Abstract
Carcinomas of the lung with benign osteoclast-like giant cells are rare. A literature search showed only 8 previously reported examples. These tumors resemble a giant cell tumor of bone. Many of these tumors, which occur in most epithelium-containing organs, are composed of an undifferentiated, sarcomatoid component that contains benign osteoclast-like giant cells and a conventional carcinoma. In some tumors the epithelial origin may be revealed by immunohistochemistry only; others lack any evidence of an epithelial component. A 59-year-old man had an inoperable tumor in the upper lobe of the left lung. The tumor did not respond to radiation therapy, and chemotherapy resulted in minimal relief of symptoms. Light microscopy of biopsy samples showed benign osteoclast-like giant cells distributed irregularly between proliferations of undifferentiated medium-sized tumor cells. Approximately one third of the undifferentiated tumor cells were cytokeratin AE1/AE3-positive, and a minor alveolar clear cell component of the tumor was cytokeratin 7-positive. The osteoclast-like giant cells were strongly CD68-positive. The clinical and histologic findings supported the diagnosis of a non–small cell carcinoma of the lung with benign osteoclast-like giant cells. The differential diagnosis is composed of giant cell carcinoma, carcinosarcoma, and mesenchymal tumors of the lung.
Introduction
Carcinomas of the lung with osteoclast-like giant cells are very rare tumors, and only 8 cases have been reported since the initial description of the entity in 1963.1-6 Carcinomas that have scattered multinucleated giant cells comparable to normal osteoclasts have been described in varied sites including the thyroid, breast, pancreas, salivary glands, and liver. 7 In most instances the osteoclast-like giant cells of these carcinomas occur in (1) sarcomatous proliferations without any epithelial element; (2) sarcomatous proliferations with a minimal epithelial component, as noted by electron microscopy or immunohistochemical evaluation; or (3) sarcomatous proliferations combined with a conventional carcinoma. 8 The sarcomatous or metaplastic portion of these carcinomas is usually described as a tumor that may include round to spindle-shaped, highly pleomorphic, neoplastic mononuclear cells and nonneoplastic, multinucleated, histiocytic giant cells. 9 Only in breast carcinomas is a sarcomatous element rare, and the osteoclast-like giant cells usually lie close to the borders of carcinomatous glands. 10
The initial diagnosis of the present case was that of an adenocarcinoma based on electron microscopic findings that were available at that time. 11 A few unstained archival slides recently allowed an, if limited, immunohistochemical reevaluation. The purpose of this report was to present the clinical and histological features of this rare lung carcinoma.
Clinical Summary
A 59-year-old man had a 5-week history of chest pain, frequent cough, hemoptysis, dyspnea, and a 5-kg weight loss. Past medical history showed no major illness. He smoked 100 g of tobacco per week. Chest radiography showed a left upper lobe pulmonary lesion, and a tumor was suspected. A computed tomography scan showed no tumor in the liver or spleen or bony destruction. Bronchoscopy showed a slight stenosis of the left main bronchus, but no intraluminal tumor was evident. Mediastinoscopy showed no tumorous infiltrate. Thoracotomy showed a 5-cm diameter tumor that adhered to the lateral thoracic wall and tumors in the central hilum; therefore, the lung tumor was deemed not amenable to curable surgery. The tumor did not respond to radiotherapy, but chemotherapy provided symptomatic relief from shortness of breath. However, the tumor metastasized to the right lung, and the general condition of the patient worsened. The patient died 4 months after the primary diagnosis. No autopsy was performed.
Material and Methods
Biopsy samples (diameter, 5 mm) were fixed in 10% formalin and embedded in paraffin. Staining (hematoxylin-eosin and periodic acid-Schiff [PAS] stains) was performed on 5-µm sections. Acid phosphatase activity was determined with 7-bromo-3-hydroxy-2-naphthoic-o-anisidide phosphate (naphthol AS-BI phosphate, Sigma-Aldrich, Taufkirchen, Germany) as substrate and pararosaniline (Sigma-Aldrich) as a coupler. Immunostaining and mucicarmine stain (Morphisto GmbH, Frankfurt am Main, Germany) were performed on 5 archived unstained slides that were retrieved 35 years after the initial diagnosis and on destained slides.
The following antibodies were applied: cytokeratin AE1/AE3 (1:100, Zytomed Systems GmbH, Berlin, Germany), cytokeratin 7 (CK7; OV-TL12/30, 1:100, Dako Denmark A/S, Glostrup, Denmark), CD68 (KP1, 1:3000, Dako Denmark A/S), p40 (1:100, Zytomed Systems GmbH), thyroid transcription factor 1 (TTF-1; 8G7G3/1, 1:100, Zytomed Systems GmbH), and napsin A (KCG1.1, 1:50, Zytomed Systems GmbH). The deparaffinized or destained and rehydrated sections were subjected to heat-induced epitope retrieval with citrate buffer (pH 6.0) at 1:10 dilution (Zytomed) in an oven (96°C). Staining was performed in an automated stainer (Dako Autostainer, Dako). For detection, a streptavidin-biotin kit was used (ZytoChem-Plus, Broad Spectrum, Zytomed Systems GmbH) and combined with a 3,3′-diaminobenzidine tetrahydrochloride chromogen (DAB substrate kit, Zytomed Systems GmbH).
Pathological Findings
Histologically, the lung tumor consisted predominantly of solid proliferation foci of polymorphous oval or spindle-shaped cells of moderate size with a small, slightly eosinophilic, ill-defined cytoplasm and polymorphic nuclei. Many nuclei had an oval shape, moderate size, and finely granular chromatin, usually with multiple small nucleoli. Mitotic figures were numerous. The tumor cells occasionally formed cohesive strands. In some areas, the tumor cells were mostly spindle-shaped and had intensely eosinophilic cytoplasm; these cells contained spindle-shaped nuclei that were polymorphic and hyperchromatic. In very few areas, tumor cells with small, water clear cytoplasm and small round nuclei were arranged in alveolar structures; these cells contained strongly PAS-positive vacuoles, and the cell membranes were intensely stained. Mucin, however, was absent in the mucicarmine stain. Osteoclast-like giant cells were irregularly distributed among the smaller undifferentiated tumor cells, and the tumor resembled a giant cell tumor of bone (Figure 1). They had a large eosinophilic cytoplasm, contained many small, uniformly round nuclei with a small nucleolus, and had an overall benign appearance. These osteoclast-like cells and many small cells with 1 or 2 nuclei stained strongly positive for acid phosphatase (Figure 2).
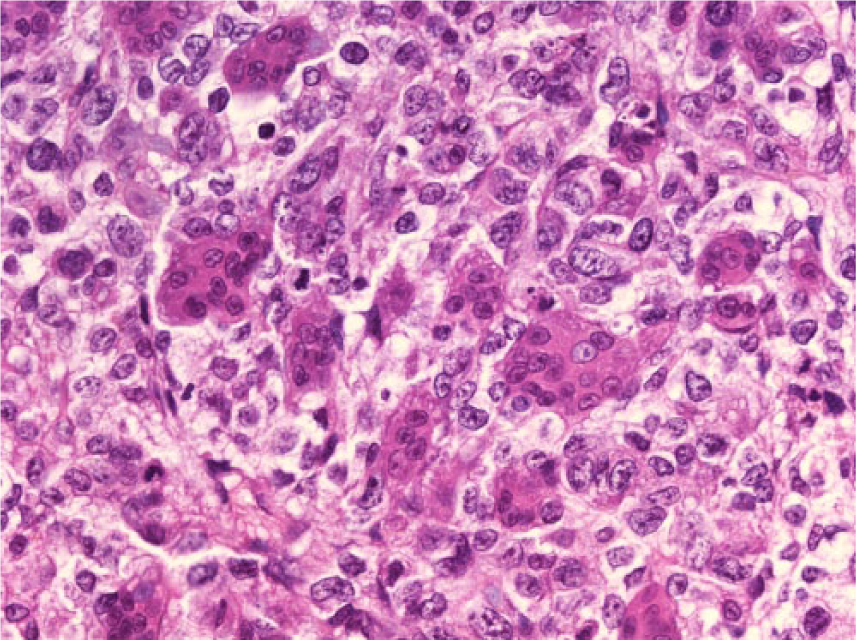
Non–small cell carcinoma of the lung with osteoclast-like giant cells (hematoxylin-eosin, original magnification ×400).
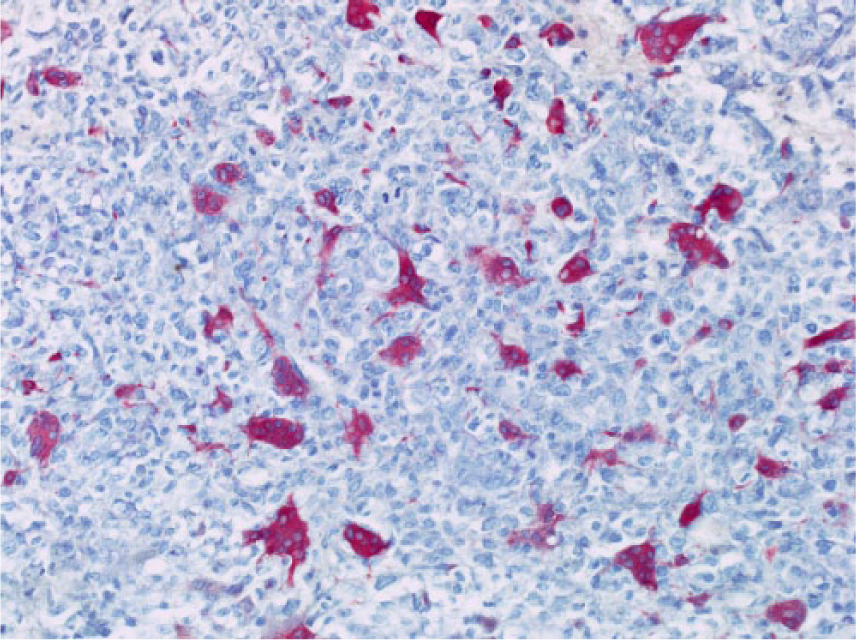
Osteoclast-like giant cells showed strong activity for acid phosphatase (original magnification ×200).
Immunohistochemically, the osteoclast-like giant cells and small mononuclear cells were also strongly CD68-positive. Approximately one third of the undifferentiated tumor cells stained positive for cytokeratin AE1/AE3 (Figure 3). The clear cell component of the tumor was positive for CK7, with the reaction limited to cell membranes (Figure 4). The reactions were negative for the pneumocyte markers TTF-1 and napsin A as well as for the squamous marker p40.
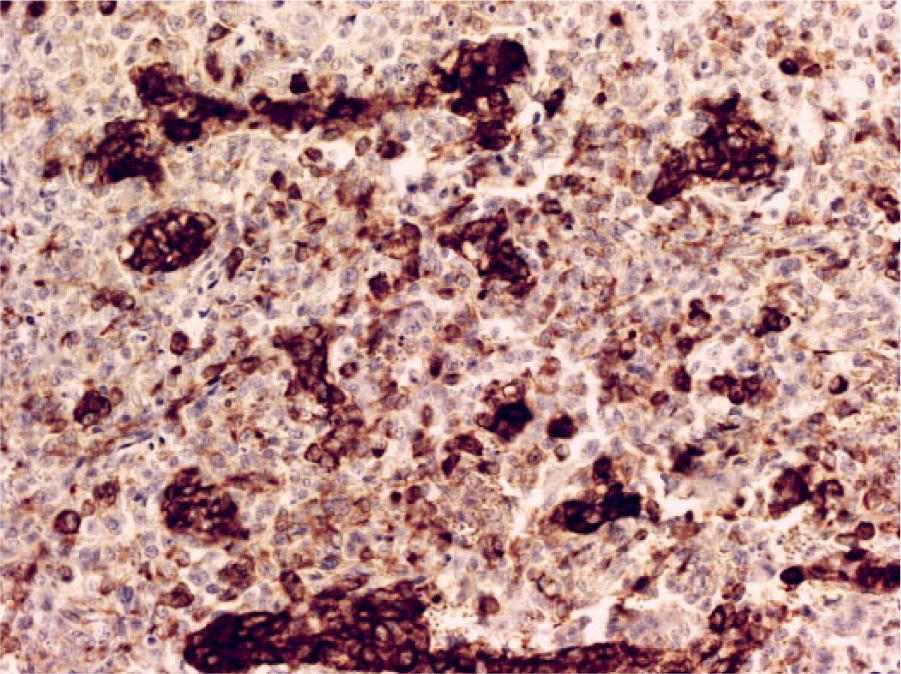
Strands and clusters of tumor cells were positive for cytokeratin AE1/AE3 (original magnification ×200).
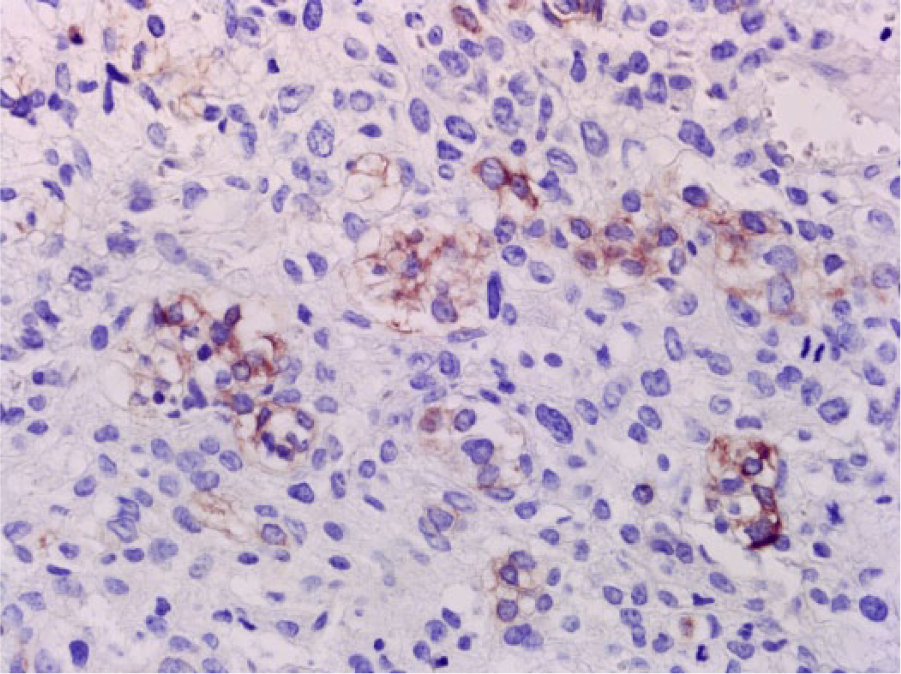
The clear cell component of the tumor was positive for cytokeratin 7 (original magnification ×400).
Discussion
This 59-year-old man had a 5-cm diameter tumor in the upper lobe of the left lung, and histologic evaluation showed a predominantly solid growth pattern of atypical cells of moderate size with a small eosinophilic cytoplasm. Numerous mitotic figures were signs of high-grade malignancy. With numerous osteoclast-like giant cells that were irregularly distributed among the malignant cells, the tumor had close similarity to a giant cell tumor of bone. The osteoclast-like giant cells and many small mononuclear cells were strongly positive for acid phosphatase and CD68, suggesting a histiocytic origin. Approximately one third of the undifferentiated tumor cells were cytokeratin AE1/AE3-positive. Groups of cells with clear cytoplasm and PAS-positive vacuoles that formed alveolar structures were positive for CK7. Mucin-containing tumor cells were not included. Staining was negative for the pneumocyte markers TTF-1 and napsin A, probably because the epitope retrieval of the aged paraffin sections was no longer successful for these antibodies. In addition, staining for p40 showed no squamous cell component of the tumor.
The diagnosis of a primary non–small cell carcinoma, favor adenocarcinoma, of the lung might have been supported by the minor CK7-positive clear cell component of the tumor. However, none of these elements is universally accepted as diagnostic in non–resection specimens, and a positive mucin stain or TTF1 reaction are required. 12 A negative reaction for TTF1 and napsin A, on the other hand, does not exclude the diagnosis of an adenocarcinoma as this is known to occur in 15% to 20% of morphological adenocarcinomas, and a carcinoma with the features of this tumor throughout a resection specimen would qualify as large cell carcinoma. 13
In the present case, the diagnosis of a primary non–small cell carcinoma of the lung with a minor clear cell component and osteoclast-like giant cells based on a biopsy specimen was supported by the clinical data, the clusters of CK7- and PAS-positive clear cells in several areas in this tumor of pleomorphic cells that to about one third were cytokeratin AE1/AE3-positive.
Carcinomas of the lung with osteoclast-like giant cells are very rare tumors, and only 8 cases have been reported since the initial description of the entity in 1963.1-6 Tumors that have scattered multinucleated giant cells comparable to normal osteoclasts have been described in varied sites. 7 The sarcomatous portion of these carcinomas is usually described as a tumor that may include round to spindle-shaped, highly pleomorphic, neoplastic mononuclear cells and nonneoplastic, multinucleated, histiocytic giant cells, 9 and is considered to represent the metaplastic or dedifferentiated part of the carcinoma. 7 There is sufficient proof for this assumption in several cases. 8
In most of the carcinomas of the lung with osteoclast-like giant cells that were previously published, the sarcomatous tumor portion was combined with a conventional squamous cell carcinoma,2,3 or adenocarcinoma.4,5 In another case, the osteoclast-like giant cells occurred in a cytokeratin-positive large cell carcinoma. 6 The description of a further case does not mention epithelial elements. 1 Two of the 8 tumors were described as sarcomatoid carcinomas, composed of malignant epithelial-appearing and spindle cell elements. 5 However, this tumor type may lack any evidence of an epithelial component. 8 Thus, a lung tumor with the same typical histologic features was depicted as of purely mesenchymal origin. 14
The characteristic nonneoplastic histiocytic multinucleated giant cells of these rare tumors resemble osteoclasts and share many features with them.7,15 The mechanism of their formation in these tumors is unknown but may represent a histiocytic reaction that is initiated by the carcinoma cells themselves.7,15
Lung carcinomas with benign osteoclast-like giant cells should be differentiated from pleomorphic and giant cell carcinomas that contain or consist essentially of malignant tumor giant cells1,16 or from carcinosarcomas with an osteosarcomatous component and malignant osteoclast-like giant cells.17,18 Resection specimens are necessary to establish the definite diagnosis of all of these tumors, too. 16 The differential diagnosis also may include primary or secondary benign pulmonary giant cell tumors of bone 19 and primary benign or malignant mesenchymal tumors that contain osteoclast-like giant cells.5,20
Footnotes
Acknowledgements
The author thanks Banu Şeker for her excellent technical assistance.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
