Abstract
Two sporadic cases of eosinophilic solid and cystic renal cell carcinoma (ESC RCC), at our institution, are presented in this study to contribute to the growing literature on this novel renal neoplasm. The first patient was a 38-year-old female with two synchronous renal masses measuring 3.5 and 1.9 cm on preoperative imaging. The second patient was a 44-year-old female with an incidental renal mass measuring 4 cm. Both patients underwent uncomplicated radical nephrectomies. The 1.9 cm mass in the first patient was consistent with clear cell RCC. The dominant mass in the first patient and the tumor in the second patient had microscopic and macroscopic findings in keeping with ESC RCC including a tan appearance, abundant eosinophilic cytoplasm, and CK20+ and CK7− staining. Both patients had an uncomplicated course following surgery with no evidence of local recurrence or distant metastatic disease for 1 and 2 years for the first and second patient accordingly. These cases contribute to a growing body of literature regarding ESC RCC including, to our knowledge, the first reported case of synchronous ESC RCC and clear cell RCC. Further research about this novel renal neoplasm is needed.
Keywords
Introduction
Recent studies and reports have identified cases of a sporadically occurring renal neoplasm known as eosinophilic solid and cystic renal cell carcinoma (ESC RCC).1-5 Previously recognized as occurring almost exclusively in patients with tuberous sclerosis complex (TSC), these eosinophilic and cystic neoplasms exhibit unique pathological, immunohistochemical, and molecular features that support their emergence as a novel and independent entity.1,2 Trpkov et al 1 proposed the term ESC RCC to describe the neoplasm they identified as occurring sporadically in the clinical setting in patients without features of or a diagnosis of TSC. Since this initial report, further cases of ESC RCC have been identified and characterized in various studies and case reports,2-6 with a current estimated prevalence of 0.07% to 0.2% among RCC diagnoses.1,2
The majority of studies classify ESC RCC as an indolent neoplasm associated with good prognosis and low metastatic potential.1,2 However, two previous cases have confirmed the potential of ESC RCC to metastasize with spread reported to the bone, liver, lung, and hilar lymph nodes.3,4,7 Currently, the rate of metastasis for this entity is estimated to be 3% to 5%.3,8 Considering these few studies, ESC RCC remains an emerging diagnosis that would benefit from further reports to help contribute to a growing body of evidence about this condition. Two such cases were identified in the Canadian province of Manitoba in the last 3 years.
Patient 1
A 38-year-old female presented to the emergency room with abdominal pain without any urinary symptoms. The abdominal pain subsequently resolved. However, abdominal ultrasound and subsequent computed tomography (CT) identified two incidental lesions in the left kidney, with the larger lesion measuring 3.5 cm (maximum) in diameter and the smaller lesion measuring 1.9 cm (maximum) in diameter. A CT of the chest showed non-specific pulmonary nodules, but no obvious metastases (Figure 1).
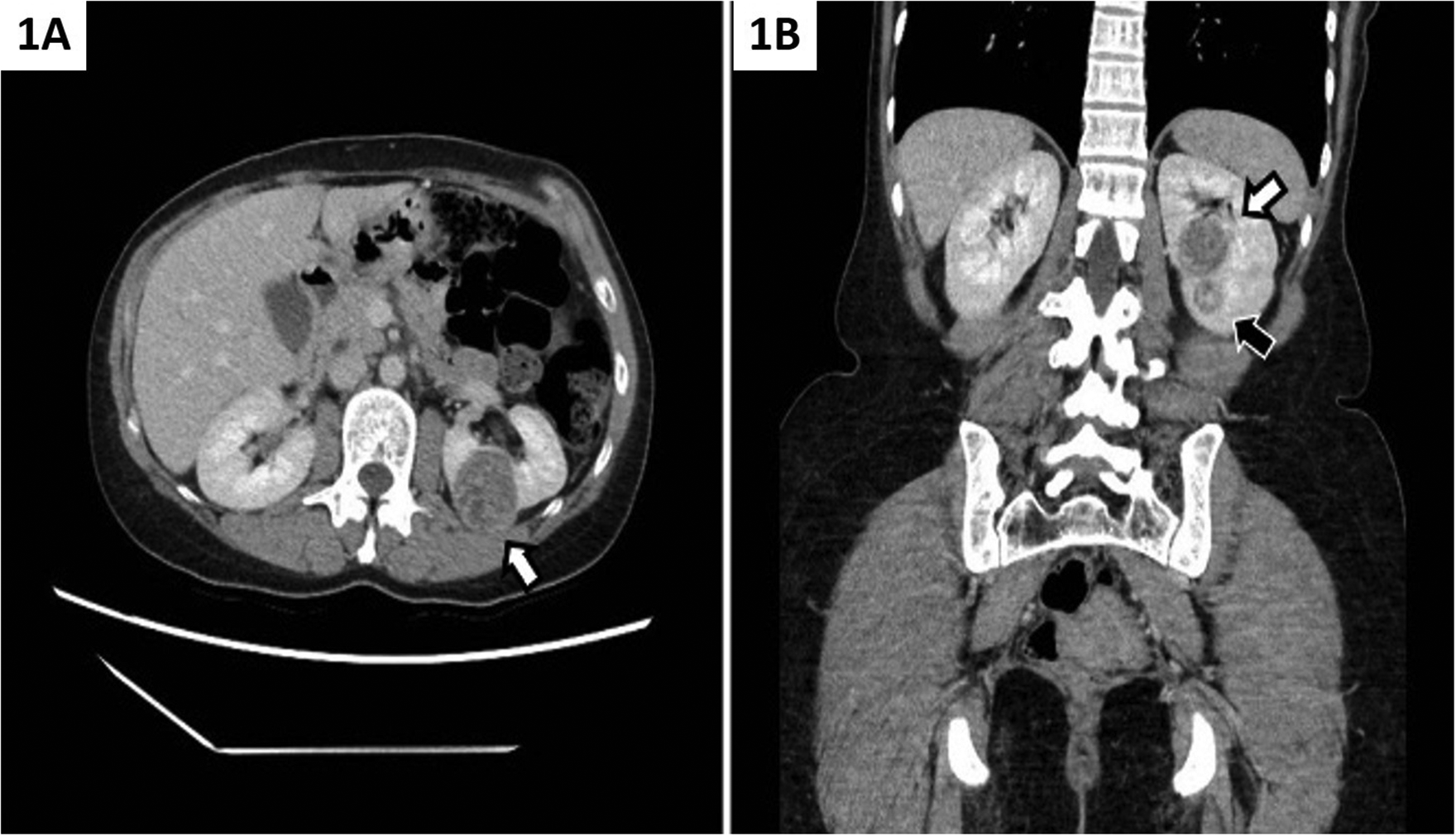
Left-sided ESC RCC in Patient 1: (A) Axial CT image showing ESC RCC (white arrow) in the middle zone of the left kidney; (B) coronal CT image showing ESC RCC (white arrow) in the middle zone of the left kidney and concurrent ccRCC (black arrow) in the lower pole of the left kidney.
A CT-guided biopsy of the dominant lesion (Figure 2) showed fragments of a neoplasm composed of sheets of cells with abundant granular eosinophilic and focally vacuolated cytoplasm and focally prominent nucleoli. Immunohistochemistry showed tumor cells positive for nuclear PAX8, CK20, CD10, RCC, and vimentin, focally positive for Alpha-methylacyl-CoA racemase (AMACR) and negative for CK7, CD117, and carbonic anhydrase IX (CAIX). The features were those of a renal cell carcinoman (World Health Organization (WHO)/International Society of Urologic Pathologists [ISUP] nuclear grade III), suggestive of ESC RCC.
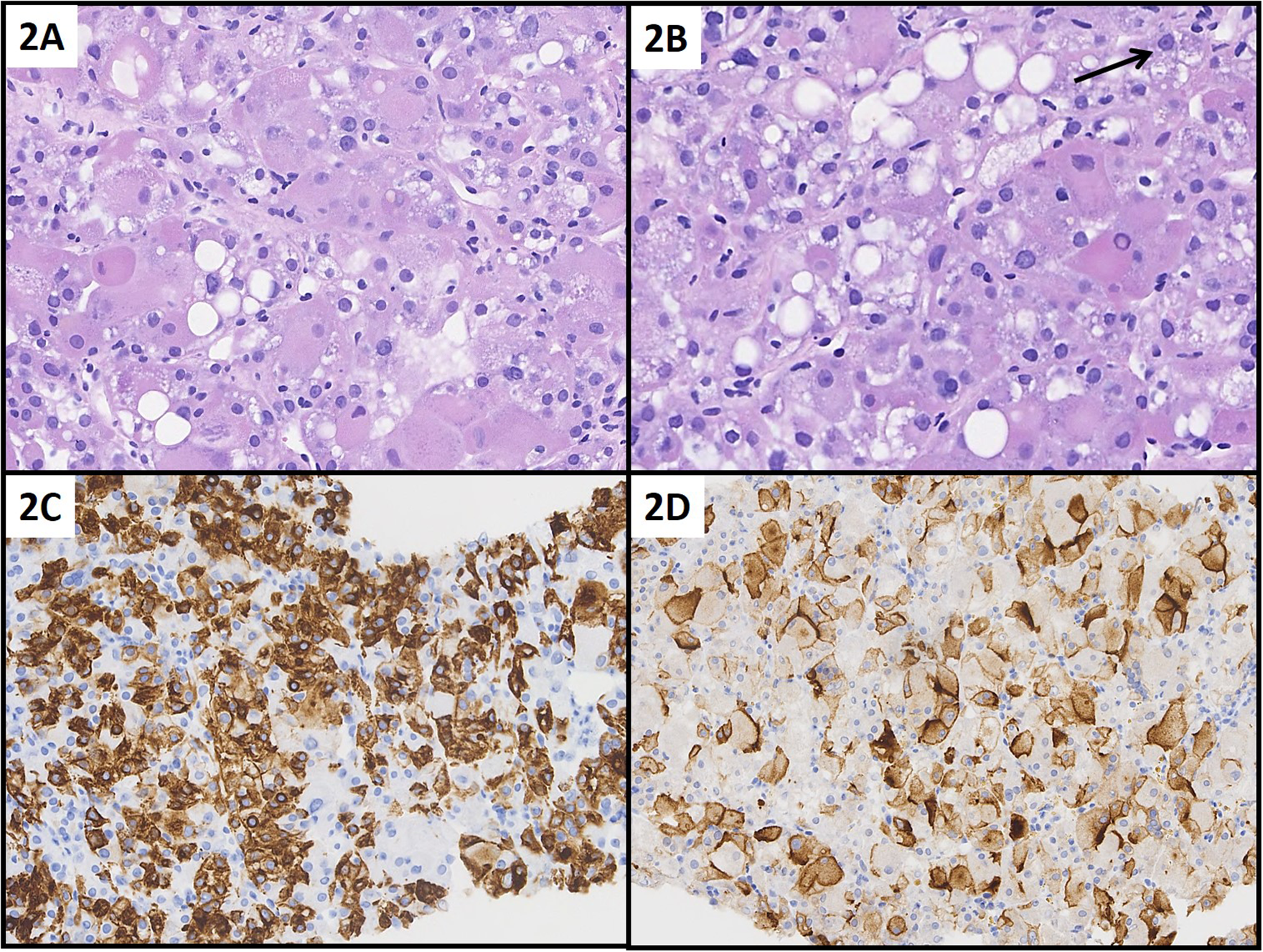
Needle core biopsy of ESC RCC in Patient 1: Neoplastic cells are large with voluminous eosinophilic and focally fine or coarse vacuolated cytoplasm (A, B). Tumor nuclei with focally prominent nucleoli (B, arrow). Tumor cells are extensively positive for CK20 (C) and CD10 (D). (A) H&E, 400×; (B) H&E, 400×; (C) CK20, 200×; (D) CD10, 200×.
The patient was evaluated by Medical Genetics and was not found to have features indicating an underlying diagnosis of TSC. Preoperative bloodwork was within normal limits. Due to the multifocal nature and size of her renal lesions, a laparoscopic left radical nephrectomy was agreed upon after a thorough discussion of risks and benefits. The patient successfully underwent an uncomplicated left laparoscopic radical nephrectomy.
A summary of the pathologic features of the tumor is seen in Table 1. The dominant lesion in the superior pole of the kidney on gross examination measured 3.5 × 3.2 × 2.8 cm, appeared tan-brown, was partly solid and cystic, and was limited to the renal parenchyma, with no lymph nodes identified. Microscopic examination (Figure 3) showed sheets and nests of large polygonal tumor cells, with abundant granular eosinophilic and focally vacuolated or microvesicular cytoplasm, and focally prominent nucleoli. Microcystic areas contained eosinophilic proteinaceous material and there were focal aggregates of foamy macrophages. Immunohistochemical studies were essentially identical to the core biopsy, with tumor cells diffusely positive for nuclear PAX8, CD10, RCC, and vimentin, strongly focally positive for CK20, weakly focally positive for AMACR (racemase), and negative for CK7, CD117, and CAIX. The tumor was classified as WHO/ISUP nuclear grade III ESC RCC.
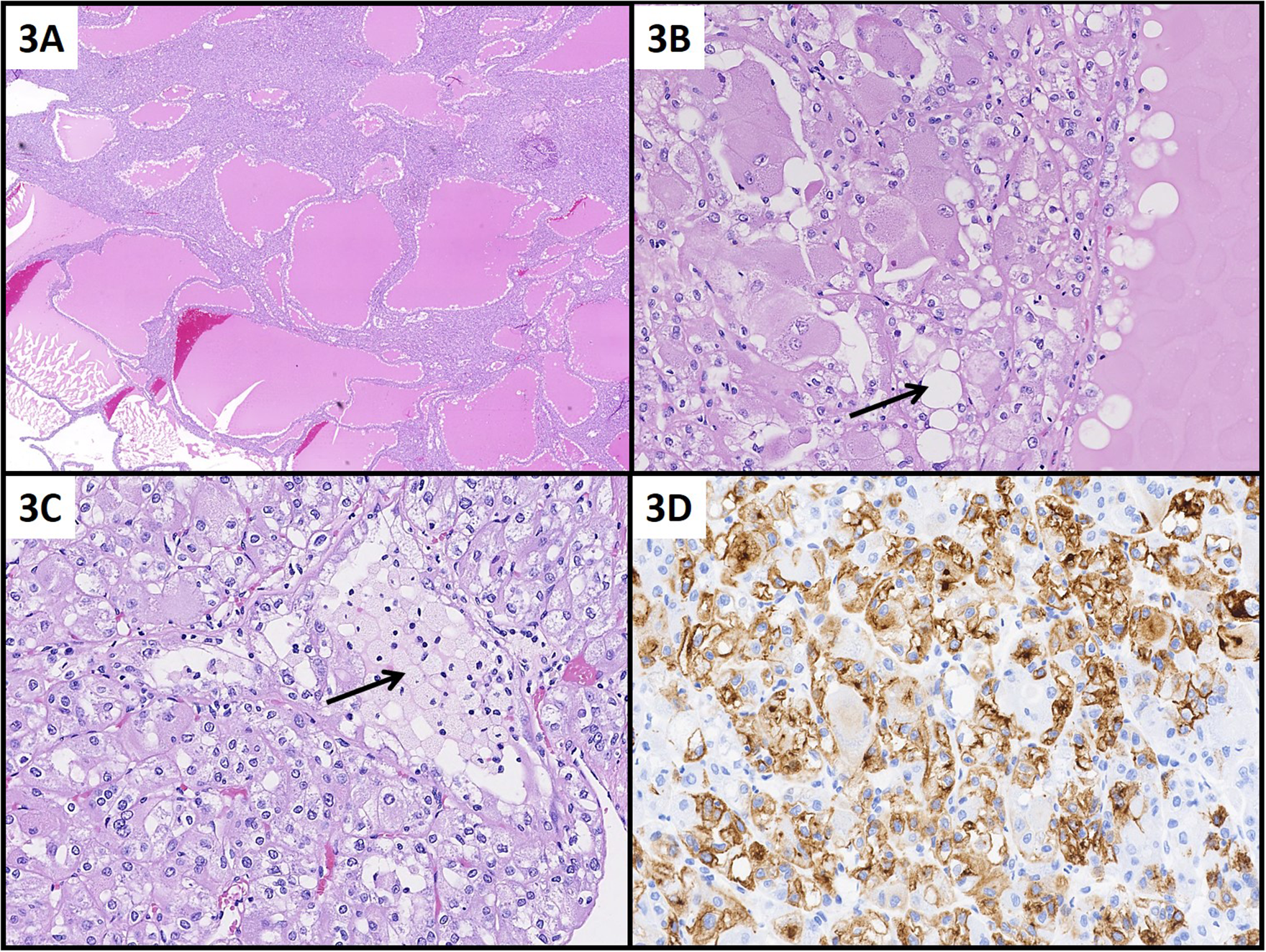
Excision specimen of ESC RCC in Patient 1: Solid and cystic architecture (A) with cystic spaces containing eosinophilic proteinaceous material and (B) with prominent vacuoles around the periphery of cystic spaces. Tumor cells have abundant eosinophilic and focally prominent cytoplasmic vacuolation (B, arrow). Small collections of foamy macrophages (C, arrow). Tumor cells are extensively positive for CK20 (D). (A) H&E, 12.5×; (B) H&E, 200×; (C) H&E, 200×; (D) CK20, 200×.
Clinical and Pathological Features of ESC RCC.

Abbreviations: ISUP, International Society of Urologic Pathologists; ESC RCC, eosinophilic solid and cystic renal cell carcinoma; F, female; RCC, renal cell carcinoma.
The second tumor, immediately inferior to the first tumor, measured 1.9 × 1.7 × 1.5 cm, was grossly bright yellow, partially solid and cystic in composition, and classified histologically (Figure 4) as conventional clear cell RCC (ccRCC, WHO/ISUP nuclear grade II). Immunohistochemistry studies of this second tumor showed tumor cells positive for CAIX, CD10, and vimentin, and negative for CK7 and CK20. Both tumors were of stage pT1a, confined within the renal capsule, and with no evidence of renal sinus, segmental renal vein, or calyceal invasion.
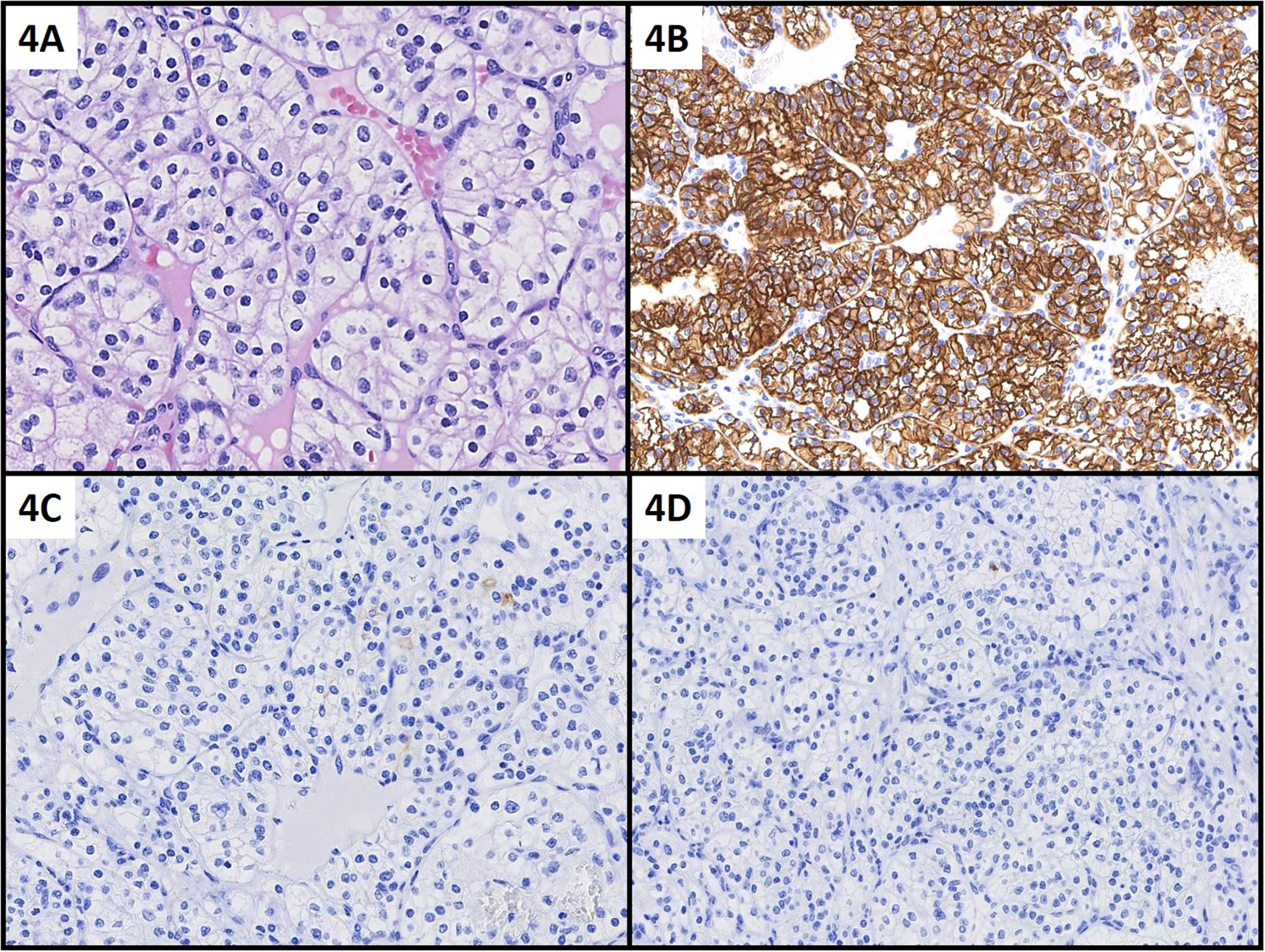
Excision specimen of separate conventional clear cell RCC in Patient 1: Nests of tumor cells with clear cytoplasm, prominent cell borders, and tumor nuclei with inconspicuous nucleoli (WHO/ISUP grade II). Tumor cells are diffusely positive for CAIX (B) and negative for CK7 (C) and CK20 (D). (A) H&E, 400×; (B) CAIX, 200×; (C) CK7, 200×; (D) CK20, 200×.
Six months following the radical nephrectomy, the patient had recovered well from their surgery and was functioning at baseline with normal bloodwork. Follow-up imaging revealed stable pulmonary nodules, consistent with non-specific pulmonary changes, and no evidence of local tumor recurrence or distant metastatic disease. The patient continues to do well as of the most recent follow-up at 1 year postnephrectomy, with no evidence of metastatic disease.
Patient 2
A 44-year-old female with an incidental right lower pole RCC was seen on an outpatient basis. This mass measured ∼4.0 cm on CT imaging and was found upon investigation for diverticulitis and abdominal pain. Previous medical history included hypothyroidism and Wolff–Parkinson–White syndrome. There was no evidence or features of an underlying diagnosis of TSC. This patient had no preoperative evidence of distant metastases. Initial bloodwork revealed an elevated white blood cell count (11.4 × 103/L), and elevated alanine aminotransferase and aspartate aminotransferase of 49 and 33 U/L, respectively. After a thorough discussion regarding the risks and benefits of intervention, radical nephrectomy was agreed upon. The patient successfully underwent an uncomplicated open right radical nephrectomy.
A summary of the pathologic features of the tumor is listed in Table 1. The radical nephrectomy specimen contained a 4.4 × 3.3 × 2.9 cm tan mass, with no extension past the renal parenchyma and no lymph nodes sampled. Microscopic evaluation (Figure 5) revealed a solid and cystic composition with tumor cells containing abundant stippled eosinophilic cytoplasm, focal hobnail arrangement of cells lining cystic spaces, scattered multinucleated cells, and focally prominent nucleoli. There were focal collections of stromal lymphoid cells. The tumor stained positively for nuclear PAX8 and vimentin. Scattered cells were positive for CK20 and tumor cells were negative for CK7 and CD117. The tumor was classified as WHO/ISUP nuclear grade III ESC RCC.
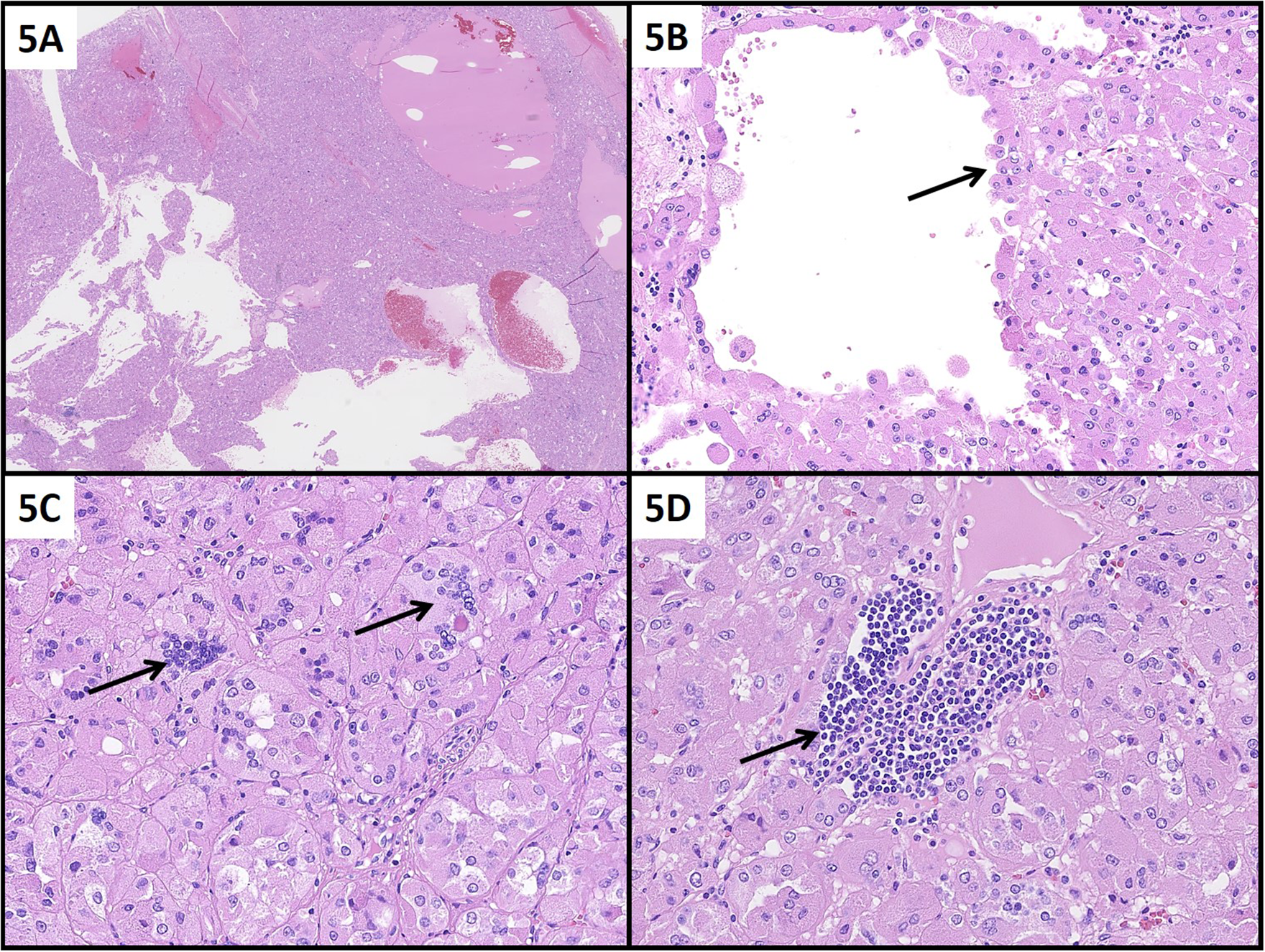
Excision specimen of ESC RCC in Patient 2: Solid and cystic architecture (A) with cystic spaces containing eosinophilic proteinaceous material and blood. Focally hobnail cells protruding into microcystic spaces (B, arrow). Tumor cells have stippled eosinophilic cytoplasm and scattered large multinucleated tumor cells (C, arrow). Focal collections of stromal lymphoid cells (D, arrow). (A) H&E, 12.5×; (B) H&E, 200×; (C) H&E, 400×; (D) H&E, 400×.
One month following the radical nephrectomy, the patient had recovered well from the procedure and was nearing her functioning at baseline. Postoperative bloodwork is listed for this patient to be within normal limits at the 6-month follow-up visit. The patient continued to do well postoperatively with no evidence of metastatic or recurrent disease at 2 years postnephrectomy.
Discussion
The clinicopathologic findings of these 2 patients’ tumors seen at our institution are consistent with the description of sporadic ESC RCC in the current literature. Table 1 summarizes the clinical and pathologic features of our 2 patients and compares them with the series reported by Trpkov et al.1,2 Our patients are both females and fall within the age range reported by Trpkov et al 1 (31-75 years). Our findings additionally correspond to reports of low-stage T1aN0M0 or T1bN0M0 disease in the majority of ESC RCC cases.1,8 Although there are reports of metastatic disease in this neoplasm, with no evidence of recurrent tumor or distant metastases, our cases, thus far, support ESC RCC as having a largely indolent clinical course. Of note, the mass in both patients was found incidentally, and features of TSC were absent. This remains consistent with emerging literature that recognizes ESC RCC as a diagnosis that may occur sporadically, as well as associated with TSC. 6 Furthermore, our cases demonstrate common gross and microscopic pathologic features found in ESC RCC including a tan appearance, abundant eosinophilic cytoplasm, and CK20+ or CK7− staining.
An incidental second primary renal tumor was additionally identified and classified as ccRCC in Patient 1. This is the first report to our knowledge of ESC RCC associated with synchronous conventional ccRCC. Whether there is a predisposition to develop both ccRCC and ESC RCC through a shared molecular pathway is unclear. The finding in the first patient may be incidental as ccRCC does occur sporadically and is the most common form of RCC (75-88%).9,10 The genetic profile of sporadic ccRCC has been thoroughly investigated, notably consisting of copy number (CN) loss at 3p, 8p, 9p, 14q, and CN gain at 5q and 7q. Several frequently mutated genes have been described in ccRCC, which are associated with syndromes including TSC 1 and 2, von Hippel–Lindau (VHL), BRCA1 associated protein-1 (BAP-1), Protein polybromo-1 (PBRM1), SET domain containing 2 (SETD2), and phosphatidylinositol 3-kinase, catalytic, alpha polypeptide (PIK3CA) among others. 11 The most common genetic alteration seen in sporadic ccRCC is chromosome 3p deletion and inactivation of the VHL suppressor gene. In comparison, the genetic profile of ESC RCC was investigated by Trpkov et al, 2 who noted CN gain at 1p, 7p-q, 10q, 13q, 16p-q, and CN loss at 19p, 19q, Xp, and Xq. Several studies have found sporadic ESC RCC to be associated with somatic TSC 1 and 2 mutations, and suggest it may arise from these mutations without associated molecular changes that cause other RCCs.6,12,13 This type of somatic genetic analysis was not carried out in our reported cases. Nonetheless, molecular karyotype analysis does not suggest significant similarities in the pathogenesis of sporadic ccRCCs and sporadic ESC RCCs. It is most likely that the occurrence of a synchronous ccRCC in Patient 1 is unrelated to the ESC RCC, and rather a consequence of ccRCC being the most common subtype of RCC.
Overall, further genetic characterization of sporadic ESC RCC would be beneficial in elucidating the relationship between sporadic ESC RCC and other common RCCs. These reported cases further characterize sporadic ESC RCC as a specific clinicopathologic entity.
Conclusion
Herein, we present two cases of sporadic localized ESC RCC with good clinical outcomes, including one case with a synchronous conventional ccRCC. Our report contributes to the growing body of evidence on this recently described rare renal tumor.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
