Abstract
Breast implant-associated anaplastic large cell lymphoma (breast implant-associated ALCL) is a recently described, distinct clinicopathological entity associated with macrotextured breast implants. The diagnostic workup of a patient suspected to have breast implant-associated ALCL includes cytological assessment of effusions and tissue biopsies of any masses or enlarged lymph nodes, with morphologic and immunophenotypic evaluation and possible flow cytometric and molecular testing. We report the case of a woman found to have breast implant-associated ALCL on fine needle aspirate and core biopsy, who on surgical resection, had extensive local disease with involvement of the resection margins and lymph nodes, requiring systemic treatment. We focus on the flow cytometric findings that identified a population of large cells on the CD30/side scatter dot plot and whose immunophenotype was consistent with breast implant-associated ALCL, highlighting the value of flow cytometry as an adjunct to morphological and immunophenotypic evaluation.
Case Report
A 45-year-old female presented to another center with a two-month history of pruritus and a palpable nodule in her left breast four years after subpectoral augmentation-mastopexy with insertion of macrotextured Silicone Implants for Medical Applications (SIMA) brand implants bilaterally in Colombia. Her only past medical history included hypothyroidism treated with levothyroxine, Cesarean sections and an abdominoplasty. Upon presentation to the outside hospital, mammography was performed and showed a mass (see Figure 1A) that, on ultrasound-guided biopsy, revealed a breast implant-associated anaplastic large cell lymphoma (breast implant-associated ALCL). Magnetic resonance imaging identified a periprosthetic effusion and a large multifocal conglomerate of masses in the inferomedial quadrant of the left breast with the largest focus measuring 5.7 cm in greatest dimension (see Figure 1B). There was capsular enhancement and extensive skin and trabecular thickening without involvement of the nipple or chest wall. Four enlarged left axillary lymph nodes were also identified.
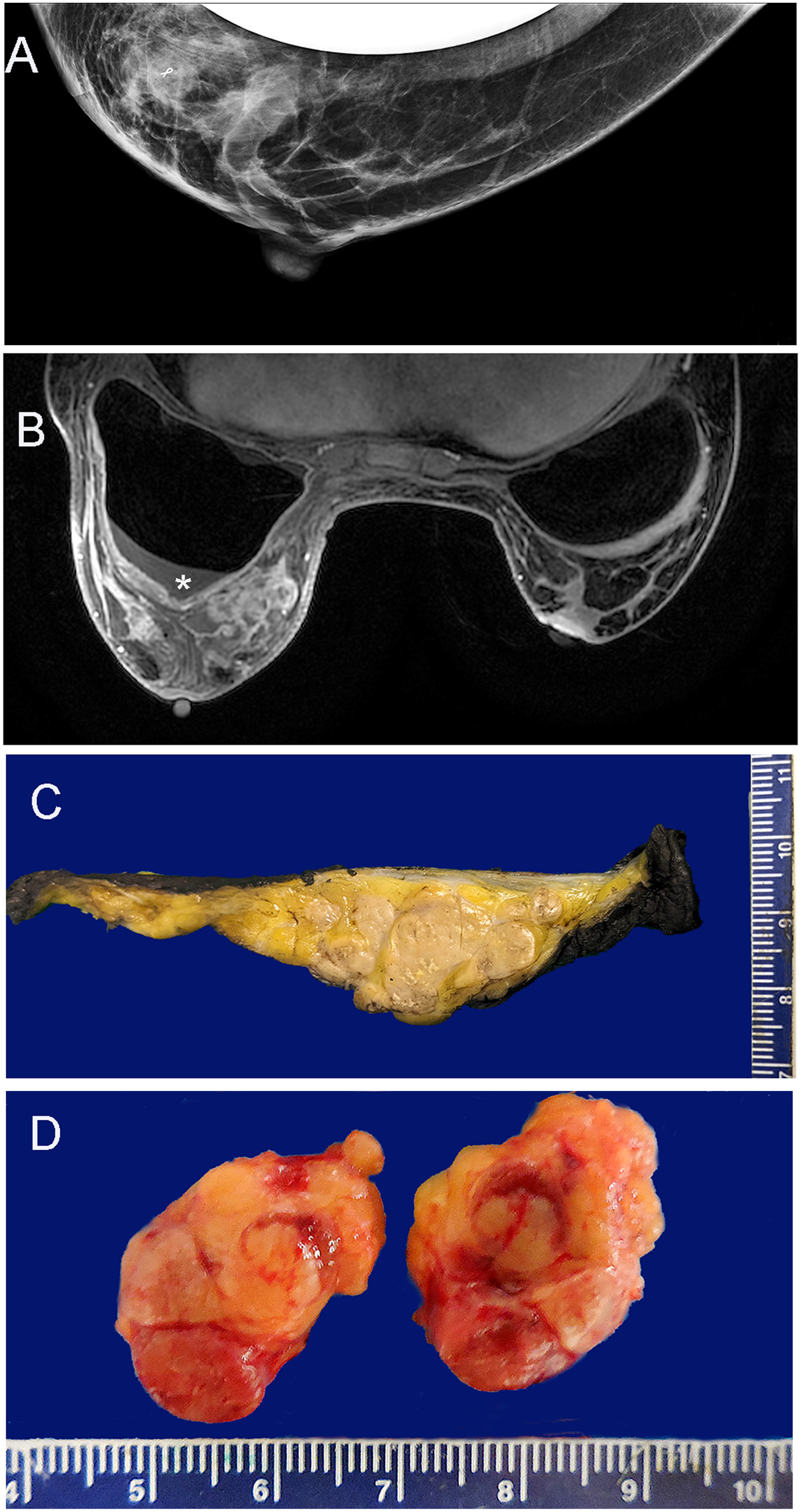
Imaging and macroscopic findings. A. Mammogram at initial presentation shows a mass anterior to the breast implant, in the lower quadrant. B. Magnetic resonance imaging reveals a conglomerate of masses in the lower inner quadrant of the left breast and enlarged lymph nodes (not seen on this slice). Note the moderate effusion (*). C. Macroscopy of one slice of the breast showing the large multilobulated mass. The smooth upper surface is the anterior surface of the implant capsule. D. Macroscopy of one lymph node, bisected, showing partial replacement by lymphoma.
Upon referral to our centre, approximately one month after initial presentation, she underwent a fine needle aspiration of the seroma that showed large anaplastic single cells (see Figure 2A and B) positive for CD45, CD30, CD2, CD4, CD43, perforin and focally EMA, consistent with breast implant-associated ALCL; these cells were negative for CD20, CD79a, PAX5, CD3, CD5, CD7, CD8, CD15, anaplastic lymphoma kinase (ALK), and HHV8. Ki-67 was positive in approximately 85% of the malignant cells. A core biopsy of the left axillary lymph node revealed a few foci of large, atypical cells consistent with focal involvement by breast implant-associated ALCL. The patient, approximately six weeks post-presentation underwent a left skin-sparing mastectomy with en bloc capsulectomy and placement of an expander in the prepectoral plane, excision of four left axillary lymph nodes, and right breast en bloc capsulectomy with implant exchange.
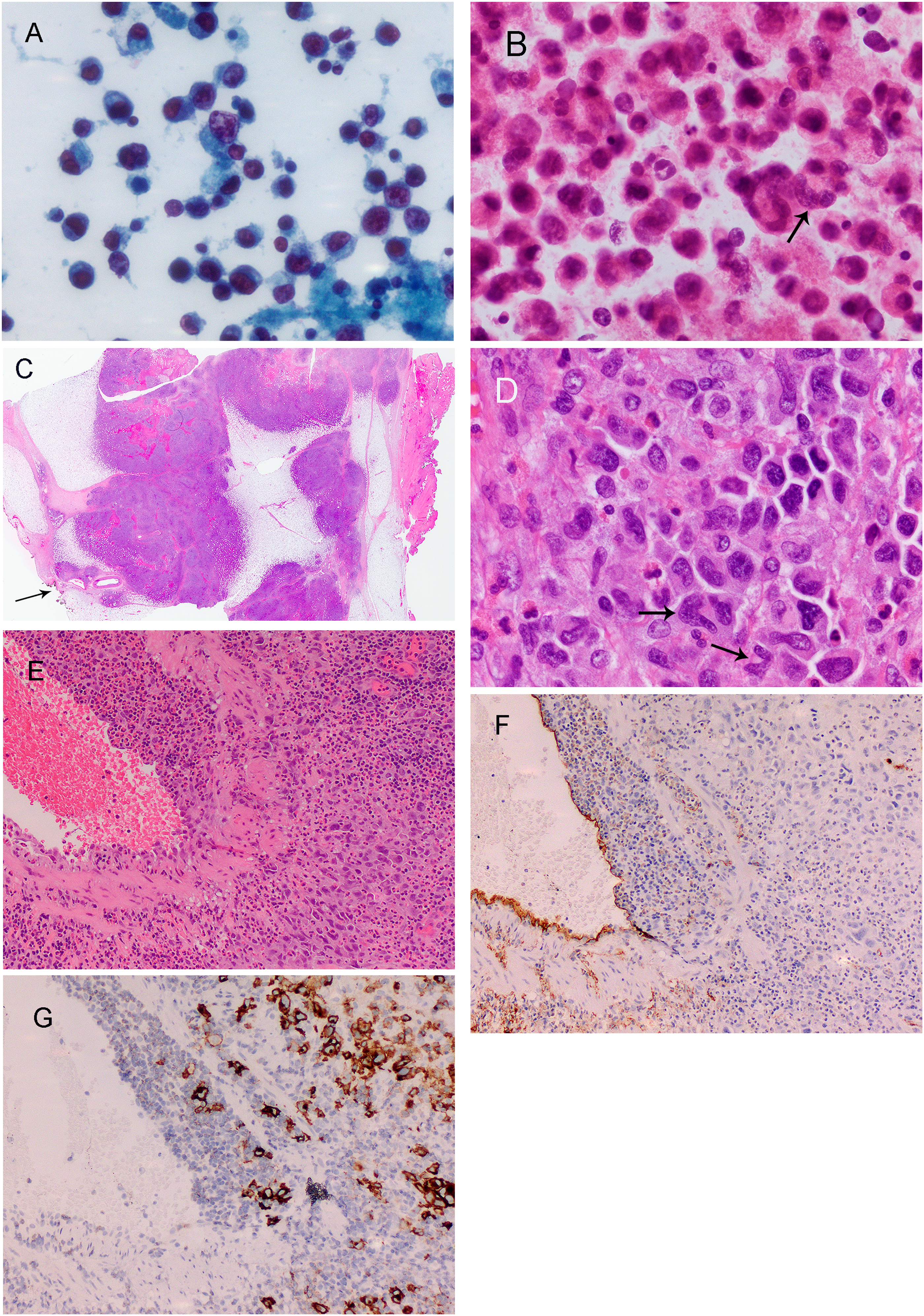
Light microscopic findings. A. High magnification image of the smear from the left breast seroma fluid showing medium to large atypical calls, including the very large one in the center. B. Analogous image from the cell block showing medium to large very atypical cells with multinucleation, prominent nucleoli, and “hallmark cells” (arrow) consistent with breast implant-associated ALCL. C. Low power of the multilobulated lymphoma in the breast with the external surface of the capsule on the right and the lymphoma very close to the inked margin on the left (arrow). D. High power of the lymphoma shows the large anaplastic cells with hallmark cells (arrows) and scattered eosinophils. E. Medium power of a vein infiltrated by anaplastic lymphoma on the right; note the prominent eosinophils. F. Immunostain for CD34 showing, along with the immunostain for CD30 (G), the anaplastic lymphoma cells infiltrating the wall of the vein.
Gross examination (see Figure 1) of the surgical resection specimen identified large multifocal involvement by the breast implant-associated ALCL in the left breast measuring 13 cm in greatest dimension and involving the peri-implant capsule, surrounding anterior breast tissue, dermis, and skeletal muscle (see Figure 1C). Microscopic examination (see Figure 2) revealed invasion of mammary tissue, including around ducts, as well as large vascular (see Figure 2E–G), lymphovascular, and perineural invasion. Margins were very close to the anterior, posterior, medial, lateral, and inferior margins. The tumor was designated pT4 and with three ipsilateral axillary nodes involved by breast implant-associated ALCL, the nodal involvement was pN2 (see Figure 1D), thus TNM stage III.1,2
Morphological assessment and immunohistochemical analyses supported the diagnosis of breast implant-associated ALCL (see Figures 2 and 3). The neoplasm consisted mostly of large cells with atypical nuclei and vesicular chromatin, conspicuous nucleoli, prominent mitoses, and moderately abundant cytoplasm. Many multinucleated giant cells as well as horseshoe, kidney-shaped or “hallmark” cells were identified, with numerous eosinophils scattered throughout. The atypical cells were positive for CD45, CD30, CD4, CD25, and perforin, with partial loss of CD2; they were negative for ALK, CD20, CD15, CD56, Epstein-Barr virus-encoded small RNAs (EBER) by in situ hybridization, and EMA. There was loss of expression of CD3, CD5, and CD7.
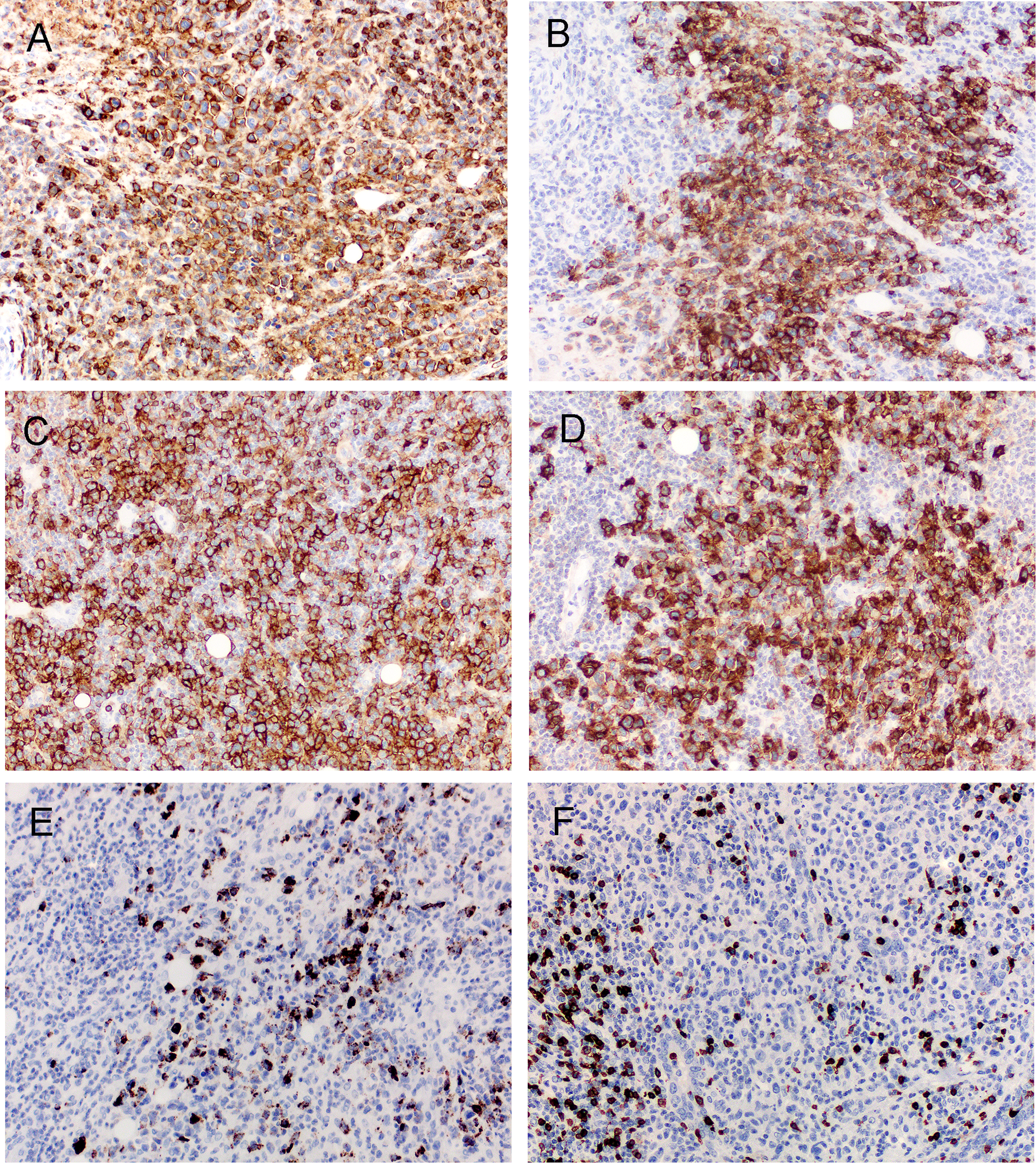
Immunohistochemical findings on sections from the excised left breast. The malignant cells were positive for CD45 (A), CD30 (B), CD4 (C), CD25 (D) and perforin (E), but negative for CD5 (F). See text for results of other immunostains.
Flow cytometry was performed on a tissue sample taken from the breast resection specimen using the Euroflow Lymphoid Screening Tube (LST) panel and four T-cell chronic lymphoproliferative disease (T-CLPD) panels 3 with Infinicyt software (version 2.0; Cytognos, S.L., Salamanca, Spain). 3 Specifically, we used the CD30/side scatter (SSC) dot plot to identify the malignant cells: these were positive for CD45, CD2, CD4, CD45RO, HLA-DR, cyTcl1, and CD25; low positive for CD3, CD7, CD8, CD26, CD28, CD197, CD45RA, and CD11c; and negative for CD5, CD27, and CD57 (see Figure 4).
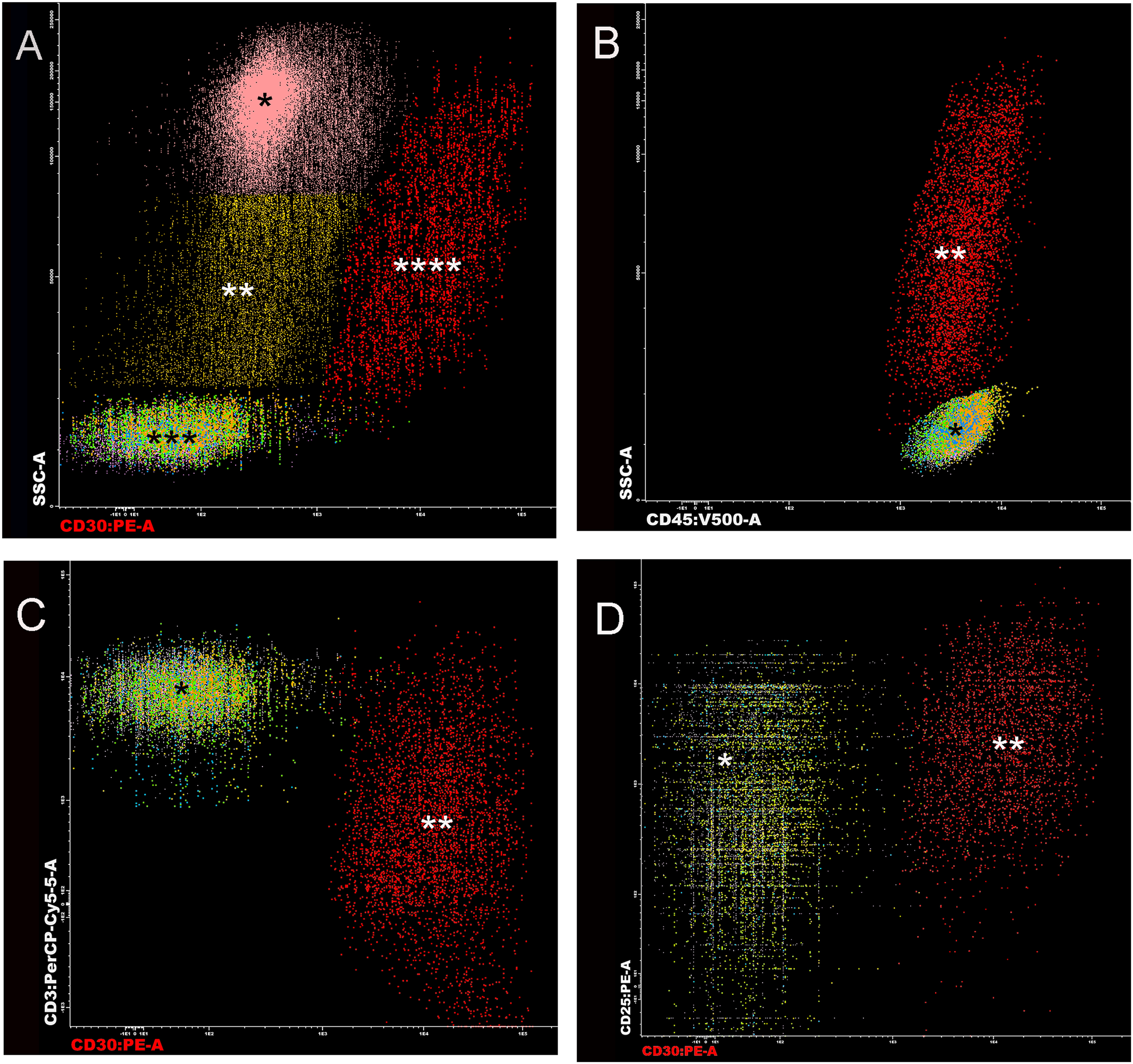
Flow cytometric findings. A. Dot plot of side scatter (SSC) versus CD30 showing the nucleated cells subdivided into eosinophils (*) 64%; monocytes (**) 12%; normal lymphocytes (***) 19%; and the abnormal CD30+ cells (****) 4.3%. B. Dot plot of SSC versus CD45 showing normal T-cells (*) and the malignant cells with high SSC (**). C. Dot plot of CD3 versus CD30 showing normal T-cells (*) with bright CD3 and the malignant cells with bright CD30 (**). D. Dot plot of CD25 versus CD30 showing normal T-cells (*) low/+ for CD25, and the lymphoma with moderate CD25 (**).
Four weeks after her surgery, the patient had a small fluid collection measuring 2.9 cm in the left axilla. Her laboratory data were within normal limits except for a mildly elevated lactate dehydrogenase level (232 U/L, normal 110-210 U/L). Postoperative positron emission tomographyscan showed residual disease. Given her stage, she was recommended post-operative chemotherapy, including brentuximab vedotin with cyclophosphamide, doxorubicin, and prednisone (BV + CHP) due to the overall survival benefit for CD30-positive peripheral T-cell lymphomas in the ECHELON-2 trial. 4 She was transferred to a centre closer to home for her chemotherapy, which started one month post surgery. A positron emission tomography scan performed after chemotherapy treatment did not identify residual hypermetabolic lesions, consistent with a complete metabolic response.
Discussion
The first case of breast-implant associated anaplastic large cell lymphoma (breast implant-associated ALCL) was described in 1997. 5 In 2011, the US Food and Drug Administration released a document summarizing 13 years of literature based on 34 unique cases, concluding that there was a possible association between breast implants and anaplastic large cell lymphoma (ALCL). 6 Since then, the US Food and Drug Administration, the American Society of Plastic Surgeons (ASPS), and the Plastic Surgery Foundation (PSF) collaborated to create the Patient Registry and Outcomes For breast Implants and anaplastic large cell Lymphoma Etiology and Epidemiology (PROFILE) Registry to faciliate data collection and research on breast implant-associated ALCL. As of January 4, 2021, there were 993 cases of breast implant-associated ALCL reported worldwide. 7
Risk estimation of breast implant-associated ALCL has been challenging due to reporting issues and misdiagnoses, which have led to underestimation of the true risk associated with textured implants. Recently published studies may better approximate this risk: a robust retrospective, single institutional analysis of 9373 patients identified the risk of breast implant-associated ALCL to be 1 in 559 patients with textured implants and a median time to diagnosis of 10.3 years. 8 Amidst growing evidence of the relationship between breast implant-associated ALCL and textured implants, Allergan recalled their Biocell textured implants in July 2019 after an FDA request. 9 Our patient had textured Colombian SIMA brand implants, which possess a macrotextured surface similar to the Allergan Biocell textured breast implants. In many places around the world, notably in Colombia and other South American countries, textured implants are used almost exclusively. 10 Although breast implant-associated ALCL has largely been underreported in South America, the first breast implant-associated ALCL registry results in South America were recently published from a multidisciplinary group in Colombia. 11 Notably, our case is the first reported SIMA-brand breast implant-associated ALCL case reported thus far in the literature.
In 2016, breast implant-associated ALCL was recognized as a provisional entity in the 2017 WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues with morphological and immunophenotypic features indistinguishable from ALK negative ALCL, but which are associated with breast implants 2 ; it is also included in the WHO classification of Tumors of the Breast. 12 breast implant-associated ALCL is characterized by large cells with abundant cytoplasm and pleomorphic, horseshoe-shaped “hallmark” nuclei expressing CD30, but negative for ALK protein expression and ALK gene rearrangement.13–16 Expression of CD4, CD43, perforin and EMA markers, identified in our case, are variably positive in breast implant-associated ALCL. 17 Loss of T-cell markers, such as CD2, CD3, CD5 and CD7, characteristic of breast implant-associated ALCL, was also seen in our case. B-cell markers (CD20, CD79a, PAX5) and CD15 were all negative, as expected in breast implant-associated ALCL. The pathogenesis is hypothesized to involve the growth of Gram-negative bacteria on the greater surface area of biofilm provided by textured implants, leading to lymphocytic stimulation and T-cell transformation. 18 In terms of molecular characteristics, breast implant-associated ALCL has been shown to have JAK1/STAT3 mutations, but consistently lacks rearrangements seen in other ALCLs such as ALK, DUSP22, and TP63. 19
Timely and accurate diagnosis as well as adequate treatment and surveillance are important as deaths due to breast implant-associated ALCL have been reported. 7 Patients generally present 8 to 10 years after placement of the implant with rapid and spontaneous onset of unilateral breast enlargement found to be due to either a seroma or an infiltrating mass lesion,1,20 but occasionally may present with capsular contracture, rash, or lymphadenopathy.21,22 Initial work-up of a unilateral enlarged breast should include imaging for fluid collection, masses and enlarged lymph nodes. If an effusion is identified, the patient should undergo fine needle aspiration for cytological evaluation with possible CD30 testing by immunocytochemistry, or flow cytometry depending on the morphologic findings and healthcare context.15,16,22,23 If a mass is identified, a biopsy should be performed with submission for lymphoma protocol evaluation.13,15 Our patient presented with unilateral breast enlargement and was found to have a seroma, mass lesions and lymphadenopathy. Accordingly, she had a fine needle aspiration of the effusion, biopsy of the breast mass and lymph node as part of her initial work-up.
Most patients present with early-stage disease (stage I) and are treated with en bloc capsulectomy and removal of the implant. At higher stages (II to IV), however, excision of any associated breast masses and suspicious lymph nodes should be performed, with addition of systemic therapy including chemotherapy and brentuximab vedotin, an anti-CD30 antibody-drug conjugate. Radiotherapy may be indicated for residual disease. Most patients have a favorable prognosis with complete surgical excision.14,17,22 Our patient, consistent with present guidelines for stage III disease, underwent surgical excision and adjuvant chemotherapy and brentuximab vedotin as per protocol for ALK-negative systemic ALCL. Although our patient had very extensive local disease and involvement of the surgical resection margins, she had a complete clinical response to chemotherapy. However, contrasting with the generally early-stage disease at presentation in breast implant-associated ALCL, our patient presented with lymph nodal involvement. Nonetheless, the patient presented soon after palpation of the mass in her breast and there was no undue delay in the diagnosis and treatment that required a multidisciplinary approach. There have been studies addressing reasons for potential delays in breast implant-associated ALCL. Evans et al 24 reported on a group of ten patients in whom pathologic specimens enabling a diagnosis were obtained 6 months to 4 years after initial non-diagnostic procedures, emphasizing that these lymphomas will persist or progress if not treated in a timely fashion.
In terms of reconstruction in the setting of breast implant-associated ALCL, one publication of the largest series states that reconstruction without textured devices can be safely performed if complete surgical ablation of the lymphoma is possible. 25 In the context of our patient, a total mastectomy with resection of overlying skin was performed, which led us to feel safe putting in a smooth tissue expander. Regardless, a shared decision-making process with the patient and oncologic surgeon was held, and the patient strongly preferred immediate tissue expander placement after discussing the risks and benefits. For the contralateral side, given the lack of clinical concern for ALCL, we replaced the textured breast implant with a smooth, round silicone breast implant according to standard of care.22,26
As described above, the role of cytopathological evaluation of late breast implant seromas has been established based on large case series. 16 More recently, various groups are assessing the role of flow cytometry analyses in the initial diagnosis and evaluation of breast implant-associated ALCL.27–31 Romero et al 30 published a case series that identified four breast implant-associated ALCL cases out of a total of 39 periprosthetic fluid samples. 30 They identified a statistically significant difference in the percentage of CD30-positive cells between breast implant-associated ALCL and those samples without tumor (54.3% in breast implant-associated ALCL vs. 0.3% in non-tumor; P = .03). They concluded by recommending consideration of flow cytometry as a first line of analysis over cytopathology in certain contexts. Although Jaffe et al17,32 acknowledge that flow cytometry has a role in the diagnostic work-up of breast implant-associated ALCLs, they emphasize that key evaluations, such as immunomorphological correlation, ALK immunohistochemistry, EBER in situ hybridization to rule out a breast implant-associated differential diagnosis of EBV-positive diffuse large B-cell lymphoma associated with chronic inflammation, 33 and molecular studies, can only be done on a cell block and therefore that priority should be given to cytology and cell blocks rather than flow cytometry. We strongly agree with this latter view.13,15
Furthermore, although flow cytometry may help in the diagnosis of breast implant-associated ALCL, there remains a paucity of literature on the topic, and most authors agree that close correlation with cell morphology and immunohistochemistry is paramount. 29 In the context of B-cell non-Hodgkin lymphomas, one study found that flow cytometry improved the sensitivity of conventional cytology for the identification of leptomeningeal disease, particularly in paucicellular samples. 34 However, this was a single institution study of 27 positive cases in the context of B-cell non-Hodgkin lymphomas and may not be broadly applicable to other diseases. Nonetheless, the panel of antibodies available for flow cytometry that, once validated, are often more numerous and different from those for immunochemistry and therefore may provide important complementary information. Further studies correlating cytology with immunophenotype by both flow cytometry and immunocytochemistry are needed to evaluate the sensitivity of flow cytometry in the context of breast implant-associated ALCL. Our case provided a unique opportunity to evaluate the flow cytometric characteristics of breast implant-associated ALCL and correlate them with the morphologic findings. We used the CD30/SSC dot plot to identify and analyze the malignant cells, a sensitive and specific strategy for breast implant-associated ALCL first described by Barr et al 29 in a case series: of the 166 seromas evaluated in their series, five were breast implant-associated ALCL with the neoplastic cells showing very bright CD30, positive for CD4, with variable CD2 and CD5, and dim or negative CD3, CD7, CD8, CD10, CD16 and CD56 by both flow cytometry and immunocytochemistry. 29 Our case showed similar findings, the malignant cells exhibiting strong CD30, CD45, CD4, and CD2 expression, negative for CD5 and low positive expression of CD3, CD7, and CD8. In other reported case series, the neoplastic CD30-positive cells were found to co-express CD25 and HLA-DR,27,31 a finding also present in our case.
Our flow cytometric findings showed a similar immunophenotype to cases reported in published series and supported the use of an “ungated” CD30/SSC plot in breast implant-associated ALCL diagnosis.27,28,30,31 However, evaluation of flow cytometric data in a larger number of cases should be undertaken to better establish its role and utility in screening seromas around breast implants. Collection of these data may be facilitated by the preexisting breast implant-associated ALCL databases to encourage research in this still largely underdeveloped field. Just as breast implant-associated ALCL registries helped identify an association between textured implants and breast implant-associated ALCL, these databases have the potential to facilitate high-powered studies to better elucidate its pathogenesis, its key pathologic characteristics to promptly diagnose this rare disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This is not required by our ERB; we have patient consent. Can be provided if required.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
