Abstract
Objective:
The objective of this study was to review the available literature for dexmedetomidine sublingual film use in the treatment of acute agitation associated with schizophrenia and bipolar disorders.
Data Sources:
A literature search of PubMed (January 2017-March 2023) and EMBASE (January 2017-March 2023) was performed using the terms: Igalmi, dexmedetomidine, schizophrenia, bipolar disorder, and agitation. Additional information sources include ClinicalTrials.gov, scientific posters, and articles identified through review of references from clinical trials publications.
Study Selection and Data Extraction:
Relevant English-language articles conducted in humans were considered, with a preference for phase 3 clinical trials. Trial analyses and articles discussing pharmacology, pharmacokinetics, efficacy, and safety were also evaluated.
Data Synthesis:
Dexmedetomidine sublingual film was evaluated for use in schizophrenia in the SERENITY 1 pivotal trial and for bipolar disorders in the SERENITY 2 pivotal trial. Both studies found treatment of mild to moderate agitation with dexmedetomidine sublingual film 180 and 120 μg to be superior to placebo in reducing the severity of agitation. Treatment effect was seen as early as 20 minutes. Somnolence was the most common adverse effect in both studies. Cardiovascular adverse effects were mild and transient in most cases.
Relevance to Patient Care and Clinical Practice:
Dexmedetomidine sublingual film is a new and novel treatment for agitation and gives clinicians an alternative to antipsychotic and benzodiazepine use. It has advantageous properties including its noninvasive route of administration, fast absorption, and rapid onset of effect. Cost may limit its use.
Conclusion:
Dexmedetomidine sublingual film provides an alternative approach to treatment of acute agitation in adults with schizophrenia and bipolar disorders based on both mechanism of action and route of administration.
Introduction
Dexmedetomidine is a selective alpha-2 adrenergic agonist approved by the US Food and Drug Administration (FDA) in 1999 as an intravenous medication for sedation in adult patients initially intubated and mechanically ventilated during treatment in the intensive care unit. The medication later received approval for use in non-intubated adult patients requiring sedation prior to and/or during surgical or medical procedures. 1 As of April 2022, dexmedetomidine has a new indication and dosage formulation. Dexmedetomidine is now available as an orally dissolving film given by sublingual or buccal administration for the treatment of acute agitation associated with schizophrenia or bipolar I or II disorder in adults. 2
Agitation is defined by the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), as “excessive motor activity associated with a feeling of inner tension.” 3 These behaviors can be repetitive and purposeless, and while not always associated with emotional distress, may lead to unintentional harm to the patient. 4 Due to excessive motor activity (eg, pacing, fidgeting, bouncing leg, moving objects without purpose), agitation is narrowly defined by the DSM-5 as “psychomotor agitation” and can be associated with psychiatric illnesses, substance intoxication/withdrawal, personality disorders, developmental disorders, and degenerative brain disorders. Agitation has been more broadly described by others as a syndrome involving restlessness, anxiety or nervousness, feelings of unease, excessive movement or speech, and irritability.3,5,6 Acute agitation is a common occurrence in psychiatric emergency settings and on inpatient psychiatry units, especially in those experiencing psychosis or mania.7,8 Of patients displaying aggression (ie, behavior associated with a potential for harm to self, other patients and health care providers) in the hospital setting, approximately a third of these cases were preceded by agitation.9,10
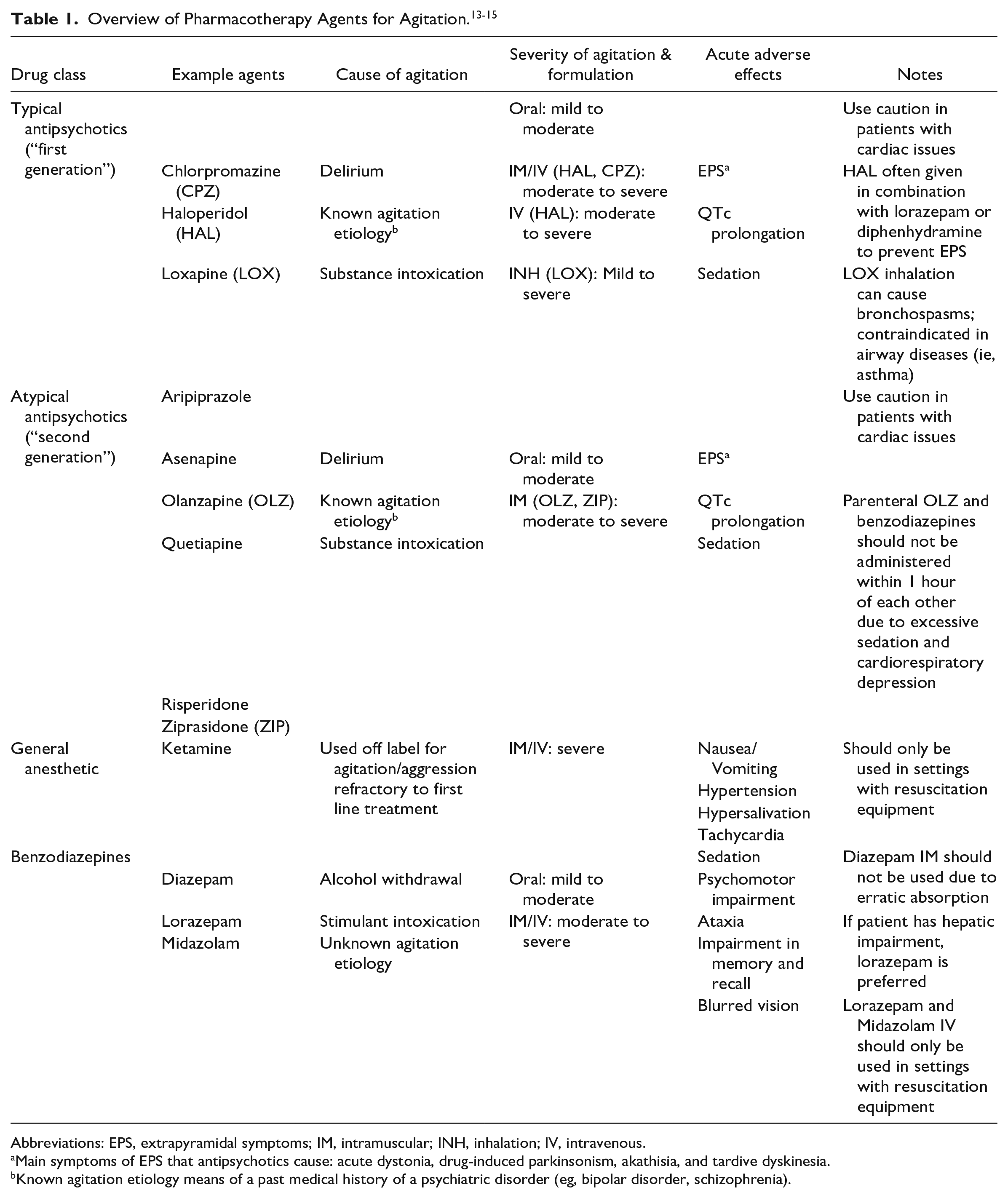
Effective and timely management of acute agitation is of utmost importance when treating patients with schizophrenia and bipolar disorders. Current treatment guidelines for the management of agitation and aggression recommend using first- and second-generation antipsychotics and/or benzodiazepines if nonpharmacologic strategies are ineffective.6,11 An overview of common antipsychotics and benzodiazepines used in clinical practice can be found in Table 1. Ideal characteristics of pharmacotherapy for acute agitation were compiled in the Expert Consensus Guideline Series: Treatment of Behavioral Emergencies which reflects core concepts in many treatment guidelines. These characteristics are as follows: (1) rapid onset, (2) noninvasive administration, (3) ability to calm without excessive sedation, (4) treating the underlying condition, and (5) tolerable adverse event profile. 12 None of the current treatments for agitation meet all stated criteria. This review will discuss the key pharmacologic, pharmacodynamic, and pharmacokinetic properties of dexmedetomidine sublingual film, clinical trials evaluating efficacy and safety leading to its approval, dosing and administration, and clinical implications for its use in the treatment of acute agitation associated with schizophrenia and bipolar disorders.
Abbreviations: EPS, extrapyramidal symptoms; IM, intramuscular; INH, inhalation; IV, intravenous.
Main symptoms of EPS that antipsychotics cause: acute dystonia, drug-induced parkinsonism, akathisia, and tardive dyskinesia.
Known agitation etiology means of a past medical history of a psychiatric disorder (eg, bipolar disorder, schizophrenia).
Data Selection
A literature search of PubMed (January 2017 to March 2023) and EMBASE (January 2017 to March 2023) was conducted in March 2023 with the following key terms: Igalmi, dexmedetomidine, schizophrenia, bipolar disorder, and agitation. Relevant English-language studies conducted in humans were considered. Primary use of Phase 3 pivotal trials was preferred; other trial data were evaluated to provide context. Additional resources included prescribing information, scientific meeting abstracts and posters, and other clinical trial data. Databases used included PubMed, EMBASE, and ClinicalTrials.gov. Bibiolography of identified articles were evaluated for other pertinent references.
Pharmacodynamics and Mechanism of Action
The mechanism of action and pharmacodynamics of dexmedetomidine differ significantly from drugs currently used to treat acute agitation in the psychiatry setting. Dexmedetomidine is a selective and potent agonist for the alpha 2-adrenergic receptor with high potency at all 3 enzymatic isoforms (A, B, C), demonstrating a Ki (inhibitor constant) of 4 to 6 nM.2,16 Agonism on the alpha2-adrenergic receptors within the brainstem, specifically alpha2a-adrenoreceptors, affects 2 pathways that contribute to a reduction in the stress response. Modulation of norepinephrine release from the locus coeruleus reduces sympathetic hyperarousal and modulation of the bed nucleus of stria terminalis decreases corticotrophin-releasing factor. 16 Through activation of presynaptic alpha2a-adrenoreceptors, it is believed that dexmedetomidine exerts its effect for the treatment of acute agitation associated with schizophrenia and bipolar disorders. 2
Pharmacokinetics
Dexmedetomidine orally dissolving film has a bioavailability of 72% and 82% when administered sublingually and buccally, respectively. 2 The bioavailability of the orally dissolving film is higher than ingested formulation of dexmedetomidine due to bypassing first-pass metabolism. 17 Drinking water as early as 15 minutes post administration has been shown to have minimal effect on the rate or extent of absorption when dexmedetomidine is given sublingually. The effect of water intake has not been assessed with buccal administration. For both sublingual and buccal administration, Cmax was reached within 2 hours. Its half-life was measured at approximately 2.8 hours. 2 Other important pharmacokinetic properties such as volume of distribution, route of elimination, and excretion parameters are only available for the intravenous formulation of dexmedetomidine and were not reported for the orally dissolving film. Table 2 contains a summary of key pharmacokinetic properties.
Select Pharmacokinetics for Dexmedetomidine Sublingual Film. 2
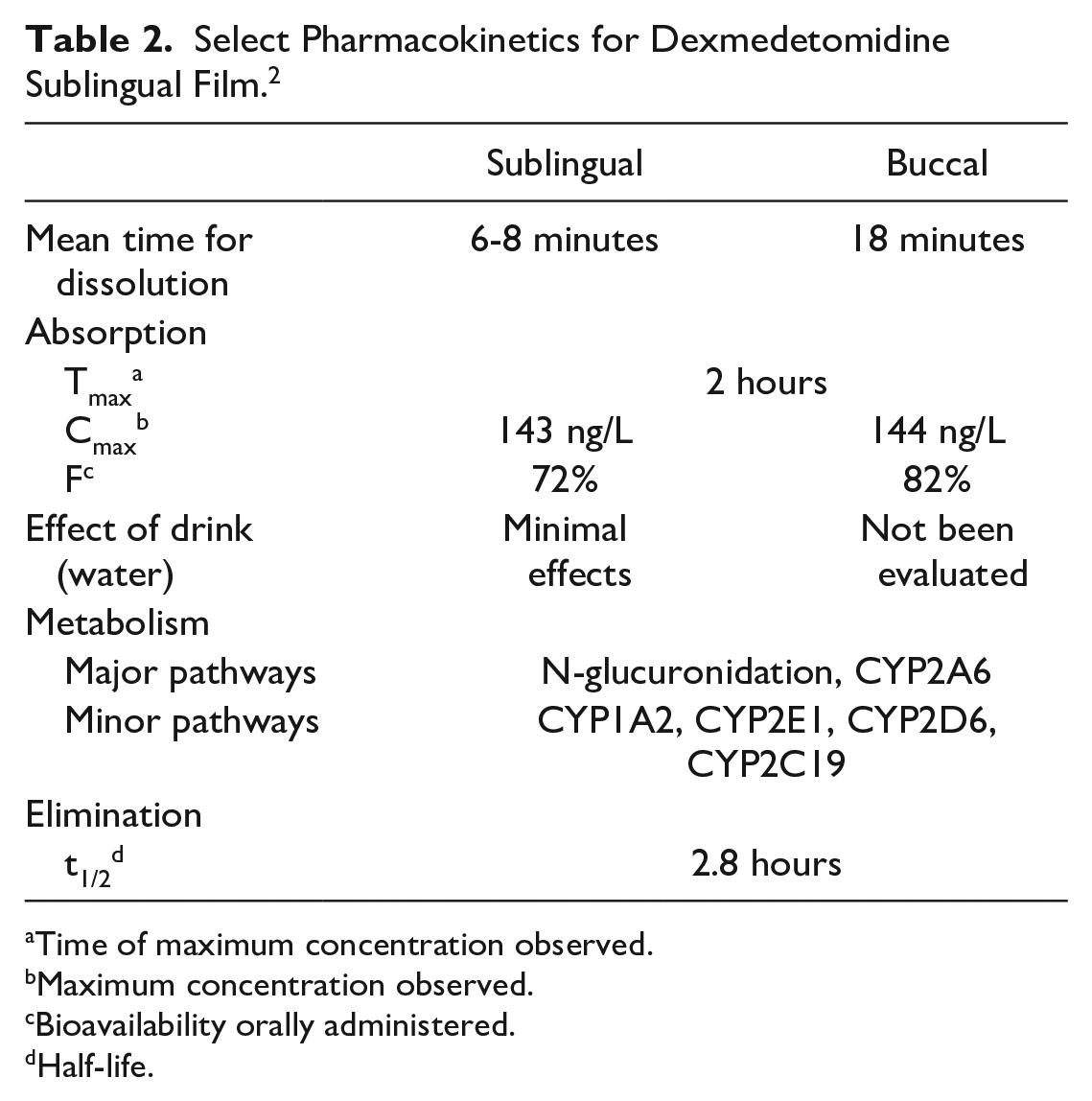
Time of maximum concentration observed.
Maximum concentration observed.
Bioavailability orally administered.
Half-life.
Dosing and Administration
The dexmedetomidine orally dissolving films are available as dexmedetomidine hydrochloride for sublingual or buccal administration in a rectangular film containing 2 microdeposits of either 60 or 90 μg providing a total dose of 120 or 180 μg, respectively. If an additional dose is needed due to uncontrolled agitation, 2 additional half-doses may be given by cutting the films at perforated lines. The time duration between the initial dose and subsequent doses must be at least 2 hours. The efficacy and safety of dexmedetomidine sublingual film has not been evaluated beyond 24 hours. The appropriate dose for an individual patient depends upon the severity of agitation, age, and hepatic function. Dosing recommendations based on patient characteristics are shown in Table 3. Product information states to avoid food and drink for at least 15 minutes after sublingual or at least 1 hour after buccal administration. 2 Although there is no FDA Risk Evaluation and Mitigation Strategy (REMS) associated with this drug, it is recommended that the drug should be used only with the supervision of a health care professional and monitoring of alertness and vital signs be completed to prevent syncope and falls.
Recommended Dosing Based on Patient Characteristics. 2
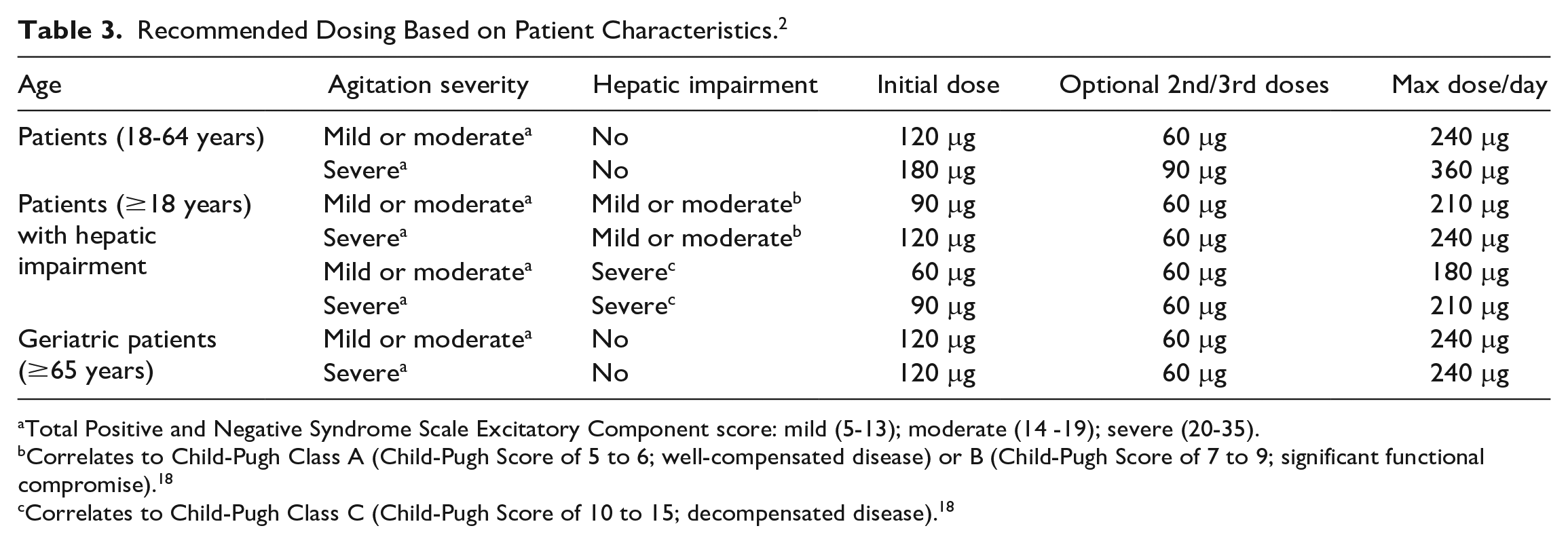
Total Positive and Negative Syndrome Scale Excitatory Component score: mild (5-13); moderate (14 -19); severe (20-35).
Correlates to Child-Pugh Class A (Child-Pugh Score of 5 to 6; well-compensated disease) or B (Child-Pugh Score of 7 to 9; significant functional compromise). 18
Correlates to Child-Pugh Class C (Child-Pugh Score of 10 to 15; decompensated disease). 18
Clinical Trials
A Phase 1b study evaluating the optimal dosage of dexmedetomidine sublingual film was available in a poster format only. 19 SERENITY I assessed the efficacy and safety of dexmedetomidine as compared with placebo in adults with agitation associated with psychotic disorders. 20 SERENITY II was similar in design to SERENITY II but included adults with agitation associated with schizophrenia. 21
Phase 1b: Sublingual Dexmedetomidine in Agitation Associated With Schizophrenia 19
The optimal dosing, efficacy, and safety of dexmedetomidine sublingual film in patients experiencing acute agitation associated with schizophrenia, schizoaffective disorder, or schizophreniform disorder as defined by the DSM-5 was evaluated in a Phase 1b, multicenter, randomized, double-blind, placebo-controlled, multiple single-ascending dose study. Efficacy was defined as the proportion of subjects who experienced a ≥40% reduction in Positive and Negative Syndrome Scale Excitatory Component (PEC) total score at 2 hours, which is a standard set by the FDA and used in pivotal trials for drugs to treat agitation. PEC is a scoring tool that measures 5 components associated with agitation: poor impulse control, tension, hostility, uncooperativeness, and excitement. Each component is scored from 1 (minimum) to 7 (maximum) and summed together to correlate to the severity of agitation: scores of 5-13 represents mild agitation, 14-19 represents moderate, and 20-35 represents severe.13,22 Five doses of dexmedetomidine sublingual film (20, 60, 80, 120, 180 μg) were compared with placebo and study participants were randomized in a 2:1 ratio to receive each dose of study drug or matching placebo. Study drug was given as an initial dose of 20 μg followed by single doses of 60, 80, 120, and 180 μg, except for the 20 μg treatment arm. Participants were required to meet standard inclusion criteria for agitation including a baseline total score on the PEC of ≥14 and a score ≥4 on at least one of the PEC items. 13
A total of 135 patients were enrolled in the study. Baseline demographic information and PEC scores were not included in the poster. The percentage of responders at 2 hours was 56% for 20 μg, 39% for 60 μg, 56% for 80 μg, 67% for 120 μg, and 89% for 180 μg doses as compared with 31% for placebo. 20 For the 2 commercially available doses (120 and 180 μg) of dexmedetomidine sublingual film as compared with placebo, the response rates were 56% and 56% vs. 22% at 1 hour, 67% and 94% vs. 20% at 4 hours, 61% and 94% vs. 29% at 6 hours, and 33% and 78% vs. 20% at 24 hours, respectively. 20 All doses separated from placebo at 1 hour on reduction in PEC total score with the exception of 20 μg as no data were presented for this dose (60 μg vs. placebo, P < 0.5; 80 and 120 μg vs. placebo, P < 0.01; 180 μg vs. placebo, P < 0.005).
This study concluded that dexmedetomidine sublingual film produced a significant and rapid reduction in agitation. All doses were safe and well tolerated. The highest dose, 180 μg, produced a lasting effect (ie, up to 6 hours) which was unexpected due to the short half-life of the drug. Based on this study, dexmedetomidine sublingual film 120 and 180 μg, were identified to move forward in clinical trials.
Phase 3: Dexmedetomidine in the Treatment of Agitation Associated With Schizophrenia or Schizoaffective Disorder 20
SERENITY I study was designed to evaluate the efficacy, safety, and tolerability of dexmedetomidine sublingual film compared with placebo in patients with acute agitation associated with schizophrenia and schizophrenia-like disorders (schizoaffective disorder and schizophreniform disorder). 21 This was a multicenter, randomized, double-blind, parallel-group, placebo-controlled study comparing dexmedetomidine sublingual film (120 or 180 μg) with placebo. The primary outcome was the mean change from baseline on the PEC total score at 2 hours. Important secondary outcomes were to determine the earliest time point a significant change from baseline in PEC total score occurred for sublingual dexmedetomidine as compared with placebo and percentage of participants experiencing response as defined by ≥40% reduction in PEC total score. Safety and tolerability were assessed through spontaneously reported adverse events, clinical laboratory tests (chemistry, hematology, urinalysis), electrocardiogram (ECG); pulse oximetry, buccal/sublingual assessment for local irritation, and vital signs. Study participants were required to meet DSM-5 diagnostic criteria for schizophrenia, schizoaffective, or schizophreniform disorder and the standard inclusion criteria for agitation stated in the previous study. Participants were randomized in a 1:1:1 ratio and stratified by age (<65 years; ≥65 years) to dexmedetomidine sublingual film 180 or 120 μg, or matching placebo. A single dose of study drug or placebo was administered upon presentation of acute agitation. If the agitation was persistent or recurrent, the change from baseline on PEC scale was <40%, and the participant was not experiencing any safety issues (eg, sedation or hypotension) then repeat administration of half the original dose (90 and 60 μg) or placebo was allowed after 2 hours.
A total of 381 patients were enrolled and randomly assigned to dexmedetomidine sublingual film 120 μg (N = 129), 180 μg (N = 125), or placebo (N = 126). Baseline demographics were similar across the groups. The study population had a median age of 45.6 years, 63% were male, 78% were black, and the majority had a diagnosis of schizophrenia (74%). The mean baseline PEC total score was 17 and similar across groups. This baseline score reflected an agitation severity of moderate. Expected behaviors seen moderate agitation would include excitement, hostility, poor impulse control, tension, and uncooperativeness. 13
Overall completion rate for this study was 97.9% (N = 372). A significant improvement in agitation was seen for both doses compared with placebo at 2 hours as reflected by a decrease in mean PEC total score from baseline. Least square (LS) mean change from baseline in PEC total score at 2 hours was −10.3 ± 0.4 with 180 μg, −8.5 ± 0.4 with 120 μg, and −4.8 ± 0.4 with placebo. For both treatment groups, the average decrease in PEC total score was clinically meaningful as severity of agitation decreased from moderate to mild. Although agitation decreased in the placebo group, the severity remained moderate. LS mean differences were significantly different from placebo at the 2 hours for both doses (P < 0.001). An improvement in agitation was observed as early as 20 minutes with 180 μg and 30 minutes with 120 μg. The response rate with sublingual dexmedetomidine 180 μg was significantly greater as compared with placebo (34.4% vs 13.5%; P = 0.0001) at 20 minutes. At 2 hours, response rates were found to be 88.8% (P < 0.0001) with 180 μg, 79.1% (P < 0.0001) for 120 μg, and 40.5% with placebo. No results were presented for participants requiring >1 dose of study drug for uncontrolled agitation.
This study concluded that dexmedetomidine sublingual film 180 and 120 μg effectively reduced agitation in adults with schizophrenia and schizophrenia-like disorders. The drug was also found to be safe and well tolerated.
Phase 3: Dexmedetomidine in the Treatment of Agitation Associated With Bipolar Disorder (SERENITY II) 21
SERENITY II study evaluated the efficacy and safety of dexmedetomidine sublingual film compared with placebo in patients with acute agitation associated with bipolar disorders. 19 The methodology of this study was identical to the SERENITY 1 study with the exception of the study population. Study participants were required to meet DSM-5 diagnostic criteria for bipolar I or II disorder (mania, mixed episode, or depressive episode) and meet the standard inclusion criteria for agitation stated previously.
A total of 378 patients were enrolled in the trial and randomly assigned to the following groups: dexmedetomidine sublingual film 180 μg (N = 127), 120 μg (N = 127), or matching placebo (N = 126). Baseline demographics were similar between groups with the exception of current agitation episode duration. Duration was longest in the 180 μg group and shortest in the placebo group (25.1 vs. 15.7 days, respectively). The mean (SD) age of the study population was 45.6 (11.6) years, and more participants self-identified as female (54.8%) and black (56.1%). Randomization was stratified for age (<65 years and ≥65 years), but only 8 participants were >65 years. Almost half of the participants were experiencing a manic episode (47.6%), while 20.9% were diagnosed with a mixed episode, 19.6% with a depressed episode, and the remaining were experiencing a hypomanic or unspecific episode associated with bipolar disorder. Baseline mean PEC total score was 18, reflecting moderate agitation, and similar across groups. Concomitant medication use was common in all groups (78.3%), and this included both psychotropic and non-psychotropic medications.
Overall completion rate for this study was 95.3% (N = 362). The majority of participants received only 1 dose of medication during the study. A second dose for persistent agitation was required for 10.3%, 23.8% and 46.0% in the 180 μg, 120 μg and placebo groups, respectively. Rescue medication with lorazepam was needed for resistant agitation in 2.4% of the 180 μg group and 1.6% in both the 120 μg and placebo groups. Dexmedetomidine sublingual film demonstrated improvement on the primary endpoint of change in mean PEC total scores at 2 hours compared with placebo. These scores decreased by 10.4 points with 180 μg, 9.0 points for 120 μg and 4.9 points for placebo. Similar to SERENITY 1, the average decrease in PEC total score was clinically meaningful in both treatment groups as severity of agitation decreased from moderate to mild. Agitation also decreased with placebo but severity remained moderate. LS mean differences were significantly different from placebo at 2 hours for both doses (P < 0.001). A significant reduction in agitation with both doses of study drug as compared with placebo was seen at 20 minutes as demonstrated by LS mean difference for 180 μg, −1.1 (97.5% confidence interval [CI], −2.0 to −0.2; P = 0.007) and for 120 μg, −1.0 (97.5% CI, −1.9 to −0.1; P = 0.009). The response rate with sublingual dexmedetomidine 180 μg was significantly greater as compared with placebo (23% vs 12.7%; P = 0.048) at 20 minutes. At 2 hours, response rates were 90.5% and 77.0% with dexmedetomidine sublingual film 180 and 120 μg compared with 46.0% with placebo (P < 0.001).
This study concluded that a single dose of dexmedetomidine sublingual film (180 and 120 μg) significantly reduced the severity of agitation in adults with acute agitation associated with bipolar I or II disorder and was well tolerated.
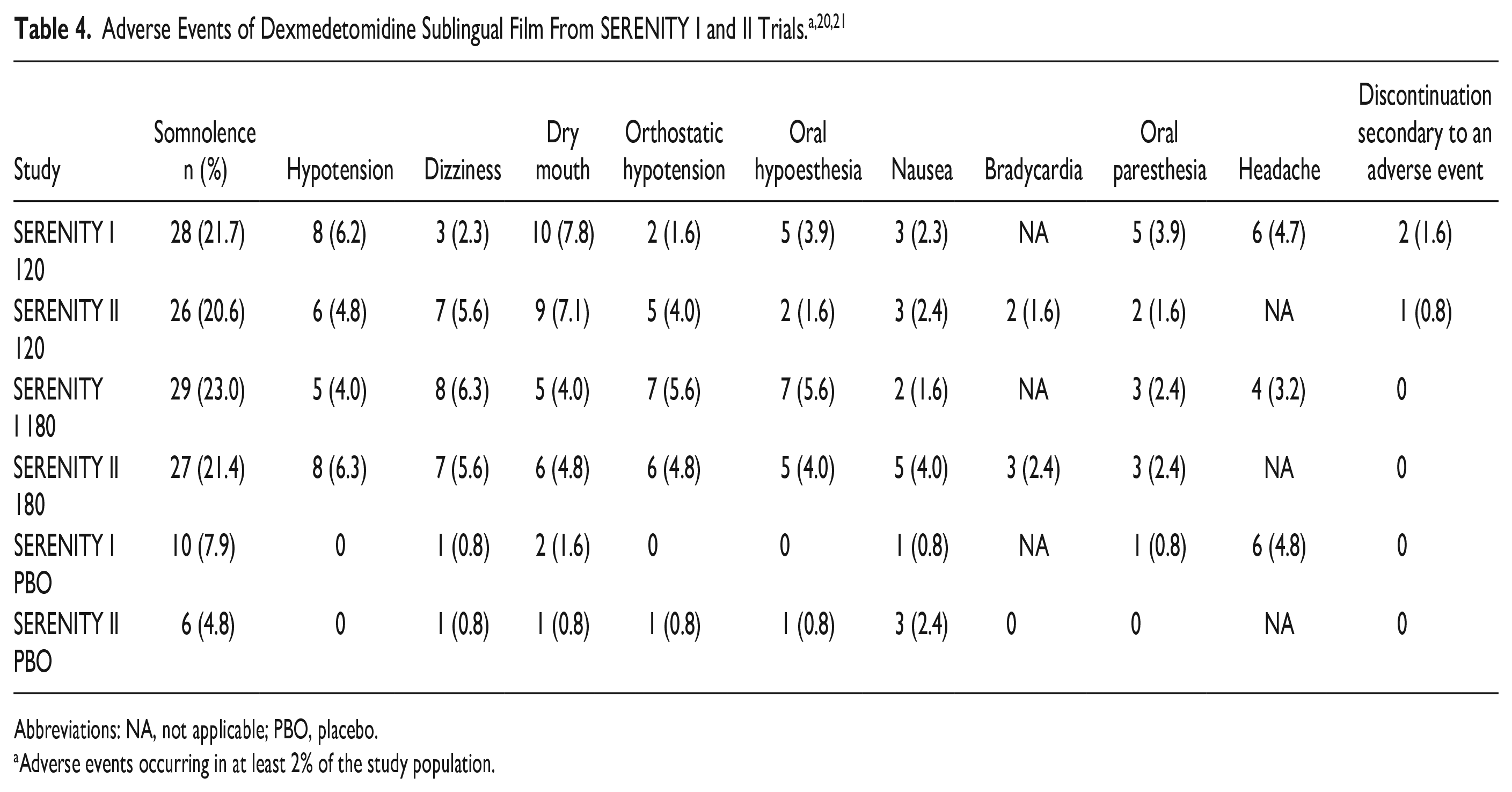
Cardiovascular Tolerability in SERENITY I and I Trials
In SERENITY 1, hypotension occurred in 4.0% of the 180 μg group and 6.2% in the 120 μg group while orthostatic hypotension occurred in 5.6% and 1.6%, respectively with none of these being classified as severe. 20 No abnormal ECG results were reported. In SERENITY II, hypotension occurred at rate of at least twice the rate of placebo (120 μg, 4.8% and 180 μg, 6.3%). 21 At the 2-hour time point, participants taking dexmedetomidine sublingual film 120 μg experienced a mean (SD) decrease of −14.81 mm Hg (15.6) in systolic blood pressure (SBP), −8.8 mm Hg (11.0) in diastolic blood pressure (DBP), and −7.9 bpm (13.0) in heart rate (HR). These measurements were obtained at baseline and after 1 minute of standing. Participants receiving the 180 μg dose had a mean (SD) decrease in SBP of −18.1 mm Hg (16.3), −11.5 mm Hg (11.1) in DBP, and −9.2 bpm (11.7) in HR. The incidence of bradycardia was low in both study drug groups (2.4%, 180 μg and 1.6%, 120 μg) but was considered to be clinically meaningful in at least half of the cases. No abnormal ECG results were reported.
Safety and Tolerability in SERENITY I and II
Dexmedetomidine sublingual film was well tolerated in both patient populations as there were no study discontinuations due to adverse events in the 180 μg and placebo groups and only 1.2% in the 120 μg group in the post hoc analysis of both SERENITY studies. 23 No reports of severe or serious adverse events were reported in either study.20,21 The overall incidence of adverse events in SERENITY I was 39.5%, 37.3%, and 15.1% in the 180 μg, 120 μg, and placebo groups, respectively. 20 Somnolence was the most common adverse effect reported in all groups with majority rating it as mild. Table 4 describes adverse effect data for both SERENITY I and II trials. No clinically meaningful changes in laboratory values were reported. Patient-reported medication acceptability (eg, flavor, no unpleasant aroma, or aftertaste) was approximately 90% for both groups. The overall incidence of adverse events in the SERENITY II trial was 35.7%, 34.9%, and 17.5% in the dexmedetomidine sublingual film 180 μg, 120 μg, and placebo group, respectively. 21 Similar to SERENITY I, somnolence was the most common adverse effect reported in all groups. It was rated as mild in 64% of cases and moderate in the remaining cases. Patient-reported medication acceptability (eg, flavor, no unpleasant aroma, or aftertaste) was 80.2% and 79.3% with dexmedetomidine sublingual film 180 μg and 120 μg, respectively. The post hoc analysis found that adverse effects did not appear to be clearly dose related except for the possibility of orthostatic hypotension (5.2%, 180 μg; 2.7%, 120 μg; 0.4%, placebo). In addition, unlike antipsychotic medications, dexmedetomidine has not been associated with the development or worsening of extrapyramidal symptoms.
Abbreviations: NA, not applicable; PBO, placebo.
Adverse events occurring in at least 2% of the study population.
Other Studies
A post hoc analysis of both SERENITY studies was performed by Citrome et al 24 using the metrics of number needed to treat (NNT) and number needed to harm (NNH) to provide clinically meaningful estimates of the efficacy and tolerability of dexmedetomidine sublingual film use in adults with acute agitation associated with schizophrenia or bipolar disorders. By combining data from both studies (N = 506), the NNT for response at 2 hours post dose for either 120 or 180 μg was found to be 3 (95% CI: 3-3). For participants with schizophrenia or bipolar disorders, NNH was >10 for both doses of dexmedetomidine sublingual film for all acute adverse effects of special interest (cardiac/vascular disorder, hypotension/bradycardia, bradycardia alone, hypotension alone). Somnolence was the most common adverse effect in both patient populations, and the NNH was 8 (120 μg, 95% CI: 5-22 and 180 μg, 95% CI: 5-23) for both doses in schizophrenia, and 7 (95% CI: 5-13) for 120 μg and 6 (95% CI: 5-12) for 180 μg in bipolar disorders. For other adverse effects including dry mouth, dizziness, oral hypoesthesia and paresthesia, headache, nausea, and orthostatic hypotension, the NNH was 17 to 33 for schizophrenia and 18 to 26 for bipolar disorders.
The authors concluded that dexmedetomidine sublingual film is appropriate for use in the treatment of adults with acute agitation associated with schizophrenia or bipolar disorders based on the following evidence: (1) NNT values were consistent and associated with a significant beneficial effect, (2) NNH values indicated the drug was reasonably well tolerated, and (3) therapeutic response was encountered more frequently than adverse effects.
Ongoing Research for Additional Indications
Dexmedetomidine sublingual film is being studied for the treatment of agitation associated with dementia, agitation associated with delirium, agitation in pediatric patients with schizophrenia and bipolar disorders, and in alcohol use disorder with comorbid posttraumatic stress disorder. 25 Clinical trials are in various stages of completeness, with no data yet available.
Precautions and Contraindications
There are no reported contraindications for the use of dexmedetomidine sublingual film. Use of the drug should be avoided in patients with known hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope due to reported dose-dependent effect of hypotension, orthostatic hypotension, and bradycardia. 2 To decrease the risk of cardiac issues from occurring after administration of the drug, patients should be adequately hydrated and should sit or lie down until vital signs are within normal range. 2 The drug should also be avoided in patients at risk of torsades de pointes or sudden death including those with known QT prolongation, history of other arrhythmias, symptomatic bradycardia, hypokalemia, hypomagnesemia, and in patients receiving other drugs known to prolong the QT interval. Dexmedetomidine has been shown to exhibit concentration-dependent QT prolongation with a mean corrected QT interval by Fredericia (QTcF) increase from baseline of 6 milliseconds (upper 90% CI: 7) for 120 μg single dose; 8 milliseconds (upper 90% CI: 9) for 120 μg plus 2 additional doses of 60 μg at 2 hours apart; 8 milliseconds (upper 90% CI: 11) for 180 μg single dose; and 11 milliseconds (upper 90% CI: 14) for 180 μg plus 2 additional doses of 90 μg at 2 hours apart. 2 Due to the risk for somnolence, patients should not perform activities requiring mental alertness for at least 8 hours after ingestion. Withdrawal reactions would be unlikely, but the drug was not studied for durations longer than 24 hours in the pivotal trials. 2
Drug Interactions
There is no evidence of any CYP-mediated drug interactions that are likely to be of clinical relevance. 2 Due to pharmacodynamic properties, drugs that prolong the QT interval and drugs that increase central nervous system (CNS) depressant effects should be avoided or use of a reduced dose of dexmedetomidine sublingual film should be considered. 2 Dexmedetomidine given concomitantly with drugs that prolong the QT interval may add to the QT-prolonging effects and increase the risk of cardiac arrhythmias. As some psychotropic medications are known to prolong the QT interval, a medication history would be recommended prior to using dexmedetomidine sublingual film. Dexmedetomidine sublingual film given concomitantly with drugs that increase CNS depressant effects, such as anesthetics, sedatives, hypnotics, or opioids, can further enhance CNS depressant effects and may worsen sedation.
Cost and Availability
The cost of a package of 10 or 30 dexmedetomidine sublingual films of either strengths of 120 or 180 μg is $126.00 per dose. 26 Currently, there are several other oral therapies used for the treatment of acute agitation associated with schizophrenia and bipolar I or II disorder that are available in inexpensive generic formulations, but none have an approved indication of acute agitation. 13
Relevance to Patient Care and Clinical Practice
Dexmedetomidine sublingual film is a new and novel option for clinicians to use in emergent situations to manage acute agitation associated with schizophrenia or bipolar I or II disorder. Studies demonstrated a significant reduction in acute agitation as compared with placebo with a treatment effect seen as early as 20 minutes. Combined outcomes and safety data suggest similar effects for both patient populations, schizophrenia and bipolar disorder, for dexmedetomidine sublingual film 120 and 180 μg. 24 It is important to note that patient cooperation with self-administration of dexmedetomidine sublingual film is required and should be taken into consideration when considering use of the medication. Given its simple and noninvasive route of administration, dexmedetomidine sublingual film use in acute agitation has the potential to improve both the overall patient experience, and cooperation with health care professionals.
Administration issues of importance associated with dexmedetomidine sublingual film that would require health care professional oversight include ensuring the patient does not consume any food or drink for at least 15 minutes following sublingual use and at least 1 hour following buccal use. Based on this restriction, sublingual route of administration would be preferred. Sublingual route of administration would also be preferred due to decreased time to complete absorption compared with buccal administration and need to monitor for “cheeking” of medication. Hypotension, bradycardia, and somnolence were seen in clinical trials so there is also a need to monitor vital signs and alertness after administration to prevent syncope and falls. These monitoring tasks are associated with a time commitment on behalf of the health care professional and may be difficult to perform especially if staffing is low. Repeat doses of dexmedetomidine sublingual film must be given at least 2 hours apart so if a patient required medication for agitation prior to this time point, an alternative agent would need to be used. In addition, the utility of dexmedetomidine orally dissolving film for management of agitation in the outpatient setting is unclear and will be limited due to these issues.
The long-term efficacy and safety of dexmedetomidine sublingual film is unknown as the studies in acute agitation associated with schizophrenia and bipolar disorder were only 24 hours in duration. Use of intravenous dexmedetomidine in clinical practice for a prolonged duration has been associated with an increased risk of adverse effects (hypotension, bradycardia, tachyphylaxis) and withdrawal reactions (nausea, vomiting, tachycardia, hypertension, agitation). 2 There is a need for efficacy and safety data in patients receiving dexmedetomidine sublingual film for >24 hours as acute agitation may be present in this patient population for an extended period of time (ie, several days) until the psychosis or mania responds to primary drug therapy.
Due to the high cost of dexmedetomidine sublingual film, it is likely clinicians will use this agent for agitation unresponsive to first-line therapy with antipsychotics and/or benzodiazepines. The timing of the last dose of antipsychotic and/or benzodiazepine prior to administration of dexmedetomidine sublingual film will be important due to an increased risk of adverse effects. Hypotension, orthostatic hypotension, bradycardia, and QTc prolongation would be the concern with dexmedetomidine sublingual film and antipsychotics while oversedation would be the issue with benzodiazepines. An advantage to using dexmedetomidine sublingual film over repeated doses of antipsychotics in resistant agitation would be that there are no extrapyramidal symptoms (eg, acute dystonia, akathisia) associated with dexmedetomidine sublingual film due to its lack of dopaminergic antagonism activity. There are no recommendations regarding specific separation of timing with antipsychotics and/or benzodiazepines and dexmedetomidine sublingual film in the product labeling. For the SERENITY trials, patients were excluded if they had treatment with an alpha-1 adrenergic antagonist, benzodiazepines, hypnotics, or antipsychotic drugs 4 hours prior to the study drug. 2
Active comparator studies including evidence-based treatments for acute agitation are needed to delineate dexmedetomidine sublingual film’s role relative to other options. Oral antipsychotics possessing the most evidence for use and recommended in treatment guidelines include haloperidol, aripiprazole, asenapine, risperidone, quetiapine, and olanzapine, with preference given to second-generation antipsychotics over haloperidol. 13 Lorazepam is the benzodiazepine possessing the most evidence for use and recommended in treatment guidelines. 13 All of the medications are available in less expensive generic formulations. Onset of efficacy with oral formulations of antipsychotics and benzodiazepines appears comparable to dexmedetomidine sublingual film. 15 The goal of pharmacologic therapy in the management of acute agitation in the psychiatric patient population is to achieve a calming effect without oversedation in a timely manner. Dexmedetomidine sublingual film meets these criteria, but its use in clinical practice may be limited due to its cost and the availability of less expensive treatment options.
Conclusion
The use of dexmedetomidine sublingual film in adults for the treatment of acute, mild to moderate agitation associated with schizophrenia or bipolar I or II disorder has robust evidence supporting its efficacy and appears to be safe and well tolerated in healthy adults. Dexmedetomidine sublingual film is a novel formulation of a preexisting drug that is self-administered with medical supervision. It represents an advancement in the treatment of acute agitation as it demonstrates rapid absorption and a fast onset of efficacy while avoiding the intramuscular route of administration. Unfortunately, the widespread use of this treatment option may be limited due to its cost. With multiple studies currently being conducted with dexmedetomidine sublingual film to address additional unmet needs in mental health, the future of this drug is evolving and may be expanding.
Supplemental Material
sj-docx-1-aop-10.1177_10600280231171179 – Supplemental material for Dexmedetomidine Sublingual Film: A New Treatment to Reduce Agitation in Schizophrenia and Bipolar Disorders
Supplemental material, sj-docx-1-aop-10.1177_10600280231171179 for Dexmedetomidine Sublingual Film: A New Treatment to Reduce Agitation in Schizophrenia and Bipolar Disorders by Danielle M. Karlin, Leigh Anne Nelson and Austin R. Campbell in Annals of Pharmacotherapy
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Leigh Anne Nelson is on the Speaker’s Bureau for Alkermes, Inc, and has received research grants from Boehringer Ingelheim, Alkermes, Inc, and Janssen Scientific Affairs, LLC. Austin R. Campbell is on the Speaker’s Bureau for Sunovion Pharmaceuticals, Inc, a paid coauthor for UpToDate, and has received grant support from Intra-Cellular Therapies and Otsuka Pharmaceutical CO Ltd.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.