Abstract
Russell bodies are accumulation of immunoglobulin in plasma cells forming intracytoplasmic inclusions. Russell body colitis is rare with only 3 cases described in the English literature up to date. We report a 78-year-old male with cirrhosis showing prominent cecal infiltration of Russell body containing plasma cells. Plasma cells showed no nuclear atypia or mitoses, and no evidence of light chain restriction. In this article, we report a fourth case of Russell body colitis, that is unique in being localized to the cecum in contrast to the other 3, 1 of which was in an inflammatory polyp in the sigmoid colon, 1 in a rectal tubulovillous adenoma and 1 as part of diffuse gastrointestinal disease. This is therefore the first report of localized Russell body typhlitis, occurring in a cirrhotic patient in whom an adjacent erosion was likely nonsteroidal anti-inflammatory drug-associated, a combination that may have facilitated the formation of Russell bodies.
Keywords
Introduction
Russell bodies are accumulation of immunoglobulin in the rough endoplasmic reticulum of overstimulated plasma cells, forming intracytoplasmic eosinophilic inclusions. Plasma cells filled with Russell bodies are referred to as Mott cells. 1
In 1998 the first case of Russell body containing lesion in the gastrointestinal (GI) tract was reported in the stomach. 2 Since then around 40 cases have been reported with the majority of cases involving the upper GI tract.3-10 Colorectal Russell body lesions are very rare with only 3 cases being described in the English literature up to date.11-13 These lesions are found in those patients whose clinical presentation varied from abdominal pain, dyspepsia, nausea, and diarrhea. The first Russell body colitis was reported in 1999 in a 53-year-old female with rectal tubulovillous adenoma and high-grade dysplasia containing Mott cells. 11 In 2015, Muthukumarana et al 12 described a case of immunocompromised patient status postkidney and pancreas transplant presenting with diffuse Mott cell infiltration throughout the GI tract. 12 A third case was reported in 2017 describing a 62-year-old male with a Mott cell infiltration within a sigmoid colon inflammatory polyp against a background of severe diverticulosis. 13 Histopathological findings in all 3 cases showed dense lamina propria Mott cell infiltration with eosinophilic intracytoplasmic inclusions.11-13 Our patient had Russell bodies in the cecum, associated with an erosion that was likely related to non steroidal anti-inflammatory drug (NSAID) use.
Case Report
Clinical Presentation
The patient was a 78-year-old male with multiple comorbidities including decompensated ethanol-associated cirrhosis, chronic kidney disease (CKD), coronary artery disease (CAD), and high-grade papillary urothelial carcinoma (noninvasive), which was not treated because of his liver disease. The patient was on many medications including NSAID. He presented with hepatic encephalopathy and melena, right flank pain and hematuria. An upper endoscopy and a colonoscopy were done to investigate the source of melena and anemia. One small varix in the esophagus and mild portal hypertensive gastropathy were noted on upper endoscopy. The colonoscopy, however, showed several small cecal erosions with surrounding mild inflammation, and an area of erythema within the ascending colon. Computerized tomography (CT) KUB (kidneys, ureters, and bladder) showed pericolonic stranding involving the ascending colon extending down the flank, suggestive of portal colopathy secondary to cirrhosis. Following that he had multiple admissions for hepatic encephalopathy. The patient died of pneumonia and progressive renal failure secondary to hepatorenal syndrome 6 months postcolonoscopy after a complicated admission.
Pathological Findings
Biopsies from the cecum showed focal erosions, corresponding to the erosions seen on endoscopy, hemorrhage, and localized expansion of the lamina propria with prominent infiltration of Mott cells containing intracytoplasmic Russell bodies, as well as prominent eosinophils that extended into the submucosa (Figure 1A to C). The erosion showed attenuated crypts similar to the withering seen in ischemia (Figure 1C). Plasma cells showed no nuclear atypia or mitosis. No significant crypt architecture distortion, granuloma, or acute inflammation was seen.
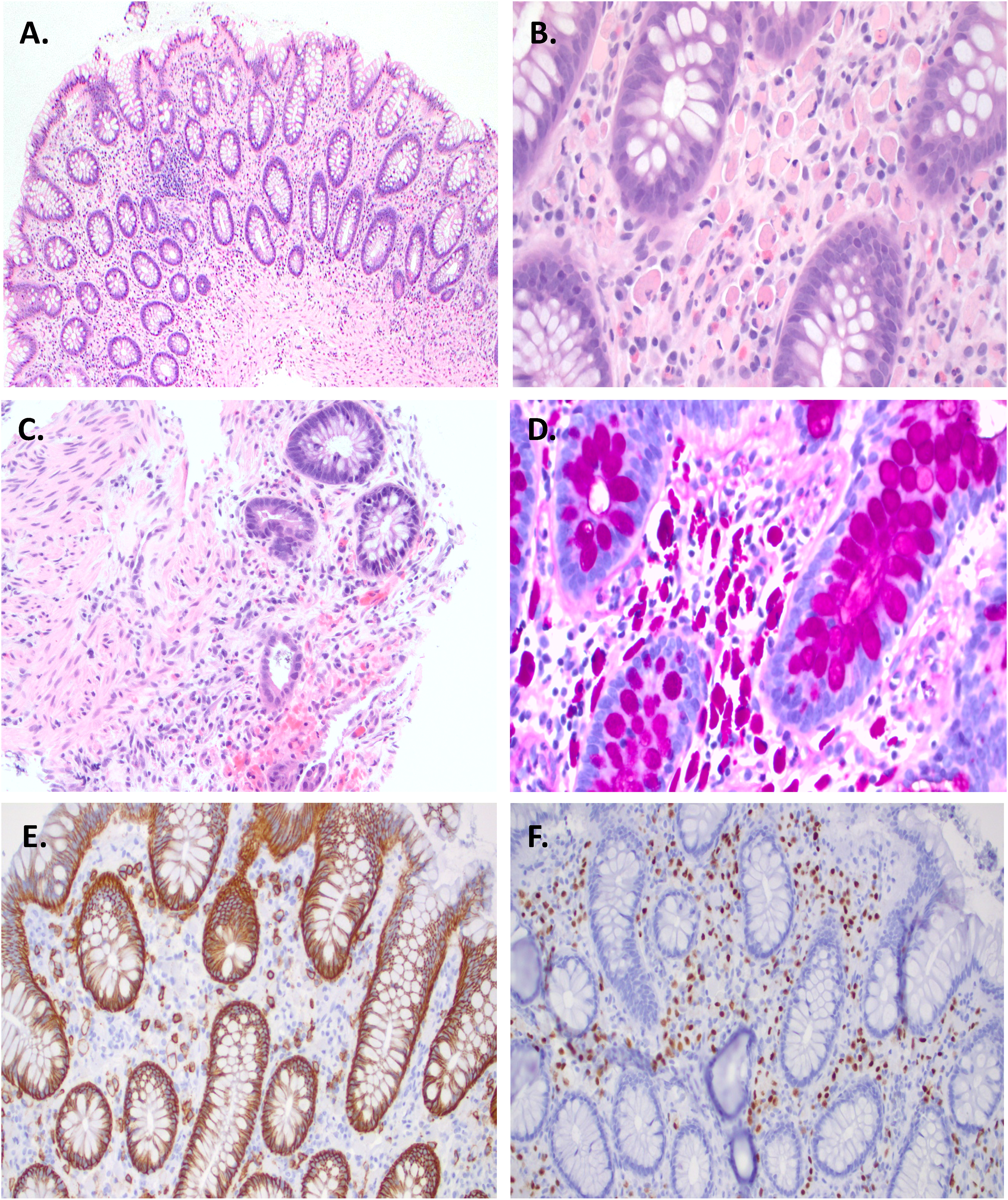
(A) Overview of Mott cells filled with Russell bodies infiltrating the lamina propria (hematoxylin-eosin, 2×). (B) Details showing Mott cells filled with Russell bodies infiltrating the lamina propria (hematoxylin-eosin, 40×). (C) Cecal mucosal erosion with attenuated reactive crypts (hematoxylin-eosin, 10×). (D) Russell bodies are periodic acid-Schiff diastase positive. (E, F) Polyclonal plasma cells seen in colon. Mott cells highlighted by CD138 (E) and MUM1 (F) (20×).
The Russell bodies stained positively with periodic acid-Schiff diastase (PASD) and were immunoreactive with cluster of differentiation 138 (CD138), multiple myeloma oncogene 1 (MUM1) (Figure 1D to F). Immunohistochemical stains showed no evidence of light chain restriction (Figure 2A and B) nor was there evidence of heavy chain restriction (Figure 2C to E). This indicates that plasma cells are of polyclonal origin.
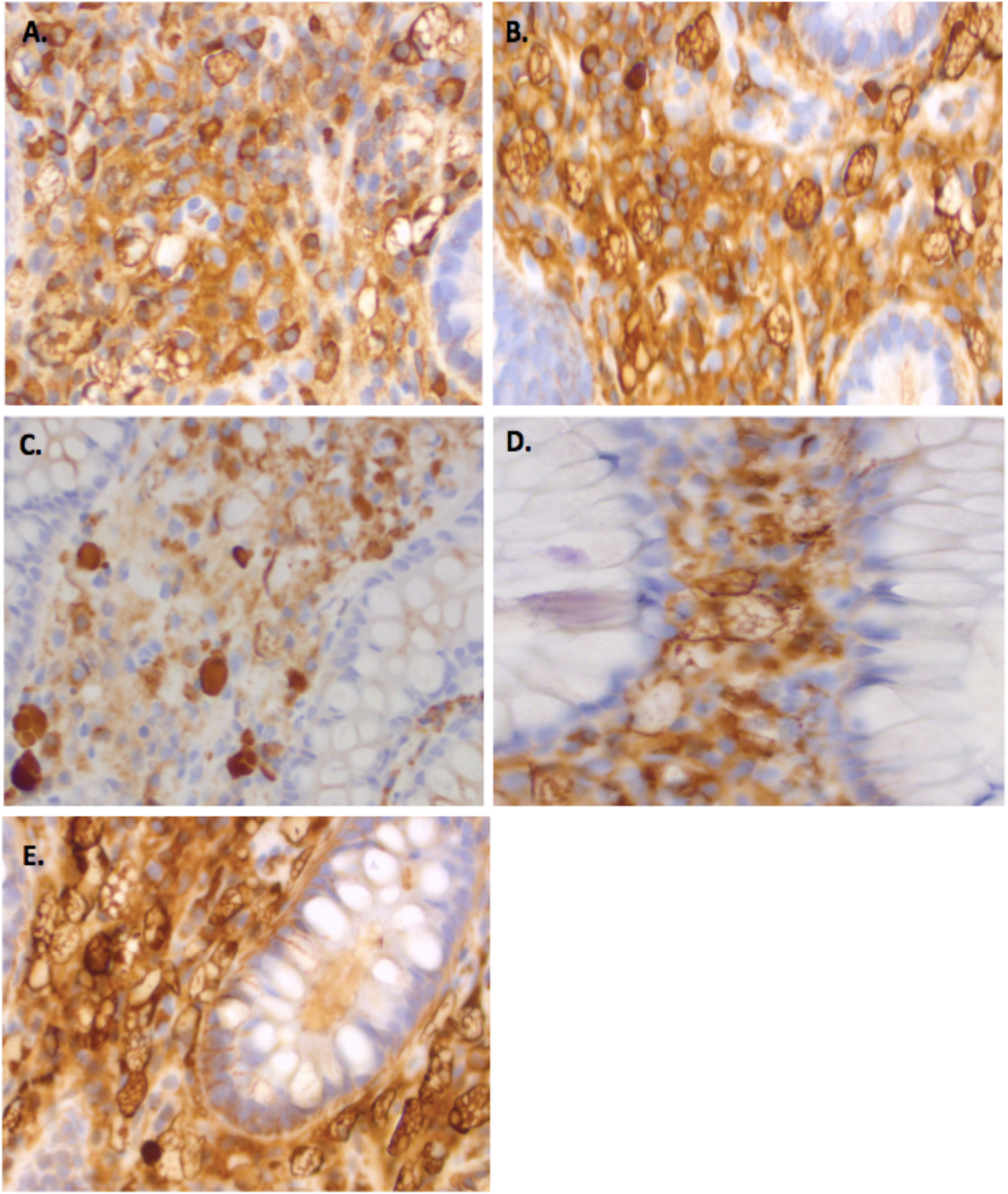
(A, B) Plasma cells were polytypic demonstrated by both kappa (A) and lambda (B) light chain expression (40×), (C–E) Mott cells staining for IgA (C), IgG (D), and IgM (E) (40×).
Biopsies from the terminal ileum, ascending colon, transverse colon, and descending colon lacked Russell body containing Mott cells. There was no evidence of acute inflammation, granuloma, or crypt architectural distortion to suggest inflammatory bowel disease, nor of an excess of intraepithelial lymphocytes, or a thickened subepithelial collagen band to suggest either major form of microscopic colitis.
Discussion
Russell body colitis is a rare benign inflammatory condition, characterized by the presence of Mott cells within the lamina propria of the colon. 3 The exact pathogenic mechanism is not fully understood. It has been postulated that chemokine production associated with chronic inflammation leads to overstimulation of plasma cells and entrapment of nondegradable immunoglobulins in the rough endoplasmic reticulum, forming Russell bodies.3,4,14 Somatic hypermutation of immunoglobulin genes have been suggested to result in the formation of abnormal immunoglobulins that are neither secreted nor degraded forming intracytoplasmic Russell body globules.15,16 This might suggest that mutated immunoglobulins fail to be recognized by the cell transport mechanism hindering their secretion across the cell.
Although rarely found in the GI tract, Russell bodies are commonly seen in malignancies of hematopoietic origin such as mucosa associated lymphoid tissue (MALT) lymphoma, plasmacytoma, lymphoplasmacytic lymphoma, and in association with chronic inflammation, autoimmune diseases such as Hashimoto's thyroiditis.17,18 Unlike plasma cell neoplasms, the benign monoclonal plasma cells seen in Russell body containing lesion lack nuclear atypia and mitotic activity.3,11 In 1 case series, Zhang et al 9 demonstrated 9 cases of monoclonal Mott cells lacking evidence of malignancy.
In the present case, a diagnosis of Russell body colitis was made based on the histological findings of dense infiltration of benign plasma cells with Russell body globules (stained with PASD). These plasma cells positively stained with CD138 and MUM1, and lacked neoplastic features such as nuclear atypia and mitosis.
The reason for cecal erosions and accumulation of Mott cells observed here is unclear. Our patient was on aspirin, an NSAID that is known to cause mucosal erosions in the lower GI tract. 19 In this case, NSAIDs could have possibly provoked a chronic inflammation and mucosal erosions, which lead to Russell body formation and Mott cell proliferation. These findings suggest a unique reactive inflammatory process driving Russell body formation in the setting of NSAID use. The alternative explanation is that the coexistence of both lesions at the same place was coincidental. As cirrhotic patients are known to be more susceptible to infections, 20 the combination of this and NSAIDs, an anti-inflammatory, may have been sufficient to produce a localized Russell body reaction in this patient.
The case presented here differs from the other Russell body colitis cases in several aspects. First, our patient presented with cecal erosions and isolated Russell body typhlitis. In contrast, previously reported cases demonstrated Russell body infiltration either within a polyp (2 inflammatory and 1 a tubulovillous adenoma) or multifocal involvement of the GI tract in an immunocompromised patient.11-13 Second, the lesion in 2 of the cases reported had polyclonal plasma cell infiltration, and 1 presented with immunoglobulin G (IgG) monoclonal plasma cells (kappa light chain restriction) with no evidence of malignancy.11-13 In comparison, our case is unique as it demonstrates Mott cells staining for IgG, immunoglobulin M (IgM), as well as immunoglobulin A (IgA), with no evidence of light chain restriction.
In conclusion, Russell body colitis is an extremely rare entity with very few cases reported in the literature. Our case is the first to occur in mucosa that is not polypoid or part of diffuse intestinal disease, presenting as an isolated Russell body typhlitis with cecal erosions that may have been NSAID associated in a clinical setting of cirrhosis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
