Abstract
Urinary bladder hamartoma is a rare benign proliferation with only 14 cases reported in the literature at present. Urinary bladder hamartoma is composed of a disorderly admixture of normal urinary bladder components, essentially represented by glands lined by transitional epithelium and a variable percentage of fibrous stroma, smooth muscle bundles, and adipose tissue. Urinary bladder hamartomas do not exhibit cytological or architectural abnormalities and show no necrosis or increase in mitotic activity. Clinical manifestations are usually represented by lower urinary tract symptoms, more or less frequently paired with gross hematuria. Several pediatric cases of urinary bladder hamartoma have been reported, sometimes with syndromic associations. Transurethral resection has been curative in all cases reported, with no evidence of recurrence. Here we report an additional rare urinary bladder hamartoma, clinically mimicking urothelial carcinoma, providing a review of the literature regarding this unusual entity.
Introduction
The term “hamartoma” originally introduced by Albrecht in 1904, 1 refers to a benign, although “tumor-like” exaggerated proliferation of elements that recapitulates the normal components of the tissue in which the hamartoma arises. 2 Among various sites where hamartomas have been described in the literature, urinary bladder localization is extremely rare, with only 14 reports in the literature (see Table 1).
A Systematic Review of Urinary Bladder Hamartoma Published Cases to Date.
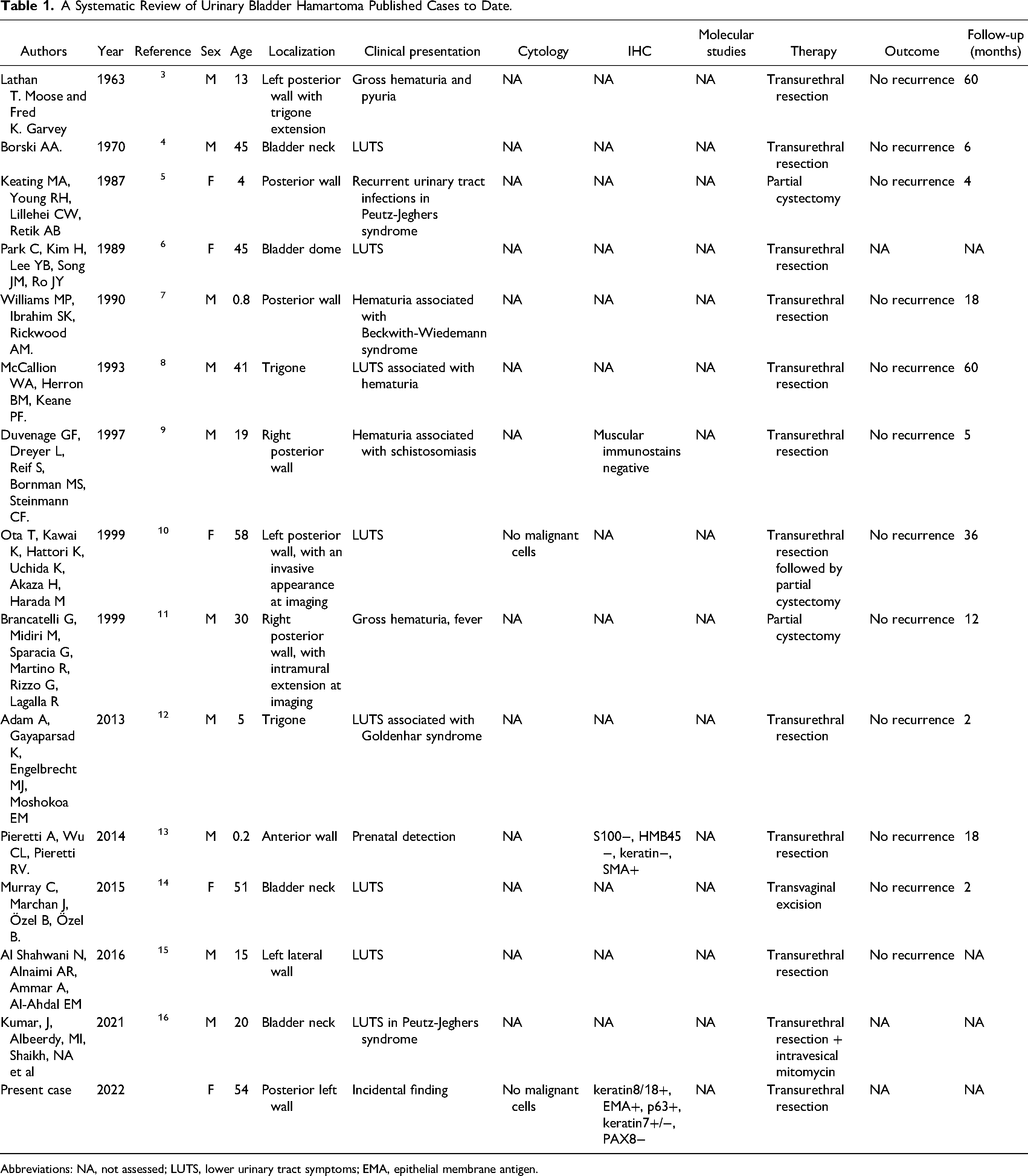
Abbreviations: NA, not assessed; LUTS, lower urinary tract symptoms; EMA, epithelial membrane antigen.
Here we describe an additional case of urinary bladder hamartoma, clinically interpreted at first as urothelial carcinoma, and we provide a detailed revision of the published literature regarding this rare entity.
Case Presentation
A 54-year-old woman with type IV Loeys-Dietz syndrome was admitted to the Nephrology Unit of our Institution for rapid onset of left flank pain with gross hematuria and elevated inflammatory markers, suspicious for left pyelonephritis. Past medical history revealed a recent aortic dissection, pulmonary emphysema, gastroesophageal reflux, and uterine leiomyomas. During hospitalization, the patient responded well to antibiotic therapy with piperacillin/tazobactam. Angio-computed tomography (CT) excluded the presence of renal infarction, while abdomen CT revealed left kidney alterations consistent with pyelonephritis complicated with renal abscesses. During the diagnostic workup for pyelonephritis, abdomen ultrasonography (US) and computed tomography (CT) with contrast revealed an 8-millimeter-wide vegetation located on the left posterior wall of the urinary bladder, which exhibited mild contrast enhancement. The lesion was considered to be at least suggestive of urothelial carcinoma. Urine cytology was negative for neoplastic cells. After acute pyelonephritis resolution, the patient underwent a cystoscopy that revealed an oval and pedunculated formation located between the trigone area and the left posterior wall, which was therefore excised by transurethral resection to exclude malignancy. At histopathological examination (Figure 1), the lesion revealed a sessile/nodular silhouette and was composed of an admixture of tubules and cystically dilated glands lined by mono- or pseudostratified urothelium and containing eosinophilic proteinaceous material, with no architectural nor cytological atypia. The tubulo-glandular elements were dispersed within a fibrotic stroma with scattered smooth muscle bundles, numerous plump fibroblasts, and increased vessel density. No necrosis nor mitotic activity was observed. Immunohistochemistry (Figure 2) revealed strong positivity for keratin 8/18, epithelial membrane antigen (EMA), keratin AE1/AE3, and p63, negativity for PAX8 and CD10, and faint/focal expression of keratin 7. The proliferative index, evaluated with MIB1/Ki67 antibody, was extremely low (< 1%).
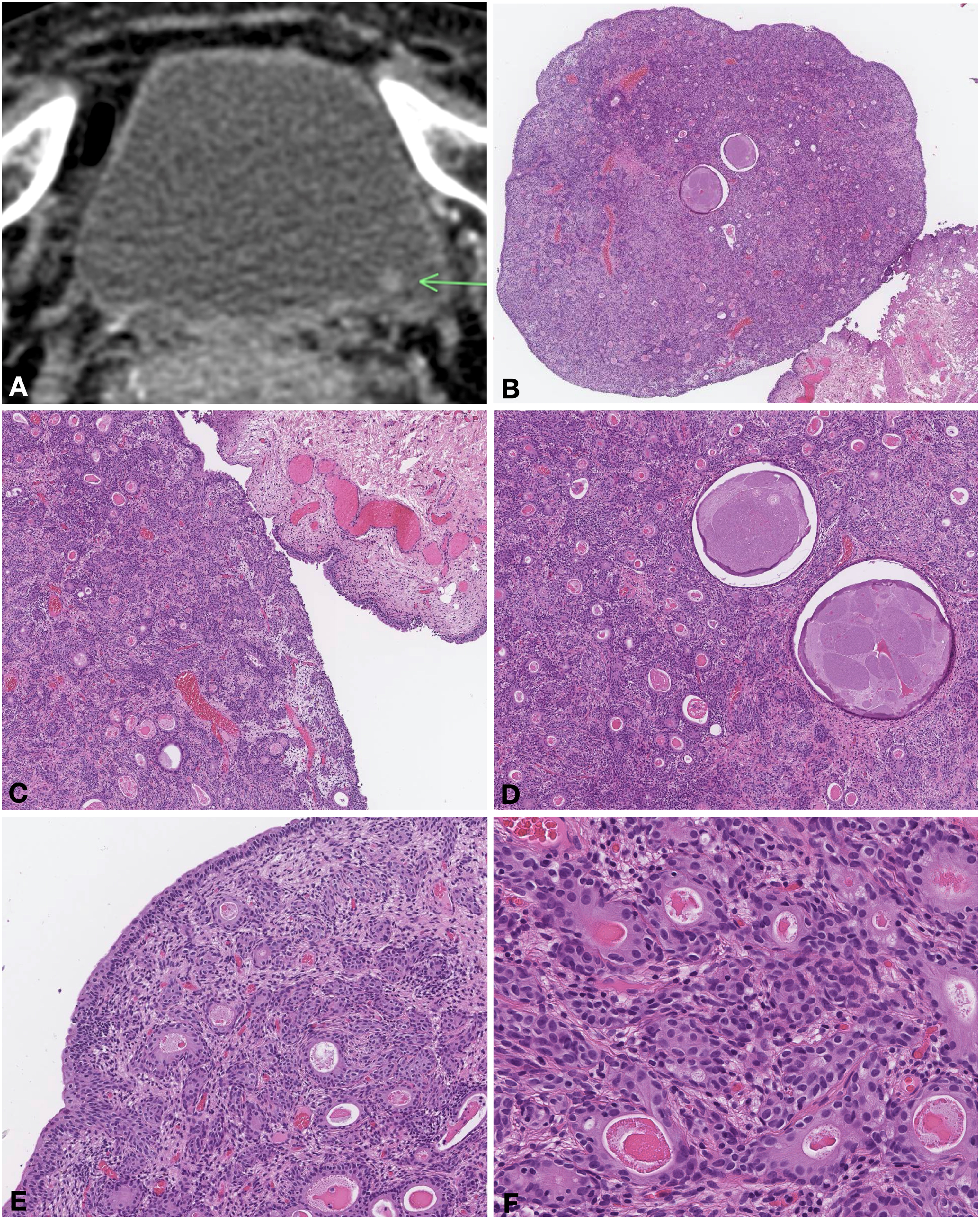
Contrast CT revealed an 8-millimeter-wide vegetation located on the left posterior wall of the urinary bladder (A, arrow). The lesion showed a nodular silhouette (B, H&E stain, 4 × magnification) and was composed of tubulo-glandular structures, often cystically dilated, lined by a single or stratified urothelium and filled with proteinaceous material, in a background of fibrotic stroma with congested capillaries, scattered smooth muscle bundles and plump fibroblasts (C-D, H&E stain, 10 × magnification; E, H&E stain, 20 × magnification; F, H&E stain, 40 × magnification).
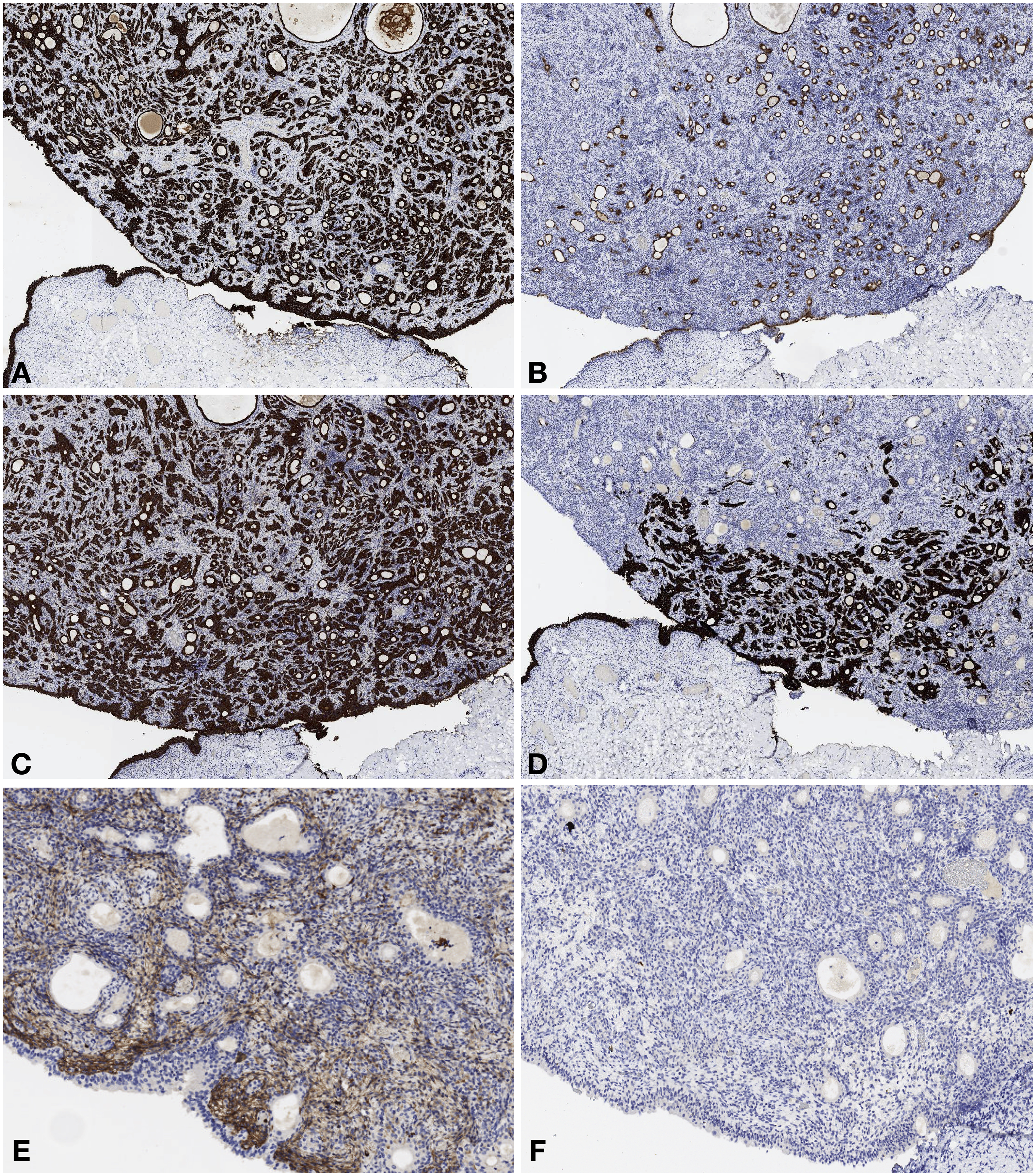
Hamartoma urothelium reveals strong positivity for keratin AE1/AE3 (A, 10 × magnification), EMA (B, 10 × magnification), and keratin 8-18 (C, 10 × magnification), focal expression of keratin 7 (D, 10 × magnification) and negativity for CD10 (E, 20 × magnification) and PAX8 (F, 20 × magnification).
Telomerase reverse transcriptase (TERT) promoter mutation analysis, performed with the Amoy Diagnostics® kit, revealed a wild-type configuration, arguing against an urothelial carcinoma diagnosis altogether with the above-mentioned features.
Given these findings, a urinary bladder hamartoma diagnosis was made.
Discussion
To systematically review the published literature regarding urinary bladder hamartoma, we performed a PubMed search using the terms: “urinary bladder hamartoma” and “bladder hamartoma,” which showed 89 results. Of these, 14 were reports of urinary bladder hamartoma, for a total of 15 individual cases (the present included), whose clinic-pathological characteristics are summarized in Table 1. The very first bladder hamartoma reported by Davis in 1949 17 was subsequently diagnosed as urinary bladder nephrogenic adenoma and was therefore excluded from our revision.
Most reported hamartomas affected male patients (10/15; 67%) with a male-to-female ratio of 2 and a mean age at diagnosis of 30 years. Bladder posterior wall was the most common localization (7/15, 47%), followed by bladder neck (3/15, 20%), trigone (2/15, 13%), anterior wall (1/15, 6.7%), left lateral wall (1/15, 6.7%), and bladder dome (1/15, 6.7%).
Clinical features were represented by irritative lower urinary tract symptoms with or without gross hematuria (8/15, 53%); gross hematuria alone was observed in 27% of patients (4/15). One case was associated with urinary bladder schistosomiasis, 9 while another one was the subject of an in-utero prenatal diagnosis. 13 Interestingly, 33% of lesions (5/15), including the present one, arose in the context of specific syndromes, such as Peutz-Jeghers syndrome,5,16 Beckwith-Wiedemann syndrome, 7 Goldenhar syndrome, 12 and, in our patient, Loeys-Dietz syndrome (LDS). LDS is an autosomal dominant connective disorder characterized by systemic involvement, mainly due to transforming growth factor beta (TGFB) ligand or receptor mutations. LDS encompasses craniofacial, skeletal, cutaneous, and vascular abnormalities, partly overlapping with Marfan syndrome manifestations: distinguishing LSD features are cleft palate/bifid uvula, hypertelorism, and arterial tortuosity. LSD is divided into 5 subtypes defined by different molecular alterations, with type IV LSD specifically bearing TGFB2 mutations and showing a milder phenotype.18,19 To the best of our knowledge, no association of hamartomas with LSD has been reported to date.
All lesions shared a common architectural and morphologic appearance: a lobulated mass composed of a mixed proliferation of tubulo-glandular, nested, or even papillary epithelial components intermingled with smooth muscle bundles, fibromyxoid stroma with plump fibroblasts, and sometimes adipose tissue. The nested or tubule-glandular elements were often cystically dilated, lined by a single or stratified urothelium with no atypia, and filled with proteinaceous material, resembling florid von Brunn nest hyperplasia or cystitis cystica glandularis. Intestinal metaplasia and hypervascularity were other potential features observed in these lesions.10,14‐16 No mitoses nor necrosis were observed.
Aside from the present case, immunohistochemical characterization has been performed only on 2 other hamartomas, one which showed negativity of muscular markers 9 and one which was positive for SMA and negative for S100, HMB45, and keratins. 13
Urine cytology was available only for 2 patients (the present included) and revealed no significant alterations. No molecular data is available regarding urinary bladder hamartomas.
Differential diagnoses of urinary bladder hamartoma may include von Brunn hyperplasia, cystitis cystica glandularis, nephrogenic adenoma, inverted urothelial papilloma, and nested/microcystic variant of urothelial carcinoma, particularly in cases with apparent infiltrative growth at imaging studies.10,11,20 Differential diagnosis with florid von Brunn hyperplasia and cystitis cystica glandularis might be problematic, although it is not clinically relevant as they all represent benign lesions. 21 Distinguishing features of urinary bladder hamartoma might be represented by the markedly exophytic and lobulated architecture, the presence of plump fibroblasts within the fibromyxoid stroma, and prominent hypervascularity. Florid von Brunn hyperplasia and cystitis cystica glandularis are usually a response to chronic inflammation and irritation, such as prolonged catheterization. 22 Our patient was catheterized during hospitalization; however, the urinary bladder lesion was found concomitantly with pyelonephritis, and we exclude a causative relationship with such a short-term catheterization.
Nephrogenic adenoma is a benign lesion presumably derived from renal tubular cells and related to prior inflammatory insult, which can exhibit a tubular, tubule-cystic, polypoid, and/or papillary growth pattern. Nephrogenic adenoma is composed of tubules of variable size with the basement membrane, luminal eosinophilic secretion, abundant intervening stroma, and cystic dilations. Despite its similarities with urinary bladder hamartoma, nephrogenic adenoma exhibits striking immunohistochemical positivity for AMACR, CD10, PAX8, EMA, keratin 7, and aquaporin-1, while it is negative for p63 and keratin 20.23–26 Our case exhibited strong p63 positivity and CD10 and PAX8 negativity, thus arguing against a nephrogenic adenoma diagnosis.
Inverted urothelial papillomas are endophytic benign neoplasms that exhibit a pushing growth pattern into the lamina propria. While they usually manifest cystoscopically as raised lesions, their histological examination reveals typical anastomosing urothelial cords of normal thickness with unequivocal endophytic growth and no necrosis, mitotic activity, or cytological atypia. Inverted urothelial papillomas are keratin 20 negative and show a low proliferative index evaluated by means of MIB1-Ki67 expression; their molecular analysis reveals KRAS or HRAS mutations.27–30 In our case, architectural and morphological features, paired with the absence of an endophytic growth pattern, were sufficient to exclude an inverted papilloma.
Nested and tubular/microcystic variants of urothelial carcinoma may mimic urinary bladder hamartoma (as well as florid von Brunn nests hyperplasia) due to their nested/microcystic architecture and bland cytology; clear evidence of infiltrative growth is of pivotal importance in the identification of these deceptive variants of urothelial carcinoma. In complex cases, as in superficial biopsies, the identification of TERT promoter mutation is of great help in diagnosing malignancy, since it is deemed the most common molecular alteration among urothelial carcinomas.30‐33 Our lesion did not exhibit infiltrative growth nor TERT promoter mutations, thus arguing against such a diagnosis. Urinary bladder rhabdomyosarcoma might also be included in the spectrum of radiological differential diagnoses, especially in pediatric patients. 5
All patients were effectively cured by complete excision via transurethral resection, partial cystectomy, or transvaginal excision; in one instance 16 simultaneous administration of intravesical mitomycin was performed in the suspicion of malignancy. No recurrence of the disease was reported, even after follow-up periods of up to 60 months.
In conclusion, urinary bladder hamartoma might be the underlying etiologic factor in a small percentage of patients experiencing gross hematuria and/or lower urinary tract symptoms. Urinary bladder hamartoma's radiological and cystoscopic features may sometimes rise suspicion of malignancy. On the other hand, its rarity and benign pathological features, resembling von Brunn nest hyperplasia or cystitis cystica glandularis, might hamper its identification, making it a potentially underdiagnosed condition. Definitive diagnosis mandatorily requires pathological examination and thorough clinical correlation, sometimes, as in our case, with the aid of molecular investigations to exclude malignancy. Of interest, its consistent yet unexplained association with certain syndromic clinical pictures might also be a useful hint for this diagnosis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
