Abstract
Fumarate hydratase deficient renal cell carcinoma (FHRCC) can exhibit a heterogenous immunoprofile. In the present case, a solitary 10.5 cm mixed cystic and solid left kidney tumor showed various growth patterns, involving renal sinus adipose tissue and the renal pelvis. Tumor cells showed prominent nucleoli and perinucleolar halos. Aberrant diffuse (>90%), strong, and membranous carbonic anhydrase 9 and variable GATA3 expression were present. Diagnostic loss of fumarate hydratase expression and 2-succinyl cysteine overexpression (cytoplasmic and nuclear) were identified. Carbonic anhydrase 9 and GATA3 expression in FHRCC is rarely reported in the literature, and may cause misdiagnosis of clear cell RCC and/or urothelial carcinoma.
Keywords
Introduction
Fumarate hydratase deficient renal cell carcinoma (FHRCC) is a rare and mostly aggressive subtype of RCC. 1 It was initially identified as a component of hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome and about 15% of syndromic patients go on to develop these tumors. 1 FHRCC has a male predominance (male-female ratio of 1.9:1) and is seen in a relatively younger population (median age 44 years), particularly in the hereditary setting. 2 Although large renal masses may present with hematuria and pain, these masses are most often identified incidentally during imaging for other reasons. Typical microscopic features include papillary, tubulopapillary, and solid formations of high nuclear grade tumor cells with frequent prominent nucleoli and perinucleolar halos; although there is a substantial morphologic heterogeneity. 2 Its diagnosis requires identification of mutations in the FH gene, either via sequencing or immunohistochemical (IHC) assay with demonstration of protein function loss. 1 Similar to the other RCC subtypes defined by a specific gene alteration, a diagnosis of FHRCC can be challenging due to morphologic and immunophenotypical overlap with other RCC subtypes. Herein, we present a case with an aberrant immunoprofile.
Case Presentation
A 42-year-old male was initially referred for urological evaluation of incidentally identified renal cysts by ultrasound, however, the patient was lost to follow-up. The patient presented again 5 years later with a 2-month history of gross hematuria with blood clots and associated left flank pain. Computed tomography and magnetic resonance imaging (Figure 1A-D) revealed an 8.3 cm mixed cystic and solid exophytic mass in the left upper pole and mid-kidney (Figure 1A-C). There was also extension of the mass into the upper pole calyces (Figure 1D). The top differential diagnoses included RCC, oncocytoma, and urothelial carcinoma, however, the findings were difficult to diagnose radiographically.
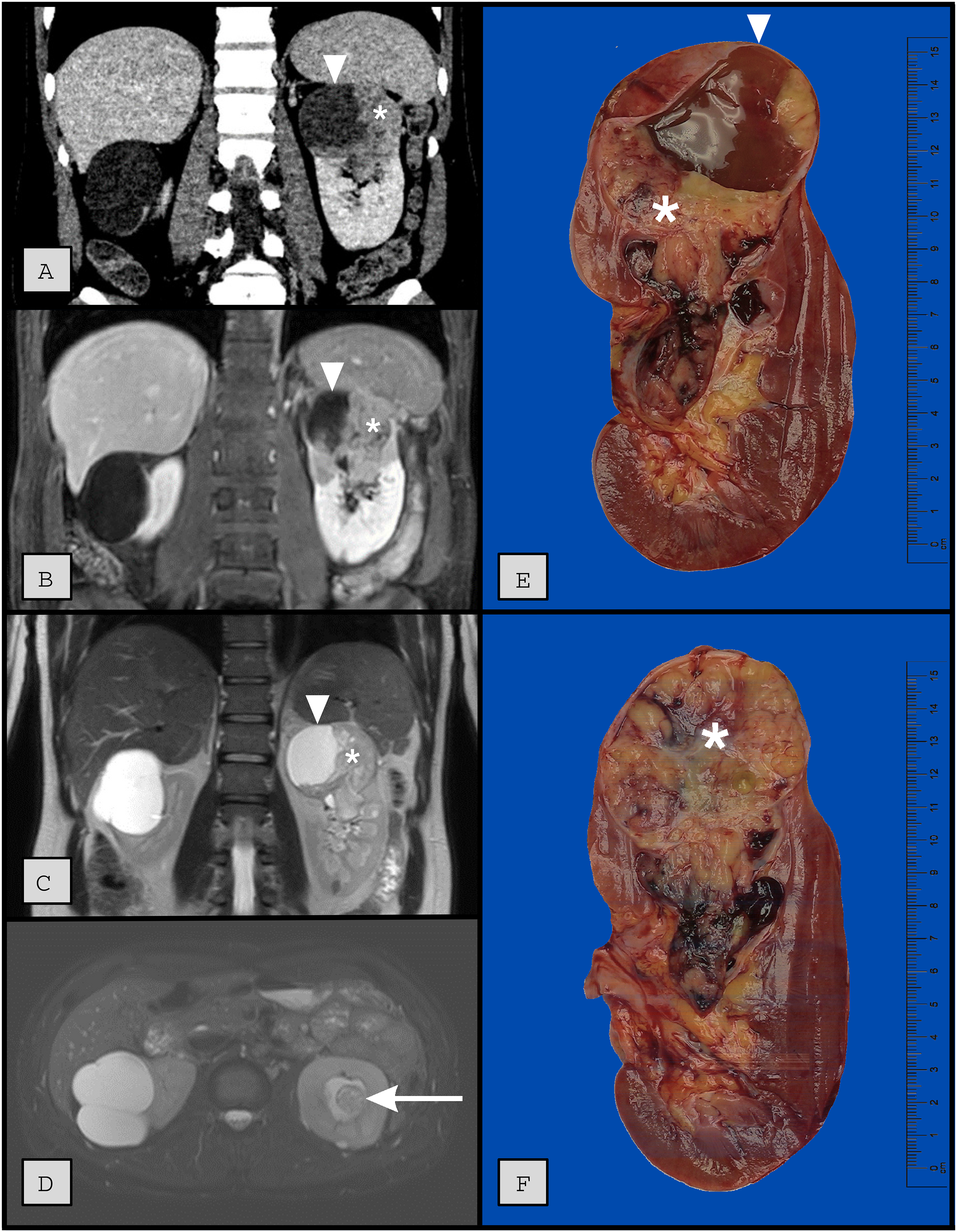
Coronal parenchymal phase contrast enhanced computed tomography (A), coronal post-contrast T1 weighted magnetic resonance imaging (B), and coronal T2 weighted MRI images (C) demonstrate an exophytic, heterogeneous mass of the left upper and mid-kidney measuring 8.3 × 7.6 × 6.0 cm. There are mixed cystic (arrowhead) and solid enhancing (*) regions throughout. The axial T2 image (D) demonstrates extension of the mass into the upper pole calyces (arrow). There is no invasion of the renal vein or surrounding structures. Of note, there is an incidental Bosniak 2F right renal cyst present on all images.
The patient underwent robotically assisted laparoscopic left radical nephrectomy. Sectioning of the kidney revealed a 10.5 cm solid and cystic mass predominating in the upper pole (Figure 1E-F). The mass demonstrated a heterogeneous tan-pink to yellow–white, soft, focally hemorrhagic cut surface with well-delineated borders. The tumor was extending into the renal sinus soft tissue and collecting system but did not penetrate into the perinephric fat or invade the renal vein.
Microscopic examination revealed tubulocystic (Figure 2A), papillary (Figure 2B) and cribriform growth patterns and occasional solid sheets. There were cysts formed by tumor cells with eosinophilic and basophilic cytoplasm and the vast majority of tumor cells exhibited prominent nucleoli with peri-nucleolar halos (Figure 2B). IHC assays showed that the tumor cells diffusely expressed pankeratin, carbonic anhydrase 9 (CA9, Figure 2C, diffuse, strong, and membranous), mostly expressed GATA3 (Figure 2D), and focally expressed CD10. Tumor cells were negative for keratin 7, keratin 20, HMB45, MART1, ALK1, and p63. Further IHC assays revealed lost expression of fumarate hydratase (Figure 2E) as well as overexpression of 2-succinyl cysteine (2SC, Figure 2F). Fluorescent in situ hybridization showed no translocations or amplifications on TFE3, TFEB, and ALK genes. With these findings, despite CA9 and GATA3 labeling, a diagnosis of FHRCC was rendered. Post-operative 3-month follow up revealed no recurrence of disease.
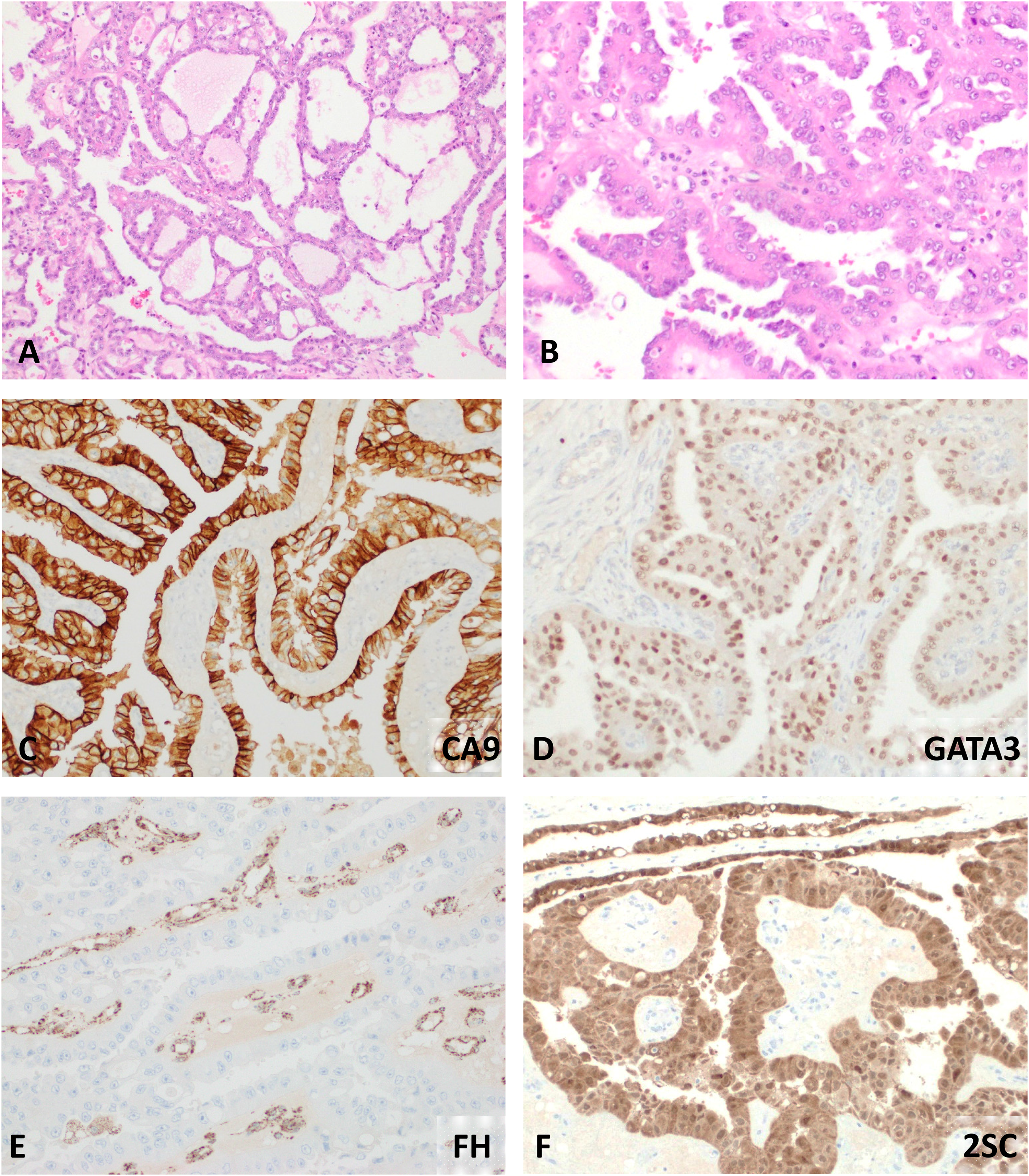
Tumor showed tubulocystic (A, 40X), and papillary features with tumor cells showing prominent nucleoli and perinucleolar halo (B, 100X). Diffuse, strong, and membranous carbonic anhydrase 9 (CA9) (C, 100X) and variable GATA3 expressions (D, 100X) were present. Diagnosis of the fumarate hydratase (FH) renal cell carcinoma was made when loss of FH (E, 100X) and overexpression of 2-succinyl cysteine (2SC, both cytoplasmic and nuclear) were demonstrated.
Discussion
The increasing utility of molecular techniques to determine the genetic background of renal neoplasms has reshaped the diagnostic approach to RCC. This change is evident in a separate sub-chapter entitled “molecularly defined renal carcinomas” in the latest edition of the World Health Organization's urinary tract and male genital system tumor classification. 1 Molecularly defined renal carcinomas include FHRCC, TFE3 rearranged RCC, TFEB altered (either rearranged or amplified) RCC, ELOC mutated RCC, succinate dehydrogenase deficient RCC, ALK rearranged RCC, and SMARCB1 deficient renal medullary carcinoma. 1 Despite alterations in different genes, these carcinomas have several common features: (1) the diagnosis depends on documentation of distinct mutations; (2) each entity provides important morphologic clues indicating defining molecular alterations; although (3) these morphologic clues can be focal; (4) molecularly defined renal carcinomas unanimously show significant morphologic and immunophenotypic heterogeneity. 1
The first identification of FHRCC was in patients with HLRCC. 3 Characteristic morphologic features in these studies were identified as “type 2 papillary RCC pattern” with eosinophilic cells with abundant cytoplasm and viral inclusion-like nucleoli, however, the “type 2” designation is no longer used. 3 Further reports with larger cohorts have expanded the morphologic spectrum, adding cystic, tubulopapillary, cribriform, and solid growth patterns.4–6 Moreover, rare occurrences of FHRCC resembling low-grade eosinophilic renal tumors have also been identified.6–8 A limited IHC panel may give further clues to the diagnosis of FHRCC, although the immunoprofile of these tumors can be variable. Keratin 7 is almost always negative. 4 Fumarate hydratase IHC is expected to be negative/lost, although expression can be retained in 20% of cases and some cases may demonstrate patchy FH loss.2,9 Although paradoxical, this heterogeneity in FH expression is thought to be a result of missense mutations that reduce FH function yet leave a variably expressed epitope that is detected by immunohistochemistry. 9 2SC appears to be the most sensitive and specific IHC, typically demonstrating both nuclear and cytoplasmic expression. 5 Aberrant IHC profiles with the risk of misdiagnosis have been reported. Baniak et al. identified diffuse, strong, and membranous CA9 expression, in 2/8 FHRCCs, a finding which is universally accepted as characteristic of clear cell RCC. 10 GATA3, most commonly used as the supporting evidence for carcinomas of the urothelial origin, was found to be variably expressed in 14/28 of cases in a separate study. 4 Recognition of this aberrant biomarker expression in FHRCC is important to prevent misinterpretation of these tumors as more common malignancies, such as high-grade clear cell RCC or urothelial carcinoma. Moreover, in this case, we could confidently exclude clear cell RCC due to lack of clear cell morphology. Similarly, the lack of keratin 7 argued against chromophobe RCC, IHC assays did not support papillary RCC, and clear loss of FH with presence of 2SC argued against papillary RCC. Finally, lack of keratin 7, keratin 20 and p63, and only focal GATA3, argued against urothelial carcinoma.
In conclusion, FHRCC is one of the molecularly defined renal carcinomas that have morphologic and immunophenotypic heterogeneity. Identification of molecular alterations such as an FH gene mutation or FH protein function loss (with FH and 2SC IHC) should be pursued when there is morphologic suspicion, even in the presence of an aberrant immunoprofile, such as CA9 and/or GATA3 expression.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This case report was approved by the Institutional Review Board at Albany Medical Center.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
This case report received a waiver for informed consent by the Institutional Review Board at Albany Medical Center.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
