-cell lymphoma may be an important mimicker of PEL 34 and both demonstrate CD30 expression, and this differentiation can be even more difficult due to the frequent aberrant expression of T-cell markers in PEL7,16,70 as demonstrated in our patient, and although these neoplasms usually have polyclonal T-cell populations, a few tumors were shown to have clonal T-cells demonstrated by DNA analysis. 71 Courville et al 46 showed in their series of 8 extracavitary PEL that aberrantly expressed T-cell markers include CD3, CD43, CD7, CD45RO, and cytotoxic granules. The expression of plasmacytic markers and the presence of HHV8 and EBV are very useful to achieve the correct diagnosis.
Important Differential Diagnoses for Extracavitary Primary Effusion Lymphoma.
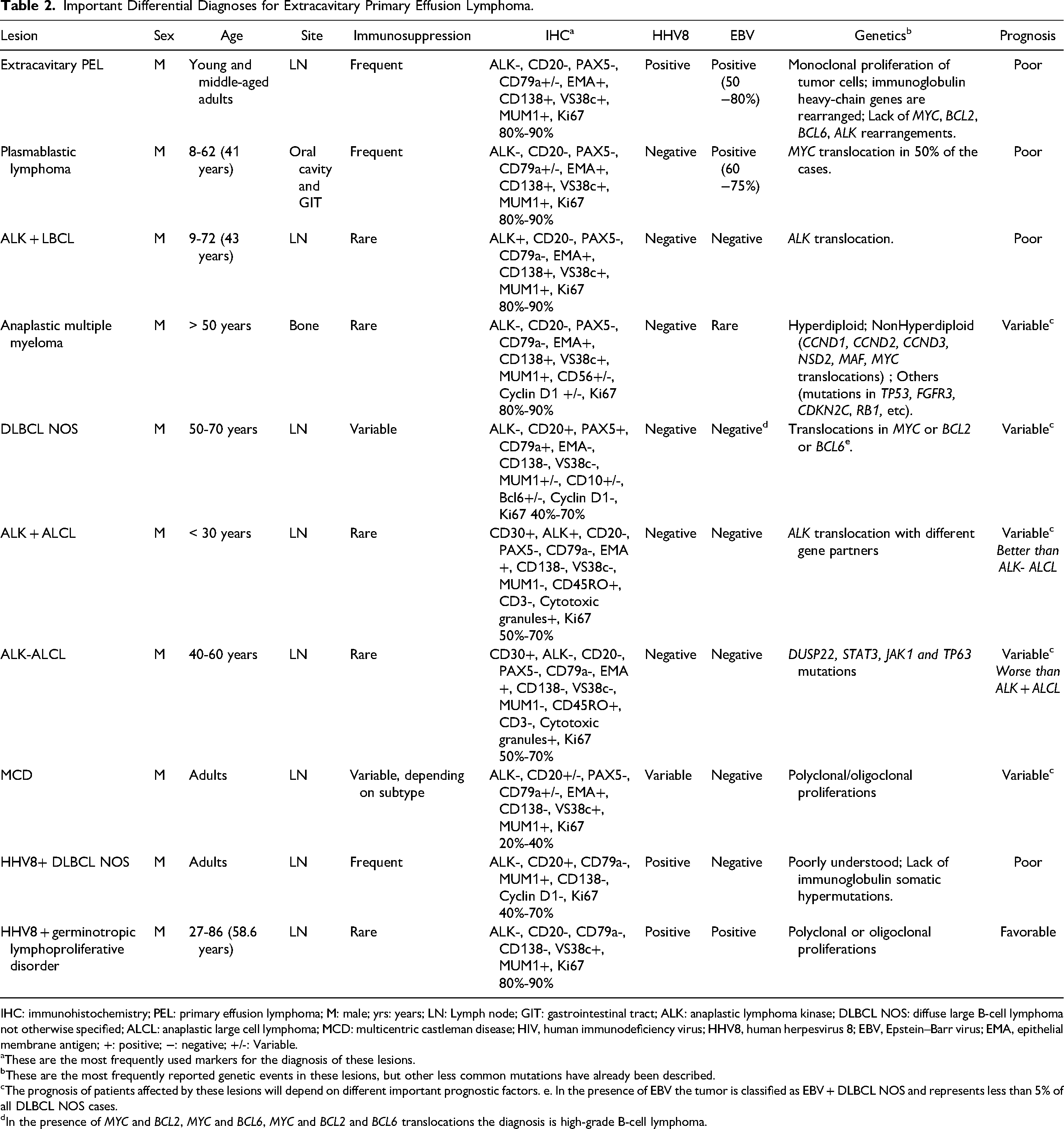
IHC: immunohistochemistry; PEL: primary effusion lymphoma; M: male; yrs: years; LN: Lymph node; GIT: gastrointestinal tract; ALK: anaplastic lymphoma kinase; DLBCL NOS: diffuse large B-cell lymphoma not otherwise specified; ALCL: anaplastic large cell lymphoma; MCD: multicentric castleman disease; HIV, human immunodeficiency virus; HHV8, human herpesvirus 8; EBV, Epstein–Barr virus; EMA, epithelial membrane antigen; +: positive; −: negative; +/-: Variable.
These are the most frequently used markers for the diagnosis of these lesions.
These are the most frequently reported genetic events in these lesions, but other less common mutations have already been described.
The prognosis of patients affected by these lesions will depend on different important prognostic factors. e. In the presence of EBV the tumor is classified as EBV + DLBCL NOS and represents less than 5% of all DLBCL NOS cases.
In the presence of MYC and BCL2, MYC and BCL6, MYC and BCL2 and BCL6 translocations the diagnosis is high-grade B-cell lymphoma.
It is known that HHV8 may be found in other lymphomas and lymphoproliferative disorders, which should be differentiated from extracavitary PEL. Multicentric castleman disease is a clinicopathological entity that encompasses a group of systemic polyclonal lymphoproliferative disorders in which there is a proliferation of morphologically benign lymphocytes, plasma cells, and vessels. 70 It is associated with HHV8, but lacks EBV infection, and its microscopic features characterized by follicles showing the so-called “onion skinning” or widened concentric rings of mantle zone lymphocytes, and prominent penetrating venules are usually very suggestive of the diagnosis. Another important differential diagnosis is HHV8 + diffuse large B-cell lymphoma that usually arises in patients with clinical features of HHV8 + multicentric Castleman disease in individuals showing a profound immunodeficiency, but these tumors express CD20 and lack CD138 protein and EBV infection. 16 More recently, it was recommended that in the absence of EBV infection and with cytoplasmic IgM, lambda, and/or associated with multicentric Castleman disease, extracavitary PEL would be better diagnosed as HHV8 + large B-cell lymphoma. 72
Finally, HHV8 + germinotropic lymphoproliferative disorder must also be ruled-out when lymph nodes are affected and this lesion may also show EBV positivity. It usually occurs in HIV-negative patients that show effacement of the germinal centers by plasmablasts in the affected lymph node, but different from PEL it does not express CD138 and often shows an indolent clinical course.70,73 Despite the differences between PEL and these HHV8-positive disorders, they have overlapped histological and clinical features and their distinction may be very difficult in some cases.5,74 The genetic basis of these lesions remains poorly understood and characterized; however, PEL has been shown to lack MYC, BCL2, and BCL6 genes translocation, as shown in our patient, but with hypermutated genes and immunoglobulin gene rearrangements, while multicentric Castleman disease and HHV8 + germinotropic lymphoproliferative disorder cells were shown to have a polyclonal/oligoclonal nature, and HHV8 + diffuse large B-cell lymphoma has no immunoglobulin somatic hypermutations. 71
In the present patient, due to its retrospective nature and because it represents a specimen received at the beginning of the 1990s, it is not possible to determine the occurrence of any cavitary effusion; however, such occurrence is not necessary to confirm the diagnosis of an extracavitary PEL since the development of a solid manifestation of this lymphoma may occur before, later or may even represent the only manifestation of the disease.10,16 Moreover, given the occurrence of an oral Kaposi's sarcoma in our patient, it is very likely that he was immunosuppressed, although it was also not possible to determine if the patient was HIV positive. The simultaneous occurrence of Kaposi's sarcoma and extracavitary PEL was previously demonstrated,6,16 but in our patient, the microscopic exam of the lymphoma did not show evidence of a simultaneous Kaposi's sarcoma, with the absence of spindle-cell proliferations and CD34 expression. Meanwhile, it is important to consider that classic and extracavitary PEL may also develop under other immunosuppressive conditions, like in transplanted patients and in elderly patients. 24
There are currently no standardized therapeutic schemes used to treat patients affected by classic or extracavitary PEL. HIV-related immunosuppression must include antiretroviral therapy, while different chemotherapeutic schemes have been applied, more often cyclophosphamide, hydroxydaunorubicin (also called doxorubicin or adriamycin), vincristine (Oncovin) and prednisolone combined or not with Rituximab, despite the lack of CD20 expression by tumor cells. Additional regimens like etoposide, prednisolone, oncovin, cyclophosphamide, and hydroxydaunorubicin (EPOCH), dose-adjusted EPOCH (DA-EPOCH), dexamethasone, high-dose arabinoside-C (cytarabine) and platinol (cisplatin), the use of methotrexate and brentuximab vedotin, among others have also been reported. The prognosis of the patients is very poor with median survival ranging from 10.2 to 22 months and a 2-years survival rate ranging from 15.4% to 71.5%.4,6,7,9,14,75 Interestingly, few individuals present a satisfactory recovery after treatment initiation and the search for new clinical and molecular prognostic parameters is highly desirable. Currently, the number of cavities involved, the type of cavity affected, the immunologic state, the presence of EBV, and elevated interleukin-6 may represent potential prognostic variables for these patients.75,76
In conclusion, extracavitary PEL is a very rare disease and its manifestation in the oral cavity is even more uncommon, demanding from pathologists a careful evaluation of lymphomas characterized by plasmablastic morphology that is more often diagnosed in this site like plasmablastic lymphoma. In these circumstances, the search for HHV8 is recommended and a careful systemic evaluation of the patient is also advisable to investigate the occurrence of possible cavitary effusions, especially in immunosuppressed patients.
Footnotes
Acknowledgments
The authors acknowledge the inputs of Dr Roman Carlos (in memorian) to the study of this case.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the local Ethical Committee of the Piracicaba Dental School of the University of Campinas and all procedures followed the ethical standards of the Helsinki Declaration of 1975, as revised in 2008.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), the São Paulo State Research Foundation (FAPESP), the Coordination for the Improvement of Higher Education Personnel (CAPES) (Financial code 001) and the National Council for Scientific and Technological Development (CNPq).
