Abstract
Superficial CD34 positive fibroblastic tumor is a rare low-grade neoplasm of the skin and subcutis with indolent behavior. This entity has been included in the current World Health Organisation (WHO) classification of soft tissue tumors. Pathological diagnosis can be challenging due to significant morphological overlap with other entities and the large spectrum of CD34 positive tumors. We report a case in a twenty-five male which showed characteristic diagnostic features, but in addition showed myxoid stroma. The presence of myxoid stroma has not been previously emphasized in this entity and broadens the histologic differential diagnosis significantly to include myxoid soft tissue tumors. A subset of these tumors harbor PRDM10-rearrangements, but a defining molecular feature has not yet been described, highlighting the need for further molecular characterization of this potentially genetically heterogenous tumor. Awareness of this entity among surgeons and pathologists is important to prevent misclassification as an aggressive sarcoma and avoid over-treatment.
Introduction
Superficial CD34 positive fibroblastic tumor has been included in the fifth edition of the World Health Organisation (WHO) classification of soft tissue and bone tumors following its first description in 2014.1,2 It typically occurs in young adults as a superficial soft tissue lesion of the lower extremities and has an excellent prognosis. 1 To date, less than forty cases have been reported. No characteristic molecular alteration has been described in this entity. We report a tumor with myxoid stroma as a novel finding in addition to the defining histologic features.
Case Report
A twenty-five-year-old previously well man presented with a soft tissue mass on the right buttock, which had recently shown accelerated growth. The lesion was superficially located, fixed to the skin, and measured ten centimeters in diameter. The clinical differential diagnosis included a deep-seated hematoma, liposarcoma and peripheral nerve sheath tumor. A magnetic resonance imaging (MRI) scan was performed, which confirmed the presence of a 9.3 × 9.6 × 9 cm well circumscribed encapsulated solid mass lesion in the gluteal subcutaneous tissue with no infiltration into skin, gluteus maximus muscle or neurovascular structures (Figure 1, T1 fat-saturated post-contrast MRI). The mass showed peritumoral vascularization, was hypointense on T1 and heterogeneously hyperintense on proton density weighted sequence with incomplete peripheral enhancement, indicating a possible sarcomatous lesion. A core needle biopsy showed a CD34 positive spindle cell lesion with scattered pleomorphic cells. The final diagnosis was deferred to the excision specimen. A CT scan of the chest did not show lung metastases.
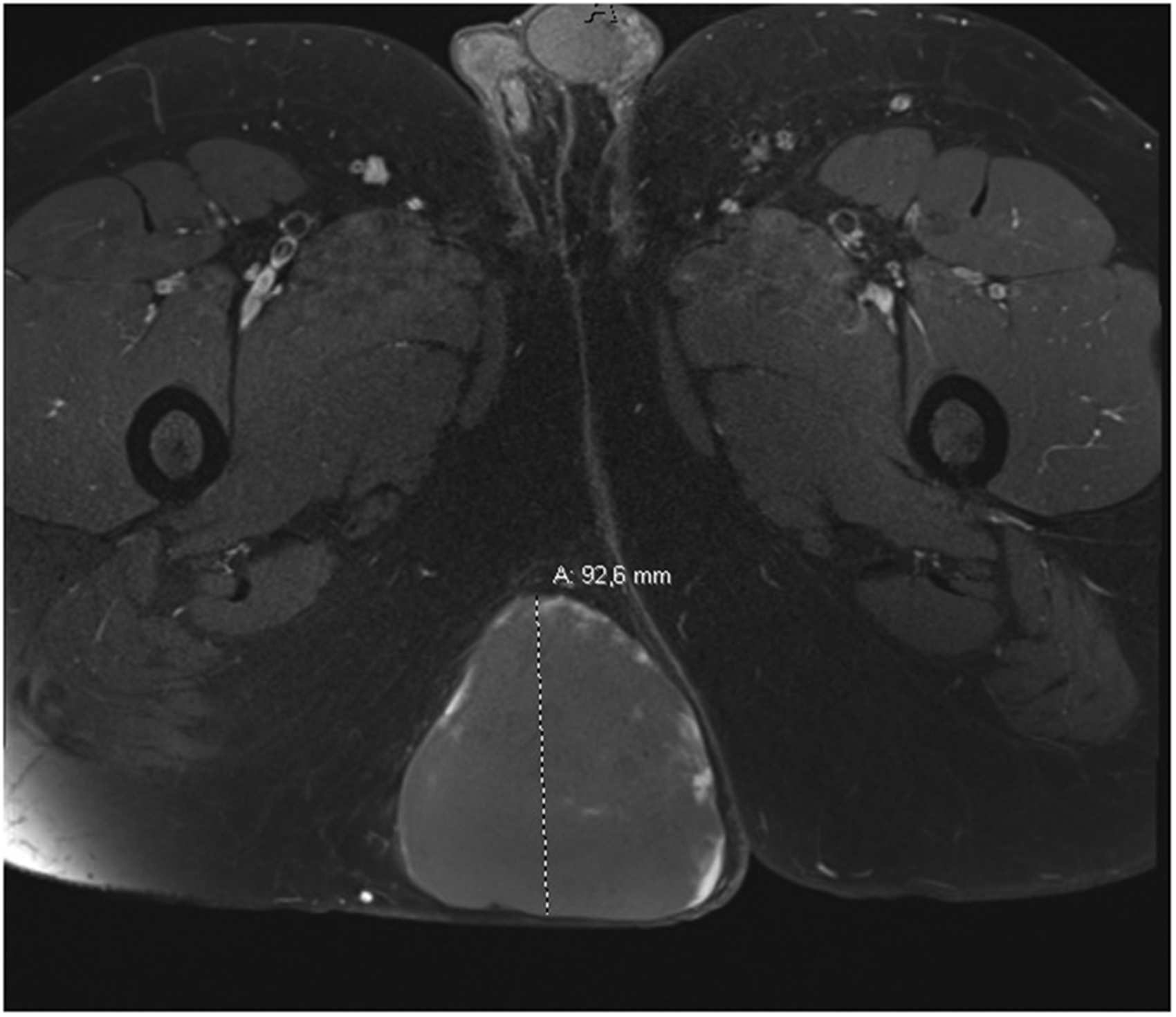
Magnetic resonance image showing a superficial soft tissue mass in the right gluteal region.
The lesion was resected with wide margins and an overlying ellipse of skin. On cut section, the lesion was well circumscribed, solid, tan colored and had a whorled appearance (Figure 2). Clinical follow-up of the patient has shown no local recurrence or distant metastases more than a year following excision.
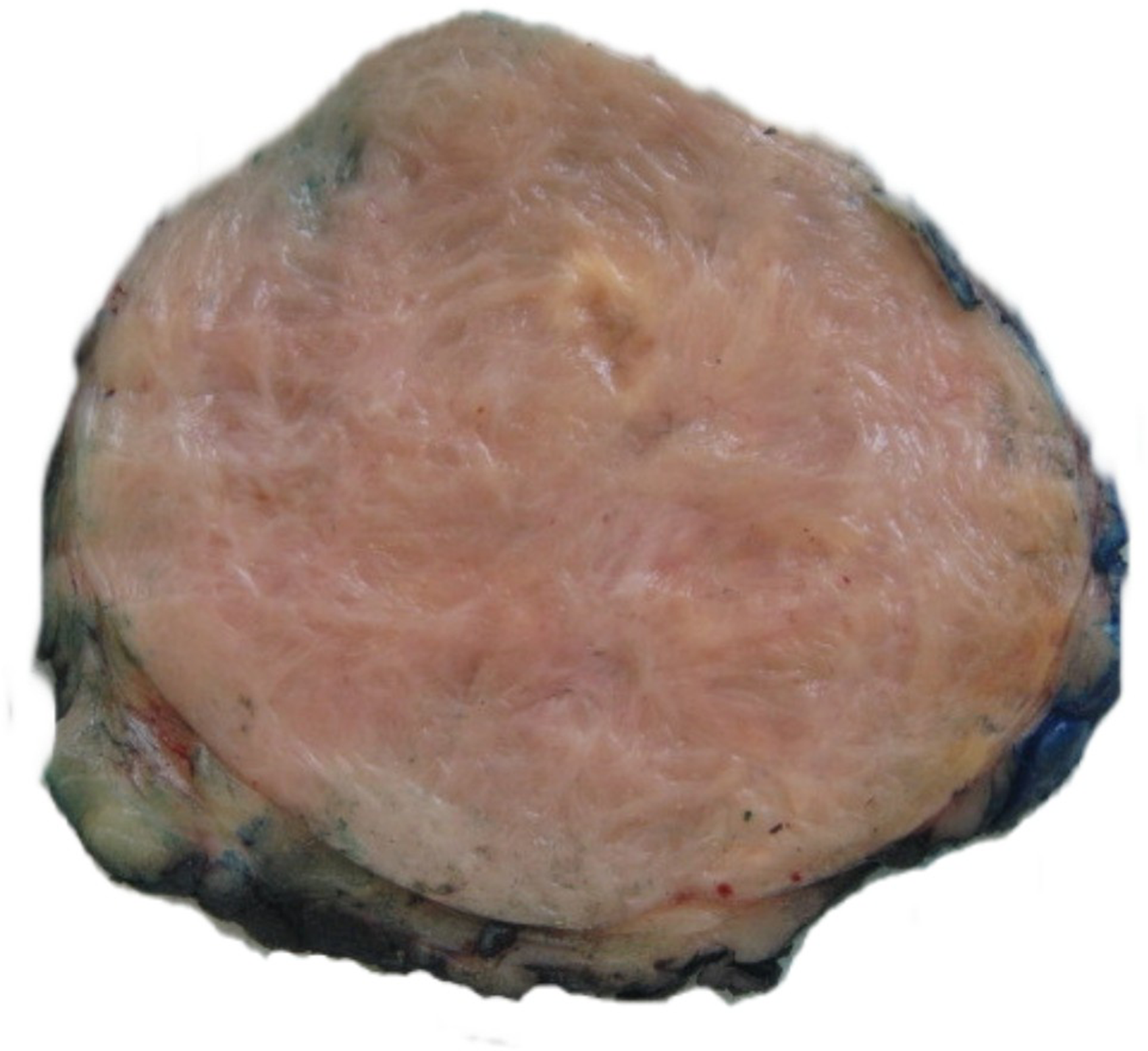
Cut section of the resection specimen shows a well circumscribed solid fibrous mass.
Findings
Histology of the specimen showed a well-circumscribed lesion located in the subcutis and deep dermis with no infiltrative growth (Figure 3A). The lesion comprised spindled cells with a fascicular and haphazard growth pattern (Figure 3A). An alcian blue periodic acid Schiff stain (ABPAS) highlighted myxoid stroma but pools of mucin or myxoid nodules were absent. The spindled cells revealed fibroblastic-like cytology with pin-point nucleoli and amphophilic cytoplasm. Scattered pleomorphic cells with more abundant granular eosinophilic cytoplasm and large irregular nuclei occurred throughout the lesion (Figure 3B, C & D). Sparse inflammatory cells, including lymphocytes and mast cells, were present. The vasculature was inconspicuous, and mitotic figures and necrosis were absent.
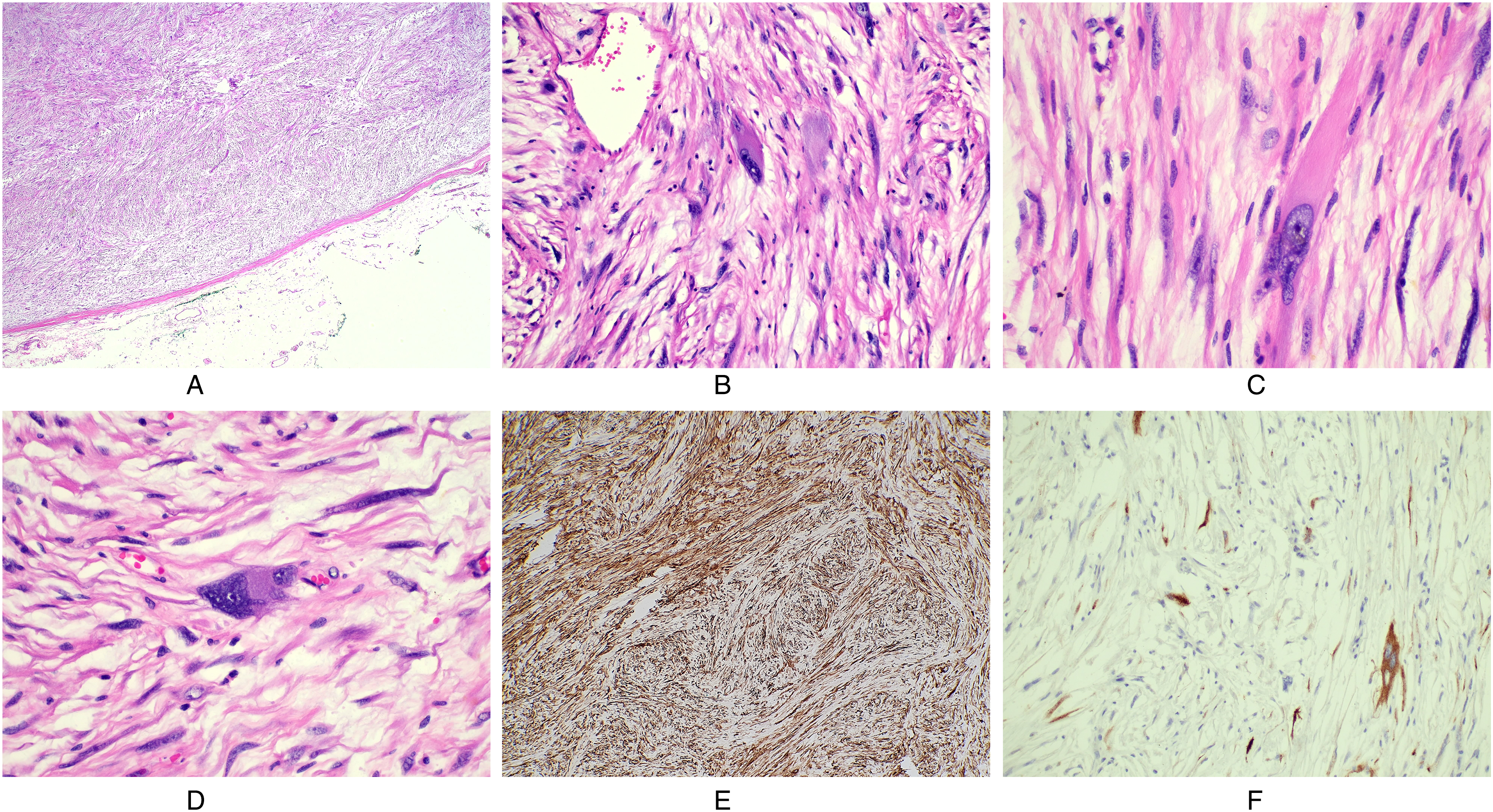
A: A spindle cell lesion with fascicular growth, myoid stroma and scattered larger pleomorphic cells is seen from low power. The well circumscribed nature of the lesion is demonstrated, hematoxylin and eosin stain, 2x. B,C and D: Scattered pleomorphic cells with abundant eosinophilic cytoplasm are present throughout the lesion, hematoxylin and eosin 40x. E: Diffuse strong CD34 immunohistochemical expression is present in the lesion. F: AE1/3 keratin staining highlights some of the cells within the lesion.
Immunohistochemistry showed positive staining of the spindled cells with CD34 and patchy positive staining (1%–5% of cells) with a pan-keratin (AE1/3) (Figures 3E and 3F). The spindled cells were negative for S100, desmin, EMA, STAT6, CD31, and MUC4, with very focal weak staining for SMA. The constellation of findings was consistent with a superficial CD34 positive fibroblastic tumor. The differential diagnoses included low-grade myxofibrosarcoma, myxoinflammatory fibroblastic sarcoma (MIFS), inflammatory myofibroblastic tumor (IMT), low-grade fibromyxoid sarcoma (LGFMS), pleomorphic hyalinising angiectatic tumor (PHAT), pseudomyogenic hemangioendothelioma and solitary fibrous tumor (SFT). These entities were excluded based on clinical, morphological and immunohistochemical grounds. Due to the CD34 positivity, a myxoid dermatofibrosarcoma protuberans (DFSP) was excluded by the lack of invasive growth, and FISH for t(17;22)(q22;q13) (COL1A1;PDGFB).
Discussion
Superficial CD34 positive fibroblastic tumor was first described in 2012 by Carter et al and has been included in the current WHO classification of soft tissue tumors.1,2 Over forty cases have been described to date.1,3–6 The essential and desirable diagnostic features are a “superficial location; large eosinophilic cells with granular to glassy cytoplasm; marked nuclear pleomorphism but a very low mitotic count; diffuse CD34 expression and frequent keratin immunoreactivity”. 2 No characteristic molecular feature was included in the diagnostic criteria. The tumor described here meets these diagnostic criteria but also shows myxoid change in the stroma, a feature not previously emphasized in this entity. While myxoid stroma is not specifically mentioned in the initial description of the lesion by Carter et al, they included in their differential diagnosis other tumors with myxoid stroma such as myxofibrosarcoma, MIFS, PHAT, and LGFMS. 1 Partial myxoid stroma was also described in a case reported by Li et al 7 The presence of myxoid stroma could be a diagnostic pitfall in diagnosing this entity, as many of the differential diagnostic considerations are characterized by myxoid stroma.
PRDM10 fusion with CITED2 or MED12 was described in three low-grade undifferentiated pleomorphic sarcomas in 2015, with nine further cases described in 2019.8,9 There is significant morphological and immunohistochemical overlap between these two entities which have not been separated in the current WHO classification. 2 Of note, myxoid stroma was present in eight of twelve PRDM10-rearranged soft tissue tumors.8,9 Puls et al investigated a cohort of seven superficial CD34 positive fibroblastic tumors for underlying molecular alterations and found PRDM10 rearrangements in three cases. 9 Four cases showed no PRDM10 rearrangements and one case had a t(2;5)(q31;q31) translocation. 9 Their findings suggest that superficial CD34 positive fibroblastic tumor is genetically heterogenous and further investigation of this entity is warranted. 9
A low-grade myxofibrosarcoma was excluded in the current tumor based on the age of the patient, the lack of characteristic curvilinear vasculature, and the well-circumscribed nature of the lesion with strong diffuse CD34 positivity and focal keratin staining. MIFS occurs at acral sites, displays myxoid nodules and pseuodolipoblasts and is typically CD34 negative. 10 IMT lacks such widespread pleomorphic spindled cells and is CD34 negative and often desmin positive. 11 LGFMS demonstrates MUC4 positivity in 99% of cases and shows collagenous hypocellular nodules alternating with cellular myxoid nodules. 12 SFT lacks the pleomorphism seen in this tumor and is STAT6 positive.13,14 PHAT is positive with CD34 but demonstrates hyalinised ectatic vasculature with prominent haemosiderin deposition. 15 DFSP typically lacks scattered pleomorphic cells and is characterized by t(17;22) COL1A1-PDGFRA fusion which was absent in this tumor. The recently described superficial ALK-rearranged myxoid spindle cell neoplasm was excluded based on negative ALK immunohistochemistry. 16
Conclusion
Superficial CD34 positive fibroblastic tumor is a relatively newly described entity with a superficial or supra-fascial location, spindled cell morphology with marked pleomorphism and diffuse CD34 expression. 1 The lesion lacks mitotic activity, has a low proliferation index, and has an excellent prognosis with only one reported case of lymph node metastases. 1 This entity should be included in the differential diagnosis for a CD34 positive soft tissue tumor. In addition to the characteristic features of this entity, we describe the occurrence of myxoid stroma as a potential diagnostic pitfall as many of the differential diagnoses of this lesion demonstrate myxoid stroma.
Footnotes
Author Contributions
All authors have contributed to the production of this manuscript and have seen and approved the content.
The authors, hereby declare that this manuscript has not been submitted or accepted elsewhere for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
HREC approval was obtained.
Informed Consent
Written informed consent was obtained from the patient.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
