Abstract
To evaluate the efficiency of pan-keratin immunostaining, tissue microarrays of 13,501 tumor samples from 121 different tumor types and subtypes as well as 608 samples of 76 different normal tissue types were analyzed by immunohistochemistry. In normal tissues, strong pan-keratin immunostaining was seen in epithelial cells. Staining intensity was lower in hepatocytes, islets of Langerhans, and pneumocytes but markedly reduced in the adrenal cortex. Pan-keratin was positive in ≥98% of samples in 62 (83%) of 75 epithelial tumor entities, including almost all adenocarcinomas, squamous cell and urothelial carcinomas. Only 17 of 121 tumor entities (13%) had a pan-keratin positivity rate between 25% and 98%, including tumors with mixed differentiation, endocrine/neuroendocrine tumors, renal cell carcinomas, adrenocortical tumors, and particularly poorly differentiated carcinoma subtypes. The 15 entities with pan-keratin positivity in 0.9%-25% were mostly of mesenchymal origin. Reduced/absent pan-keratin immunostaining was associated with high UICC stage (p = 0.0001), high Thoenes grade (p = 0.0183), high Fuhrman grade (p = 0.0049), advanced tumor stage (p < 0.0001) and lymph node metastasis (p = 0.0114) in clear cell renal cell carcinoma, advanced pT stage (p = 0.0007) in papillary renal cell carcinoma, and with advanced stage (p = 0.0023), high grade (p = 0.0005) as well as loss of ER and PR expression (each p < 0.0001) in invasive breast carcinoma of no special type (NST). In summary, pan-keratin can consistently be detected in the vast majority of epithelial tumors, although pan-keratin can be negative a fraction of renal cell, adrenocortical and neuroendocrine neoplasms. The data also link reduced pan-keratin immunostaining to unfavorable tumor phenotype in in epithelial neoplasms.
Introduction
Pan-keratin antibodies are mixtures of two or several antibodies that detect multiple low and high molecular weight keratins. These antibody cocktails have been designed to immunohistochemically detect all epithelial cell types irrespective of their tissues of origin with one single diagnostic tool. In surgical pathology they are typically employed to document the epithelial origin of neoplastic or non-neoplastic tissue or for detection of small metastases in lymph nodes. There are, however, limitations to the concept that pan-keratin antibodies stain all epithelial tumors and that non-epithelial tissues are “keratin negative”. For a large variety of different epithelial tumors, pan-keratin negative tumors have been described 1–6 and “keratin positive” mesenchymal tumors have also been reported across various mesenchymal tumor entities.7–12 The frequencies reported for pan-keratin negative carcinomas and pan-keratin positive non-epithelial tumors varies considerably in the literature, however. For example, pan-keratin positivity has been described in 15% to 45% of hepatocellular carcinoma carcinomas,3,5 0% to 95% of adrenocortical carcinomas,2,13,14 33% to 100% of clear cell15,16% and 73% to 100% of papillary renal cell carcinomas,17,18 20% to 78% of angiosarcomas,19,20 and 17% to 100% of leiomyosarcomas.21,22 These conflicting data may be caused by the use of different antibodies, immunostaining protocols, and criteria to determine “positivity” in these studies.
To generate a comprehensive dataset on the prevalence of pan-keratin positivity in epithelial and non-epithelial neoplasms, a set of tissue microarray (TMAs) was analyzed in this study that contained more than 15,500 tumor tissue samples from 121 different tumor types and subtypes as well as 76 different non-neoplastic tissue types.
Materials and Methods
Results
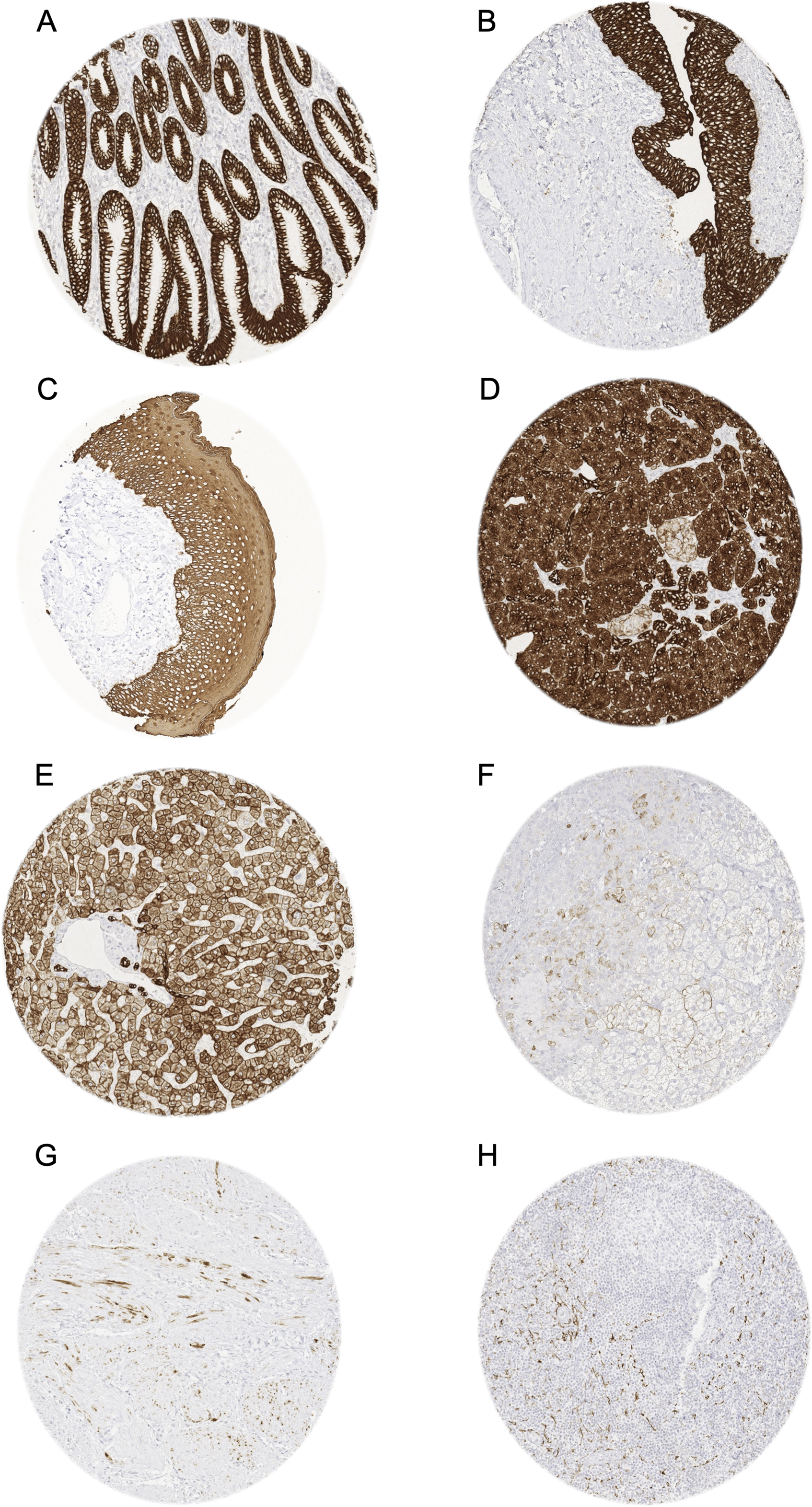
Pan-keratin immunostaining of normal tissues. The panels show a strong pan-keratin positivity of epithelial cells of the stomach (A), the urothelium of the urinary bladder (B), and the squamous epithelium of the oral cavity (C). In the pancreas, acinar cells show a strong staining while cells of islets of Langerhans stain only weakly (D). In the liver pan-keratin staining is variable (weak to moderate) in hepatocytes but strong in bile ducts (E). In the adrenal gland, only a subset of cortical cells shows a weak staining (F). In the myometrium, groups of spindle-shaped pan-keratin positive cells are found (G). In lymph nodes, a delicate fibrillar staining caused by fibroblastic reticulum cells occurs mainly in the interfollicular area (H).
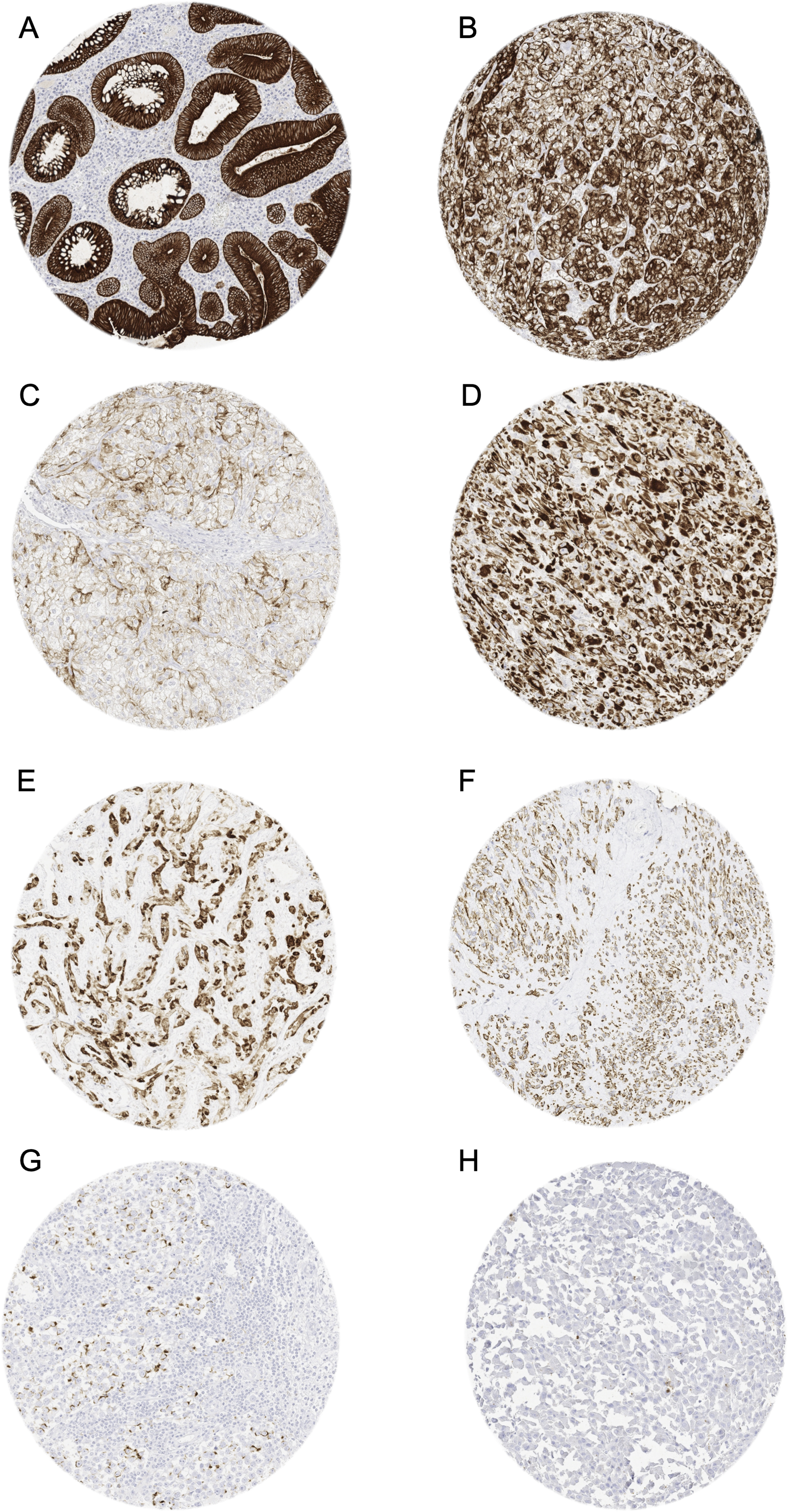
Pan-keratin immunostaining in tumors. The panels show a cytoplasmatic pan-keratin immunostaining of variable intensity in samples from a colorectal adenoma (A: strong staining), two clear cell renal cell carcinomas (B: strong; C: weak), a sarcoma NOS (D: strong), an angiosarcoma (E: strong), a gastrointestinal stromal tumor (GIST) (F: moderate), and a seminoma (G: weak). Pan-keratin immunostaining is lacking in an adrenocortical carcinoma (H).
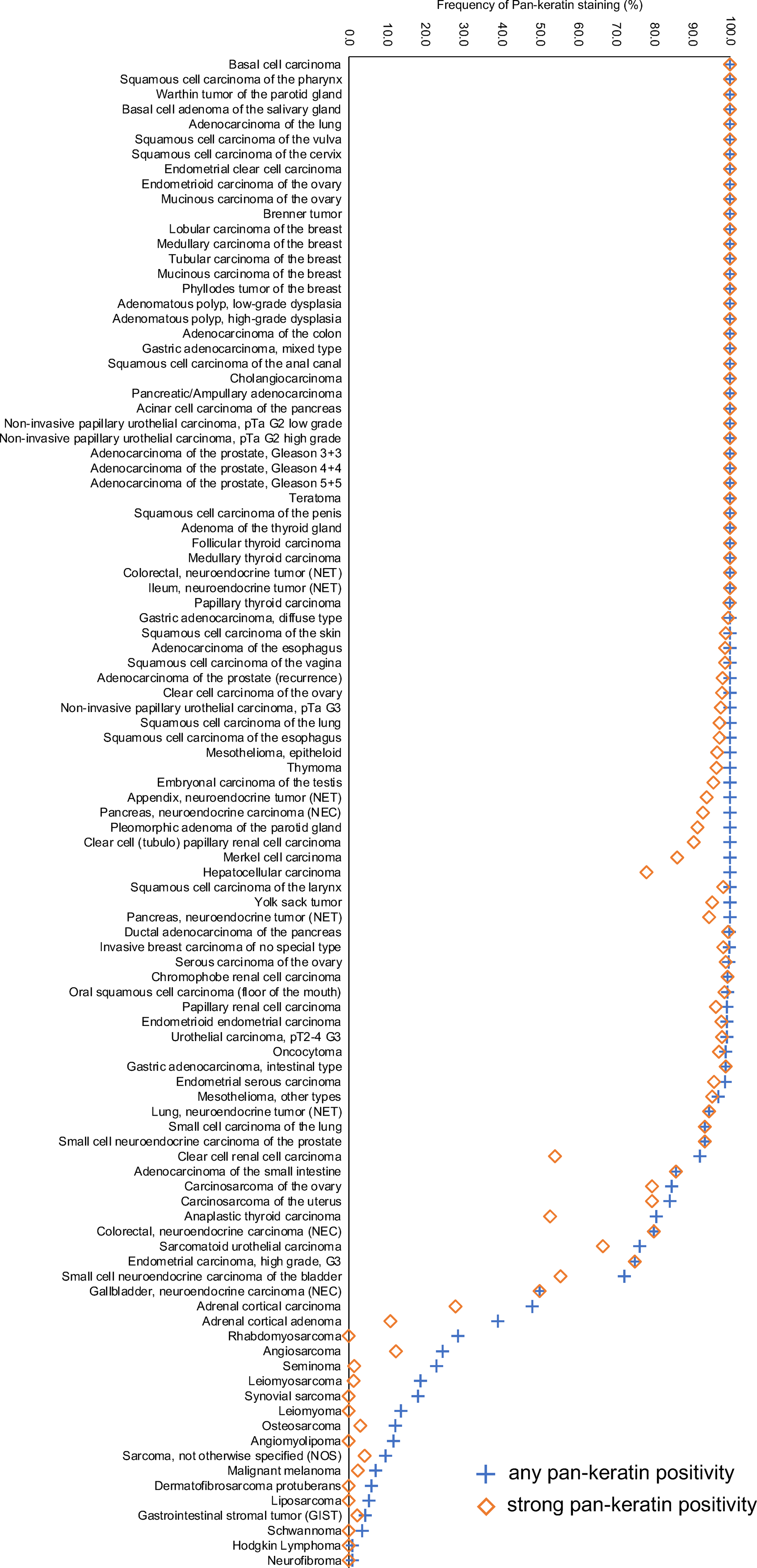
Ranking order of pan-keratin immunostaining in tumors. Both the frequency of positive tumors (blue cross) and the frequency of strongly positive tumors (orange rhombus) are shown.
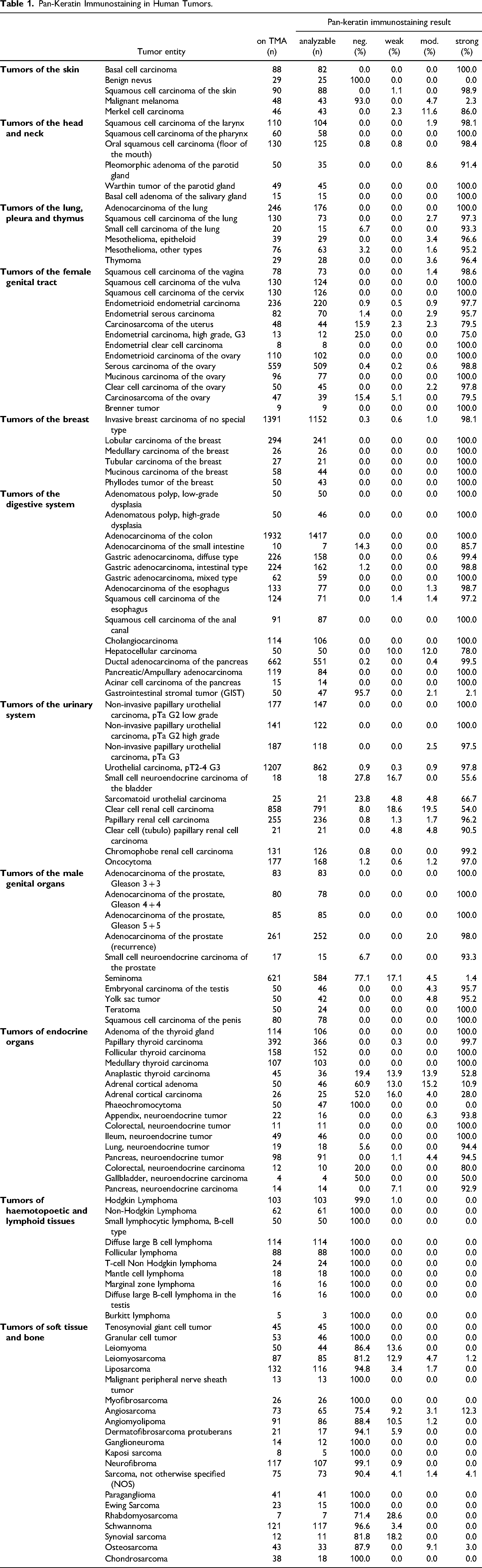
Pan-Keratin Immunostaining in Human Tumors.
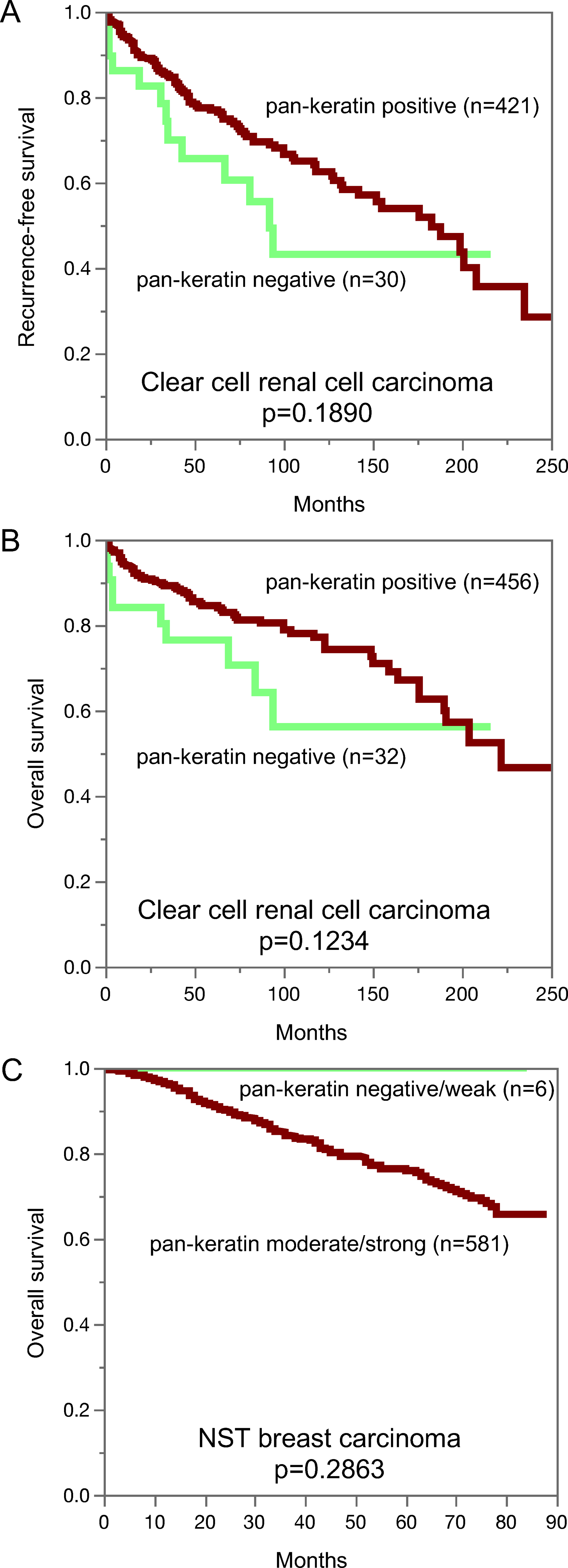
Pan-keratin immunostaining and recurrence-free survival (A) and overall survival (B) in clear cell renal cancer. Pan-keratin immunostaining in NST breast cancer and overall survival (C).
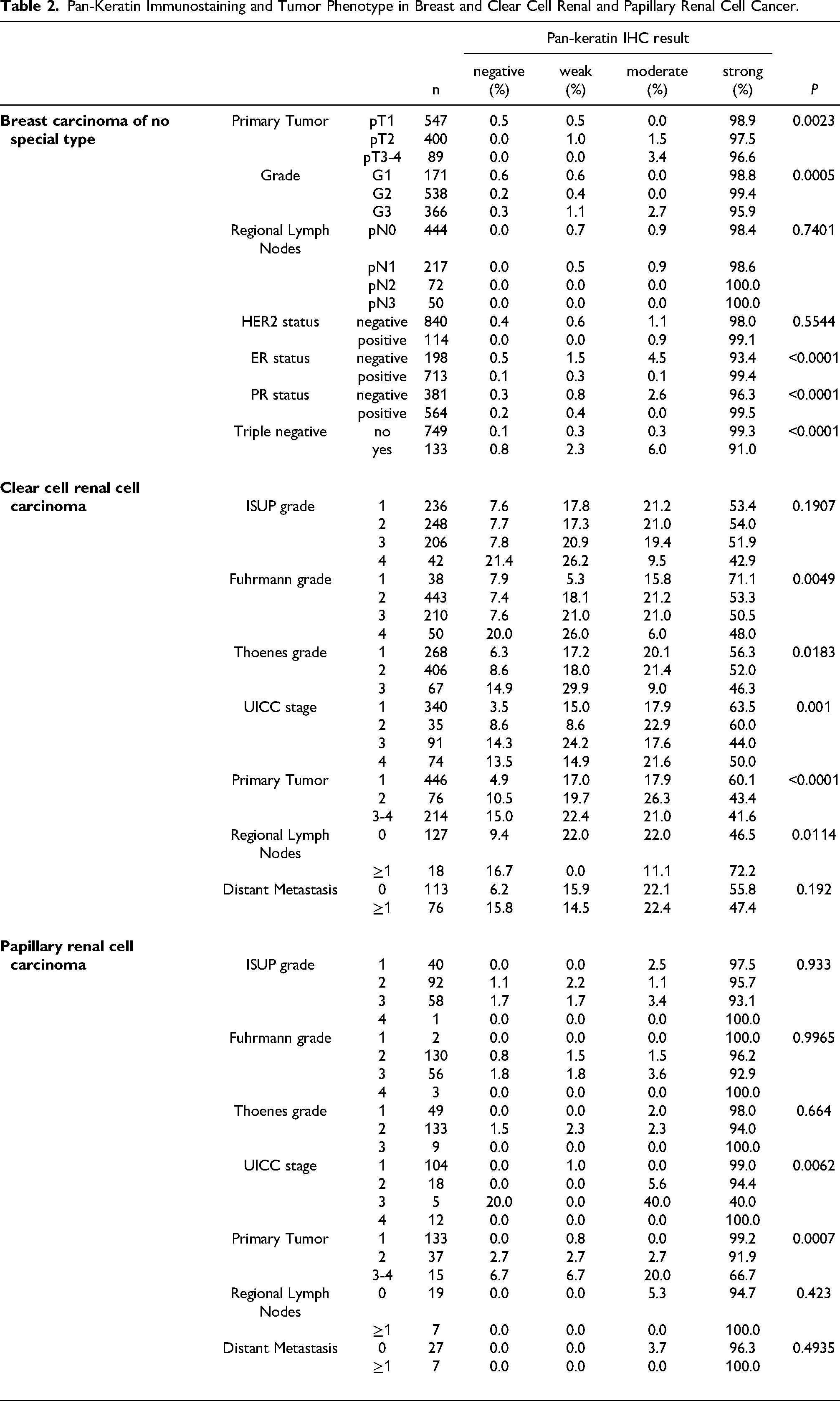
Pan-Keratin Immunostaining and Tumor Phenotype in Breast and Clear Cell Renal and Papillary Renal Cell Cancer.
Discussion
The standardized analysis of 13,501 tumors provided a comprehensive overview on pan-keratin immunostaining in different tumor types. That the graphical representation of the frequencies of pan-keratin immunostaining among 121 different tumor entities resulted in an S-shaped curve reflects the fact that intense pan-keratin immunostaining is common in epithelial neoplasms while non-epithelial tumors are usually pan-keratin negative.
Among epithelial tumor entities, 50 of 75 (67%) showed Pan-keratin positivity in 100% of tumors and 12 (16%) were positive in ≥98% of tumors. These entities include virtually all important types of adenocarcinomas and squamous cell carcinomas. We assume that a fraction of the few negative tumors in these cancer types may be caused by technical issues. Some unexpected negative immunostaining results always occur in TMAs because not all tissues are properly fixed in all areas. 25 Unequal immunostaining in tissues can results in an immunostaining gradient across a tissue block and can thus cause false negative immunostaining, if TMA cores are taken from areas with poor reactivity. 26 It is likely that – in at least a fraction of these tumors - some immunoreactive areas will be found if larger tissue samples, perhaps derived from different blocks, are analyzed.
The group of tumor entities with a pan-keratin positivity in 25% to 98% of analyzed tumors made up for only 17 (13%) of analyzed entities. Most of these tumors could be categorized into the following 5 groups: tumors with mixed differentiation, endocrine/neuroendocrine tumors, kidney tumors, adrenocortical tumors, and particularly poorly differentiated carcinoma subtypes. The group of tumors with mixed epithelial-mesenchymal differentiation includes carcinosarcoma of the uterus and the ovary, phyllodes tumor of the breast, teratoma of the testis, and malignant mesothelioma. In these tumors, epithelial but not mesenchymal tumor areas are pan-keratin positive. The pan-keratin TMA result therefore depends on whether epithelial components are present in the TMA spot or not. The rather low positivity rate in kidney cancers reflects the fact that these tumors have low cytokeratin levels and tend to express vimentin instead.27,28
For adrenocortical, neuroendocrine and endocrine tumors, the intermediate positivity rate appears to mirror the rather low pan-keratin immunostaining in corresponding normal cells. Hepatocellular carcinoma, another tumor for which reduced cytokeratin has often been reported 5,29,30 was always pan-keratin positive in this study, although staining was only weak or moderate in 22% of tumors.
Very poorly differentiated cancers, such as small cell and sarcomatoid cancers as well as anaplastic cancers of the thyroid, often showed lower pan-keratin immunostaining rates as compared to corresponding normal tissues and more differentiated tumors from these organs. This may reflect that a reduced expressions of keratins is a feature of tumors dedifferentiation that can occur during cancer progression. This interpretation is also in line with our findings in kidney and breast carcinomas showing significant associations between reduced pan-keratin immunostaining and several unfavorable phenotypic tumor features. Other investigators had previously also described significant correlations between reduced expression of specific keratins 31–34 or reduced pan-keratin immunostaining 35 and poor patient prognosis or unfavorable tumor phenotype in various tumor types. A reduced expression of keratins in tumors derived from keratin positive progenitor cells is likely to represent a feature of cellular dedifferentiation which regularly goes along with cancer progression. Our data also emphasize that pan-keratin immunostaining is not uncommon in mesenchymal tumors although the expression levels are usually lower in these neoplasms as compared to carcinomas. In this study, pan-keratin immunostaining was observed in 13 mesenchymal tumor entities with highest rates in rhabdomyosarcomas (29%) and angiosarcomas (25%). These findings are in line with earlier studies describing keratin expression in 20%, 36%, 100% of rhabdomyosarcomas 36–38 and in 20%, 33%, and 88% of angiosarcomas.19,20,39 Given the significant pan-keratin staining in the majority of cells is several sarcomas of different types, cytokeratin positivity – even if strong – should not automatically lead to a diagnosis of “sarcomatoid carcinoma”.
In summary, our data show that pan-keratin can consistently be detected in the vast majority of epithelial tumors but also identify renal cell, hepatocellular, adrenocortical and neuroendocrine neoplasms as tumors lacking pan-keratin immunostaining in a fraction of tumors. Moreover, pan-keratin immunostaining - usually at lower levels - can also occur in a broad range of mesenchymal tumors.
Supplemental Material
sj-pdf-1-ijs-10.1177_10668969221117243 - Supplemental material for Pan-keratin Immunostaining in Human Tumors: A Tissue Microarray Study of 15,940 Tumors
Supplemental material, sj-pdf-1-ijs-10.1177_10668969221117243 for Pan-keratin Immunostaining in Human Tumors: A Tissue Microarray Study of 15,940 Tumors by Anne Menz, Natalia Gorbokon, Florian Viehweger, Maximilian Lennartz, Claudia Hube-Magg, Lisa Hornsteiner, Martina Kluth, Cosima Völkel, Andreas M. Luebke, Christoph Fraune, Ria Uhlig, Sarah Minner, David Dum, Doris Höflmayer, Guido Sauter, Ronald Simon, Eike Burandt, Till S. Clauditz, Patrick Lebok, Frank Jacobsen, Stefan Steurer, Till Krech, Andreas H. Marx and Christian Bernreuther in International Journal of Surgical Pathology
Supplemental Material
sj-docx-2-ijs-10.1177_10668969221117243 - Supplemental material for Pan-keratin Immunostaining in Human Tumors: A Tissue Microarray Study of 15,940 Tumors
Supplemental material, sj-docx-2-ijs-10.1177_10668969221117243 for Pan-keratin Immunostaining in Human Tumors: A Tissue Microarray Study of 15,940 Tumors by Anne Menz, Natalia Gorbokon, Florian Viehweger, Maximilian Lennartz, Claudia Hube-Magg, Lisa Hornsteiner, Martina Kluth, Cosima Völkel, Andreas M. Luebke, Christoph Fraune, Ria Uhlig, Sarah Minner, David Dum, Doris Höflmayer, Guido Sauter, Ronald Simon, Eike Burandt, Till S. Clauditz, Patrick Lebok, Frank Jacobsen, Stefan Steurer, Till Krech, Andreas H. Marx and Christian Bernreuther in International Journal of Surgical Pathology
Footnotes
Acknowledgements
We are grateful to Melanie Witt, Inge Brandt, Maren Eisenberg, and Sünje Seekamp for excellent technical assistance.
Contributiors
AM, RS, GS, CB: contributed to conception, design, data collection, data analysis and manuscript writing. NG, FV, ML, AML, EB, DH, CF, PL, RU, FJ, SM, TSC, DD: participated in pathology data analysis and data interpretation. TK, AHM collection of samples
AM, CB: immunohistochemistry analysis. RS, LH, MK, CHM: data analysis. AM, RS, GS, CB: study supervision. All authors agree to be accountable for the content of the work.
Data Availability Statement
All data generated or analyzed during this study are included in this published article
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The Pan-keratin antibody clone MSVA-000R was received from MS Validated Antibodies GmbH (owned by a family member of GS).
Ethical Approval
The use of archived remnants of diagnostic tissues for TMA manufacturing, their analysis for research purposes, and use of patient data were according to local laws (HmbKHG, §12) and analysis had been approved by the local ethics committee (Ethics commission Hamburg, WF-049/09). All work has been carried out in compliance with the Helsinki Declaration.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
