Abstract
Objective:
To determine whether implementation of antimicrobial stewardship (AMS) interventions improve management of bacteriuria in hospitalized adults.
Data Sources:
EMBASE, MEDLINE, CINAHL, and Clinical Trials Registries via Cochrane CENTRAL were searched from inception through May 2021. Reference lists of included studies were searched, and Scopus was used to retrieve articles that cited included references.
Study Selection and Data Extraction:
Randomized and nonrandomized trials, controlled before-after studies, interrupted time-series studies, and repeated measures studies evaluating AMS interventions for hospitalized adult inpatients with bacteriuria were included. Risk of bias was assessed independently by 3 team members and compared. Results were summarized descriptively.
Data Synthesis:
The search yielded 5509 articles, of which 13 met inclusion criteria. Most common interventions included education (N = 8) and audit and feedback (N = 5) alone or in combination with other interventions. Where assessed, resource and antimicrobial use primarily decreased and appropriateness of antimicrobial use improved; however, impact on guideline adherence was variable. All studies were rated as having unclear or serious risk of bias. This review summarizes and assesses the quality of evidence for AMS interventions to improve the management of bacteriuria. Results provide guidance to both AMS teams and researchers aiming to develop and/or evaluate AMS interventions for management of bacteriuria.
Conclusions:
This review demonstrated benefit of AMS interventions on management of bacteriuria. However, most studies had some risk of bias, and an overall effect across studies is unclear due to heterogeneity in outcome measures.
Introduction and Objectives
Bacteriuria is a common occurrence in many populations. Bacteriuria is defined as the presence of bacteria in the urine in quantitative counts ≥105 colony-forming units/mL. 1 Individuals with bacteriuria may develop symptoms of urinary tract infection (UTI); however, many individuals remain asymptomatic. While treatment with antimicrobial agents is recommended for UTIs, current guidelines recommend against antimicrobial therapy for asymptomatic bacteriuria (ASB) except in specific populations, including pregnant individuals and those undergoing endoscopic urologic procedures associated with mucosal trauma.1,2 The Choosing Wisely® and Choosing Wisely Canada® campaigns also recommend against treating bacteriuria unless symptoms are present.3,4 Despite clear evidence-based recommendations for managing individuals with bacteriuria, suboptimal treatment for UTIs and inappropriate prescribing for ASB have been widely reported in the literature.5-7
Unnecessary use of antimicrobial agents is considered a major driver of antimicrobial resistance, 8 one of the greatest global public health challenges today. 9 In particular, high rates of resistance have been observed in bacteria that cause UTIs. 10 In hospitalized patients, UTIs are one of the most common reasons for prescribing antibiotics, accounting for up to 17% of antimicrobial use.11,12 Detection of bacteriuria has been shown to lead to unnecessary therapy in 30% to 50% of ASB patients.13,14 and treatment of ASB has been found to account for half of all unnecessary fluoroquinolone regimens in hospitalized patients. 15 To minimize the negative consequences of antimicrobial use, including antimicrobial resistance, antibiotics must be used judiciously. 16
As a result of growing rates of antimicrobial resistance, antimicrobial stewardship (AMS) initiatives are recommended to improve the use of antibiotics.17,18 Antimicrobial stewardship is defined as “coordinated interventions designed to improve and measure the appropriate use of [antibiotic] agents by promoting the selection of the optimal [antibiotic] drug regimen including dosing, duration of therapy, and route of administration.” 17 A Cochrane review of AMS interventions has shown improved compliance with antibiotic policy and reduced duration of antimicrobial use. 19 The review however, did not focus on AMS interventions specifically for patients with bacteriuria. We aimed to determine whether implementation of AMS interventions improves management of bacteriuria (UTIs and ASB) in hospitalized adults, as compared to usual care.
Methods
This systematic review was completed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) methods 20 (http://www.prisma-statement.org/). The protocol was registered with PROSPERO 2020 CRD42020159051. 21
Data Sources and Search Strategy
We systematically searched EMBASE (Elsevier), MEDLINE (Ovid), and Cumulative Index to Nursing and Allied Health Literature (CINAHL, EBSCO). ClinicalTrials.gov and World Health Organization (WHO)’s International Clinical Trials Registry Platform were both searched through the Cochrane Central Register of Controlled Trials (CENTRAL). In keeping with the Cochrane Effective Practice and Organization of Care (EPOC) recommendations, 22 eligible study types were randomized and nonrandomized trials, controlled before-after studies, interrupted time series (ITS) studies, and repeated measures studies. An EPOC MEDLINE method filter was used in the search process to limit the results to the eligible study types.22,23 Reference lists from the 13 included studies were reviewed to identify additional studies and Scopus was searched to retrieve any citing articles of these included studies. The search was developed and performed by one of our authors (MH) who is a librarian. The search was peer reviewed by a second librarian.
The searches were conducted on July 12, 2019 and updated on May 29, 2021. Search results were imported into Covidence® software and duplicates were identified and removed. Our search appendix and search data are available at https://doi.org/10.5683/SP3/D9XYSU.
Study Selection and Data Extraction
Two authors or research assistants (MH, HM, EB, HN, TR, and/or MS) independently reviewed the title/abstract of all eligible studies and completed full-text review of all included studies in Covidence software. A third author (EB or HN) resolved disagreements independently. Studies were included if they (1) described an AMS intervention delivered by any healthcare provider to adult inpatients 18 years of age and older with symptomatic UTIs and/or ASB and (2) reported the impact of the intervention. Studies that were exclusively completed in outpatient settings, long-term care homes, intensive care units, and emergency departments were excluded. Studies published in languages other than English were excluded.
Data were extracted from included studies by a member of the research team (GM, MH, and HM) using a standardized data extraction tool adapted from the EPOC guidelines. 22 All data extracted were reviewed by a second member of the research team. Study data were extracted as well as detailed information on interventions, which were compared to other interventions or usual care.
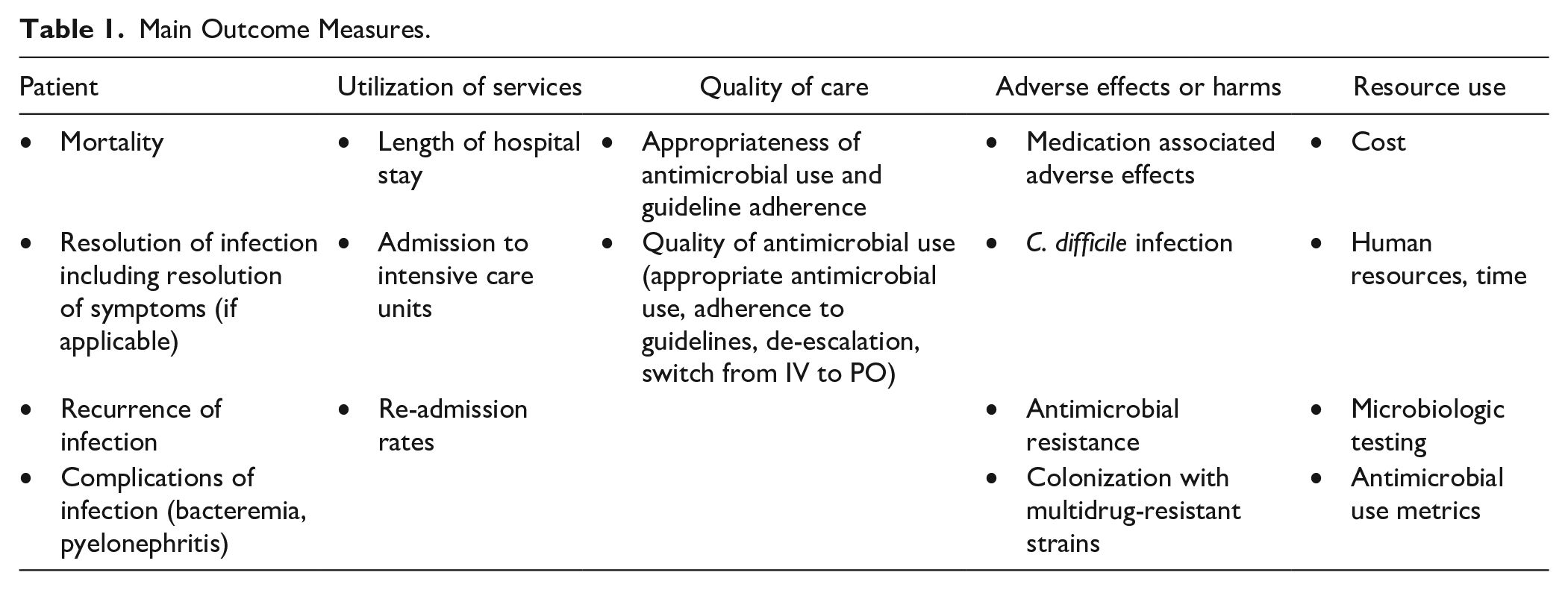
We classified intervention components according to the EPOC taxonomy. 22 Intervention components were also categorized as outlined by Davey et al 19 (education, persuasion, enablement, restriction, and environmental restructuring). Outcomes of interest as recommended by EPOC included any patient outcomes (including health behavior[s] and health status), utilization of services, quality of care, adverse effects or harms, and resource use outcomes. 22 Main outcome measures are listed in Table 1.
Main Outcome Measures.
Risk of bias was assessed independently by 3 members of the research team (EB, HN, and GM or MH) using the Cochrane risk of bias (RoB 2) assessment tool for randomized trials 24 and the risk of bias in nonrandomized studies of interventions (ROBINS-I) tool. 25 Disagreements were resolved by consensus. We had intended to complete a meta-analysis on main outcome measures; however, there were many differences in the populations, interventions, comparisons, and methods. As a result of heterogeneity, we conducted a descriptive synthesis of the findings.
Results
Data Synthesis
A total of 3539 studies were screened in July 2019 by titles and abstracts after duplicates were removed. In May 2020, 144 references of included studies were reviewed, and Scopus was used to identify, 143 articles that had cited the included studies after duplicates were removed. As well a total of 142 Clinical Trials were screened by titles and abstracts after duplicates were removed. The literature search including clinical trials search was updated in May 2021 and an additional 1017 studies were screened after duplicates were removed. Full-texts were reviewed for 157 studies and 13 studies met inclusion criteria (Figure 1) and were included in the analysis.
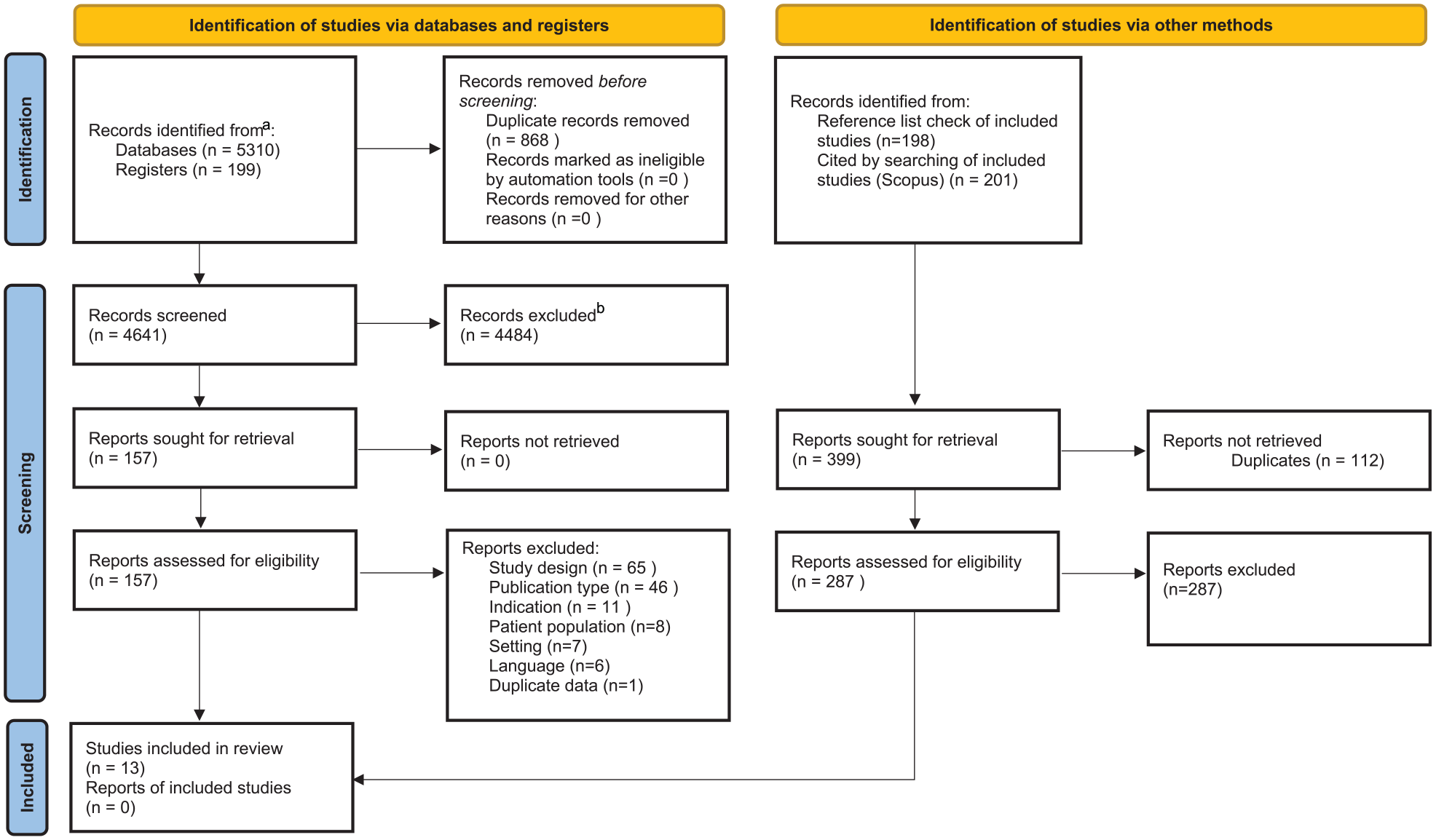
PRISMA 2020 flow diagram for new systematic reviews which included searches of databases, registers, and other sources.
A summary of the included studies is provided in Table 2. There were 10 nonrandomised studies (NRSs),26-35 2 randomized controlled trials (RCTs),36,37 and 1 randomized trial with no control. 38 Of the NRS, 6 were controlled studies,26-30,35 3 were ITS analyses,31,32,34 and 1 was a before-and-after study with a secondary analysis of main outcomes by ITS analysis. 33 All studies were completed in high-income countries as defined by the World Bank Atlas method. 39 Seven were carried out in the United States,27,30-34,37 3 in Canada,28,29,36 1 in France, 26 1 in the Netherlands, 38 and 1 in New Zealand. 35 Most were undertaken at tertiary and/or teaching hospitals (N = 10)26-32,34,36,37 or a combination of teaching and nonteaching hospitals (N = 3).33,35,38 Two studies included hospitalized and nonhospitalized individuals.30,38
Comprehensive Summary of Results.

Quality of care: Appropriateness of antimicrobial use, guideline adherence. Patient outcomes: Mortality, resolution of infection (including resolution of symptoms, if applicable), complications of infection, recurrence of infection, bacteremia. Utilization of services: Length of hospital stay, admission to intensive care units, re-admission rates. Adverse effects or harms: Medication associated adverse effects, antimicrobial resistance, colonization with multidrug resistant strains, C. difficile infection. Resource use: Cost, human resources, time, microbiologic testing, antimicrobial use metrics.
Abbreviations: ASB, asymptomatic bacteriuria; ASP, Antimicrobial stewardship physician; B, baseline; C, control; I, intervention; M, maintenance; FU, follow-up; CAUTI, catheter-associated urinary tract infection; CDI, C. difficile infection; CDS, computer decision support; CFU, colony-forming units; DDD, defined daily doses; IDSA, Infectious Diseases Society of America; IRR, incidence rate ratios; ITS, interrupted time series; ITT, intention-to-treat; LOS, length of stay; QI, quality indicator; RCT, randomized controlled trial; RR, rate ratio; UTI, urinary tract infection; WISCA, weighted incidence syndromic combination antibiogram; NR, not reported.
Results of the intervention for patients with skin and soft tissue infections reported separately and not included in this review.
All of the NRS were rated as having serious risk of bias26-29,31,33-35or lacked sufficient detail to assess.30-32 Of the 3 randomized trials, 2 were considered to have some concern36,38 and 1 RCT had a high risk of bias 37 (Table 2). Common reasons for a rating of “some concern” in a bias domain included lack of blinding (both recipients and individuals delivering the interventions) or no information on prespecified analysis plan of data. Ridgeway et al 37 was assessed as having a high risk of bias due to baseline differences between intervention groups, suggestive of a problem with randomization.
Intervention types
Many studies assessed multifaceted interventions.29,30,32-34,37,38 The most common intervention described was provider education alone or in combination with other interventions (N = 8)26,27,29,30,32,34,38 followed by audit and feedback (N = 4).29,30,24,28 A fifth study by Ridgeway et al 37 also described a feedback intervention that did not meet the EPOC definition of “a summary of health workers’ performance over a specified period of time.” The intervention in this study was educational outreach consisting of review and recommended changes. Jenkins et al 30 also suggested participating study sites use audit and feedback as one strategy to promote uptake of implemented guidelines; however, the number of sites that used this strategy is unknown. Details of all identified interventions targeted at healthcare workers classified using EPOC taxonomy subcategories are outlined in Table 2.
Outcomes
Quality of care
Six studies assessed quality-of-care outcomes as defined by appropriateness of antimicrobial use26,28-30,36,38 or local guideline adherence 35 as a primary outcome. The studies used a variety of interventions; most used education either alone 26 or combined with audit and feedback.29,30,38 Six studies demonstrated a statistically significant change in target AMS practice compared to baseline or usual care after intervention delivery, including decrease in inappropriate antibiotic therapy26,28-30,36 or improvement in overall quality indicator sum score. 37 Spoorenberg et al 38 was the only study to also compare the effectiveness of 2 strategies: they compared a multifaceted strategy including feedback, education, and reminders to a less time-consuming competitive audit and feedback strategy that included individual feedback with nonanonymous ranking of departments and found no difference between the 2 arms in quality of care measures. Only 2 studies assessed the effectiveness of a modified reporting intervention on appropriateness of antimicrobial use. These studies assessed modified reporting of positive urine cultures and showed statistically significant improvement of appropriate treatment of ASB compared to control.28,35 One study that assessed implementation of an electronic phone app failed to show improvement in adherence to guidelines compared to usual care. 35
Resource use and utilization of services
Resource use was reported frequently, with 9 studies assessing the impact of AMS interventions on resource use29-34,36,38 or utilization of services33,37 as a primary or secondary outcome. Most of these studies were multifaceted and included education with audit and feedback, reminders, clinical decision support, or guideline implementation.29,30,32-34,37,38 Other studies assessed formulary restriction 31 and modified reporting of urine cultures.28,36 The impact of these interventions on resource use primarily resulted in less microbiologic testing29,30,32 and a decline in antibiotic utilization.28,33,34 Cost savings of modified culture reporting was assessed by Daley et al 36 who found no significant benefit. One study assessed the impact of electronic decision support on hospital length of stay and found no significant decrease compared to control groups. 37
Adverse effects or harms
One study reported adverse effects or harms as outcomes. 31 O’Brien et al 31 evaluated restriction of fluoroquinolones and reported a decline in antimicrobial resistance after the intervention was implemented, with the proportion of E. coli isolates nonsusceptible to ciprofloxacin decreasing from 41.5% to 32.8%, and there were no other infection control policies or procedures that may have impacted resistance patterns implemented concurrently.
Patient Outcomes
Three studies reported patient outcomes. Daley et al 36 reported a similar rate of bacteremia between intervention and control groups when modified urine culture reporting was implemented. Leis et al 28 also reported no patients with clinical signs of a UTI or sepsis with modified urine culture reporting. Bacteremia, sepsis, and clinical signs of a UTI were categorized as a patient outcome, but could equally be considered an adverse effect or harm as the negative consequence of an AMS intervention. As well. Ridgway et al 37 assessed the odds of 30-day mortality as a secondary outcome. They implemented an electronic clinical decision support tool and educational outreach through review of individual patients and recommendations for change. This intervention did not result in a statistically significant decrease in mortality.
Interpretation of the Data
This systematic review is the first to summarize the results of AMS interventions that specifically aimed to improve management of bacteriuria in adults admitted to hospital. Most of the included studies implemented interventions that demonstrated improved quality of care and/or a decrease in use of antimicrobials and microbiologic testing.
Our findings are consistent with a large Cochrane review by Davey et al 19 that evaluated AMS interventions for all infectious syndromes and found many restrictive and enabling interventions were successful in reducing unnecessary antibiotic use in hospitals. Davey et al 19 highlighted that while both enablement and restriction were independently associated with increased effectiveness of interventions, including an enabling component further enhanced the effect of restrictive interventions. Interventions that showed the greatest benefit in the Cochrane review were those that included the addition of feedback to enabling interventions. 19
A systematic review of pharmacist-led education-based AMS interventions also reported consistent improvement in antimicrobial use. Forty-five of 52 studies in their review included an educational intervention. The authors additionally reported that combined interventions, particularly with audit and feedback, were more effective than single educational interventions. 40 Consistent with these findings, 5 studies in our review reported use of some form of audit and feedback as a component of multifaceted interventions. Where reported, this intervention led to improved quality of care and decreased antimicrobial use.29,30,34,38
Strengths and Limitations
Our review has several strengths that should be considered. We completed a systematic review using a standardized approach as outlined by EPOC 22 and PRISMA. 20 This systematic review was authored by an interdisciplinary team of clinicians (pharmacists and a physician) with infectious disease expertise and researchers with experience performing systematic reviews. Our search was designed by members of the team with expertise in library services and was peer reviewed. In addition, risk of bias was assessed independently by 3 members of our research team.
Despite these strengths, several limitations should be noted. While we completed a comprehensive search, for practical reasons we only included studies published in English and relevant studies in other languages may have been missed. Only 3 randomized trials met inclusion criteria. In additional, the quality of included studies was generally low, with all NRS at serious risk of bias or receiving a rating of no information. This is, however, consistent with the quality of evidence in the general AMS literature. A systematic review by Schweitzer and colleagues from 2019 reported generally low quality of evidence for studies evaluating AMS interventions, which did not change over time. 41 Finally, differences in context, patient population, study design, interventions implemented, and outcome measures made it difficult to compare the effectiveness of interventions across studies, such that we were unable to perform a meta-analysis to identify which intervention(s) are more effective.
Generalizability is also limited as all studies were completed in high-income countries primarily in North America. Furthermore, studies were mainly completed at teaching or tertiary care hospitals and only 3 studies included community hospitals.33,35,38 Where described, most sites had a formal AMS team or pharmacists with infectious diseases and/or microbiology expertise. An established network of clinicians delivering AMS may influence acceptability or practicability of implementing interventions at other institutions.
Relevance to Patient Care and Clinical Practice
This review adds to the literature that is focused on delivery of AMS interventions for individuals with bacteriuria. When designing interventions for inpatients with bacteriuria, institutions may consider implementation of an intervention that includes audit and feedback in combination with education or other stewardship strategies. Where assessed in this systematic review, studies that incorporated audit and feedback into their intervention consistently demonstrated improved antimicrobial use. Further evidence is needed to determine the best format for audit and feedback; however, limited results from this study suggest a less time consuming competitive feedback strategy may result in similar benefit to anonymous feedback in conjunction with a multifaceted intervention. 37 Greatest benefit may also be observed for indications or on hospital units where baseline adherence to best practice is low. 30 For sites aiming to see more judicious use of microbiology testing, implementation of a multifaceted intervention as described by Keller and colleagues that includes education combined with clinical decision support may be considered. 32
When multifaceted interventions that are resource intensive may not be feasible, another strategy consistently resulting in positive outcomes that could be considered is modified culture reporting. As identified by Leis et al 28 and Daley et al, 36 this strategy may decrease treatment of asymptomatic bacteriuria with antibiotics.
To develop an AMS intervention at our institution, results of this review will be considered in conjunction with practical considerations and previous qualitative research that our team completed on local antimicrobial use and stewardship. Health care providers in Nova Scotia have reported audit and feedback in conjunction with other initiatives such as education as possible facilitators to improving antimicrobial use.42,43 Antimicrobial stewardship teams at other institutions may also consider this approach to develop a tailored intervention that meets local needs.
Conclusion
While findings from our review provide evidence to support some AMS interventions, future studies should consider study designs that limit the risk of bias such as randomized controlled trials or stepped wedge designs. 44 Pragmatic clinical trials and strong observational designs including studies that use ITS analysis, a control site, or repeated measures may also further contribute to the literature on effectiveness of implementing AMS initiatives for hospitalized adults with bacteriuria.
Supplemental Material
sj-docx-1-aop-10.1177_10600280221134539 – Supplemental material for A Systematic Review of Antimicrobial Stewardship Interventions to Improve Management of Bacteriuria in Hospitalized Adults
Supplemental material, sj-docx-1-aop-10.1177_10600280221134539 for A Systematic Review of Antimicrobial Stewardship Interventions to Improve Management of Bacteriuria in Hospitalized Adults by Mari Humphrey, Gemma MacDonald, Heather Neville, Melissa Helwig, Tasha Ramsey, Holly MacKinnon, Ingrid Sketris, Lynn Johnston and Emily K. Black in Annals of Pharmacotherapy
Footnotes
Acknowledgements
The authors of this review would like to acknowledge Robin Parker for their peer review of the literature search. They would also like to acknowledge Morgan Sproul for assistant with title and abstract screening.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GM, MH, and HM received salary support for their work on this review from Research Nova Scotia. EB has received research funding from Research Nova Scotia, Dalhousie Pharmacy Endowment Fund, Dalhousie University Faculty of Health, and the Drug Evaluation Alliance of Nova Scotia. She has also received speaker/consulting fees from Dalhousie Continuing Pharmacy Education, Ontario Pharmacists Association, and Pear Healthcare Solutions and Prime Event Partners dba Prime Strategies Inc., a division of MGME.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by an Establishment grant from Research Nova Scotia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
