Abstract
Background:
Clostridioides difficile infection (CDI) causes a significant national health care burden. Literature has demonstrated lower rates of CDI recurrence with fidaxomicin compared with oral vancomycin. However, patients are sometimes switched to oral vancomycin before completing a fidaxomicin course.
Objective:
The objective of this study is to evaluate rates of CDI recurrence in full courses of fidaxomicin versus partial courses of fidaxomicin followed by a switch to oral vancomycin.
Methods:
In this single-center, retrospective, cohort study of adults with CDI, patients were screened for inclusion if they received either a full 10-day course of fidaxomicin or partial course of fidaxomicin followed by a switch to oral vancomycin. The primary outcome was the rate of CDI recurrence within 30 days after completion of initial therapy determined by a positive CDI test and initiation of treatment.
Results:
Ninety-nine patients received a full course of fidaxomicin, and 95 patients received a partial course of fidaxomicin followed by oral vancomycin. Mean age was lower in the full course group compared with the partial course (65.3 years vs 71.5 years, P < 0.002). Clostridioides difficile infection recurrence occurred in 5.1% of the full course group and 7.4% of the partial therapy group (P = 0.503) at 30 days and 13.1% versus 14.7% (P = 0.747) at 90 days. Clostridioides difficile infection–related readmissions at 30 days were similar in the full course and partial course groups (7.1% vs 4.2%, P = 0.389).
Conclusion and Relevance:
Partial courses of fidaxomicin followed by oral vancomycin had similar 30-day CDI recurrence compared with full course fidaxomicin.
Background
Clostridioides difficile infection (CDI) is the most common cause of nosocomial diarrhea in developed countries. Data published by the Centers for Disease Control and Prevention estimate that the national burden of CDI is over 450 000 cases and 8000 hospitalizations per year. 1 Despite the inpatient incidence of CDI in the United States decreasing since 2011, outpatient incidence and mortality have not improved. 1 Of patients who experience CDI, rates of recurrence are reported to range from 25% to 65% in people who have experienced multiple previous episodes. 2 Beyond the quality of life burden for these patients, CDI recurrences also increase health care costs. 3 Risk factors for CDI recurrence include age ≥65 years, proton pump inhibitor use, severe disease presentation, continued use of non-CDI antibiotics, and colonization with hypervirulent strains. 4
Fidaxomicin is a macrocyclic antibiotic recommended as the preferred agent for initial and recurrent CDI episodes, by both the Infectious Diseases Society of America (IDSA) and European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines.5,6 Compared with the glycopeptide antibiotic vancomycin, fidaxomicin has a relatively narrow spectrum of activity and is notable for inhibition of sporulation, which contributes to rapid recovery of disrupted microbiota in patients with CDI and theoretically should result in a decreased disruption of other enteric commensal bacteria. 7 Previous literature has demonstrated lower rates of CDI recurrence with fidaxomicin compared with oral vancomycin.8 -11 Despite acquisition cost being higher, impact from reduction in recurrences and admissions demonstrate overall cost-effectiveness.3,12,13
In January 2018, Carilion Roanoke Memorial Hospital (CRMH) began utilizing fidaxomicin as the first line CDI agent for patients with a high risk of recurrence. Published literature from CRMH was significant for improved clinical and economical outcomes with fidaxomicin. 14 However, despite increased availability and financial assistance, there continue to be barriers that exist and may impede successful completion of a full course of fidaxomicin. A transition to oral vancomycin during fidaxomicin therapy could exacerbate the probable dysbiosis associated with oral vancomycin and reduce the positive impact fidaxomicin has on recurrence reduction. The utility of fidaxomicin partial courses is not established in the literature, and there is a question of whether reduction in recurrences is preserved. The objective of this study was to evaluate rates of recurrence in full courses of fidaxomicin versus partial courses of fidaxomicin where partial therapy was completed with oral vancomycin.
Methods
This was a single-center, retrospective, cohort study of adults with CDI conducted in Southwest Virginia at CRMH. The study protocol was approved by the Institutional Review Board prior to commencement. Patients were screened for inclusion if they were diagnosed with CDI between May 1, 2018, and May 1, 2023, and received either a full 10-day course of fidaxomicin or partial course of fidaxomicin followed by a switch to oral vancomycin. Patient exclusion criteria were the presence of fulminant CDI, greater than 2 CDI episodes within 1 year, pregnancy, lactation, incarceration and transitioning to comfort care or hospice during CDI treatment.
The confirmed CDI diagnosis was based on documented episodes of diarrhea within the electronic health record (EHR) and 2 diagnostic assays. Before 2021, the institution utilized a glutamate dehydrogenase antigen (GDH) and toxin A/B antigen test, followed by CDI toxin polymerase chain reaction (PCR) if the tests were discordant. A positive CDI case could be due to the following results: GDH and toxin antigen positive, GDH and PCR positive, or toxin antigen positive and PCR positive. Starting in 2021, the institution transitioned to utilizing a positive toxin PCR as the first step and if positive it would be reflexed to the cytotoxin assay (CTA). A positive CDI case determined by positive PCR and positive CTA. The origin of CDI was determined as community onset, community-onset healthcare facility-associated or healthcare facility-onset by the infection control team utilizing definitions from the National Healthcare Safety Network. 15
The process for obtaining fidaxomicin when patients are transitioning from inpatient to outpatient care begins on day 1 of therapy. First, a CDI order set is utilized by an ordering provider which includes prompts for contact isolation, nursing care, treatment and more. When a provider chooses treatment with fidaxomicin, a case management consult is automatically linked to the order. After financial investigation has taken place by case management, the team may pursue a Carilion Medication Assistance Program (CMAP) application if fidaxomicin is too expensive and the patient meets criteria. Manufacturer assistance is the primary method of assistance utilized by CMAP. Reasons for partial course of fidaxomicin were collected by sorting through the EHR note documentation from either case managers or social workers. Fidaxomicin cost on discharge was documented in the EHR by the case management team as either covered with no cost, covered with a copay, not covered or patient received medication assistance.
Patient data were obtained from a list of all positive CDI cases maintained by Infection Control and Prevention. Study participants who met inclusion and exclusion criteria were divided into 2 groups, full course fidaxomicin or partial course fidaxomicin. The full course group consisted of fidaxomicin 200 mg twice daily for 10 days. Patients included in the partial group received at least 1 day and no more than 9 days of fidaxomicin 200 mg twice daily followed by a switch to oral vancomycin to complete 10 days total.
The primary outcome was the rate of CDI recurrence within 30 days after completion of initial therapy determined by a positive CDI test and initiation of treatment. Secondary outcomes included CDI recurrence at 90-day, clinical cure, and sustained response. Clinical cure was defined as resolution of symptoms at end of therapy for index episode and no requirement for continued treatment. 6 Sustained response was defined as clinical cure with no recurrences at 90 days following completion of therapy. 6 In addition, CDI-related readmissions and all-cause mortality were evaluated at 30 and 90 days.
Known risk factors for CDI recurrence were collected as baseline characteristics and include age ≥65 years, concomitant antibiotic use during CDI treatment, proton pump inhibitor (PPI) use, immunosuppression, history of cancer, baseline renal insufficiency and episode of CDI within previous 12 months. Proton pump inhibitor use was defined as concomitant inpatient use or outpatient prescription filled within previous 12 months. 16 Immunosuppression was defined as an active hematologic malignancy or solid tumor, HIV with CD4 <200 cells/mm3, absolute neutrophil count <500 cells/mm3, concomitant use of immunosuppressive agents. Concomitant non-CDI antibiotics were defined as at the time of CDI diagnosis or any point on therapy. Fecal microbiota transplant (FMT) and bezlotoxumab therapies were also collected as they are known to reduce the rate of CDI recurrence. Receipt of bezlotozumab or FMT were evaluated in the 30 days from index episode. Severe CDI was defined as a leukocytosis with white blood cells > 15 000 cells/mL or SCr≥1.5x baseline. 17
A pre-determined sample of 197 total participants (99 in each group) was deemed required to meet 80% power to detect a 20% relative risk reduction in incidence of 30-day recurrence using an alpha of 0.05. Categorical data were analyzed using chi-square or Fisher’s exact test as appropriate while continuous data were analyzed using Student’s t-test. P-values of <0.05 were considered statistically significant and data was analyzed using SAS Enterprise Guide 8.3 (64-bit). Post-hoc a subgroup analysis of the partial courses was run for those receiving ≤ 3 days compared with > 3 days and full course of fidaxomicin therapy. Data was stratified by age ≥ 65 years or less and impact of recurrence at 30 days in the partial group. In addition, post-hoc drivers of partial course utilization were evaluated including Medicare status, severity of CDI and concomitant antibiotics.
Results
The entire study period (May 2018-April 2023) was screened for inclusion with the final cohort resulting in 194 patients who were treated with fidaxomicin for CDI. The reasons for exclusion included oral vancomycin initially received (n = 100), presence of fulminant CDI (n = 50), transitioned to comfort care during treatment (n = 47), greater than 2 CDI episodes in 1 year prior (n = 10), age less than 18 years (n = 27), and incarceration (n = 2). Ninety-nine patients received a full course of fidaxomicin, and 95 patients received a partial course of fidaxomicin followed by oral vancomycin. Mean age was lower in the full course group compared with the partial course (65.3 years vs 71.5 years, P < 0.002). The number of severe CDI was numerically greater in the partial therapy group (34.3% vs 45.3%, P = 0.120) and nearly a quarter of patients from both groups had experienced a prior episode of CDI (22.2% vs 17.9%, P = 0.452). Most of the positive CDI cases were determined through the initial treatment algorithm prior to institutional change at 72.1% in the full course versus 81.1% in the partial course (Table 1). The median length of fidaxomicin therapy in the partial course arm was 3 days (IQR, 2-5 days).
Baseline Characteristics.
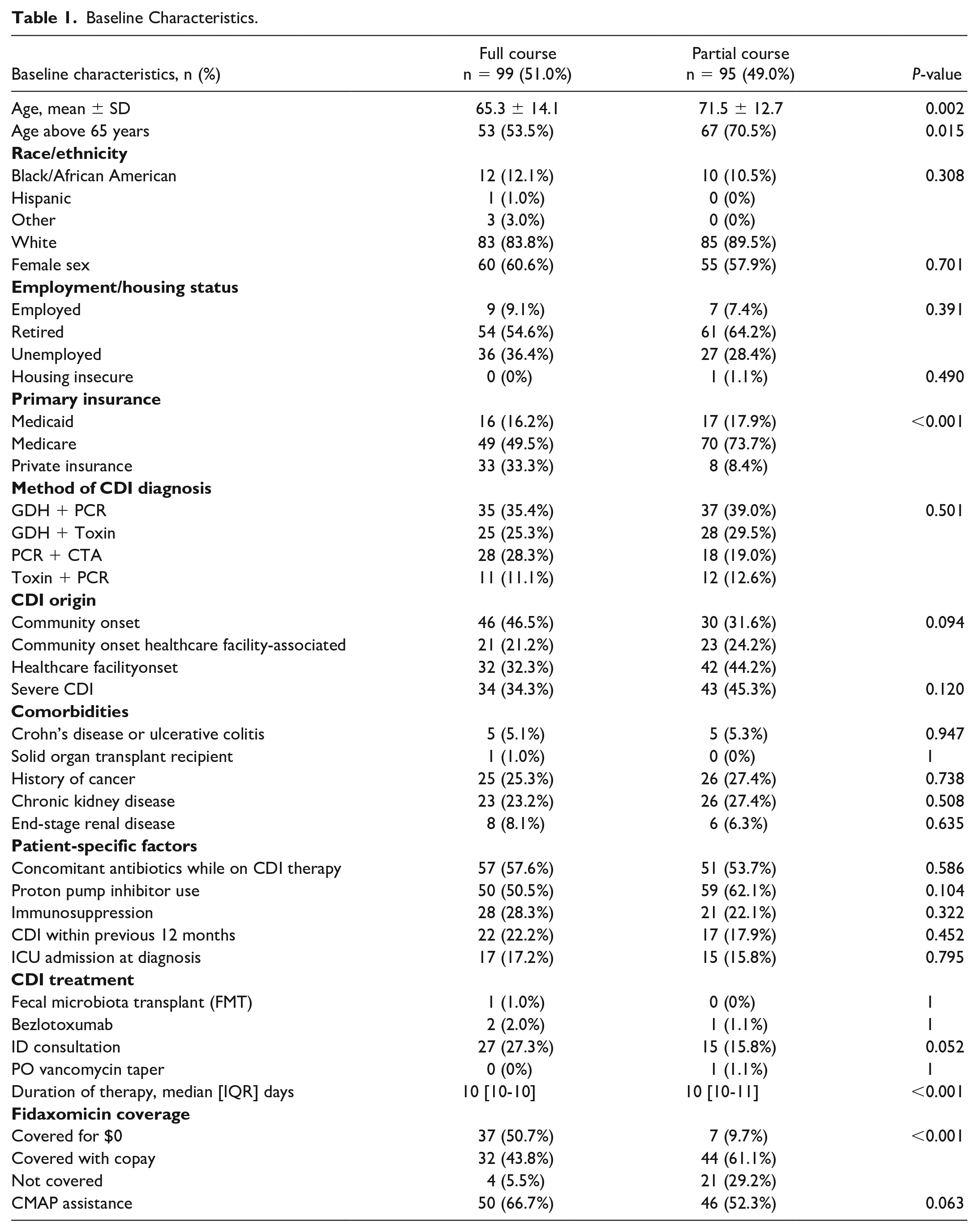
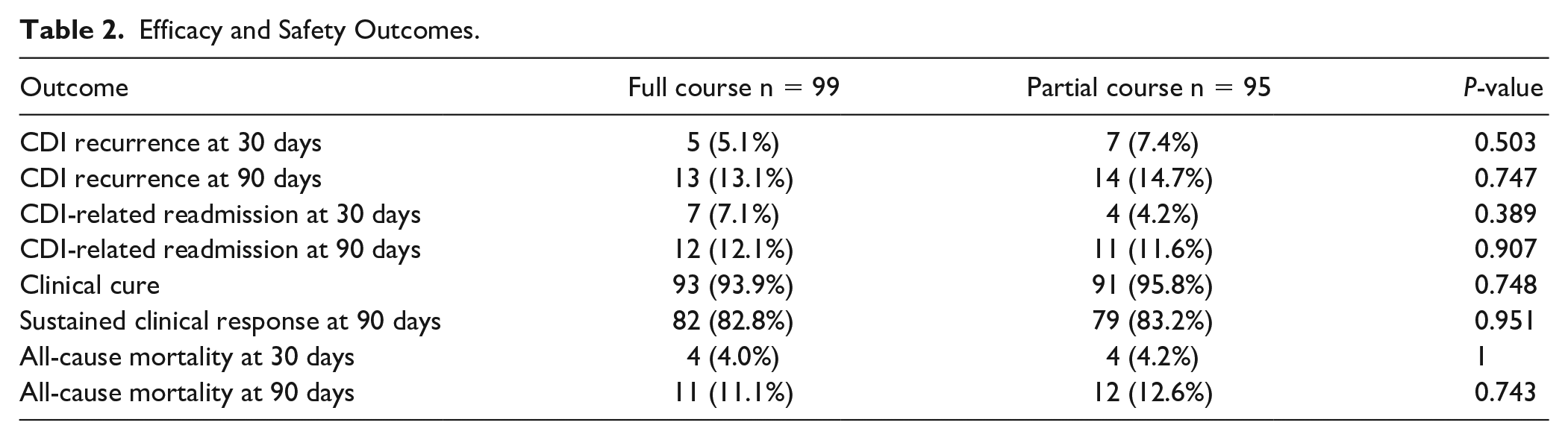
The primary outcome of CDI recurrence within 30 days occurred in 5.1% of the full course group and 7.4% of the partial therapy group, P = 0.503 (Table 2). CDI recurrence at 90 days was similar between the full course and partial course group (13.1% vs 14.7%, P = 0.747). Similarly, CDI-related readmissions at 30 days were similar in the full course and partial course groups (7.1% vs 4.2%, P = 0.389). In addition, sustained clinical response at 90 days was similar between both full course and partial course groups (82.8% vs 83.2%, P = 0.951).
Efficacy and Safety Outcomes.
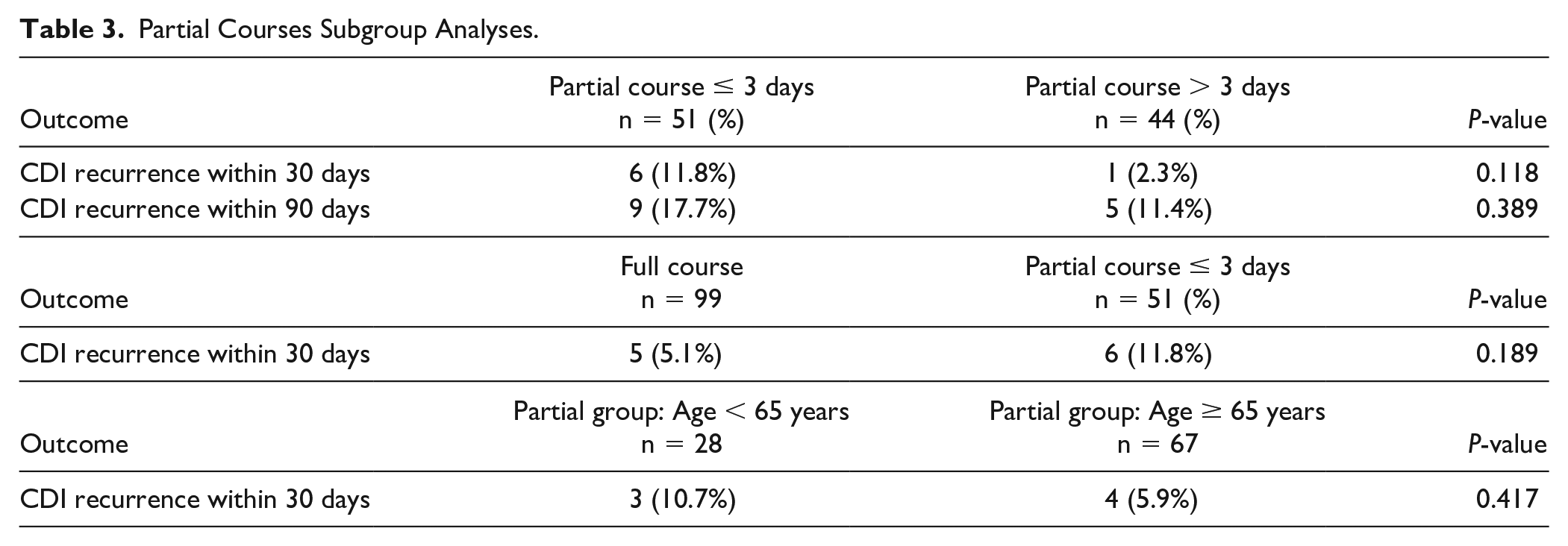
In the partial therapy arm, 51 patients received ≤3 days of fidaxomicin while the remaining 44 received > 3 days of fidaxomicin before being switched to oral vancomycin (53.6% vs 46.3%) (Figure 1). Fifty-five percent of the partial therapy arm switched to oral vancomycin due to patients transitioning to a skilled nursing facility while 47% of patients were switched due to cost prohibition to the individual. Alternative reasons for partial therapy included provider decision, difficulty swallowing pills, and switching to oral vancomycin taper. In the subgroup of patients that received partial courses of therapy, fidaxomicin less than or equal to 3 days in duration had a CDI recurrence at 30 days of 11.8% compared with 2.3% in those with greater than 3 days, P = 0.118 (Table 3). Post-hoc a subgroup analysis of the partial course of fidaxomicin ≤3 days compared with full course found 30 day CDI recurrence rate, 6/51 (11.8%) versus 5/99 (5.1%), P = 0.189, respectively. Within the partial group, patients ≥ 65 years did not display a difference in 30-day recurrence compared with those <65 years (6.0% vs 10.7%, P = 0.417).
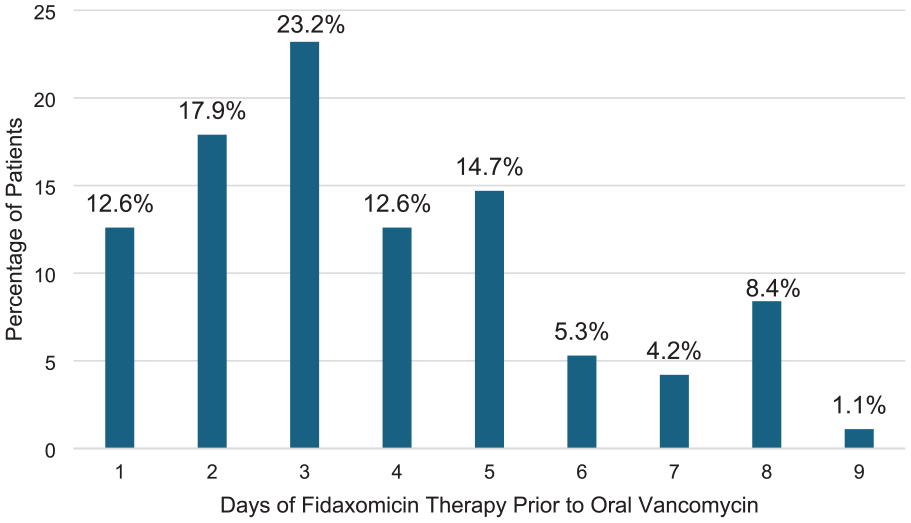
Days of fidaxomicin prior to therapy change.
Partial Courses Subgroup Analyses.
Although most patients had Medicare as their primary insurance, there was a greater percentage of Medicare patients in the partial group (49.5% vs 73.7%, P < 0.001) and a greater percentage of private insurance in the full course group (33.3% vs 8.4%, P < 0.001). Medicare patients had a higher odds of being treated with a partial course than patients with other insurances (OR: 2.85, CI 1.563-5.208). There were more patients in the partial therapy arm who either had co-pays associated with fidaxomicin (43.8% vs 61.1%, P < 0.001) or the treatment was not covered at all (5.5% vs 29.2%, P < 0.001). Respectively, there were more patients in the full course arm who had fidaxomicin covered for $0 (50.7% vs 9.7% patients, P < 0.001).
Discussion
Both groups, fidaxomicin followed by oral vancomycin and full courses of fidaxomicin, had low rates of CDI recurrence at 30 days and 90 days (5.1% vs 7.4%, P = 0.5029, 13.1% vs 14.7%, P = 0.747). All-cause mortality at 30 days and 90 days were similar between treatment groups (4.0% vs 4.2%, P = 1.00) and (11.1% vs 12.6%, P = 0.743). These findings suggest that patients who are unable to afford a full course of fidaxomicin or transitioning to a facility that does not accept fidaxomicin, may not necessarily experience worse clinical outcomes in terms of recurrence. This study supports the idea that being unable to complete a full course of fidaxomicin should not preclude it from being started but additional studies are needed to confirm this hypothesis and practice.
To the authors’ knowledge, no previous studies of this size have analyzed fidaxomicin treatment as full course versus partial course to assess recurrence rates of CDI. Few studies evaluate alternative treatment strategies or combinatory regimens and their impact on CDI recurrence. A small study by Duricek et al 18 evaluated the impact of shorter courses for CDI, however it does not address the impact of the use of partial regimens despite observing a low rate of recurrence in the study group. Of patients who have had an episode of CDI, recurrence can occur in up to 15% to 25% with recurrence as high as 65% in patients with multiple previous episodes. 2 Previous studies have shown that fidaxomicin offers benefits over traditional oral vancomycin due to improvement in recurrence rates.8 -11 Lower recurrence rates are likely due to fidaxomicin’s unique microflora sparing features. Fidaxomicin has minimal systemic absorption, high fecal concentrations, bactericidal activity, and a prolonged post antibiotic effect against C difficile. 19 Louie et al 9 compared oral vancomycin to fidaxomicin, and results suggested a 45% relative reduction in CDI recurrences. Particularly, presence of organisms such as Bacteroides species and Prevotella 19 species after completion of fidaxomicin treatment may suggest lower rates of CDI recurrence. Finally, a meta-analysis performed by Al Momani et al 10 included 4 observational studies with over 2300 patients in which fidaxomicin was found to be associated with a significantly lower recurrence with a pooled OR of 0.47 (95% CI, 0.37-0.60).
Partial fidaxomicin courses were defined as at least 1 day before switching therapy, since the impact of oral vancomycin dysbiosis is not completely known but potentially impacted by 1 day of therapy. Due to there being a wide range of acceptable partial courses, we conducted a subgroup analysis of partial courses 3 days or less versus partial courses greater than 3 days. The results demonstrate CDI recurrence within 30 days for patients treated with a partial course of fidaxomicin ≤ 3 days 11.8% versus partial courses > 3 days, 2.3% (P = 0.118). Similarly, there was not a statistical difference when the ≤ 3 days was compared with a full course. These subgroup analyses may suggest that patients who completed more days of fidaxomicin partial therapy are less likely to experience recurrence at 30 days, but further studies would be necessary as the study was not powered to detect a difference. These results may suggest the importance of the duration of fidaxomicin treatment in correlating with preservation of gut microbiome and decreased recurrence. The results of the present study do not suggest the purposeful utilization of partial fidaxomicin courses over complete fidaxomicin courses.
Patients with Medicare as their primary insurance had 2.85 times higher odds of receiving a partial course compared with other insurances [OR: 2.85, CI 1.563-5.208]. Similarly, we found a greater percentage of patients in the partial course group had fidaxomicin co-pays or no coverage for fidaxomicin (43.8% vs 61.1%, P < 0.001; 5.5% vs 29.2%, P < 0.001). Conversely, the full course arm had more patients that were covered for fidaxomicin with a zero-dollar copay (50.7% vs 9.7%, P < 0.001). Despite the efforts of the CMAP program, challenges with acquiring coverage for high-cost drugs in a timely fashion remain. These results highlight potential disparities of care that can arise due to health insurance and financial barriers. Patients who received a partial course had a higher mean age than the patients in the full course arm (65.3 ± 14.1 years vs 71.5 ± 12.7 years, P = 0.002). This information may suggest that due to more advanced age, patients have a greater chance of requiring skilled nursing facility care, which is a potential barrier to completion of a course of fidaxomicin.
There were several study limitations given the small sample size and retrospective study design. Low CDI recurrence rates and high proportion of patients with sustained response were seen in both treatment groups, requiring a larger than anticipated sample size; therefore, power was not met. Patients were included based on the positive testing algorithm and presence of documented diarrhea; however, the specific quantity of stools was not collected. Therefore, overdiagnosis may have occurred for those patients with transient diarrhea. Although, this misclassification bias would theoretically be similar in both treatment arms and part of the study period included CTA testing which has high specificity for true infection. The presence of concomitant antibiotics was collected but there was no differentiation between broad spectrum, high-risk agent or duration of treatment. Not every variable that has the potential to impact recurrence was able to be collected, but some known risk factors were similar between the two groups and include: PPI use, previous episode of CDI, history of cancer, immunosuppression and concomitant antibiotics. Some previously recognized factors associated with higher rates of recurrence were higher in the partial arm include age ≥ 65 year and severe disease diagnosis. Lastly, there were 2 patients in the full treatment group and 1 patient in the partial treatment group who received bezlotoxumab therapy and 1 patient in the full treatment arm also received FMT, which are both known to reduce the rate of recurrence.
Partial courses were considered as anything less than the 10-day course, and although the days of therapy varied, the majority received <5 days. Given the structure of this study, selection bias existed, yet the partial therapy arm with older patients did not experience a higher rate of CDI recurrence at 30 and 90 days. It is also important to note that due to the retrospective nature of this study, we could not account for patients seeking follow up care for CDI recurrences at other institutions.
Generalizability may be limited given that this study was conducted at a single center with a patient population that may not be reflected at other hospitals. Carilion Roanoke Memorial Hospital has a medication assistance program which enables patients with financial barriers to gain access to more expensive medications such as fidaxomicin, which may not be feasible at all institutions. Carilion Medication Assistance Program primarily utilizes manufacturer programs and could potentially be replicated. However, the availability of manufacturer programs may change over time. Involvement of rounding pharmacists including medication specific interventions was not collected and therefore their impact on fidaxomicin acquisition at discharge cannot be quantified. Information on previous treatment courses, specifically choice of agent, was not collected. Although this information may have offered perspective on quantifying baseline dysbiosis, the full course and partial course groups had a minority of patients who had experienced a CDI episode in the previous 12 months (22.2% vs 17.9%, P = 0.452).
Conclusion and Relevance
Partial courses of fidaxomicin followed by oral vancomycin to complete therapy, had similar 30-day CDI recurrence compared with full course fidaxomicin. The study findings do not suggest a benefit of purposeful utilization of partial courses of fidaxomicin over full courses. However, in a real-world setting, there are transitions of care and financial barriers that may preclude completing a full course of fidaxomicin. These findings may suggest switching to oral vancomycin therapy is not detrimental. Concerns regarding affordability at discharge should not be a contraindication to the initiation of fidaxomicin. Additional studies are needed to confirm this hypothesis and practice.
Footnotes
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study protocol was reviewed by the Carilion Clinic’s Institutional Review Board prior to commencement and determined to be exempt as it did not meet the definition of human subjects’ research.
