Abstract
Background:
Rasburicase is used to prevent and treat tumor lysis syndrome (TLS) by breaking down uric acid. Administration methods vary, including fixed, weight-based (0.15-0.2 mg/kg), single and multiple doses. Our primary objective is to describe rasburicase use in both adults and children for TLS prevention and treatment. Secondary objectives are to describe the efficacy and safety of different regimens inventoried. Exploratory objective is to estimate savings associated with use of a single fixed dose.
Methods:
Retrospective descriptive study conducted among patients who received rasburicase for TLS prevention or treatment between January 2014 and April 2024 in 3 oncology services of the Hospital Center.
Results:
A total of 133 adults and 60 children were included, of which 42.9% and 5.0%, respectively, presented biochemical TLS pre-rasburicase. A single 6 mg dose was administered in 86.5% of adults and 21.7% of children. Multiple daily doses were used in 24.8% of adults and 76.7% of children. Few adults (1.5%) received weight-based doses, contrary to children (76.7%). Normalized uric acid (< 476 μmol/L) was observed in 97.9% of patients 24 hours after a first dose, with no serious adverse events. Estimated savings were 86 542.92 CA$ if all patients included (n = 193) received a single 0.15 mg/kg dose, capped at 6 mg.
Conclusion and relevance:
In the adult and pediatric population, a single rasburicase dose of 0.15 to 0.2 mg/kg, capped at 6 mg, represents an effective, safe, and more cost-effective option for both the prevention and treatment of TLS. Further studies are warranted to compare single versus multiple dosing in pediatric populations and to identify potential risk factors for nonresponders.
Keywords
Introduction
Tumor lysis syndrome (TLS) is an oncological emergency where rapid tumor cell breakdown releases potassium, phosphorus, and uric acid precursors into the bloodstream, risking arrhythmias, tetany, seizures, and kidney failure.1-5 Up to a third of TLS cases require dialysis, and around 15% are fatal.2,5 TLS incidence varies from 2% to 30% in hematological cancer patients, depending on risk factors.1-3 In particular, certain hematological cancers are at increased risk of TLS due to a high proliferation rate, rapid cell turnover, large tumor burden, or high sensitivity to chemotherapy.1-3 These include acute lymphoblastic lymphoma (ALL), acute myeloid leukemia (AML), Burkitt’s lymphoma, diffuse large B-cell lymphoma, and high-grade tumors.1,2,5,6 Other risk factors include pre-existing renal insufficiency or hyperuricemia, dehydration, or use of drugs that can increase blood uric acid levels.1-5,7
In current practice, hydration and fluid management are essential for both TLS treatment and prevention, along with careful monitoring and aggressive correction of relevant biochemical parameters. 5 If needed for uric acid reduction, allopurinol can be used in low- to moderate-risk patients, or rasburicase for high-risk patients.1,3,5 For established TLS, rasburicase is preferred due to its superior efficacy and time to action compared with allopurinol.3,5 Some experts discourage combining rasburicase with allopurinol. 5
Rasburicase is a recombinant urate oxidase approved for hyperuricemia prevention and treatment in oncology patients.8,9 In a phase 3 randomized study in adults with hematological malignancies (N = 275), the percentage of patients with normalized uric acid ≤ 446 μmol/L at 3 to 7 days was 87% with rasburicase 0.2 mg/kg/day for 5 days, versus 66% with allopurinol 300 mg/day for 5 days. Time to uric acid control in hyperuricemic patients after a first dose was 4 hours for rasburicase and 27 hours for allopurinol. 10
Limited data exist regarding common characteristics of rasburicase nonresponders, including a retrospective study of 92 patients, of which 5 were nonresponders, defined as having uric acid was > 446 μmol/L at 24 to 36 hours after a first dose or those requiring a second dose within 24 hours. In these patients, baseline uric acid was higher among nonresponders, with no difference found in body mass index (BMI), creatinine, or creatinine clearance. 11
Rasburicase is well tolerated, with rare serious adverse events, including hypersensitivity reactions, methemoglobinemia and hemolytic anemia.8,9 The main contraindication to rasburicase administration is the presence of glucose-6-phosphate dehydrogenase (G6PD) deficiency due to the risk of causing hemolytic anemia in these patients.9,12,13 Screening for G6PD deficiency is thus advised in patients receiving rasburicase, especially in populations at higher risk, such as African, Asian, Middle Eastern, and Mediterranean descent. 9
Many studies have explored describing and comparing fixed, weight-based, single and repeated rasburicase doses in the prevention and treatment of TLS to reduce costs while maintaining efficacy and safety.5,14-17 However, most studies focus on limited dosing regimens, often exclude either adult or pediatric populations, or address only TLS prevention or treatment.14,15,17 Pediatric studies identified also had relatively small sample sizes (n ≤ 53). 17
The primary objective of our study is to describe real-world rasburicase use for TLS prevention and treatment in adult and pediatric populations in an academic Canadian network of hospitals offering specialized cancer care. Secondary objectives are to compare the efficacy and safety of the rasburicase regimens inventoried. Additional exploratory objectives are to describe the rasburicase nonresponders and to estimate the costs that could have been saved by the use of single fixed dosing.
Method
Study Design and Population
A retrospective, longitudinal, descriptive study was conducted at a network of 5 North American academic hospitals. The medical records of patients who received rasburicase between January 1, 2014, and April 1, 2024, were reviewed in 3 of these hospitals offering specialized cancer care, 2 of which to adults, 1 to children. The lists of eligible patients in each of the hospitals were retrieved from the hospital’s medico-administrative database and medical records were manually reviewed to confirm inclusion criteria and collect relevant data. All eligible patients at the pediatric center were screened. For the 2 adult centers, the lists of eligible patients ordinated randomly were successively screened until an approximately equal number of patients, comparable with that of the pediatric center, were included for each of the 2 hospitals. Inclusion criteria required receiving rasburicase for the prevention or treatment of TLS. Patients who received rasburicase for any other indication were excluded. Only the first treatment course was included, defined as the 7-day period from the initial dose. Children were defined as aged < 18 years.
Data Collection
Collected data included patient characteristics, rasburicase-related efficacy criteria, adverse events (methemoglobinemia, hemolytic anemia and grade ≥ 3 hypersensitivity reactions defined by Common Terminology Criteria for Adverse Events Version 5.0), the use of drugs known to affect serum uric acid (Supplemental Table A), TLS complications (arrhythmias, tetany, seizures, dialysis, and death), and biochemical results. 18 Data were collected up to 7 days following a first rasburicase dose.
Biochemical TLS and risk classification followed Cairo et al 7 (Table 1 19 and Supplemental Table B, respectively). Renal impairment was defined as creatinine > 2 times the upper institutional laboratory limit in patients aged < 13 years and as > 123 μmol/L otherwise.20,21 A bulky mass was defined as measuring ≥ 10 cm using a computed tomography scan or a positron emission tomography scan. 22 Normalized uric acid was defined as < 476 μmol/L 24 hours post first rasburicase dose. 10 Calcium values corrected for albumin (calcium: 0.025 [albumin: 40]) were used. The most recent biochemical value of the collected periods was recorded. Baseline values correspond to the most recent values measured a week or more before the first dose of rasburicase.
Cairo-Bishop Definition of Biochemical Tumor Lysis Syndrome.

Based on: Cairo and Bishop. 19
Data collection and management was performed with REDCap (Research Electronic Data Capture) software. No patients were excluded due to missing data, which was marked as “unknown” and accounted for in the statistical analyses. Two hospital pharmacy residents and an undergraduate student collected data. For the latter, a systematic validation of collected data for every tenth record was performed by the pharmacy residents.
The estimated cost saved per patient was determined by comparing the cost of a scenario where all patients in the study would have received a single dose (0.15 mg/kg/dose, up to 6 mg/dose) versus the actual cost of all doses administered during the treatment course of all patients in the study, using the following equation: Cost saved ($CAD) per patient = ([total cost of capped single dosing scenario] – [actual cost of all doses administered])/total number of patients. The cost per dose of rasburicase, rounded up to the nearest vial of 1.5 mg, was calculated based on $141.41 CAD per vial, as per the negotiated price agreement in effect for hospitals in the province of Quebec.
Statistical Analysis
Descriptive statistics were performed using IBM SPSS Statistics 29, and group comparisons and correlations were done in SAS 9.4. Analyses were stratified by age (pediatric vs adults) and by reason to initiate rasburicase (TLS prevention vs and treatment). Continuous variables were compared using the Wilcoxon-Mann-Whitney or Student’s t-test and categorical variables with the exact Pearson chi-squared and Wald tests. The Pearson and Spearman tests assessed correlation between rasburicase dosing and efficacy outcomes. Statistical significance was set at a 2-tailed P value < 0.05. The study was approved by CHU de Québec-University Laval ethics committee (no. 2024-7388) and the Quebec Commission for Access to Information to collect health data from medical records without patients’ written consent.
Results
Study Population
Of the 249 screened patients, 193 were included, with their characteristics detailed in Table 2. All 6.7% tested for G6PD deficiency were negative.
Patient, Malignancy, Treatment, and Tumor Lysis Syndrome Characteristics Before Administration of a First Dose of Rasburicase.
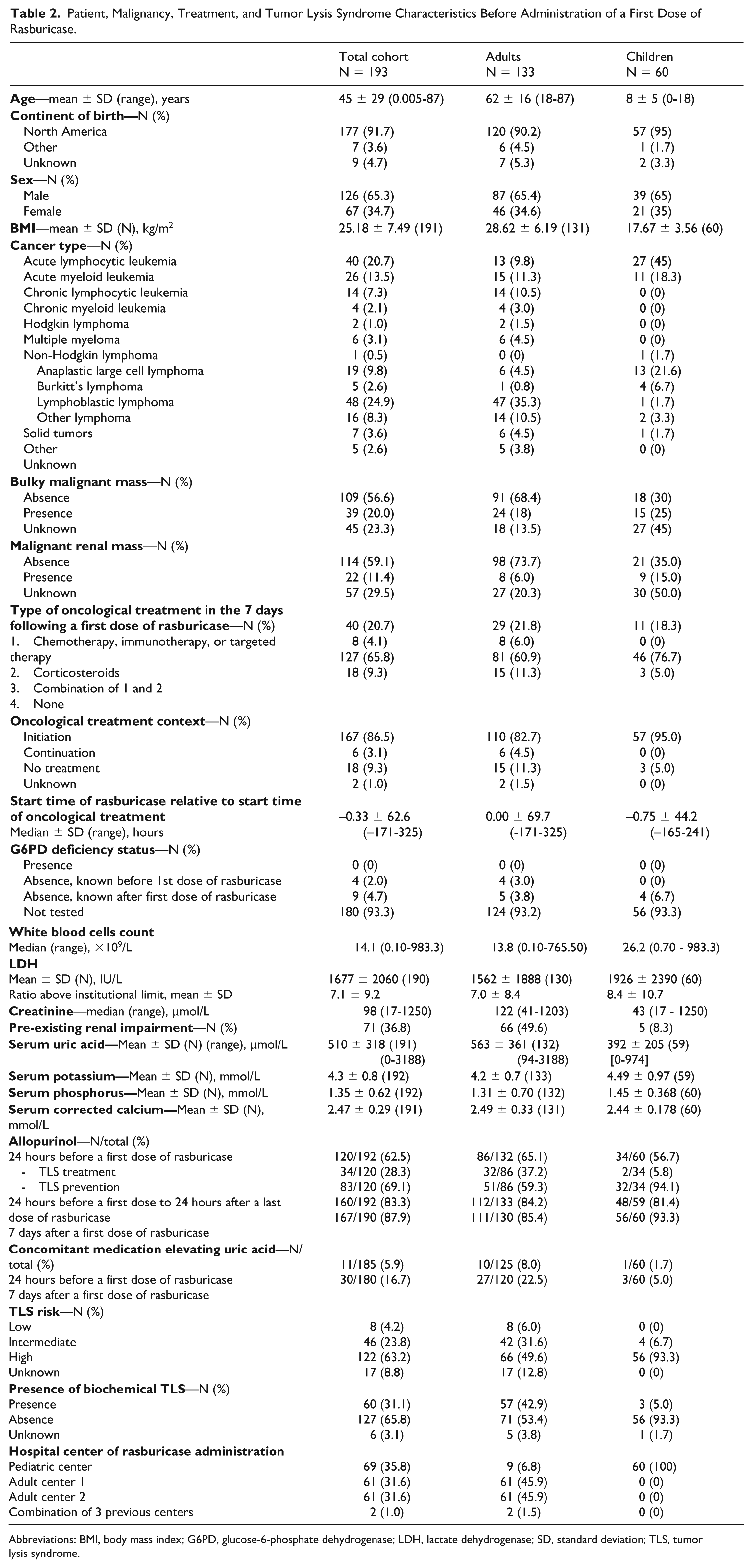
Abbreviations: BMI, body mass index; G6PD, glucose-6-phosphate dehydrogenase; LDH, lactate dehydrogenase; SD, standard deviation; TLS, tumor lysis syndrome.
Adult population
Among the 133 adults included (mean age of 62 years, range 18-87 years), the most common cancers were non-Hodgkin lymphomas (NHL) (40.6%), AML (11.3%), solid tumors (10.5%), and chronic lymphocytic leukemias (10.5%). Nearly, half were at high risk for TLS (48.9%) and presented biochemical TLS pre-rasburicase (42.9%). Common TLS risk factors included elevated uric acid (> 476 µmol/L, 57.9%), pre-existing renal injury (49.6%), and bulky tumors (18%).
Pediatric population
Sixty children (mean age of 8 years, range 2 days-18 years) were included. The most common cancers were ALL (45.0%), Burkitt’s lymphoma (21.6%), and AML (18.3%). Most (93.3%) were at high risk for TLS, 21.7% had elevated uric acid and 5% had biochemical TLS before rasburicase.
Rasburicase Usage Profiles
In most adults, a fixed dose of 6 mg was chosen (86.5%) and a single dose was received (75.2%). In contrast, most children received multiple doses (76.7%) with a weight-based dose of 0.15 to 0.20 mg/kg (78.3%). As illustrated in Figure 1, children weighing > 50 kg (n = 12) generally received a 6 mg fixed first dose instead. Dose changes during the course of treatment occurred in 18 (10.7%) cases. As shown in Table 3, in adults, dosing practices between TLS prevention and treatment were similar. In children, those needing TLS treatment (n = 3) received only 1 dose and multiple doses were more frequent in TLS prevention (80.4%).
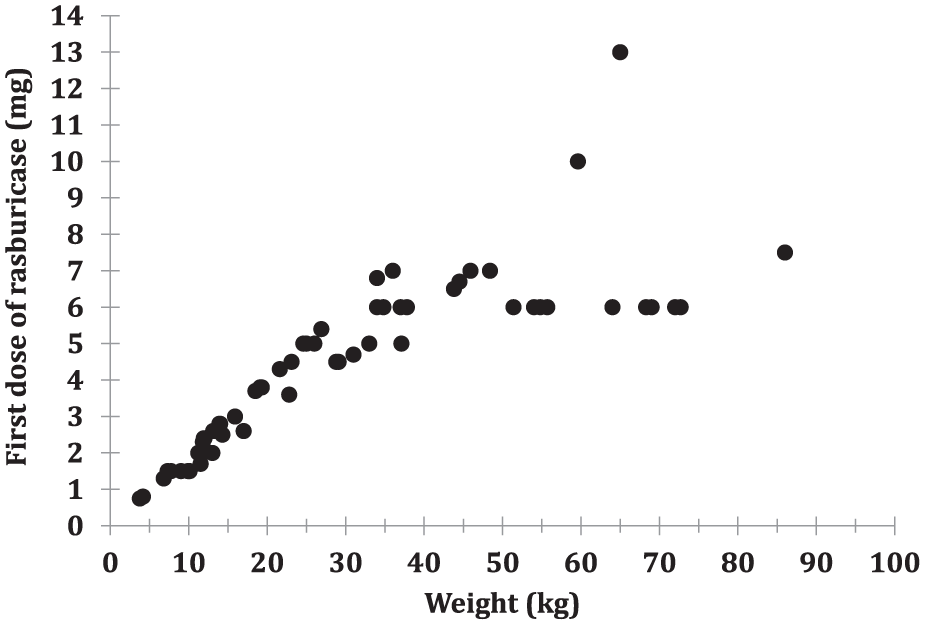
Rasburicase first dose received by weight in pediatric patients (N = 60).
Rasburicase Dosing and Administration Characteristics.
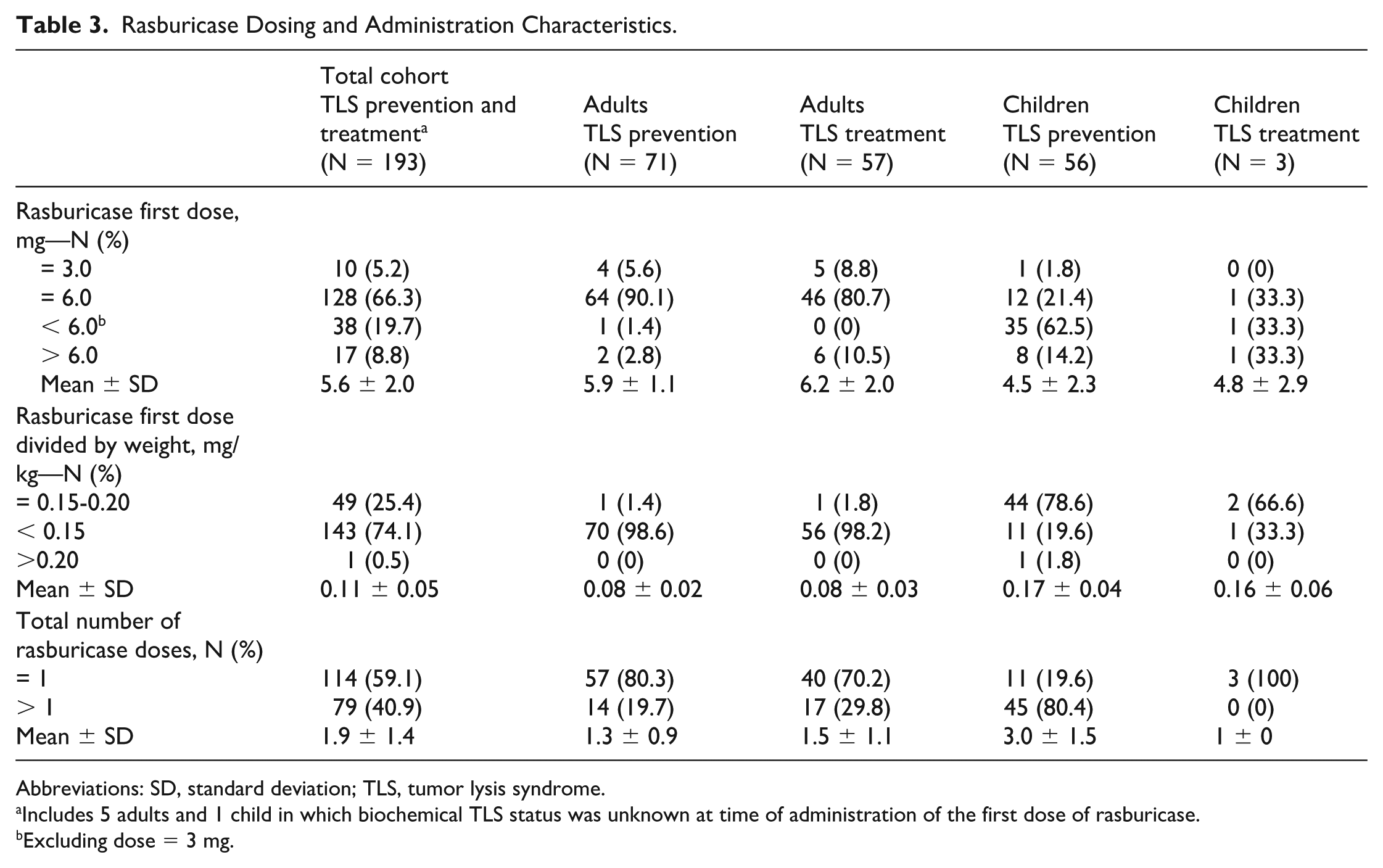
Abbreviations: SD, standard deviation; TLS, tumor lysis syndrome.
Includes 5 adults and 1 child in which biochemical TLS status was unknown at time of administration of the first dose of rasburicase.
Excluding dose = 3 mg.
Allopurinol was commonly used before and after rasburicase in all patients, with 62.5% receiving it within 24 hours preceding a first dose of rasburicase and 83.3% receiving it 24 hours either before or after a first dose of rasburicase, which did not impact the rate of normalization of uric acid and creatinine at 24 hours. Uric acid raising medication use was more common in adults (22.5% adults vs 5.0% children) but had no significant impact on rasburicase uric acid reduction.
Efficacy of Various Rasburicase Administration Regimens
The mean (range) pre-rasburicase level of uric acid in adult and pediatric patients were, respectively, 563 (94-3188) µmol/L and 392 (0-974) µmol/L (Table 2). Rasburicase effectively reduced or maintained serum uric acid to ≤ 476 µmol/L in 184/188 (97.8%) of patients with available data within 24 hours after a first dose (Table 4) and serum creatinine levels also improved during the studied period. The uric acid lowering effect was sustained througt 168 hours for 98,7% of patients (Figures 2 and 3) a period during which 87,9 % of the total cohort was still using allopurinol. Only 4 patients (3 adults and 1 child) did not achieve uric acid levels of ≤ 476 µmol/L within 24 hours. In adults, biochemical TLS rates rapidly declined following a first dose of rasburicase. Children had lower pre-rasburicase rates of TLS (5.0% vs 42.9% in adults), with rates rising slightly before declining after 72 hours (Figure 4). No significant difference in uric acid normalization rate or absolute change was found between the first doses of 0.15 to 0.2 mg/kg up to 6 mg and > 6 mg at 24 hours in all patients. However, relative change in serum uric acid at 24 hours compared with pre-rasburicase values was significantly different (–69.5% vs –85.8%, P = 0.015).
Evolution of Biochemical Tumor Lysis Syndrome, Serum Uric Acid, and Serum Creatinine and Throughout Time.
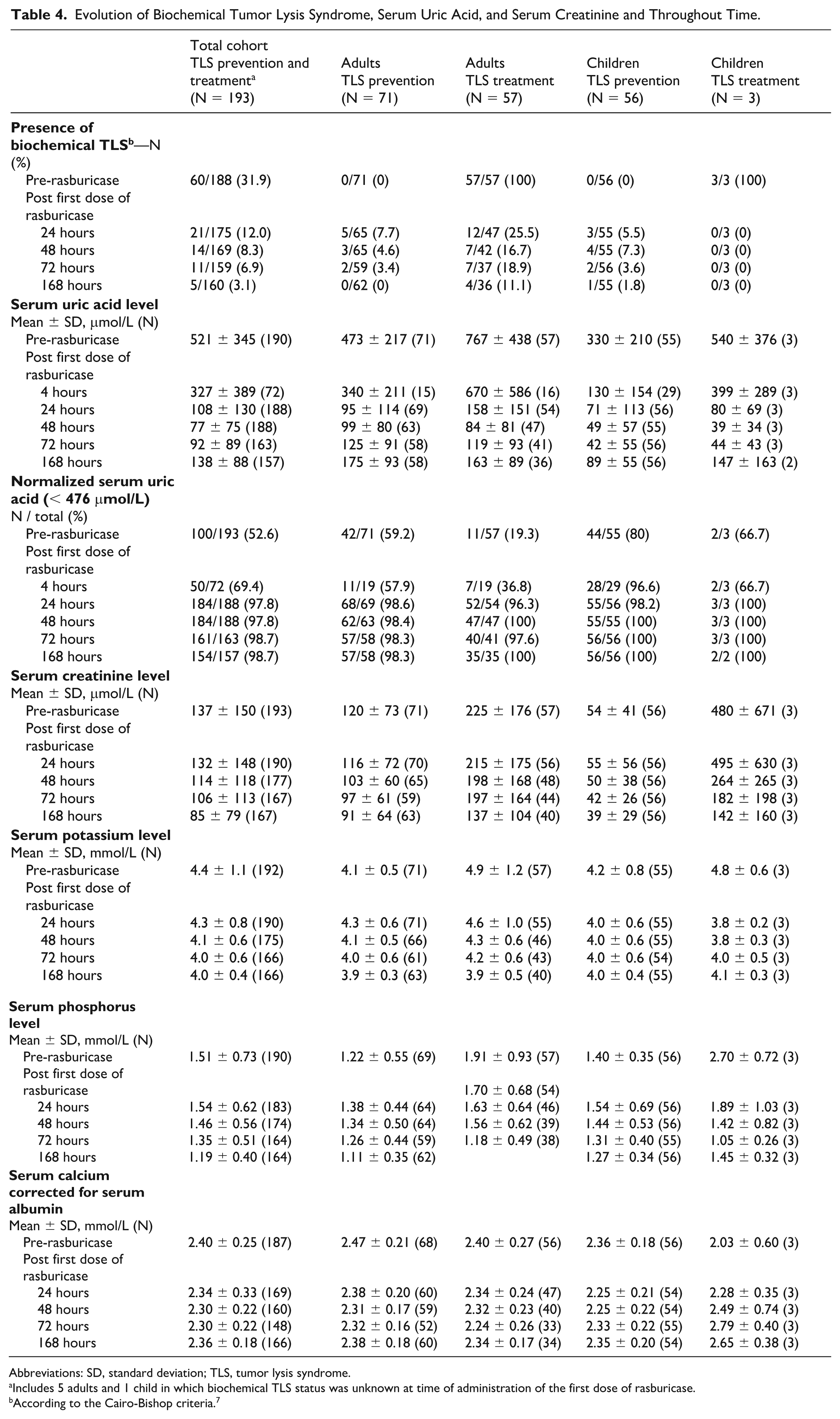
Abbreviations: SD, standard deviation; TLS, tumor lysis syndrome.
Includes 5 adults and 1 child in which biochemical TLS status was unknown at time of administration of the first dose of rasburicase.
According to the Cairo-Bishop criteria. 7
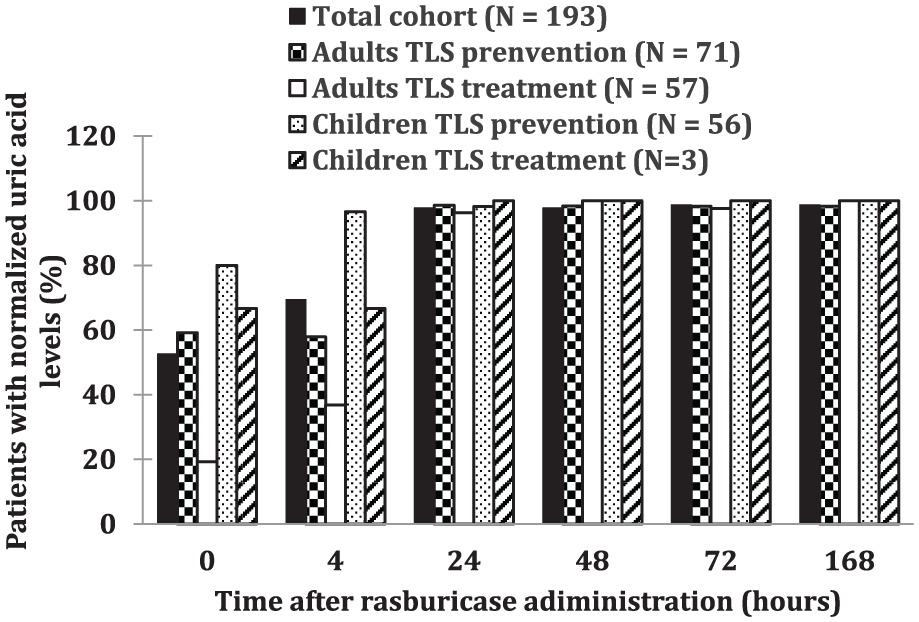
Normalization of uric acid throughout time after administration of a first dose of rasburicase.
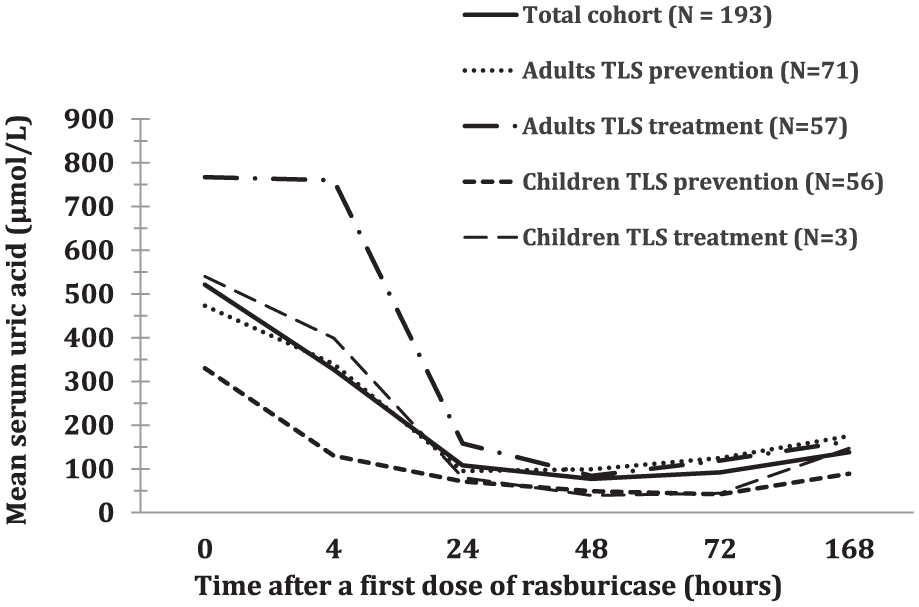
Evolution of serum uric acid before (time: 0 hour) and after rasburicase administration.
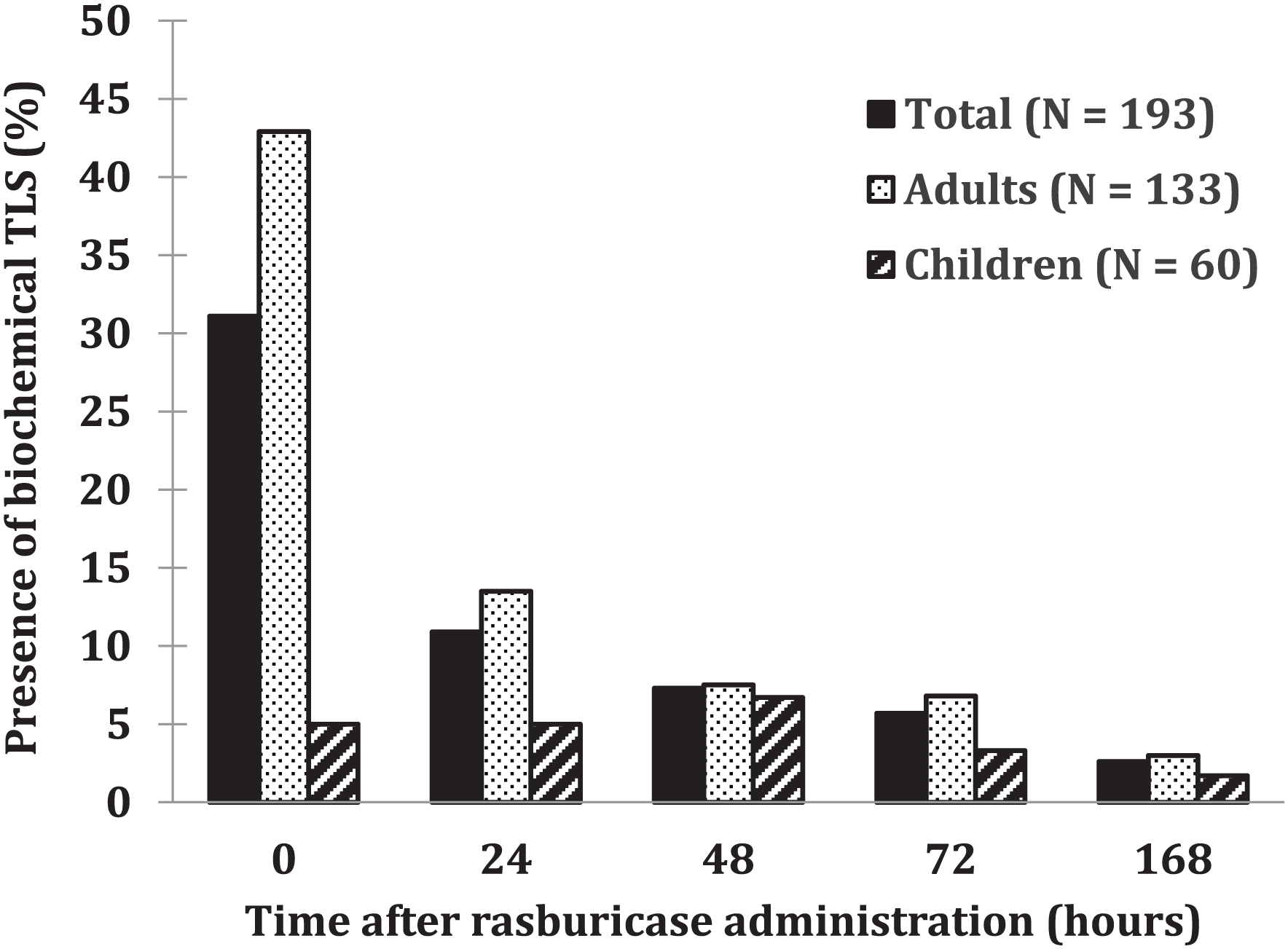
Presence of biochemical tumor lysis syndrome before and throughout time after administration of a first dose of rasburicase.
Initial rasburicase dosing (mg/kg) did not significantly affect absolute or relative changes in uric acid at 24 hours (Figure 5).
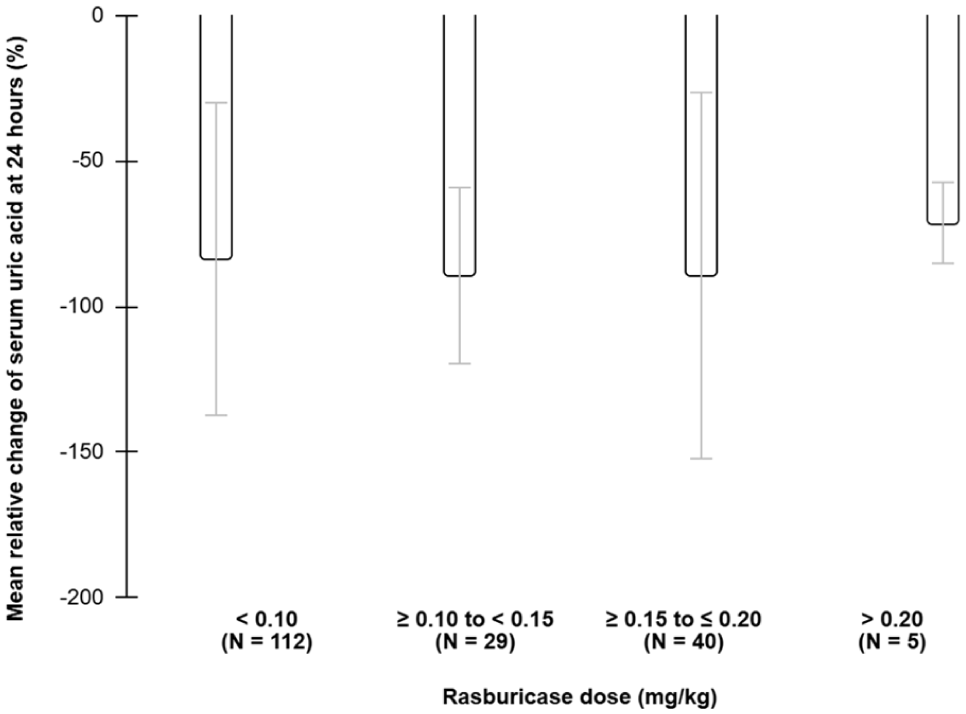
Relative change in serum uric acid by dose of rasburicase in mg/kg at 24 hours after administration of a first dose of rasburicase. Error bars represent standard deviations.
There were 11 possible arrhythmias cases suspected to be related to TLS (6 in adults and 5 in children), with 5 occurring in patients with biochemical TLS pre-rasburicase. All had normalized uric acid within 24 hours, though 2 still met biochemical TLS criteria in the 7 days after rasburicase use due to other ionic abnormalities, and 5 had renal impairment within the same timeframe.
In the 7 days after the first rasburicase dose, 21.8% of patients spent time in an intensive care unit (ICU) (45.0% children and 11.3% adults) for a median of 2.8 days (median: 2.0 days in adults and 3.2 days in children). Hemodialysis was required for 6.7% of patients (6.0% adults and 8.3% children) for a median duration of 3.0 days (median: 4.7 days in adults and 2.3 days in children). In the 30 days post-rasburicase initiation, 39 (29.3%) adults and 1 (1.7%) child died. Of these, 10.3% of adult deaths were TLS-related. The sole child death was not TLS-related.
Safety of Rasburicase Administration Regimens
No serious rasburicase-related adverse events were observed.
Characteristics of Rasburicase Nonresponders
Nonresponders, defined as patients whose uric acid levels did not normalize within 24 hours post-rasburicase, included 4 patients, as shown in Figure 6.
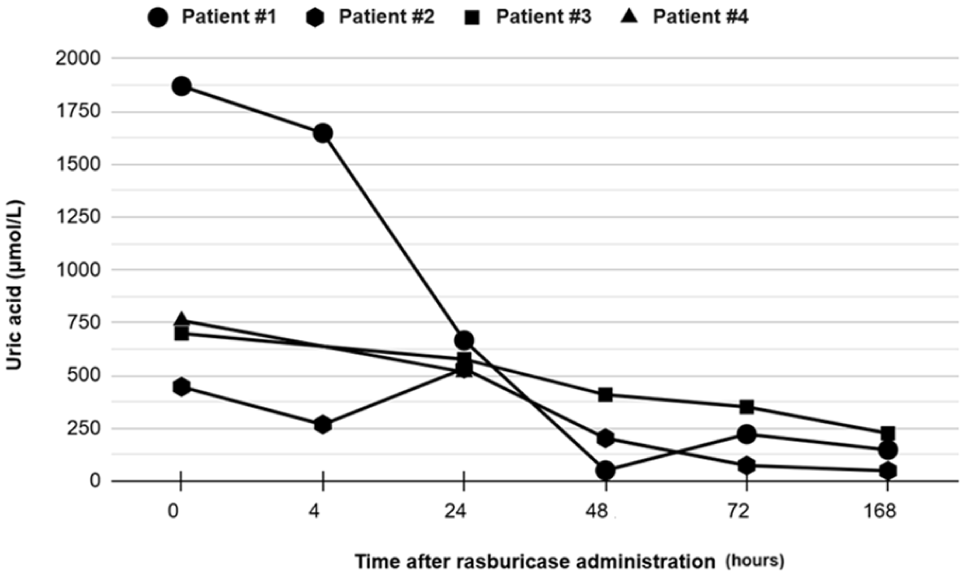
Evolution of uric acid in the 4 nonresponders to rasburicase. Patient 2 is a pediatric patient, while all others are adults.
All cases except patient 2 were men aged > 60 years with stage IV aggressive NHL and pre-existing renal injury. Of these cases, 2 were initially given rasburicase to treat biochemical TLS, 2 had bulky tumors, and 1 had a malignant renal mass. Patient 2 was a toddler with ALL initially receiving rasburicase for TLS prevention.
Patients 1 and 3 received a single 6 mg dose, patient 2 received five 3 mg (~0.2 mg/kg) doses, and patient 4 received 3 mg on day 1, 6 mg on day 2, and died on day 2 from multiple causes including TLS.
Nonresponders had higher mean pre-rasburicase uric acid (1015 vs 506 µmol/L, P = 0.003), but similar BMI (24.6 vs 25.1 kg/m2, P = 0.91) and creatinine (250 vs 132 µmol/L, P = 0.12) compared with responders.
Estimation of Potential Savings With a Fixed Single Dose of Rasburicase
Total real cost (n = 360 doses administered) calculated for all patients (n = 193) was 185 954.15 $CAD while the total theoretical cost for the single dose scenario was $99 411.23 CAD (n = 193 doses). Thus, it is estimated that the latter scenario would have saved 448.41 $CAD per patient.
Discussion
Rasburicase Usage
This study primarily aimed to describe rasburicase use for treating and preventing TLS in adults and children at our centers. In addition to previous literature, it inventories various dosing approaches in all these subpopulations, from single fixed doses to multiple weight-based doses, across a large sample of patients.
In contrast to inconsistent practices found in a recent study on adults within a Canadian health authority, our center predominantly used a 6 mg single fixed dose for both prevention and treatment, consistent with emerging literature indicating this dosage regimen effectively lowers and maintains uric acid levels in malignancy-associated hyperuricemia.17,23 Fixed doses of 3 mg, proposed by certain guidelines for the prevention of TLS, were rarely used at our center, administered in only 5.2% of patients, despite 65.8% of the cohort receiving rasburicase for TLS prevention. 5
In our pediatric population, we observed that rather than the fixed doses suggested for TLS prevention and treatment in current literature of 1.5 mg (regardless of weight) or fixed 6 mg doses (≥30 kg), a majority of patients in both prevention and treatment of TLS received multiple weight-based (0.15-0.2 mg/kg) doses of rasburicase, with fixed dosing of 6 mg becoming more common at weights > 50 kg.23-25 Thus, further studies are warranted to evaluate the safety and efficacy of fixed doses in the specific context of the pediatric population at our institution.
Rasburicase Efficacy
This study’s high 24-hour post-rasburicase uric acid normalization rate (> 90%) is similar to prior findings, including a meta-analysis of 213 studies in adults, in both TLS prevention and treatment, reporting a 93.4% normalization rate (timing unspecified) after rasburicase initiation, regardless of regimen. 15 Similar high rates observed in children reflect previous results, such as a meta-analysis of 92 children, in both TLS prevention and treatment, showing 93.6% normalization within 24 hours of a single 0.15 mg/kg dose. 17
Similar to a prior study in patients > 30 kg in TLS prevention or treatment, which found no difference in 24-hour uric acid normalization (< 476 µmol/L) rates between 0.1 and 0.2 mg/kg and mg doses, respectively, our study showed similar results between doses > 6 and ≤ 6 mg. 24 This supports the emerging practice of capping rasburicase doses at 6 mg in patients > 30 kg.
Post-rasburicase peak biochemical TLS rates observed in adults (7.7%) and children (7.3%) match the 7.4% incidence reported in the meta-analysis. The relationship between rasburicase and reported arrhythmias remains unclear, as other factors like ionic imbalances likely contributed, considering all cases achieved uric acid normalization within 24 hours. Half of the patients presenting arrhythmias met biochemical TLS criteria, emphasizing the importance of timely TLS identification and treatment. 15 Hemodialysis rates (6.7%), ICU admission (20%), and death within 30 days (10%) observed corresponded to results found within existing literature, though influenced by precautionary ICU transfers and critical illness unrelated to TLS.11,15,26
British guidelines advise against combining allopurinol and rasburicase due to possible reduced rasburicase efficacy by blocking hypoxanthine and xanthine metabolism into uric acid and a possible increased risk of acute renal injury through accumulation of these same precursors. 5 However, in this study, allopurinol use within 24 hours before rasburicase did not affect uric acid normalization rates nor serum creatinine, supporting that rasburicase maintains its efficacy and safety when escalating from allopurinol.
Rasburicase Safety
The lack of severe adverse events from rasburicase was similar to results found in the meta-analysis previously described, which reported a low adverse event rate of 2.6%. 15 Low G6PD deficiency testing rates at our institution reflect the priority of avoiding delays in TLS management given that rapid turnover testing is not available at our institution. Despite the ethnicity of patients being unknown in this study, most were North American-born, a region where G6PD deficiency is less prevalent, which helps to mitigate the risk of hemolytic anemia combined with close monitoring strategies. 12 The absence of hemolytic anemia observed in our study supported a low prevalence of a clinically significant G6PD deficiency in the cohorts studied. 11
Rasburicase Nonresponse
A retrospective study by Nauffal et al (2019, N = 92 adults) assessed the efficacy of a single 6 mg dose of rasburicase and identified risk factors for treatment failure in TLS. Their nonresponder rate (5%, 5/95) was similar to ours (2%, 4/188), though broader nonresponse criteria were used, including uric acid not normalizing within 24 to 36 hours or requiring a second dose within 24 hours. 11 In our cohort, 19.2% of adults received multiple doses, likely due to institutional prescribing tools suggesting daily dosing rather than rasburicase nonresponse, as indicated by high 24-hour uric acid normalization rates. Thus, our definition of nonresponse did not consider additional dosing. Nauffal et al 11 also reported higher pre-rasburicase uric acid in nonresponders, with no significant BMI differences. As our study lacked the statistical power to detect more significant differences between responders and nonresponders, further research is needed to better identify high-risk patients who may benefit from alternative dosing.
Study Strengths and Limitations
This study’s strengths include broad inclusion criteria and patients from various oncology services, enhancing generalizability to routine practice in Quebec university centers. Consecutive patient selection minimizes selection bias, and the large sample size provides a greater statistical power than most previous retrospective studies, covering 4 key subgroups: children or adults receiving rasburicase for TLS treatment or prevention. Variables, such as rasburicase response, laboratory TLS, and TLS risk align with current practices and literature, allowing for meaningful comparisons with prior research.
Limitations include missing data on key TLS management parameters, such as intravenous hydration and ionic normalization medications. The timing of dialysis relative to rasburicase was not collected, possibly introducing a bias in favor of rasburicase efficacy in the small subset of patients who received dialysis (6.7 %), as dialysis initiated within 24 hours of rasburicase administration could independently contribute to the normalization of uric acid levels. The sample size was insufficient for determining differences in characteristics between responders and nonresponders. Ethnicity of patients was not collected, making the risk for deficiency in G6PD activity not assessable. The impact of uric acid sample handling, which consists of ideally preserving samples on ice in heparin tubes and analyzing samples within 4 hours, on reliability of uric acid results could not be verified despite institutional protocols in place. 9 Data collection by multiple individuals introduced potential information bias, mitigated by systematic audits of collected data for one tenth of the total cohort. The retrospective design led to missing or incomplete data, though sample sizes were provided where applicable.
Research and Clinical Implications
This study supports the safe and effective use of fixed single 6 mg doses of rasburicase for TLS prevention and treatment in adults and children over 50 kg, consistent with emerging literature, as well as the maintenance of rasburicase efficacy and safety when escalating from allopurinol.
While elevated pre-rasburicase uric acid levels were more prevalent for rasburicase nonresponders in this study, further research is needed to identify populations that may benefit from alternative dosing regimens to avoid treatment failure. Additional studies are also needed to assess the clinical impact of concurrent rasburicase and allopurinol use, along with the efficacy and safety of fixed single dosing in children under 50 kg.
Locally, this study highlights the safe, effective, and cost-efficient approach of single fixed doses of rasburicase, supporting the revision of local guidelines and prescription forms that feature weight-based dosing. The findings provide a foundation for updating clinicians on optimal rasburicase use. With a large sample size and generalizable data, this study aims to improve clinical practices beyond Quebec by adding to the current evidence in favor of a more affordable rasburicase single fixed dose regimen in adults and pediatric patients.
Conclusion and Relevance
This study reinforces existing literature showing that multiple or uncapped dosing regimens of rasburicase in adults do not appear to provide additional benefit in terms of efficacy. A single dose of 0.15 to 0.20 mg/kg, capped at 6 mg, may therefore represent an effective, safe, and more cost-effective option for both the treatment and prevention of TLS. In pediatrics, this study, which includes one of the largest reported pediatric populations on this topic, suggests that capping rasburicase doses at 6 mg also appears to be an adequate strategy in terms of efficacy. However, the study design does not allow firm conclusions regarding comparative efficacy of a single versus multiple doses, as most children received more than one dose. Further studies are warranted to clarify this aspect, as well as to identify potential risk factors for rasburicase nonresponse and to evaluate the impact of concomitant allopurinol use on rasburicase efficacy.
Supplemental Material
sj-docx-1-aop-10.1177_10600280251391871 – Supplemental material for Rasburicase Real-World Usage for the Prevention and Treatment of Tumor Lysis Syndrome in Adults and Children
Supplemental material, sj-docx-1-aop-10.1177_10600280251391871 for Rasburicase Real-World Usage for the Prevention and Treatment of Tumor Lysis Syndrome in Adults and Children by Sandra Labonté, Émilie Bergeron, Isabelle Laverdière, Julien Valois-Demers and Louis-Philippe Grenier in Annals of Pharmacotherapy
Footnotes
Acknowledgements
The authors would like to thank Michel Dorval for providing critical feedback on the study protocol; David Simonyan, Mahukpe Narcisse Ulrich Singbo, and Pierre-Hugues Carmichael for their help with statistical analysis; and the Faculty of Pharmacy at Université Laval for financial and material support and SCIENTA at CHU de Québec-Université Laval for providing a targeted list of patients for screening.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
