Abstract
Nodular fasciitis is usually a benign lesion genetically characterized by ubiquitin-specific protease 6 (USP6) rearrangements. We present a case of a 10-year-old boy with a 1.5-week history of a painless mass on the right chest wall, which was excised. A histomorphologically malignant tumor with pronounced pleomorphism, atypical mitotic figures, and a myoid immunophenotype was observed. The methylation profile was consistent with nodular fasciitis and fluorescence in situ hybridization confirmed USP6 rearrangement. Using Archer Fusion Plex (Sarcoma Panel) and RNA sequencing, a collagen, type VI, alpha 2 (COL6A2)–USP6 gene fusion was subsequently identified. Furthermore, DNA clustering analysis also showed a match with nodular fasciitis. During the follow-up of 22 months, no recurrence or metastasis occurred. In conclusion, we describe a clinically benign, histomorphologically malignant mesenchymal neoplasm with a myoid immunophenotype, and a genetic and epigenetic profile consistent with nodular fasciitis. In such cases, molecular analysis is a useful adjunct to avoid unnecessary overtreatment.
Keywords
Introduction
Nodular fasciitis (NF) is a benign rapidly growing myofibroblastic lesion, usually arising in the superficial soft tissue of young adult patients. Involvement of the upper limbs and trunk is most common but localization at any other site is possible.1-5 The identification of ubiquitin-specific protease 6 (USP6) rearrangement in over 90% of NF cases has not only supported its clonal neoplastic nature1,6-8 but also serves as a reliable diagnostic confirmation in respective lesions, especially when small biopsies are taken, to avoid overtreatment. Malignant behavior of NF has been reported exceptionally in cases with classical morphology.9,10 However, according to our knowledge, NF cases with pleomorphic sarcoma-like morphology are hitherto not described.
In this report, we present an unusual case of a clinically benign but morphologically malignant lesion in a 10-year-old boy, which is genetically and epigenetically consistent with NF.
Clinical Summary
A 10-year-old, otherwise healthy, boy presented with a 1.5-week history of a painless swelling on the right dorsal chest wall. Clinically, it was a firm, slightly mobile mass, not adherent to the overlying skin but fixed to the muscle. On magnetic resonance imaging an intramuscular lobulated lesion in the serratus anterior muscle was seen, measuring 24 × 12 × 22 mm. The lesion showed slightly high signal intensity compared to the surrounding muscles on T1-weighted images and moderately high signal intensity on T2-weighted images. Hypointensity of the tumor border was observed in both T1-weighted and T2-weighted images. The mass extended along the superficial fascia of the muscle (fascial tail sign). Positron emission tomography scan localized the metabolic activity. After a core needle biopsy was taken, yielding a diagnosis of pleomorphic sarcoma possibly leiomyogenic in type, wide resection including the deep fascia of the serratus anterior muscle and part of the latissimus dorsi muscle was performed. The tumor was completely removed with oncological margins. No adjuvant therapy was administered. Follow-up was performed every 3 months and the patient underwent annual computed tomography scanning and chest X-ray on the right chest wall. In the most recent follow-up, 22 months after surgery, the child had no subjective symptoms including pain and there was no sign of recurrence or metastasis.
Material and Methods
The tissue was fixed in 4% buffered formalin, routinely processed, and embedded in paraffin; 2 to 4 µm thick sections were stained with hematoxylin and eosin and immunohistochemistry was performed using standard protocols and commercially available antibodies.
Fluorescence in situ hybridization (FISH) was previously reported by Bekers et al, 11 whereas Archer Fusion Plex (Sarcoma Panel) was used analogously to Lam et al. 12 RNA sequencing and DNA clustering analysis were described by Karanian et al. 13 DNA methylation profiling was performed as published by Koelsche et al. 14 A next-generation sequencing (NGS) cancer hotspot panel as described by de Leng et al 15 was additionally used.
Pathological Findings
The core needle biopsy revealed a pleomorphic tumor with a fascicular growth pattern. The atypical cells had enlarged, irregular, and vesicular nuclei with prominent nucleoli. Mitotic figures were numerous including atypical forms. Osteoclast-like giant cells were seen, scattered throughout. The resection specimen showed a relatively circumscribed lesion incompletely surrounded by a collagenous pseudo-capsule. Histology was similar to the biopsy. In addition, there was focally peripheral ossification and hemosiderin deposition throughout the lesion. Vascular invasion was observed focally (Figures 1 to 3).

Low magnification shows a high cellular, infiltrative, and well-circumscribed soft tissue lesion.
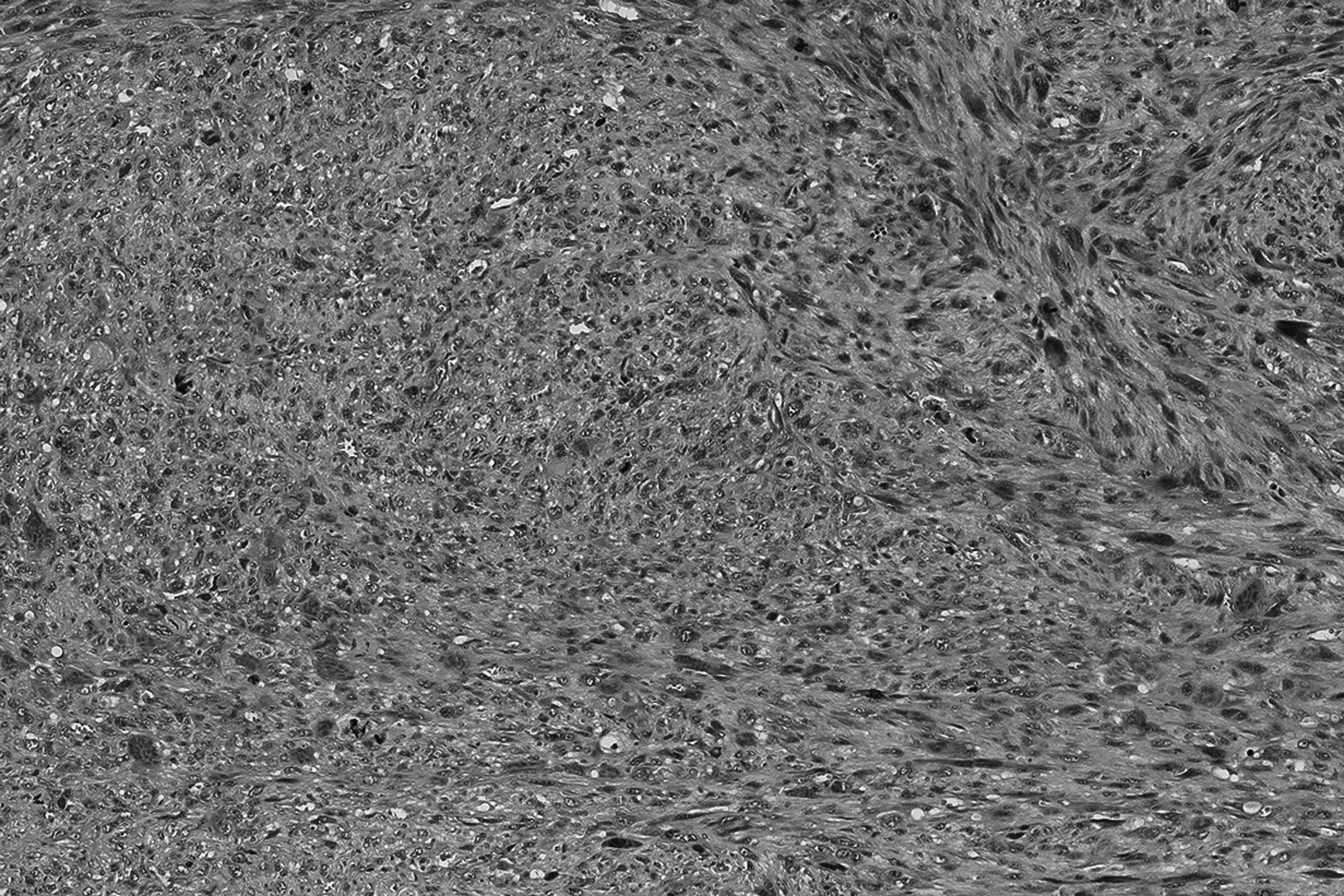
High magnification illustrates fascicular growth and pronounced pleomorphism.
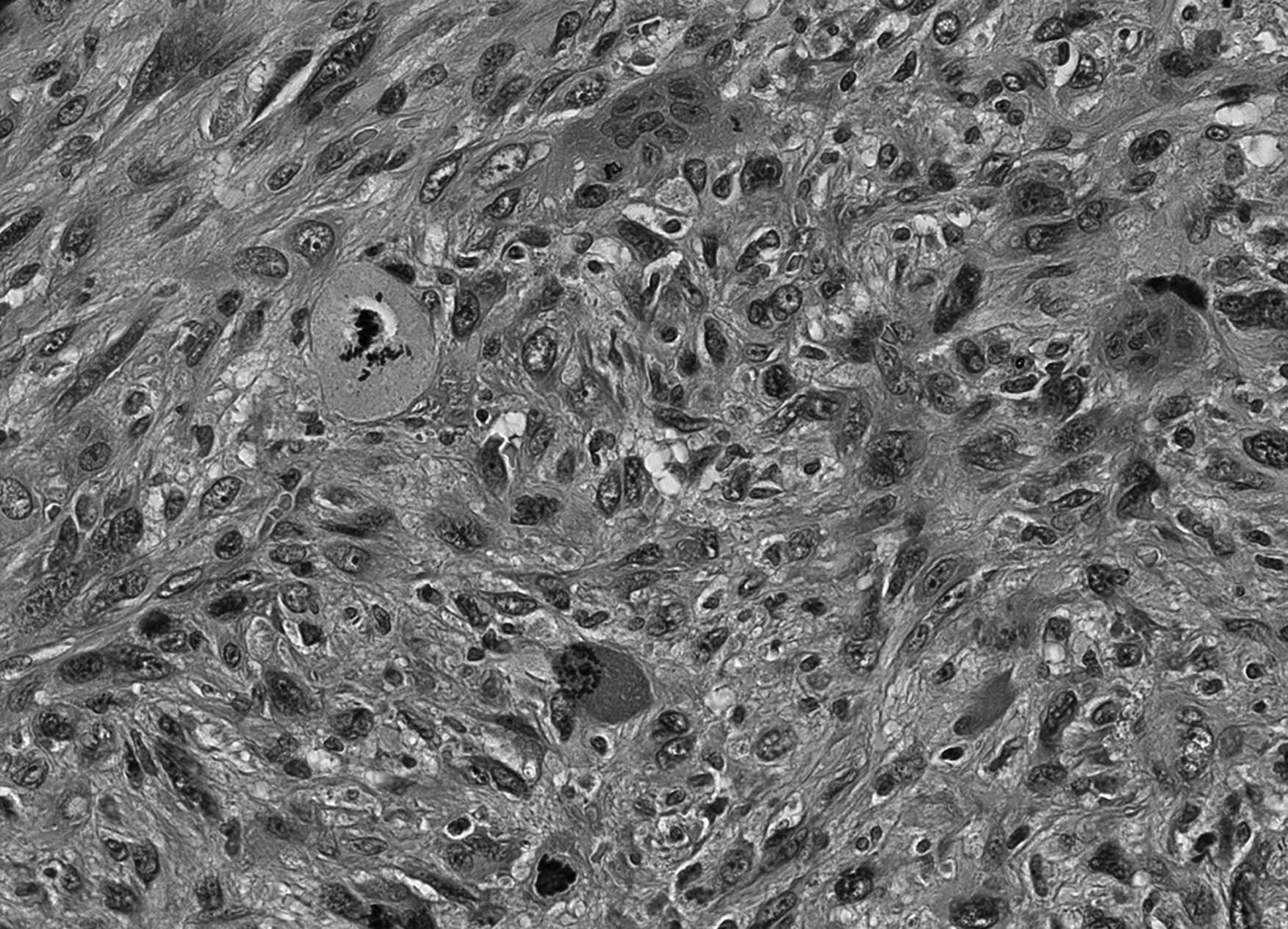
Atypical mitoses are frequent.
Immunohistochemically, the spindle cells were positive for smooth muscle actin (SMA) and caldesmon and negative for desmin, S100, cytokeratin MNF116, epithelial membrane antigen, and CD34 (Figure 4).
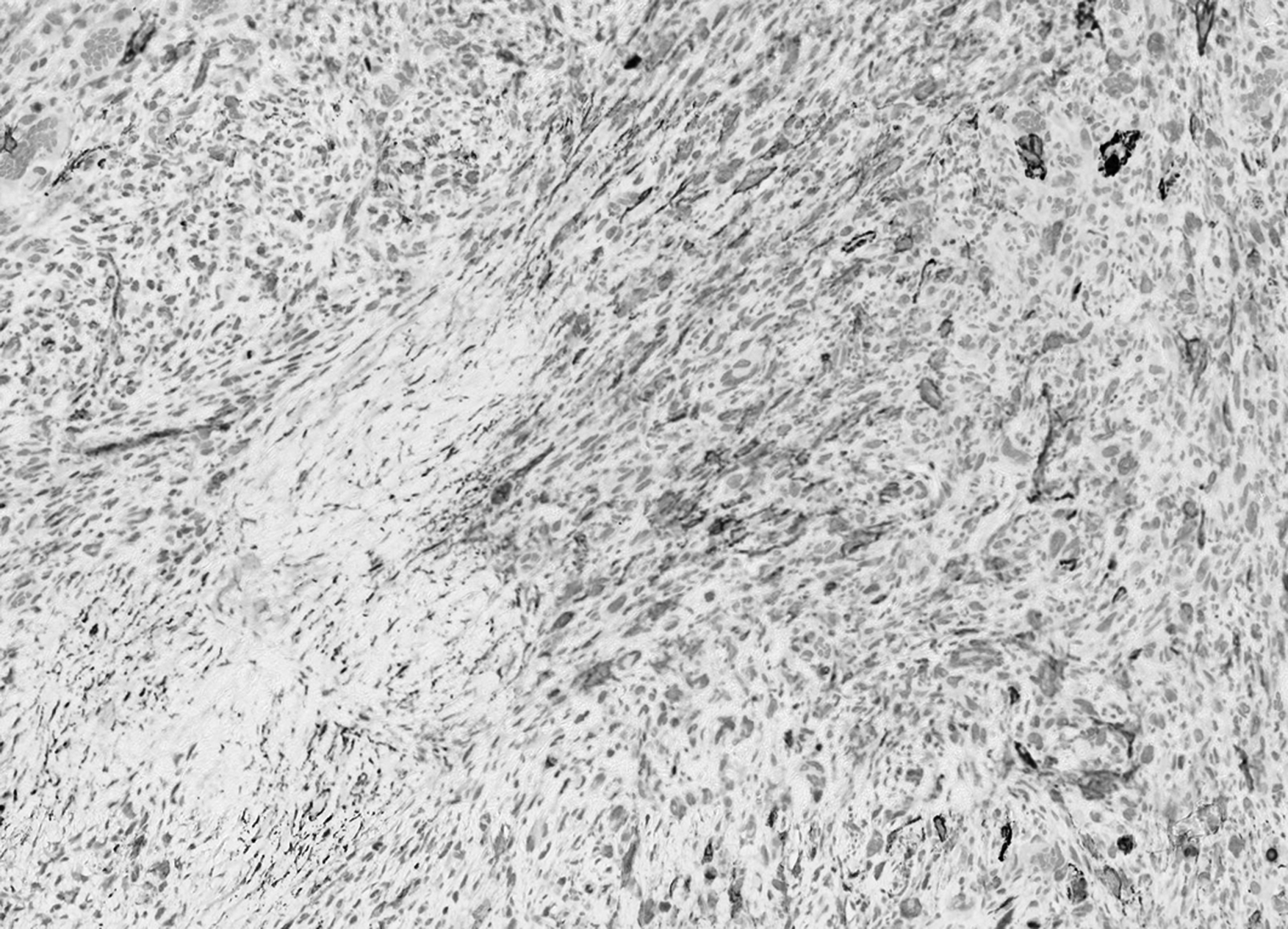
Smooth muscle actin (SMA) expression is inconsistent.
Molecular Findings
The methylation profile matched with NF (calibration score: 0.94). Therefore, FISH, Archer Fusion Plex (Sarcoma Panel) and RNA sequencing were performed identifying USP6 rearrangement (break apart signal in 49 of 100 cells) with a collagen, type VI, alpha 2 (COL6A2)–USP6 (ex1–ex9) fusion (Figures 5 and 6). DNA clustering analysis (uniform manifold approximation and projection [UMAP] three-dimensional plot) of 1434 sarcoma samples in 124 molecular subgroups allocated the lesion within the NF cluster (Figure 7). An NGS cancer hotspot panel with complete coding regions of the genes F-box and WD repeat containing 7 (FBXW7), SWI/SNF related, matrix associated, actin dependent regulator of chromatin, subfamily a, member 4 (SMARCA4), SWI/SNF related, matrix associated, actin dependent regulator of chromatin, subfamily b, member 1 (SMARCB1), Tumor protein P53 (TP53), von Hippel-Lindau tumor suppressor (VHL), and WT1 transcription factor (WT1) revealed no aberrations.
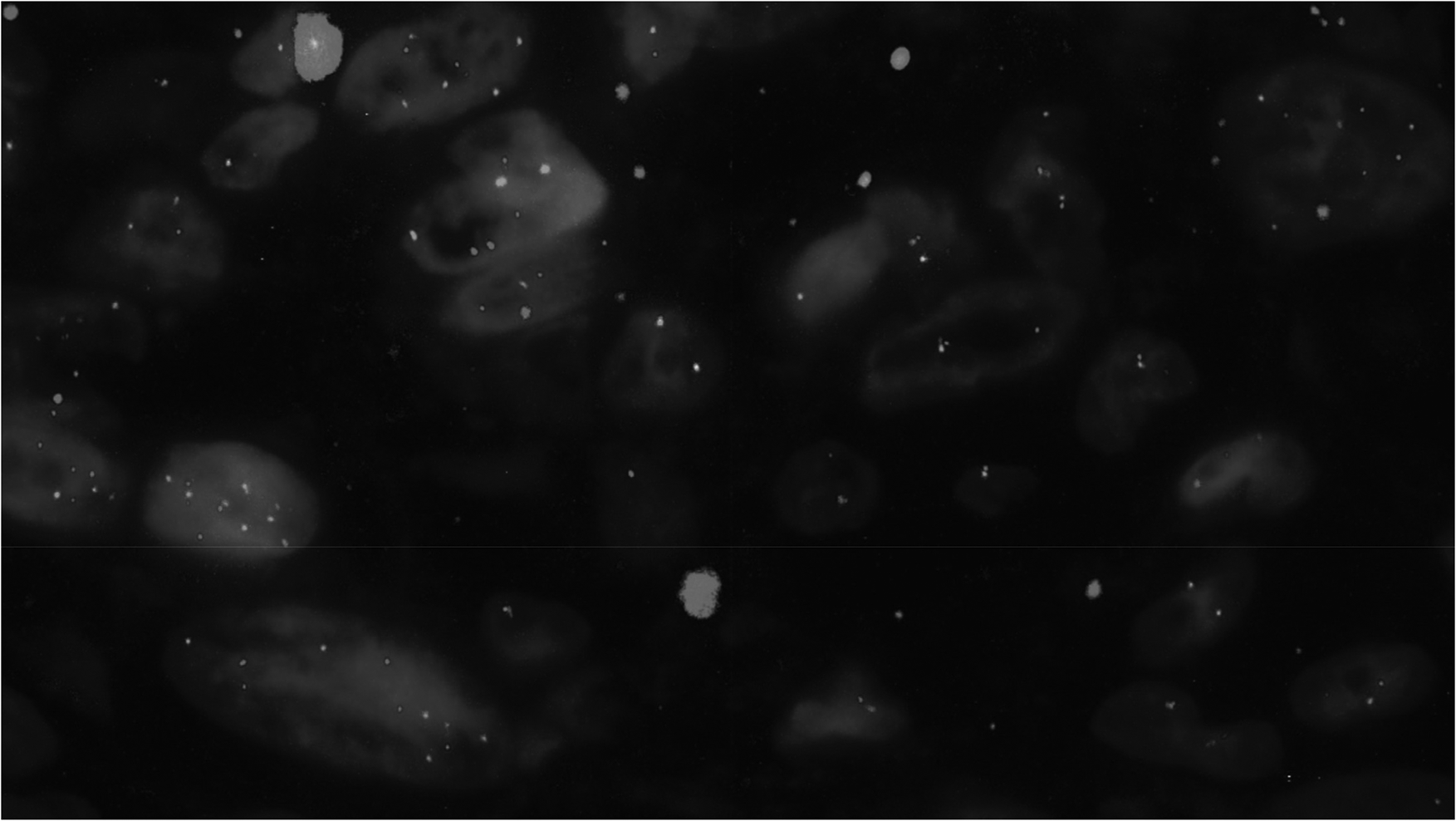
By USP6–FISH, a break-apart signal indicating rearrangement was shown.
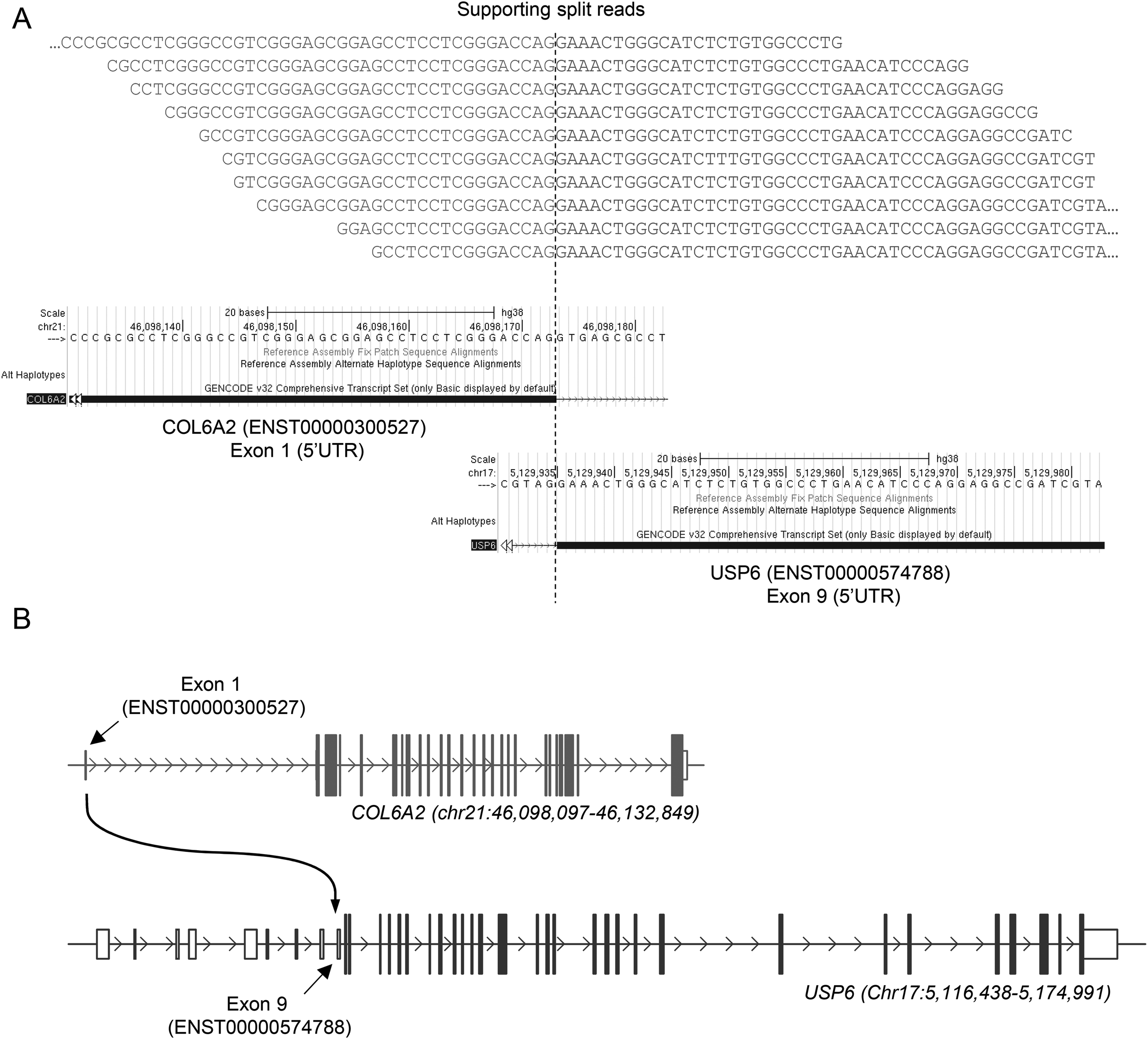
COL6A2–USP6 fusion gene as detected by RNAseq. (A) Ten unique reads supporting the COL6A2–USP6 fusion were detected to align both at the COL6A2 (ENST00000300527) exon 1 boundary and the exon 9 boundary of USP6 (ENST00000574788). Positions on UCSC Genome Browser are shown for both genes. (B) Schema of the genomic region of COL6A2 (blue) and USP6 (red) genes. Genomic regions and positions indicated are related to the GRCh38 genome assembly.
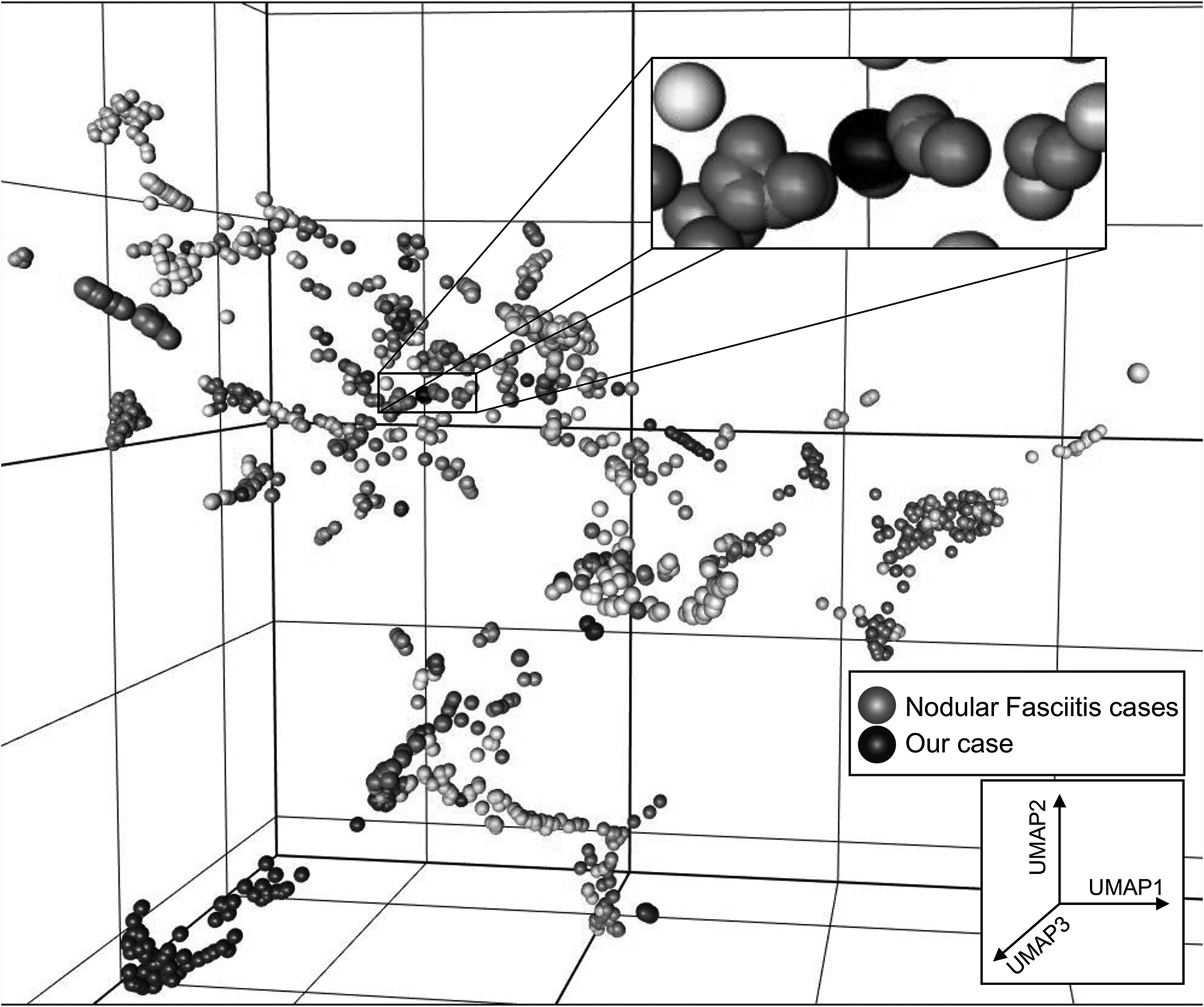
Uniform manifold approximation and projection (UMAP) analysis represented nodular fasciitis cases in red and our case in black.
Diagnosis
Based on morphological and immunohistochemical findings, the initial diagnosis on the biopsy specimen was high-grade sarcoma possibly leiomyogenic in type, which was confirmed on the resection specimen showing a similar histological and immunohistochemical profile (Figures 1 to 4). USP6 rearrangement with a COL6A2–USP6 fusion, DNA clustering analysis, and methylation profiling, however, led to the diagnosis of NF (Figures 5 to 7).
Discussion
NF usually is a benign superficially located soft tissue lesion molecularly defined by the presence of a USP6 rearrangement.1,6 Currently, several USP6 fusion partners including ribosome binding protein 1 (RRBP1), myosin heavy chain 9 (MYH9), calumenin (CALU), catenin beta 1 (CTNNB1), MIR22 host gene (MIR22HG), secreted protein acidic and cystein rich (SPARC), thromobospodin 2 (THBS2), and COL6A2 are known 8 and their number is still growing due to improved molecular techniques.
USP6 fusions also occur in other nonmalignant soft tissue and bone abnormalities such as myositis ossificans, fibro-osseous pseudotumor of the digits, a subset of fibromas of the tendon sheath, possibly better classified as NF, primary aneurysmal bone cyst, and giant-cell tumors of the small bones.11,16-20 All these lesions probably form a spectrum of one entity called USP6-associated neoplasms. 20 As our lesion was intramuscularly situated and showed focally ossification one could argue whether this case better could be classified as myositis ossificans. However, this is clinically not of relevance.
The current available comprehensive molecular analysis allowed us to make the diagnosis of NF in a histomorphological pleomorphic sarcoma-like lesion in a child. We identified for NF known COL6A2–USP6 fusion. 8 Also, methylation profiling and DNA-clustering analysis were consistent with NF. Furthermore, we did not find secondary genetic changes using a NGS oncology panel. To our knowledge, this is the first NF case showing pleomorphism with bizarre nuclei and atypical mitotic figures, not in concordance with the classical morphology of NF. However, the lesion behaved benignly like ordinary NF cases.
In the literature, we encountered two cases with malignant clinical course.9,10 In contrast to our case, both showed classical histologic features of NF and a PPP6R3–USP6 fusion with additional amplification suggesting that this specific amplified fusion gene is causative for the aggressive clinical course of the disease. One of these cases was characterized by multiple recurrences and metastatic behavior whereas the other case showed aggressive growth with local invasion.9,10
In summary, we present a soft tissue tumor in a child with morphological features of a high-grade sarcoma but with molecular characteristics of NF representing the first case of a morphologically malignant NF, albeit with benign clinical features.
This case illustrates that the morphology of soft tissue tumors does not predict per se clinical behavior and genetic analysis is of added value to get a better insight into the morphological spectrum of an entity. The latter can help to avoid over- or undertreatment.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Confirm that all the research meets the ethical guidelines, including adherence to the legal requirements of the study country.
Informed Consent
Informed consent was obtained.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
