Abstract
Development of mesothelioma is associated with asbestos exposure. Common presentations are with pleural-based plaques invading the chest wall and/or pleural effusion on chest imaging. The intent of this case report is to describe a rare presentation of mesothelioma, which presented atypically as a large tension pneumothorax. A 93-year-old male presented with a history of dyspnea that started after a coughing episode. On physical examination he was hemodynamically stable, but was hypoxic requiring 2L of supplemental oxygen. Computed tomography of the chest revealed a large right tension pneumothorax. A chest tube was placed and connected to suction (−20cmH20), but he continued to have an unresolving air leak over the following 2-week period. Upon video-assisted thoracotomy there were no blebs or adhesions seen. Right apical wedge resection and talc pleurodesis were performed. Pathologic examination revealed an atypical mesothelial cell proliferation with minimal, focal invasion into the pulmonary parenchyma. Tumor spread along the visceral pleura was thought to be the underlying cause of the pneumothorax. The surgical margins were uninvolved by the tumor, and the patient was later discharged home in stable condition. This was a rare presentation of what could best be described as minimally invasive mesothelioma arising in a background of probable mesothelioma in situ, which presented atypically as a large tension pneumothorax. This case highlighted the importance of establishing a pathologic diagnosis from pleural effusion cytology and/or pleural biopsy in persons presenting with spontaneous pneumothorax, and the difficulty in confirming a pathologic diagnosis of early mesothelial neoplasia.
Introduction
Development of mesothelioma is associated with asbestos exposure in roofing and ship-building.1–5 Common presentations of mesothelioma are with pleural-based plaques invading the chest wall and/or pleural effusion on chest imaging.1,5 The intent of this case report is to describe a rare presentation of mesothelioma, that presented atypically as a large right tension pneumothorax.
Case Report
A 93-year-old male presented with a history of dyspnea that started after a coughing episode. His past medical history was significant for low-grade papillary urothelial bladder cancer treated with docetaxel and gemcitabine, and prostate cancer treated with surgical resection. Of note, during his years of employment, he worked in a naval shipyard.
On physical examination, he was hemodynamically stable, but was hypoxic requiring 2L of supplemental oxygen. Breath sounds on the right side were absent. Computed tomographic imaging of the chest revealed a large right tension pneumothorax (Figure 1). A chest tube was placed and connected to suction (−20cmH20). However, he continued to have an unresolving air leak and small right apical pneumothorax over the following 2-week period.
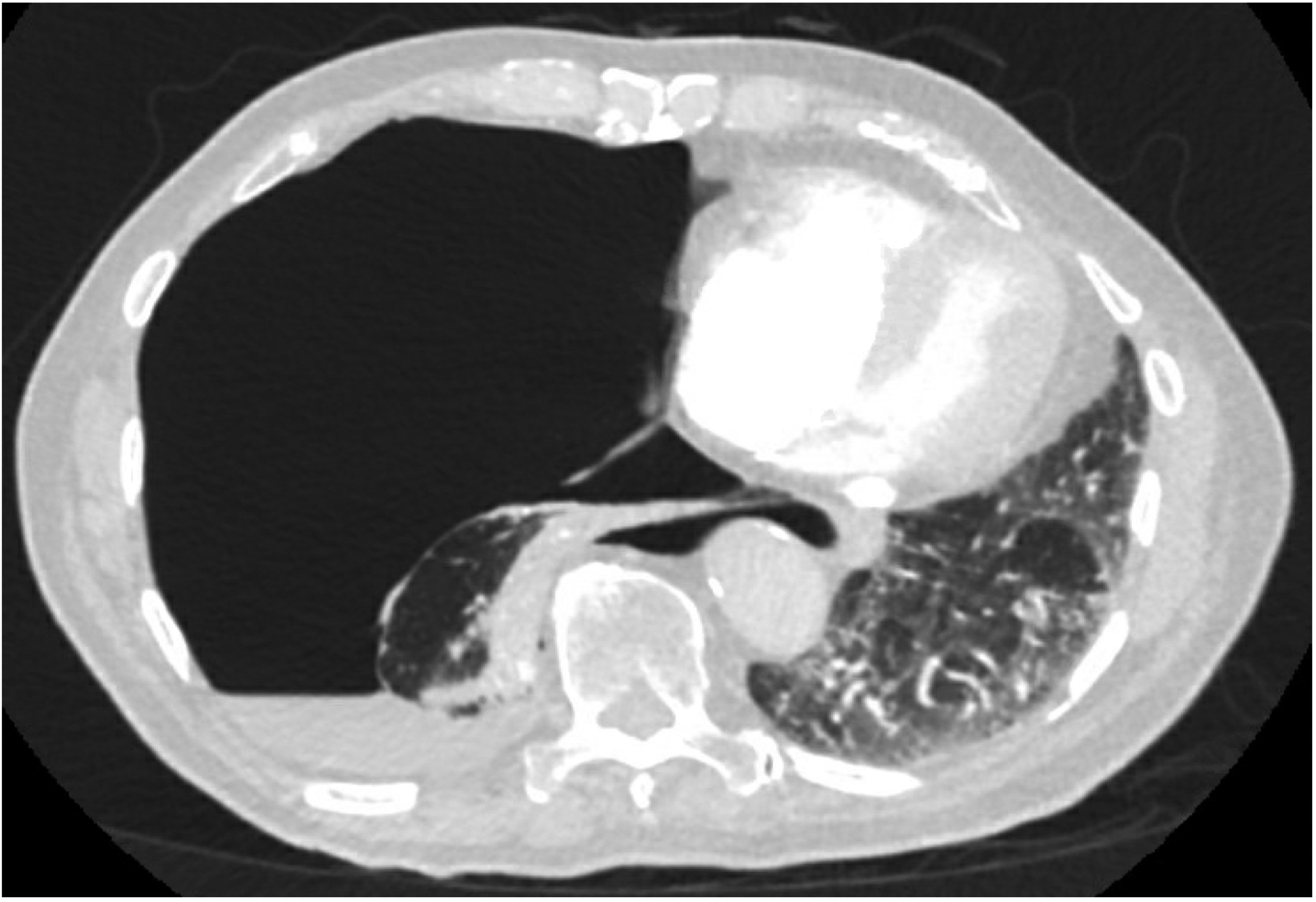
Transverse view of the large right tension pneumothorax, with massive shift of the heart into the left hemithorax and small right sided pleural effusion on CT-Chest.
Upon video-assisted thoracotomy, no blebs or adhesions were seen. However, a right apical wedge resection and talc pleurodesis were performed for his persistent air leak. Received in the pathology department was a lung wedge specimen measuring 7.8 cm×4 cm×1.5 cm. The pleural surface was pink and generally unremarkable without adhesions or pleural retraction. Sectioning of the specimen revealed pink-red, spongy parenchyma without lesions or masses and the entire specimen was ultimately submitted for histologic examination.
Hematoxylin and eosin (H&E) stained sections revealed an atypical mesothelial cell proliferation involving the visceral pleura (Figure 2). The atypical mesothelial cells were cuboidal to tall columnar with amphophilic cytoplasm. Cytologic atypia was overall mild with bland nuclear features, but slightly prominent nucleoli and few identifiable mitotic figures. Of important note, the atypical mesothelial cells demonstrated loss of BRCA associated protein-1 (BAP1) nuclear expression (Figure 3). Immunohistochemical staining confirmed the mesothelial origin of the atypical cells, which were found to be WT1, keratin 5/6, and calretinin positive with negativity for D2-40, TTF1, and Napsin-A (Figure 4).
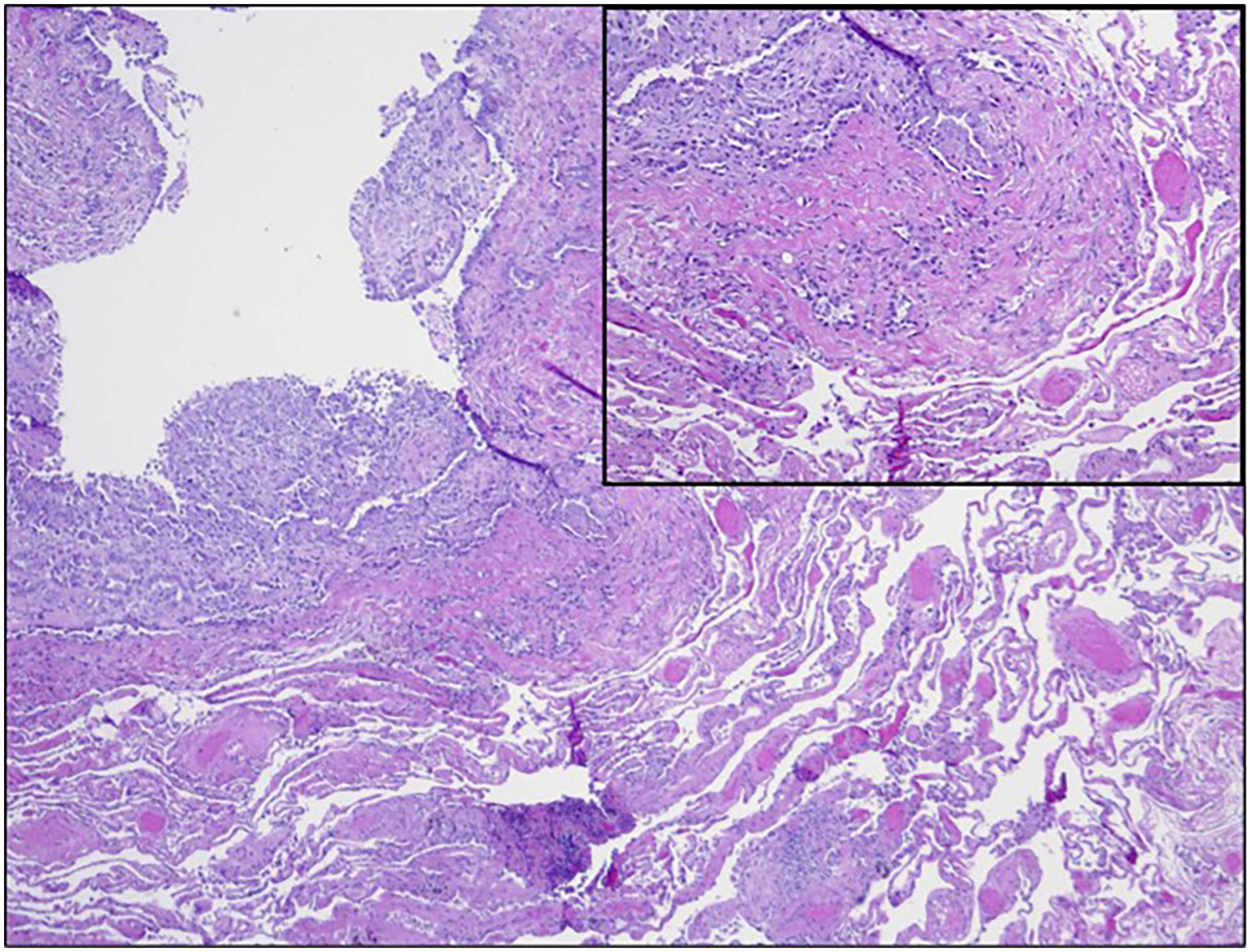
H&E stained section (40× total magnification) highlighting the architectural features of the atypical mesothelial proliferation which focally forms tubules that appear in close approximation to alveolated lung parenchyma proper (inset, 100× total magnification).
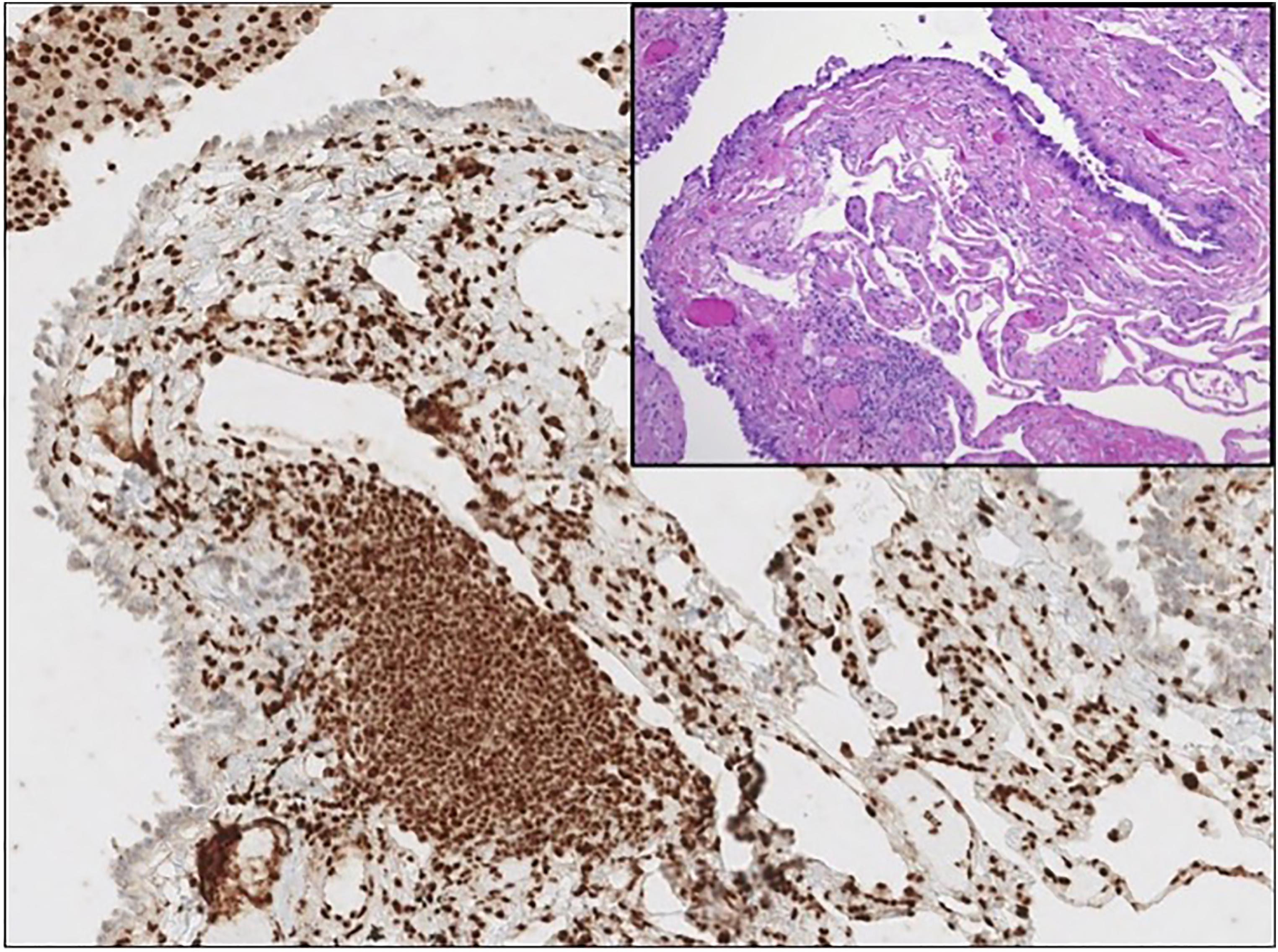
Immunohistochemical stain for BAP1 (100× total magnification) demonstrating mesothelial cells with loss of BAP1 nuclear expression supportive of a component of mesothelioma in situ. A paired H&E section demonstrates an absence of invasive mesothelioma at this location (inset, 100× total magnification).
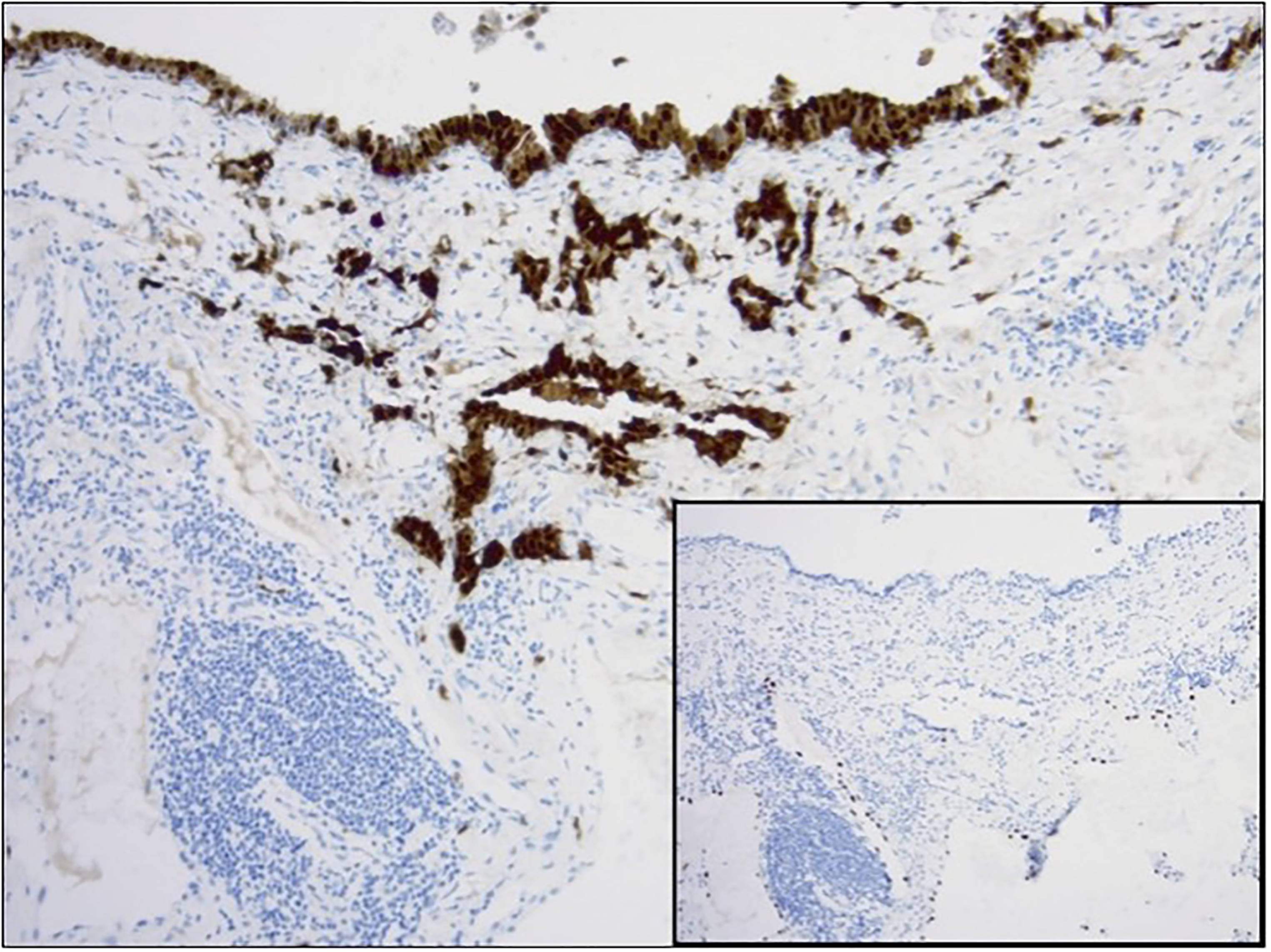
Calretinin immunostain (100× total magnification) demonstrates the invasive cells as being mesothelial cells with negative TTF1 expression (inset, paired section, 100× total magnification).
There were several foci identified whereby the atypical mesothelial cells formed small nests, tubules, and were distributed as single cells that appeared in close approximation to, if not fully involved, alveolated lung parenchyma proper. These foci were further evaluated for possible invasion by a histochemical Movat stain, which demonstrated focal invasion of the atypical mesothelial cells beyond the visceral pleura elastic lamina layer and into the pulmonary parenchyma. Overall, the features suggested invasive mesothelioma with only minimal invasion into the pulmonary parenchyma arising in a background of probable mesothelioma in situ. The surgical margins of the wedge resection appeared uninvolved by tumor. Following this surgery, the patient was discharged home in stable condition.
Discussion
This case was a rare presentation of what could best be described as minimally invasive mesothelioma, which presented atypically as a large right tension pneumothorax. The typical presentation of mesothelioma is with dyspnea, cough, and chest pain. Chest imaging generally reveals pleural-based plaques invading the chest wall and/or pleural effusion.1–5 Rarely malignant mesothelioma has presented with spontaneous pneumothorax. However, these cases were locally advanced with significant pleural and chest wall invasion.1,6,7 Rupture of necrotic tumor nodules was the most common explanation for the development of pneumothorax in these patients. 1 To our knowledge, this is the first presentation of early invasive mesothelioma as a spontaneous pneumothorax. We suspect tumor spread along the visceral pleura was the underlying cause.
The development of mesothelioma has been most commonly associated with asbestos exposure in roofing and ship-building.1,2,8 Other associations have been made with therapeutic radiation using thorium dioxide and zeolite, erionite exposure in construction and road paving, and other fibrous minerals such as fluoro-edenite, winchite, and richterite.2,8 Simian virus 40 (SV 40) infection and various genetic aberrations such as the BAP1 tumor predisposition syndrome (BAP1-TPDS) have also been associated with increased predisposition to the development of mesothelioma. Individuals with SV 40 exposure and these genetic aberrations develop mesothelioma at a younger age, and at lower levels of asbestos exposure than would typically be expected.2,3,8,9 Relevant authorities have been enforcing regulations aimed at reducing asbestos exposure and the subsequent development of mesothelioma. However, it is highly likely that we will continue to rarely see cases of mesothelioma in the genetically predisposed if they continue to have what would otherwise be considered low level asbestos exposure.
Comprehensive genomic analyses have demonstrated that the most frequently mutated genes in mesothelioma include BAP1, NF2, TP53, SETD2, DDX3X, ULK2, RYR2, CFAP45, SETDB1, and DDX51. 10 Mutations in the BAP1 gene encoding this protein can be somatic mutations within tumor cells or occur as heterozygous germline pathogenic variants as part of BAP1-TPDS. This syndrome is inherited in an autosomal dominant pattern and is associated with peritoneal and pleural mesotheliomas, as well as melanocytic tumors and clear cell renal cell carcinoma. 11 In addition to BAP1 inactivation, CDKN2A homozygous deletions are considered to occur early in mesothelioma development. 12 The presence of germline mutations on their own do not appear to predispose to the development of mesothelioma. However, these mutations make it difficult to repair DNA damages accumulated during asbestos and other carcinogenic exposure and may lead to the development of mesothelioma at levels of exposure much lower than would be typically expected.2,3,9 Multiple studies have demonstrated that the median age at diagnosis for germline BAP1-TPDS associated mesothelioma is significantly younger (reported as 55-58 years) than for sporadic mesothelioma (reported as 68-72 years).13–15 Though our patient's tumor demonstrated loss of BAP1 nuclear expression by immunohistochemistry, his advanced age at the time of diagnosis made BAP1-TPDS appear unlikely. The tumor he developed was most likely sporadic in nature and related to his history of occupational asbestos exposure while working in a naval shipyard.
The current categories of pleural mesothelial neoplasia established by the World Health Organization (WHO) are diffuse pleural mesothelioma, localized pleural mesothelioma, mesothelioma in situ, and well-differentiated papillary mesothelial tumor of the pleura. Essential criteria for diagnosis of diffuse pleural mesothelioma are diffuse pleural thickening by a malignant neoplasm with epithelioid, sarcomatoid, or biphasic histology confirmed by immunohistochemistry to be of mesothelial origin and invasion of adjacent structures (ie, adipose tissue, skeletal muscle, and/or lung parenchyma), as well as identification of tumor necrosis or formation of unequivocal malignant tumor nodules. 10 The rare localized pleural mesothelioma has the same histological features of diffuse mesothelioma, but presents as a solitary localized mass by imaging, intraoperative findings, and histology with lack of invasion beyond the circumscribed borders of the tumor. 16 The very rare mesothelioma in situ is diagnosed in cases of non-resolving pleural effusion in the absence of mass when a single layer of mesothelial cells lining the pleural surface exhibit loss of BAP1 and/or methylthioadenosine phosphorylase (MTAP) by immunohistochemistry and/or CDKN2A homozygous deletion by fluorescence in situ hybridization (FISH) and all this followed by multidisciplinary discussion of the diagnosis. 12 The inclusion of mesothelioma in situ as a diagnostic category was a major change in the most recent WHO Classification of Thoracic Tumors (2021). 17
With this fairly recent introduction of mesothelioma in situ as a bona fide diagnostic category, the presentation of this case is timely and underscores for clinicians that a recurrent pleural or even peritoneal effusion without a clear etiology should be investigated by thoracoscopy or laparoscopy, and pleural or peritoneal biopsy performed even if the serosal surfaces appear completely normal upon visual inspection.
For pathologists, the important conclusion that can be drawn from this case is that in a patient with recurrent effusions or a biopsy that looks like non-specific pleuritis or peritonitis, immunohistochemical staining for BAP1 is essential in spite of a bland appearance of the surface mesothelial cells on routine H&E stain. For cases in which there is no loss of BAP1, immunohistochemical staining for MTAP should be performed and FISH for CDKN2A should at least be considered. In our case, the atypical mesothelial cells demonstrated loss of BAP1 nuclear expression. The entire specimen was submitted for histologic examination and revealed several small foci of invasive mesothelioma, in a lesion that would have otherwise been classified as mesothelioma in situ. This pattern of invasion of the lung parenchyma was minimal which made the pathologic diagnosis challenging and somewhat atypical.
After wedge resection, the margins were uninvolved by the invasive mesothelioma. Surgical resection in this case appeared adequate without need for further treatment. Otherwise, for invasive mesothelioma of localized or diffuse type, the usual treatment approach is surgery, with or without chemoradiation in cases of advanced disease. The prognosis in these cases is uniformly poor at 6 to 17 months.3,4,18 For mesothelioma in situ, there are no clear recommendations yet on the optimal treatment approach.19,20 Close follow up and monitoring is recommended at a minimum due to the potential of progression of mesothelioma in situ to invasive mesothelioma. 19 Resection of the pleura followed by intracavitary chemotherapy, photodynamic therapy, or cryoablation may also be a reasonable approach. 20
In summary, this case was a rare pathologic presentation of invasive mesothelioma with minimal focal invasion arising in a background of probable mesothelioma in situ, in combination with an atypical clinical presentation as a large tension pneumothorax. Tumor spread along the visceral pleura was thought to be the underlying cause of the pneumothorax.1,19 This case highlights the difficulty in confirming a pathologic diagnosis of early mesothelial neoplasia and the importance of obtaining a pathologic diagnosis from pleural effusion cytology and/or pleural biopsy in older persons presenting with spontaneous pneumothorax. 1 Histologic examination of the entire specimen is encouraged to avoid missing focal areas of invasive mesothelioma in lesions that would otherwise be classified as mesothelioma in situ.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
