Abstract
Background:
The use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) in nephrology practice is increasingly becoming standard of care in patients with diabetes or those with proteinuria.
Objectives:
The primary outcome was to identify the proportion of pre-dialysis patients with chronic kidney disease (CKD) G3a, G3b, or G4 prescribed an SGLT2i and describe their characteristics.
Methods:
This was a retrospective, multicentric, cross-sectional study of patients with CKD followed at 4 pre-dialysis clinics in the province of Quebec, Canada. We collected data of multiple covariates associated with prescribing SGLT2i in patients over 18 years of age with CKD G3a, G3b, or G4. We then performed a multivariate logistic regression to assess their associations.
Results:
Of the 874 patients included, 22.7% were prescribed an SGLT2i. Factors most strongly associated included male sex (odds ratio [OR] = 4.88, 95% CI = 2.38-10.03), being prescribed metformin (OR = 4.30, 95% CI = 2.23-8.31), having type 2 diabetes (OR = 4.00, 95% CI = 1.86-8.62), or having an albumin-to-creatinine ratio greater than 300 mg/g (OR = 1.84, 95% CI = 1.08-3.14). The majority of patients (60.4%) had their SGLT2i initiated by the pre-dialysis clinic and the most frequent adverse event was an initial increase in serum creatinine 1 week after starting treatment (33.9%).
Conclusion and relevance:
An increasing number of patients with CKD are being prescribed SGLT2i. Nonetheless, significant disparities in sex, severity of disease, and comorbidities remain. We suggest that specific strategies be put in place to promote prescribing of SGLT2i in women and other at-risk populations, in particular among nephrology teams, to improve patient care.
Keywords
Introduction
Chronic kidney disease (CKD) is an irreversible health condition that affects approximately 1 in 10 Canadians. 1 As the Canadian population continues to age, its prevalence is bound to increase along with its numerous comorbidities and health care burden. 1 Until recently, angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARBs) were the only pharmacological intervention recommended to slow the decline of the estimated glomerular filtration rate (eGFR) independent of blood pressure control, albeit in patients with diabetes or those with proteinuria.2,3 Nonetheless, eGFR will continue to decline with pharmacotherapy, and not all patients are candidates for ACEi or ARBs.
Sodium-glucose cotransporter-2 inhibitors (SGLT2i) have increasingly become standard of care to slow the progression of CKD. In 2019, the Canagliflozin and Renal Endpoints in Diabetes with Established Nephropathy Clinical Evaluation (CREDENCE) trial demonstrated a reduction in composite outcome of kidney failure, doubling of serum creatinine, or kidney or cardiovascular (CV)-related death irrespective of HbA1c improvement in patients with diabetes with proteinuria. 4 In 2020, the Dapagliflozin and Prevention of Adverse Outcomes in CKD (DAPA-CKD) trial would increase the eligibility to non-diabetic patients with CKD and proteinuria with an eGFR down to 25 mL/min/1.73 m2. 5 Finally in 2022, the Empagliflozin in Patients with Chronic Kidney Disease (EMPA-KIDNEY) trial proved that patients with an eGFR down to 20 mL/min/1.73 m2, regardless of proteinuria, could also be prescribed an SGLT2i to lower the risk of progression of kidney disease and reduce CV-related death. 6
A recent retrospective cohort study demonstrated that 17.5% of patients followed in nephrology care in British Columbia between 2014 and 2017 would be candidates for SGLT2i based on CREDENCE and DAPA-CKD inclusion criteria. 7 Currently, the Canadian Diabetes guidelines recommend initiating an SGLT2i in patients with type 2 diabetes and CKD stage 3b to slow the progression of CKD, whereas the KDIGO 2022 diabetes guidelines recommend initiating SGTL2i in patients with type 2 diabetes and an eGFR >20 mL/min/1.73 m2.8,9 Considering the widening eligibility of SGLT2i for patients with CKD and the limited therapeutic options, it is important to understand their real-time use in pre-dialysis clinics. The primary aim of this study was to identify the proportion of patients with CKD G3a, G3b, or G4 prescribed an SGLT2i and to describe their characteristics to better identify practice gaps and propose solutions to improve nephrology practice.
Methods
Study Design and Population
This multicenter cross-sectional retrospective study was performed using health data collected between March 1 and August 4, 2023, of patients with CKD from 4 pre-dialysis clinics in the province of Québec, Canada: l’Institut Universitaire de Cardiologie et de Pneumologie de Québec (IUCPQ), CSSS Memphrémagog Centre Hospitalier, Centre Hospitalier de Rimouski, and Centre Hospitalier Universitaire de Sherbrooke (CHUS). The centers represent 4 out of the 5 dedicated multidisciplinary pre-dialysis clinics in Québec, as well as being representative of a broad proportion of the Québec population at risk of CKD progression.
Patients frequenting pre-dialysis clinics in the province of Québec have CKD G3a, G3b, G4, and G5. Patients are followed by a multidisciplinary team, which includes a pharmacist, a nephrologist, a nurse, and a nutritionist who meet yearly with the patients to conduct a detailed review of their clinical condition. Patients may be prescribed an SGLT2i by their nephrologist after these medical visits. Furthermore, clinic pharmacists, in collaboration with a nurse clinician, are responsible for the management of hypertension, diabetes, and laboratory monitoring periodically (approximately every 3 months). During these follow-ups, which are mainly conducted by phone, pharmacists may identify patients eligible to receive an SGLT2i and have the professional autonomy to independently initiate therapy. Medical notes, including current pharmacotherapy and laboratory values, are updated by a member of the multidisciplinary team at each follow-up.
Patients enrolled in the study were screened to ensure they were followed for CKD and not for other renal diseases. Chronic kidney disease was stratified per the KDIGO 2012 classification based on non-adjusted eGFR as it was the most accessible laboratory reference used in all participating pre-dialysis clinics. 7 Patients were included if they were more than 18 years old by the collection date and had visited one of the designated pre-dialysis clinics at least once since the month of April 2021. The date of April 2021 was chosen as an inclusionary criterion as it corresponds to 6 months after the publication of DAPA-CKD (October 2020), which would allow time for prescribers to incorporate the findings of this study into their practice. Our inclusion criteria were broader than those in the CREDENCE, DAPA-CKD, and EMPA-KIDNEY studies to include as many patients followed for CKD as possible to best capture real-world use of SGLT2i.
Patients with CKD G5 or who were followed for less than 3 months by the pre-dialysis clinic were excluded from the study at the time of data collection. We considered that new patients would likely not be prescribed an SGLT2i at their first appointment as prescribers are likely to focus on other interventions first.
Data Collection and Study Variables
A non-validated data collection questionnaire was used to compile information from patient files.
We classified patients currently prescribed SGLT2i as users, patients previously prescribed an SGLT2i which had since been discontinued to be previous users, and all other patients as non-users. Previous use of SGTL2i was assessed based on the clinical notes. For users, data were collected as per the start date of when the medication was prescribed by the pre-dialysis clinic. For users who started their medication by an outside prescriber or before referral to a pre-dialysis clinic, the information was collected as per their first visit to the clinic where the medication was recorded. For any users with missing information, data were instead collected as per the closest time preceding the follow-up. For previous users and non-users, the variable data was collected based on the last clinic follow-up.
The presence or absence of a prescription for an SGLT2i (canagliflozin, dapagliflozin, or empagliflozin) was considered the only dependent variable. The independent variables, or covariates, used to describe the characteristics of enrolled patients include demographic data (age, gender, pre-dialysis center). In addition, clinical data likely to influence prescribing was collected such as the category of CKD, cause of CKD (diabetic nephropathy, hypertension or renovascular disease, glomerular disease, lithiasis, polycystic kidney disease other, unknown), body mass index (BMI), comorbidities (hypertension, type 1 or 2 diabetes, heart failure, coronary artery disease [CAD], peripheral artery disease [PAD]), recent laboratory values (serum creatinine, non-adjusted eGFR, HbA1c, albuminuria category), and the use of concomitant medications with similar physiological effects (diuretics, antihypertensives, antidiabetics).
To better understand prescribing patterns and barriers, several other independent variables were also collected such as the name and dose of the SGLT2i, if it was initiated by the pre-dialysis clinic, the variation of the eGFR 1 week, 1 month, and 3 months after introducing an SGLT2i, any adjustment of concomitant medications in the month following the start of an SGLT2i, tolerability of the SGLT2i (presence or absence of known adverse reactions), as well as if the patient was a previous user and the cause of cessation if it was recorded.
The collection was carried out by a team of 5 research collaborators, including pharmacists and interns. A training session was organized before starting to limit inter-individual variability. Furthermore, a mini collection of data of 5 patients was completed at each participating center by 2 research collaborators to detect discrepancies in the interpretation of the data to be collected and make corrections if necessary. The data were then anonymized by being assigned a random number before being compiled using Redcap software. Each center kept a password-protected Excel file associating the patient file with its randomized number, so that the collaborators could refer to it if any data appeared to be missing.
Ethics Approval
This study was approved by the research ethics board at the CIUSSS de l’Estrie-CHUS as a multicentric project. The study was assigned the research code MP-31-2023-4993.
Statistical Analysis
Descriptive statistics were used to portray patient characteristics in each group (current users, non-users, and previous users). Previous users were considered as non-users when descriptive statistical analyses were performed. We summarized categorical variables as proportions and compared between groups with chi-square tests. We summarized continuous variables with means and standard deviations (SD) and compared between groups with independent t tests. We reported the proportion of missing data when it exceeded 5% of the total values collected.
We performed univariate logistic regressions on all the covariates to ascertain the most significant variables involved in being prescribed an SGLT2i. The results were presented as odds ratios (ORs) and 95% CIs. A multivariate logistic regression was then produced with the most significant covariates for all patients enrolled in the study. We considered previous users as current users for the multivariate regression. We also completed a second regression analysis only with patients with diabetes to determine the effects of the HbA1c on SGLT2i use. The results are presented as adjusted OR with 95% CI. Statistical analyses were completed by a statistician using SPSS version 28.
The sample size calculation is based on our primary outcome where we wanted to estimate the proportion of patients with stage 3 or 4 CKD who are prescribed an SGLT2i. According to recent Canadian studies, the proportion of SGLT2i users with CKD ranges from 7.1% to 17% in nephrology care which includes diabetic and non-diabetic patients.7,10 However, we estimate that we will have a higher proportion, around 20%, considering criteria was not restricted only to patients with diabetes and the data were collected after the release of the DAPA-CKD trial. We initially estimated in our research protocol a sample size of 600 patients with a 95% CI and a user proportion of 20%, which gave a margin of error of 3.4%. As the study evolved to include a larger number of patients, we recalculated a sample size of 900 patients and estimated with NCSS software a margin of error of 2.5% with a CI of 17.6% to 22.6%.
Results
The pre-dialysis clinics included in the study consisted of 1116 patient files that were reviewed individually by the research team. Of these files, 4 did not meet the inclusion criteria and 242 were excluded on the day the data were collected (Figure 1). From the 874 patients included in the study, 22.7% were prescribed an SGLT2i, 2.2% had previously been prescribed an SGLT2i but had ceased taking it, and 75.1% had never been prescribed an SGLT2i (Figure 1).
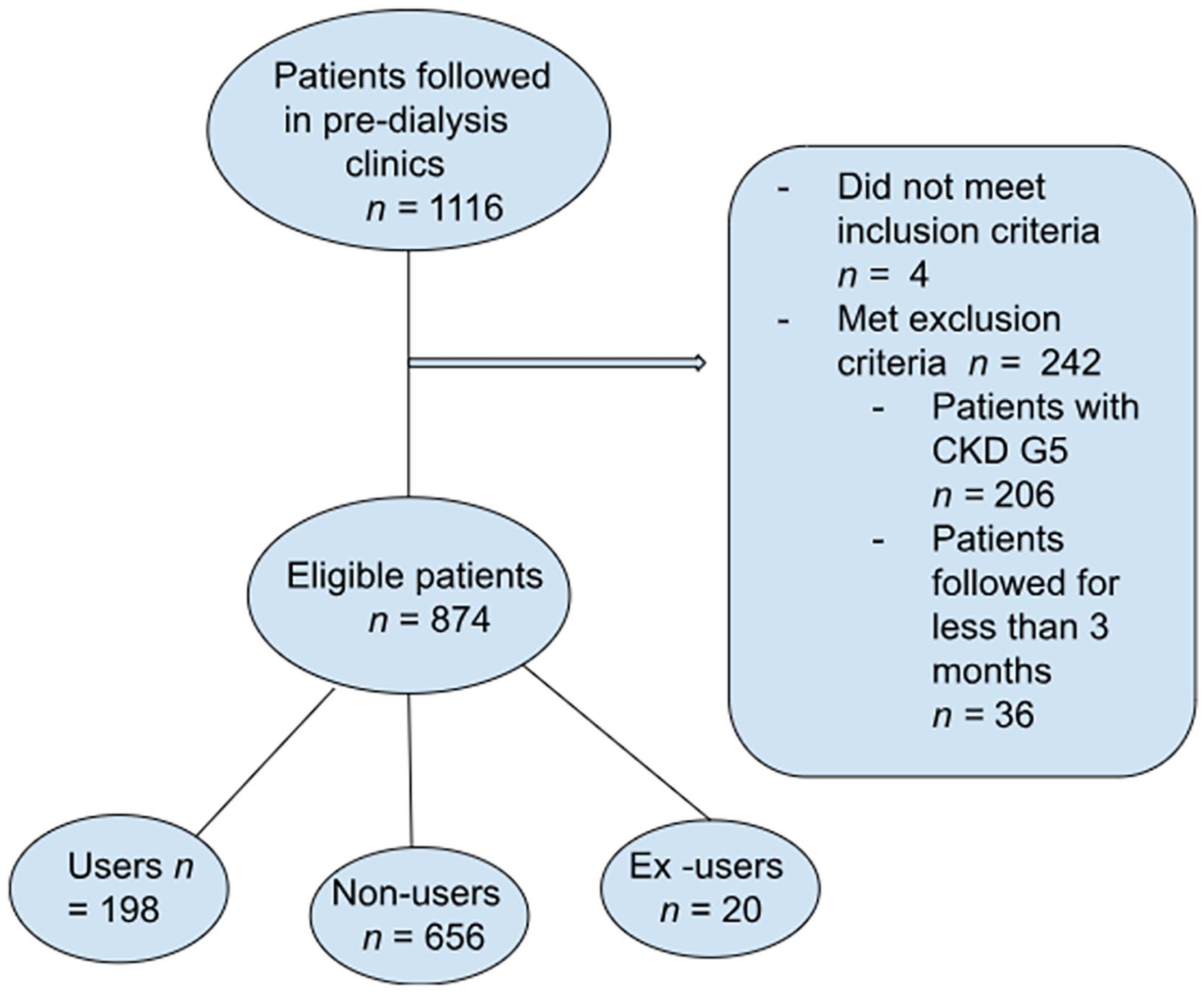
Selection of SGLT2i patients.
Factors Associated With Prescribing SGLT2i
The baseline characteristics of patients included in the study are presented in Table 1. When compared with patients not receiving an SGLT2i, those receiving an SGLT2i were on average slightly younger, predominantly male, and have a greater mean BMI. Overall, patients prescribed an SGLT2i have a better estimated renal clearance, have an albumin-to-creatinine ratio greater than 300 mg/g, be prescribed an ACEi/ARB, and are more likely to have CKD secondary to diabetic nephropathy. Patients with diabetes were more likely to be prescribed SGLT2i, have higher mean HbA1c, and be prescribed concomitant antidiabetic medications such as metformin, sulfonylureas, DPP4 inhibitors, GLP1 agonists, or insulin. Sodium-glucose cotransporter 2 inhibitor users were also more likely to be hypertensive as well as be prescribed a thiazide diuretic (21.8% vs 12.2%).
Baseline Characteristics of SGLT2i Users Compared With Non-Users.
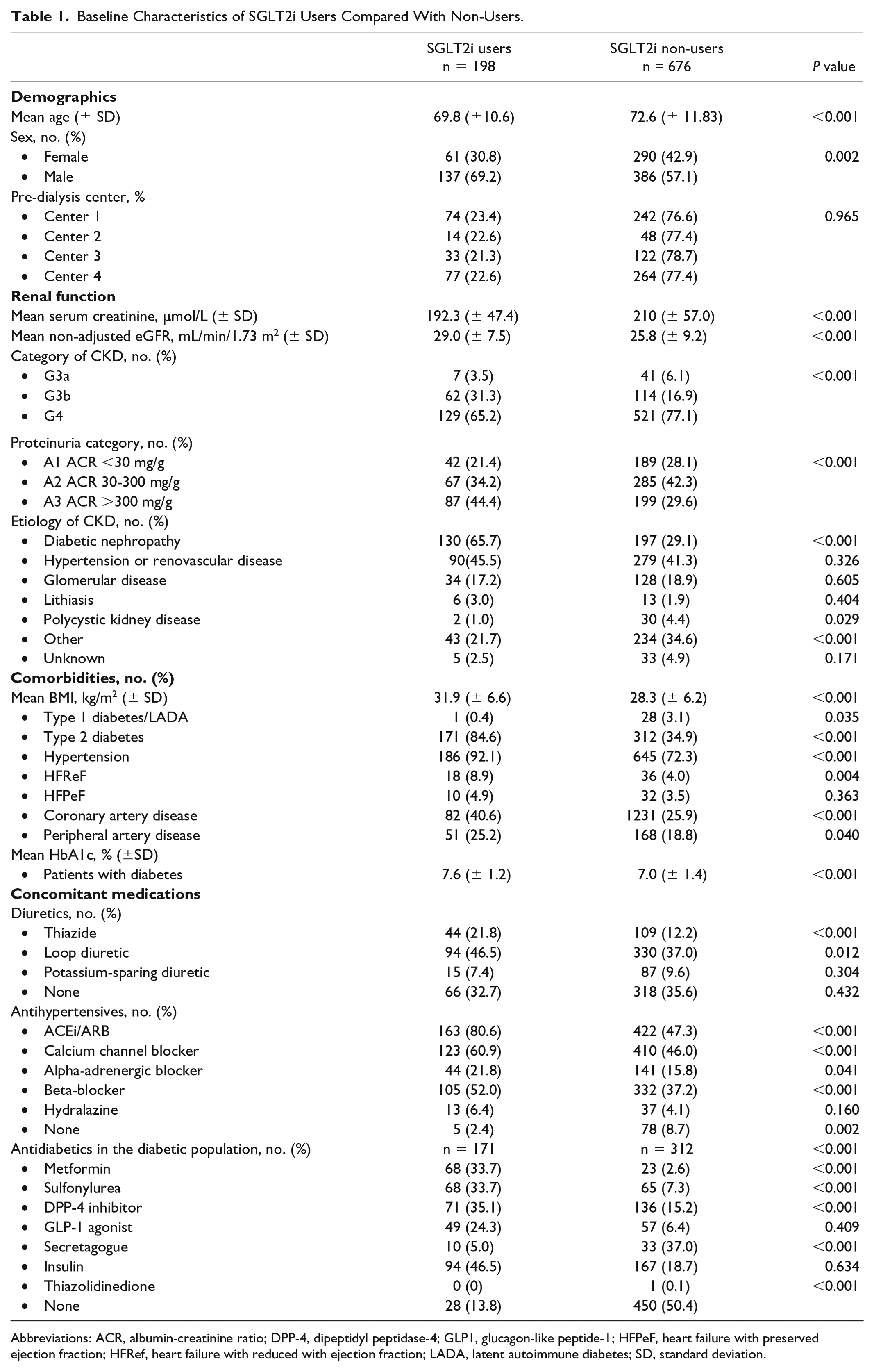
Abbreviations: ACR, albumin-creatinine ratio; DPP-4, dipeptidyl peptidase-4; GLP1, glucagon-like peptide-1; HFPeF, heart failure with preserved ejection fraction; HFRef, heart failure with reduced with ejection fraction; LADA, latent autoimmune diabetes; SD, standard deviation.
The multivariate logistic regression included 857 patients with complete data on all variables (Figure 2). Factors most strongly associated with being prescribed an SGLT2i included male sex (OR = 4.89, 95% CI = 2.38-10.03), being prescribed metformin (OR = 4.30, 95% CI = 2.23-8.31), having type 2 diabetes (OR = 4.01, 95% CI = 1.86-8.62), or having an albumin-to-creatinine ratio greater than 300 mg/g (OR = 1.84, 95% CI = 1.08-3.14; Figure 2). Developing CKD secondary to renovascular hypertension (OR = 0.470, 95% CI = 0.728-1.993) or a different etiology (OR = 0.322, 95% CI = 0.790-2.053) were least associated with being prescribed an SGLT2i, although these findings were not statistically significant (Figure 2). In a secondary analysis of patients with diabetes, HbA1c was associated with an increased likelihood of taking an SGLT2i (OR = 1.57, 95% CI = 1.27-1.93), and male sex had a stronger association (OR = 6.13, 95% CI = 2.70-13.89; Figure 3).
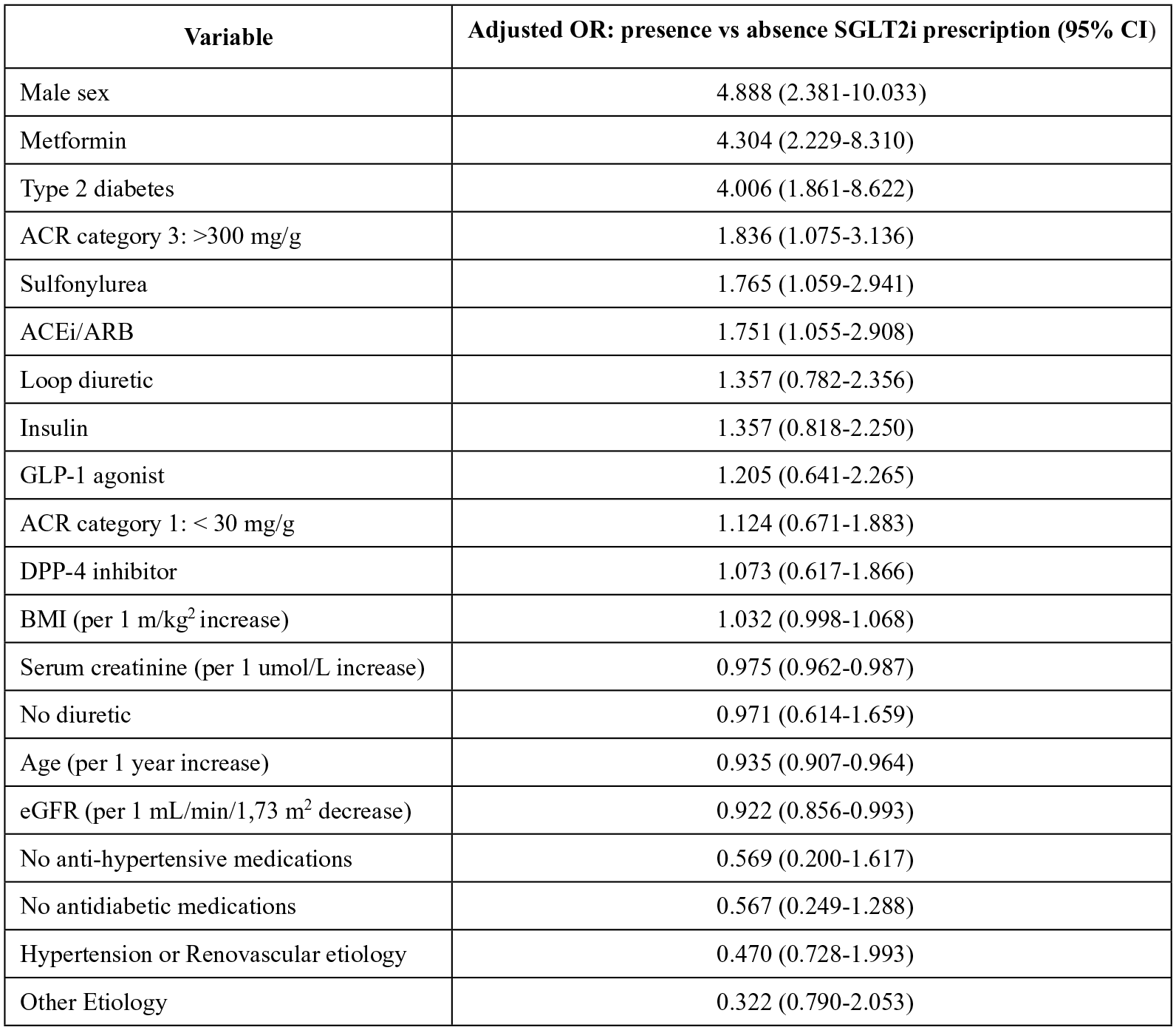
Factors associated with SGLT2i prescribing in all patients followed in pre-dialysis clinics.
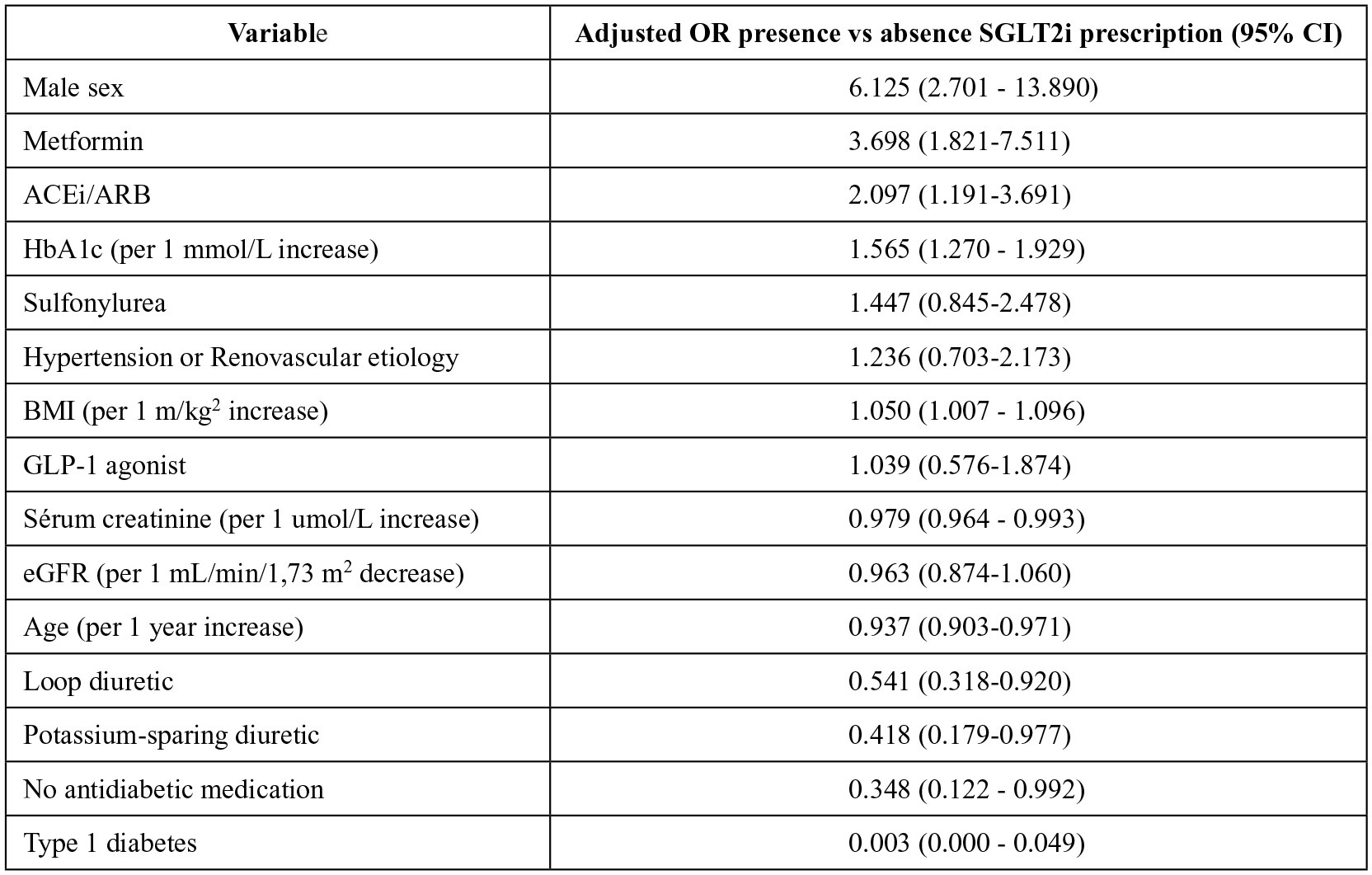
Factors associated with SGLT2i prescribing in patients with diabetes followed in pre-dialysis clinics.
Characteristics of Patients Prescribed an SGLT2i
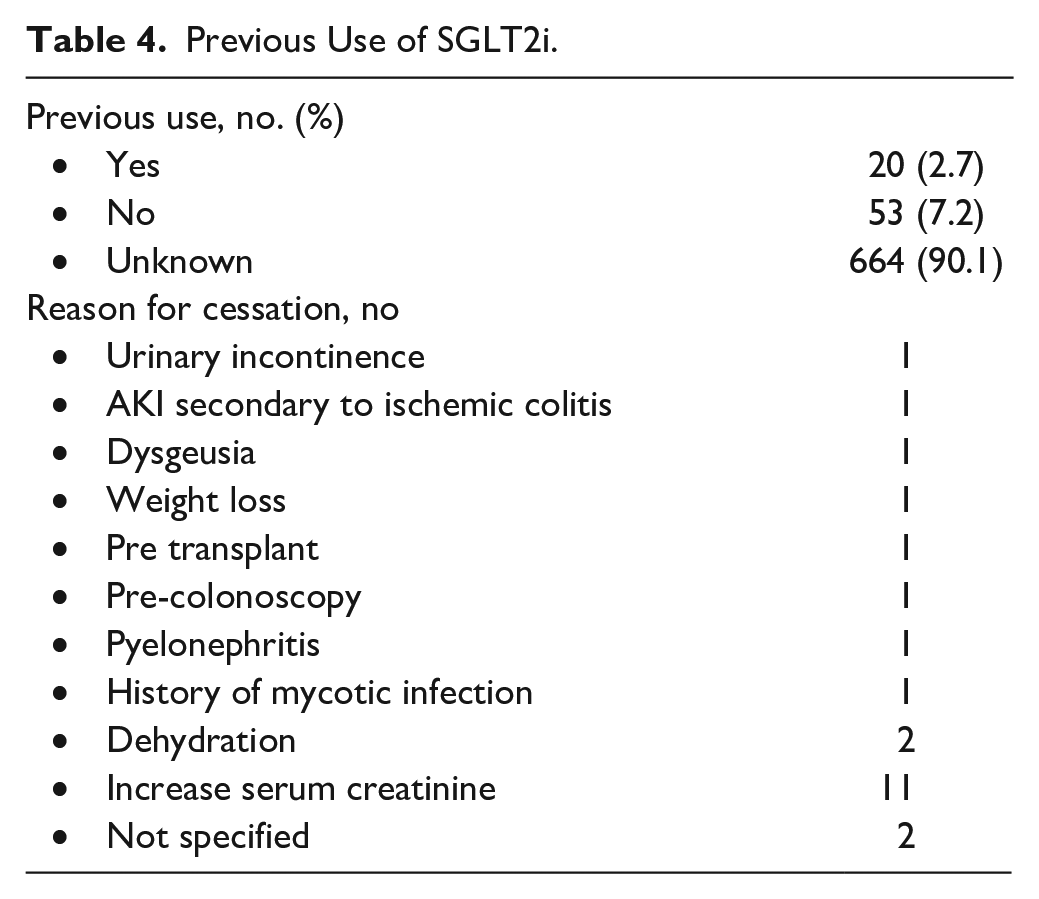
From patients followed in pre-dialysis clinics, 60.4% of them had an SGLT2i initiated by the nephrology team (Table 2). The largest proportion of SGLT2is prescribed were dapagliflozin 10 mg and empagliflozin 10 mg (Table 2). The mean non-adjusted eGFR at baseline of initiating an SGLT2i was 28.7 mL/min/1.73 m2, which decreased slightly 1 week after initiating an SGLT2i to 26.3 mL/min/1.73 m2, and subsequently returned to a mean of 28.8 mL/min/1.73 m2 after 3 months of use (Table 2). The most common adverse event was any increase in creatinine 1 week after starting an SGLT2i, with the majority being less than a 20% increase or between 20% and 30% (Table 2). A further 7.4% were found to have asymptomatic hypotension after starting the medication (Table 2). Aside from the rate of adverse events after initiating an SGLT2i, which was missing in 48.3% of patients, missing data for all other variables recorded was less than 2%. The majority of patients had a concomitant medication adjusted when starting an SGLT2i (Table 3). Decreasing a concomitant antidiabetic medication, stopping an antidiabetic, stopping a thiazide, or decreasing the dose of a loop diuretic were the most common interventions noted (Table 3). From all patients enrolled in this study, 2.7% were confirmed to have previously taken SGLT2i, but had since ceased. However, in 90.1% of patients, previous use could not be confirmed as this information was infrequently recorded in medical files (Table 4). The most common reason for cessation was due to increased creatinine levels, although the percentage of increase was not documented (Table 4).
Characteristics of SGLT2i Prescribing in the Province of Québec.

Interventions on Concomitant Medications When Initiating SGLT2i.

Previous Use of SGLT2i.
Discussion
To the best of our knowledge, this is the first observational study showing how SGLT2is are being used in a CKD population frequenting pre-dialysis clinics in a publicly funded health care system. This study is exploratory in nature, examining the relation between current prescribing patterns and the increasing medical literature regarding the best use of SGLT2i for renal protection. The study demonstrated that 22.7% of patients followed by nephrology teams in Québec were prescribed an SGLT2i, which markedly surpasses the estimated number of eligible patients from other Canadian studies that examined patients with CKD after the results of DAPA-CKD were published.7,10 Although current Canadian guidelines recommend starting SGLT2i in patients with diabetes with CKD G3b or greater, it is interesting to note that a majority of patients in this study had their SGLT2i initiated by the pre-dialysis clinics who follow almost exclusively patients with an eGFR of G3b or less.8,10,11 As noted by Ozaki et al, 12 it can take many years before optimal use of medications are achieved, such as with statins in patients with CAD.
In addition, only 80.6% of patients were also prescribed an ACEi or ARB, and yet it is only the most recent published trial in 2023, EMPA-KIDNEY, that included patients not on renin-angiotensin-aldosterone system inhibitors. 6 Furthermore, a significant proportion of patients who would be excluded from randomized control trials, such as those with polycystic kidney disease, were nonetheless still prescribed an SGLT2i.4-6 These findings seem to indicate an increased ease with starting these medications in the pre-dialysis population and that prescribing practice may either precede guideline updates or supersede the best available evidence. 13
SGLT2i prescribing in our eligible population at risk of CKD progression was nonetheless less optimal than seen in randomized controlled trials. In this study, being a male was associated with an almost 5-fold increased odds to receive an SGLT2i compared with being a female after adjusting for other covariables. Alas, the wide CIs are likely to minimize the significance of our result. Lau et al, 10 in a cross-sectional study of adults with CKD, also reported an almost 2-fold odds of receiving an SGLT2i in male patients. Whereas sex-based disparities in monitoring and management of CKD have increasingly come to light in recent years, it is still unclear as to why this exists among similarly high-risk groups.14,15 An increased rate of mycotic infections has been proposed as a possible factor; it is only recommended to avoid SGLT2i in patients with a history of severe, recurrent infections.16,17 Increasing awareness of this unrecognized imbalance presents an opportunity for nephrology teams to increase SGLT2i prescribing for a substantial portion of patients.
Furthermore, although patients with diabetes make up 55% of patients treated in pre-dialysis clinics, they represent 85.7% of patients prescribed an SGLT2i. This could most likely be attributed to multiple factors, such as the timing of drug approval for renal protection in non-diabetic patients with proteinuria only being recognized in recent years.5,6 Poor glycemic control is considered another covariable often associated with SGLT2i prescribing. This study found one and a half times increased odds of receiving an SGLT2i in regard to HbA1c levels, which is less significant than the increased odds found by Lau et al. 10 This could possibly be due to the increasing evidence of using SGLT2i for renal protection, especially considering that the glycemia lowering effect is inefficient in CKD G3b but most insurances will only reimburse payment for patients with diabetes.18,19 Our finding also correlates with the SGLT2i doses prescribed in this study, which better reflect doses used for nephroprotection regardless of glycemic control.5,6,16 Efforts to update nephrology guidelines should be made to support increasing insurance coverage of SGLT2i for non-diabetic patients.
Although not examined in our study, socioeconomic disparities are important variables in SGLT2i prescribing. 12 As a publicly funded health care system, the Québec provincial formulary, the RAMQ, currently reimburses patients for SGLT2i in cases of uncontrolled diabetes or certain cases of heart failure with reduced ejection failure (HFrEF), thus causing a barrier to prescribe in CKD without diabetes or heart failure. 19 Patients without access to a private insurance plan and not eligible for provincial coverage would have to disburse between $78 and $83 monthly before dispensing fees. 20 Prescribers can request a special authorization for non-diabetic or HFrEF patients, although it is not known how frequently these requests are approved. Our study showed that patients with diabetes were twice as likely to being prescribed an SGLT2i than a patient with severe proteinuria, although elevated proteinuria was itself associated with an almost 2-fold chance of being prescribed an SGLT2i. This difference between patients with diabetes and patients with proteinuria could be explained by the fact that both patients are the most likely to benefit from this medication, yet patients with proteinuria would be less likely reimbursed by the provincial health care system. 20
Patients with heart failure with reduced ejection fraction also had a 2-fold rate of being prescribed an SGLT2i. It is however hard to draw further conclusions considering they comprised only 6.3% of the patient population. Nevertheless, considering the limited pharmacotherapeutic options that exist to limit CKD progression, efforts will be needed to increase SGLT2i prescribing in non-diabetic patients with CKD. As noted by previous researchers, a focus on net cost savings associated with SGLT2i prescribing in patients with CKD could be aid in the campaign to expand public drug insurance criteria.7,21
A majority of patients had concomitant medication adjusted when starting an SGLT2i. This included any adjustments done to loop or thiazide diuretics, ACEi or ARBs, other antihypertensives, and antihyperglycemic medications (Table 3). Even though SGLT2i demonstrate diuretic, antihypertensive, and antidiabetic effects, there does not seem to be a consensus between expert opinion and clinical experience on how to introduce these medications.16,22 Further research in how to ideally adjust patients’ pharmacotherapy in this population is warranted.
This study’s strengths are 2-fold. First, it is representative of current nephrology practice as it was multicentric and derived from clinical notes. Unlike studies derived from large medical databases, this study was able to correlate more precisely lab monitoring and pharmacotherapy interventions in relation to SGTL2i prescribing. Second, it was not limited to the inclusion and exclusion criteria of randomized controlled trials. Thus, it was able to capture more patients who are being treated with SGLT2i and therefore be more representative of real-world practice.
Our study did have several limitations. We were not able to capture information from patients managed in private practice or by other health care professionals. This may have led to a selection bias as these patients may be considered at higher risk of CKD progression and complications regardless of SGLT2i eligibility or best practice guidelines. There is also missing data due to either a lack of documentation or that the data sought had not yet been collected, though this is a limit of cross-sectional studies and was confined in this study to previous SGLT2i use and the rate of adverse events. It is also possible that patients prescribed an SGLT2i were not adherent to their treatment, as this was limited to information recorded in patients’ medical files during follow-ups. Finally, the data collected by each clinic is not homogeneous, and therefore, some variables could not be collected for analysis while other variables presented with large CIs limit the precision of our results. Nonetheless, our study is representative of a broad proportion of Québec patients at risk of CKD progression and provides insight into factors affecting the probability of being prescribed an SGLT2i.
Conclusion and Relevance
Among patients frequenting pre-dialysis clinics in a publicly funded health care system, an increasing number is being started on SGLT2i to slow the progression of CKD. Although rates of prescribing surpass earlier estimates, disparities in sex, severity of disease, and comorbidities remain an impediment to further improving patient care. Future research should address sex inequalities in prescribing SGLT2i as this remains the least explained gap in practice. Barriers to prescribing also seem to include antiquated guidelines, prescribers’ discomfort with initiating medications in earlier stages of CKD, as well as lack of coverage by public health insurance. Specific strategies should promote prescribing of SGLT2i in women and other at-risk populations, in particular among nephrology teams, to optimize the use of these nephroprotective agents.
Footnotes
Acknowledgements
The authors would like to thank Gabriela Maria Zuniga and Sarah-Maude Bachand for their support in collecting information from patient files, as well as Catherine Allard for her statistical expertise.
Author’s Note
Aylon Wisbaum is also affiliated Centre Intégré Universitaire de Santé et de Services Sociaux de la Montéregie Centre,Longeuil, QC, Canada.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sandrine Gaudreau, Aylon Wisbaum, Isabelle Cloutier, Pascale Robert, Regina Kolment, and Marie-France Beauchesne have no conflicts of interest to declare. Jodianne Couture has received funding from AstraZeneca, Amgen, and Otsuka Pharmaceuticals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by unrestricted research grants from AstraZeneca, Amgen, and Otsuka Pharmaceuticals as well as a grant by the University of Montreal for the statistician services. The funders had no role in the design of the study, the collection, the analyses, or the interpretation of data, the writing of the manuscript, or the decision to publish the results.
