Abstract
We report a 32-year-old male 14 years post-living-related kidney transplant presenting with new-onset hematuria and BK viremia. He was found to have BK virus-associated urothelial carcinoma originating in the renal allograft with locally advanced disease and metastases to multiple sites. He also developed acute T-cell-mediated rejection in the setting of immunosuppression reduction for BK viremia prior to undergoing transplant nephrectomy. Eight months following transplant nephrectomy and immunosuppression cessation, distant metastases persisted with partial response to chemotherapy and immunotherapy. Here, we discuss this unique presentation and compare it with other BK virus-associated allograft carcinomas reported in the literature, in addition to discussing evidence for the role of BK virus in oncogenesis.
Introduction
BK virus, a member of the family Polyomaviridae, is a ubiquitous double-stranded DNA virus related to JC virus and simian virus 40 (SV40). 1 In fact, because BK virus shows significant homology with SV40, immunohistochemical (IHC) staining for SV40 is routinely used as a surrogate for the presence of BK virus. 1 Following primary infection and viremia, BK virus develops latency in many organs, primarily the renal tubular epithelium and uroepithelium. 2
Although BK virus reactivation is known to cause nephropathy and ureteral stenosis in renal transplant recipients, its’ role in the development of posttransplant malignancies remains unclear.1,3 In general, immunosuppressed transplant recipients are at higher risk for developing cancers than the general population. 1 This can be attributed to immunosuppression causing decreased immune surveillance and increased susceptibility to infection by known oncogenic viruses, including Epstein–Barr virus and human papillomavirus. 3 BK virus-associated carcinomas have been reported in patients posttransplant, and many have postulated that BK virus, too, may play a role in oncogenesis.1,2,4‐11 Of note, however, there have also been reports of carcinomas in renal transplant patients that appear to lack BK virus involvement. 3 This has led some to postulate that rather than playing a role in oncogenesis, BK virus is merely an “innocent passenger” infection in tumor cells. 5
Case Presentation
A 32-year-old man with end-stage kidney disease due to focal segmental glomerulosclerosis (FSGS) underwent living-related donor kidney transplant without significant postoperative complications. His baseline creatinine was approximately 1.7 to 1.9 mg/dL with immunosuppression regimen of tacrolimus, mycophenolate, and prednisone. Fourteen years posttransplant, he developed intermittent hematuria; imaging revealed an exophytic, 2.3 cm mass at the inferior anterior transplant kidney (tumor score Bosniak 2F). Although new-onset BK viremia was found (∼26,000 copies/mL) with subsequent follow-up, creatinine was stable at 1.8 mg/dL, and urine protein:creatinine ratio (UPCR) was ∼0.9. Mycophenolate was held. Repeat imaging in 3 months showed an increase in mass to 3.1 cm.
Four months post-initial presentation for hematuria, BK viremia worsened (∼38,500 copies/mL), and IVIG and leflunomide were given, without improvement. When he developed fluid overload, elevated creatinine (∼8 mg/dL), and UPCR of 11.6, allograft biopsy showed suspicious/borderline T-cell-mediated rejection, Banff 2019 Criteria, 90% effacement of podocytes suggestive of FSGS recurrence, mild acute tubular injury, moderate (40%) interstitial fibrosis and tubular atrophy, moderate-to-severe hyaline atherosclerosis, and moderate-to-severe arteriosclerosis. Imaging noted bilateral lung masses, and computed tomography (CT)-guided biopsy demonstrated poorly differentiated carcinoma. fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT demonstrated likely additional metastases to pelvic lymph nodes. He completed steroid pulse for rejection, slightly improving creatinine from 8 to 6 mg/dL, and was discharged on prednisone taper with plans for transplant nephrectomy.
Shortly thereafter, he presented to our institution seeking a second opinion. With worsening allograft function, nephrotic-range proteinuria, and decreased viremia (3566 copies/mL), immunosuppression was reduced to low-dose prednisone only. Imaging revealed allograft mass increase to 3.3 cm. CT-guided core biopsies of allograft renal mass and lung mass showed tumor cells in sheets with eosinophilic cytoplasm, nuclear pleomorphism, prominent nucleoli, and some with nuclear vacuoles. There were increased mitotic figures and areas of tumor necrosis and apoptotic debris. Tumor cells positively stained for AE1/AE3, PAX8 (variable), GATA3, and SV40 (MRQ-4 mouse monoclonal antibody, IgG2a, 351 M series, Cell Marque) with negative staining for CA9. In the allograft biopsy, PD-L1 immunostain labeled approximately 30% of tumor cells and IHC demonstrated retained DNA mismatch repair proteins MLH1, PMS2, MSH2, and MSH6. In the lung mass biopsy, there were adjacent areas of lymphoplasmacytic inflammatory infiltrate, and further IHC was negative for TTF1, napsin A, and keratin 5/6 with focal p40 positivity. These were both diagnosed as poorly differentiated urothelial carcinoma, BK virus present (SV40 + ) in carcinoma cells. Representative microscopic images of allograft and lung mass biopsies are shown in Figures 1 A-D and 2A-D, respectively.
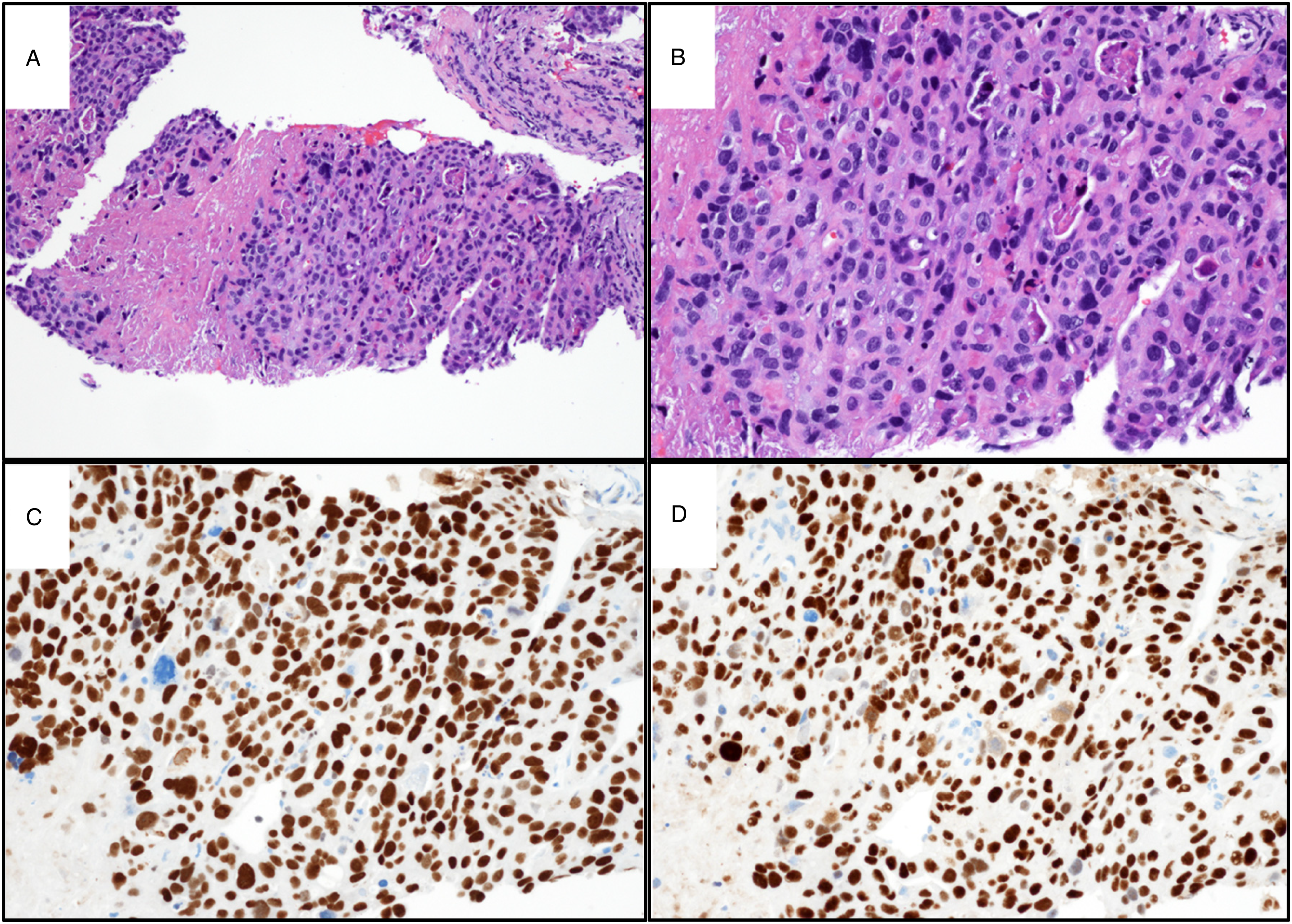
Microscopic images of renal allograft pelvic mass biopsy, diagnosed as poorly differentiated urothelial carcinoma with BK virus present (simian virus 40 [SV40] + ) in carcinoma cells. (A) H&E-stained tumor with focal necrosis (10x). An area of crushed uninvolved renal parenchyma is present in the right upper corner. (B) H&E-stained tumor with adjacent area of necrosis (20x). (C) GATA3-positive tumor cells (20x). (D) SV40-positive tumor cells (20x).
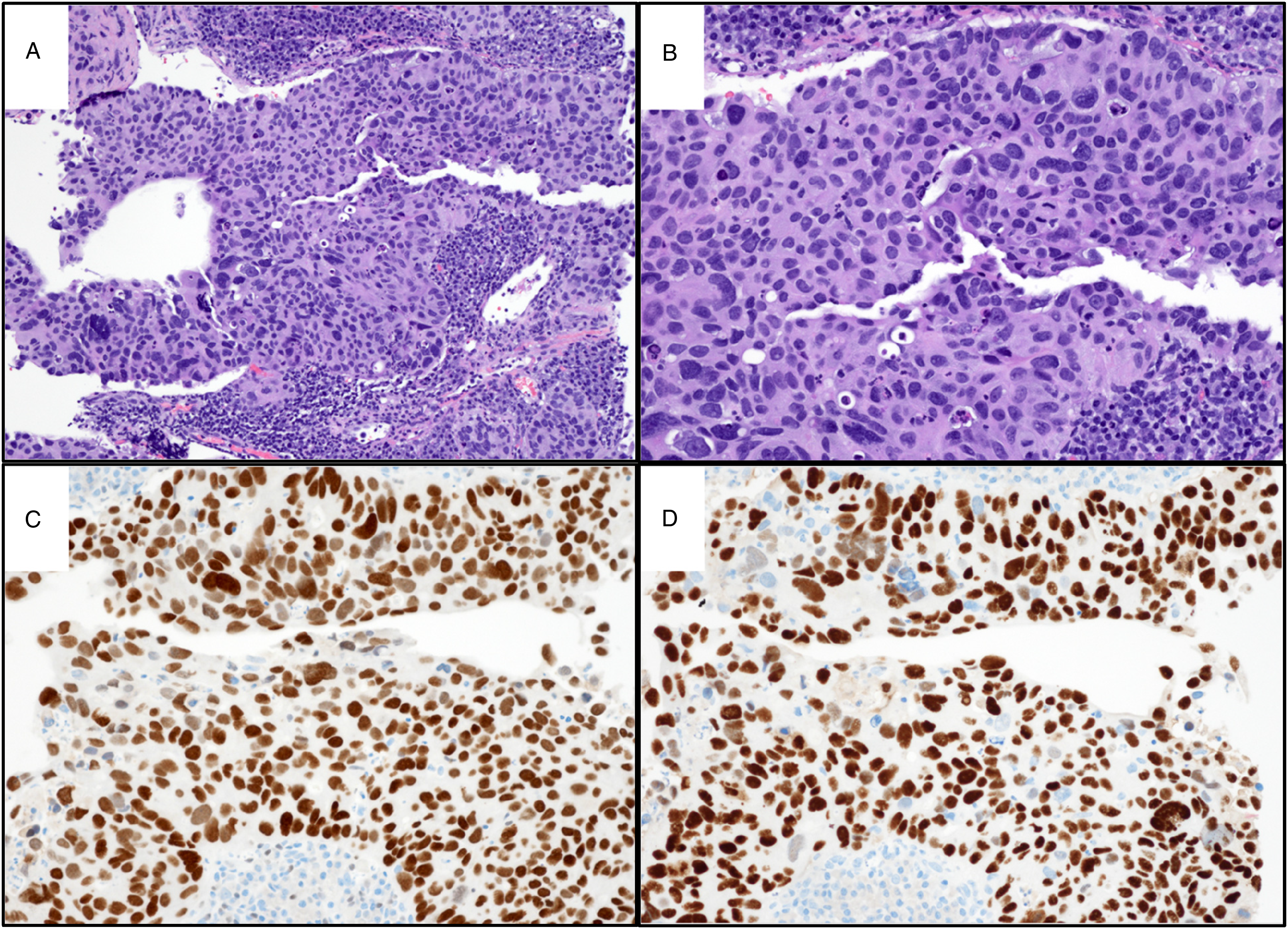
Microscopic images of left upper lobe mass biopsy, diagnosed as consistent with metastatic urothelial carcinoma, BK virus present (simian virus 40 [SV40] + ) in carcinoma cells. (A) H&E-stained tumor with adjacent lymphoplasmacytic inflammatory infiltrate (10x). (B) H&E-stained tumor with adjacent inflammatory infiltrate (20x). (C) GATA3-positive tumor cells with adjacent inflammatory cells acting as internal negative control (20x). (D) SV40-positive tumor cells with adjacent inflammatory cells acting as internal negative control (20x).
He was initiated on hemodialysis and underwent transplant nephroureterectomy with pelvic lymph node dissection, which showed pT3N2M1 high-grade urothelial carcinoma with carcinoma in situ of renal pelvis and invasive carcinoma involving the hilar adipose and renal parenchyma. Extensive tumor necrosis (∼70%), lymphovascular invasion, and a lymph node metastasis (3.4 cm) with extranodal extension were identified. Both p53 overexpression and SV40 positivity by IHC were limited to carcinoma and negative in benign urothelium. Representative microscopic images are shown in Figure 3 A-I. Molecular testing (TEMPUS xT 648 gene panel, Tempus Labs, Inc.) revealed a TERT promoter mutation, c.-124C > T, with 36.0% variant allele fraction. Tumor mutational burden was 8.9 m/MB (84th percentile) without microsatellite instability. Non-neoplastic allograft displayed tubulitis, interstitial infiltrate of lymphocytes, and intimal arteritis, consistent with Banff grade 2 chronic active T-cell-mediated rejection. There was also acute tubular injury, as shown in Figure 4A,B.
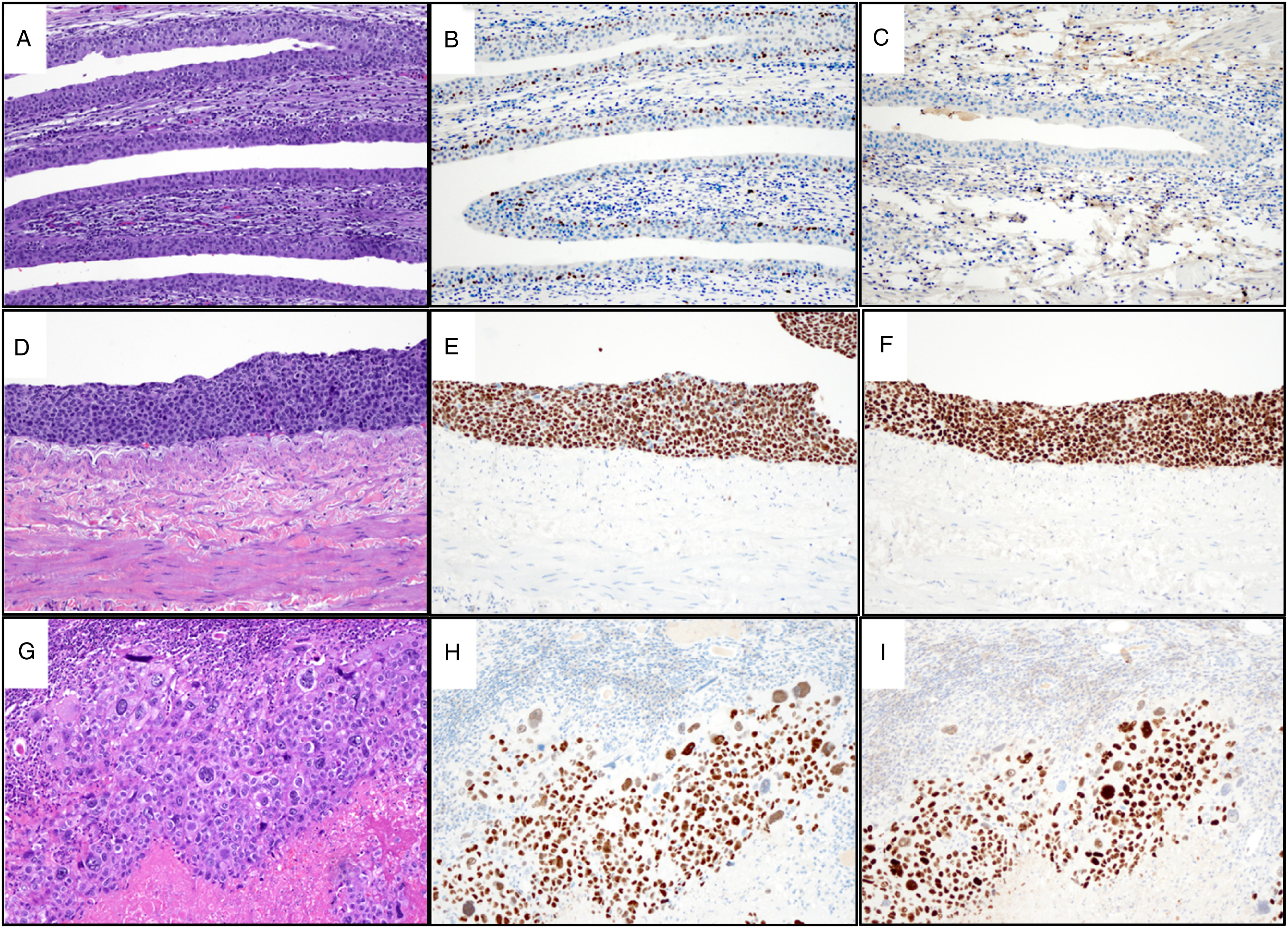
Microscopic images of allograft pelvic high-grade urothelial carcinoma. (A) H&E-stained urothelium uninvolved by tumor (10x). (B) Uninvolved urothelium, negative for p53 overexpression (10x). (C) Uninvolved urothelium, negative for simian virus 40 (SV40) (10x). (D) H&E-stained carcinoma in situ component. (E) p53-positive carcinoma in situ. (F) SV40-positive carcinoma in situ. (G) H&E-stained carcinoma invading renal parenchyma. (H) p53-positive invasive carcinoma. (I) SV40-positive invasive carcinoma.
Non-neoplastic allograft tissue displaying Banff grade 2 chronic active T-cell-mediated rejection and acute tubular injury. (A) Chronic active rejection with vasculopathy (4x). (B) Chronic active rejection (Banff grade 2) and acute tubular injury (10x).
The patient then underwent 2 cycles of carboplatin and gemcitabine with several pulmonary nodules decreasing in size and cavitating. Patient was unable to tolerate further chemotherapy and began avelumab. With 6 cycles completed, there has been some further cavitation and size decrease of pulmonary metastases. He has experienced no immune-related adverse events 8 months post-nephrectomy.
Discussion
There have been other reports of BK virus-associated carcinoma arising in the renal allograft, as summarized in Table 1. While urothelial carcinoma is the most frequently reported BK virus-associated carcinoma arising in the allograft (7 of 10 reports), renal cell carcinoma has also been reported (3 of 10 reports) .1,2,4‐11 They include transplants from deceased (7 of 10 reports) and living donors (3 of 10 reports). Although most patients had a known BK nephropathy history (7 of 10 reports), the interval between BK nephropathy and development of a BK virus-associated allograft carcinoma is unpredictable. Remarkably, the vast majority of BK virus-associated carcinomas of the allograft either improve or do not recur after treatment only with transplant nephrectomy and cessation of immunosuppression (7 of 8 reports; 1 excluded for lack of follow-up data; 1 excluded for maintained immunosuppression), highlighting the major role of immunosuppression in these malignancies.1,2,5‐10 However, for the few patients with disease progression or persistence despite these initial measures, immune checkpoint inhibitors appear to be a promising option, although data is admittedly very limited. For the patient reported by Chiang et al., administration of pembrolizumab, a PD-1 inhibitor, resulted in eventual complete remission 21 months post-nephrectomy, despite multiple sites of metastasis. 4 Additionally, our patient's pulmonary metastases have shown further improvement with avelumab, a PD-L1 inhibitor, following initial chemotherapy.
Summary of Clinicopathological Features of Reported BK Virus-Associated Carcinoma in Transplant Kidneys.
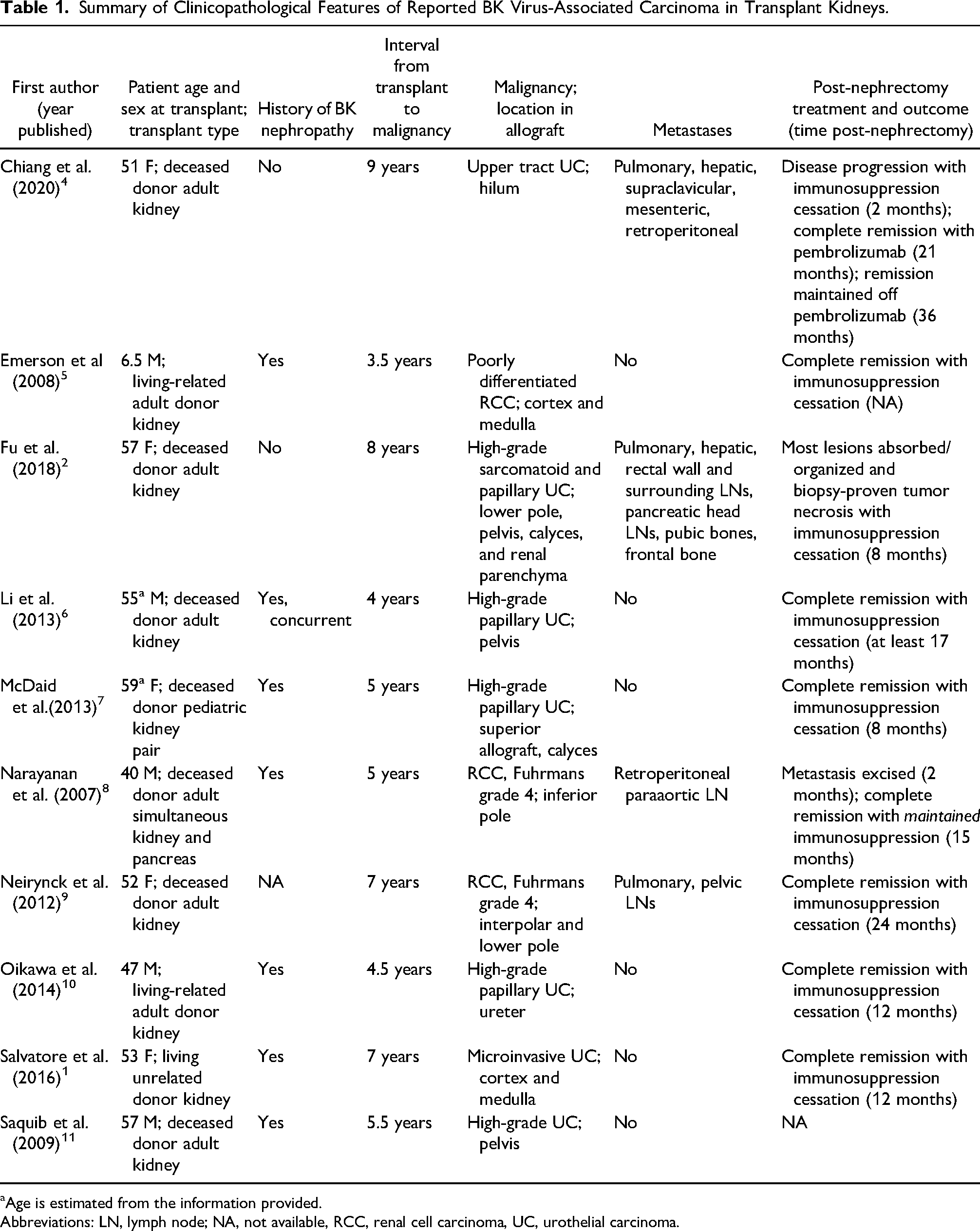
Age is estimated from the information provided.
Abbreviations: LN, lymph node; NA, not available, RCC, renal cell carcinoma, UC, urothelial carcinoma.
Although the causative role of BK virus in these tumors is debated, studies have demonstrated the oncogenic role of BK virus in vivo with animal models, identifying viral T-Ag protein as a likely culprit.12,13 The T-Ag protein, produced by both SV40 and BK virus, binds to and inactivates pRB and p53 proteins involved in cell cycle control.8,14 This hypothesized mechanism of oncogenesis is supported by the findings in our tumor. Staining for SV40 and overexpression of p53 is limited to the carcinoma, including carcinoma in situ component, with the background normal urothelium showing no SV40 staining or p53 overexpression. A similar pattern of SV40 positivity with or without accompanying aberrant p53 has been reported in several other BK virus-associated tumors.1,7,14,15 Of note, the SV40 positivity is maintained in the lung metastasis as well, as also noted in some other reports.8,16 Further, NGS did not identify a TP53 mutation in our tumor, strengthening the argument that virus-derived T-Ag is causing the abnormal expression of p53. This supports the hypothesis that BK virus is playing a role in driving tumor cell division in these tumors, not merely indiscriminately infecting background cells that may randomly undergo tumorigenesis via an unrelated process. Although the utility of SV40 IHC for identifying viral involvement has been called into question by some, particularly 1 study examining gastrointestinal tumors, 17 SV40 (T-Ag) IHC positivity in the context of posttransplant urinary tract carcinomas in immunosuppressed patients is highly concordant with actual BK viral integration into the tumor genome. 14
As seen in our patient, TERT promoter mutations have been reported in many BK virus-associated carcinomas.14,15 HER2 amplification has also been reported. 15 Initially, this may be unsurprising since TERT promoter and HER2 mutations are common in urothelial carcinomas generally, alongside TP53, PIK3CA, TSC1, FGFR3, and HRAS mutations. 18 However, it is notable that, as in our case, these mutations frequently occur in isolation or with few associated pathogenic/likely pathogenic mutations.14,15 On average, BK virus-associated carcinomas have fewer mutations identified per a tumor versus comparable non-BK virus-associated tumors.14,15 In a small comparison of 30 urinary tract carcinomas (8 BK virus-associated carcinomas in immunosuppressed patients, 4 non-BK virus-associated carcinomas immunosuppressed patients, and 18 carcinomas non-immunosuppressed patients), Sirohi et al. found that non-BK virus-associated carcinomas demonstrated 2 to 4 times as many pathogenic/likely pathogenic mutations per tumor on average compared to the less complex genetic alterations of BK virus-associated carcinomas. 15 This may further support the assertion that T-Ag is directly interfering with tumor suppressor proteins to drive tumorigenesis, compared to traditional tumors, which result from a culmination of oncogenic mutations.
In conclusion, BK virus-associated carcinoma arising in the allograft in kidney transplant recipients is uncommon. However, the growing number of reports describing this entity raises the possibility that this entity is more common than previously thought and may even go undetected and be underreported in immunosuppressed populations. Latent BK virus infection in urinary epithelium is known to be reactivated in immunosuppression, causing nephropathy and other diseases. It is hypothesized that reactivation of BK virus may also lead to tumorigenesis driven largely via T-Ag protein inactivation of pRB and p53 proteins. Fortunately, most patients with BK virus-associated carcinomas arising in the allograft respond well to allograft nephrectomy and cessation of immunosuppression, and immunotherapy may be a good option for the smaller subset of refractory cancers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any studies with human or animal subjects.
Data Availability Statement
Data sharing is not applicable – no new data are generated.
