Abstract
Introduction
Septic shock remains a significant contributor to the morbidity and mortality of critically ill patients. 1 Initial management includes fluid resuscitation, antimicrobials targeted at the most likely pathogen(s), and vasoactive agents. 2 Catecholamines, specifically norepinephrine, are recommended for hemodynamic support in patients with septic shock unresponsive to adequate fluid resuscitation. 2
Norepinephrine and other catecholamines are associated with peripheral and tissue ischemia due to their potent vasoconstrictive properties and cardiac arrhythmias due to beta-adrenergic receptor agonism.3-5 New-onset arrhythmias (NOAs), particularly new-onset atrial fibrillation (NOAF), have been observed in up to 40% of septic shock patients and have been associated with poor outcomes including increased mortality, length of stay (LOS), hospital cost, hemodynamic decompensation, and long-term stroke risk.6-9 Studies of sepsis and septic shock patients have observed exposure to sepsis-related factors including higher catecholamine dose and longer catecholamine exposure were associated with increased odds of NOA development.6-16
Vasopressin is a non-catecholamine vasopressor recommended as adjunctive treatment to norepinephrine in septic shock. 2 Physiologically, vasopressin may improve hemodynamics via V1a receptor-mediated vasoconstriction, reduction of nitric oxide-mediated vasodilation, and restoration of septic shock–induced vasopressin deficiency. 17 Additional activation of vasopressin receptors in the kidney may promote glomerular filtration rate maintenance in septic shock. Clinical data support the ability of vasopressin to improve hemodynamics, reduce catecholamine exposure, and reduce the incidence of NOA.18-23
The optimal timing of vasopressin initiation in septic shock patients receiving norepinephrine remains unknown. Studies of early vasopressin mono- or adjunctive therapy have observed hemodynamic benefits and mixed results related to clinical outcomes.20,24-29 Reardon et al 24 conducted a single-center, retrospective cohort study and observed a significant association with reduced incidence of NOA in septic shock patients initiated on vasopressin within 6 hours of septic shock onset. The purpose of this study was to further assess the association of early administration of vasopressin on NOA in patients with septic shock.
Materials and Methods
Study Setting and Population
A retrospective, single-center, cohort study of adult patients with International Classification of Diseases, clinical modification (ICD)-9 or 10 diagnosis code(s) for septic shock at an urban, 1300-bed quaternary referral, academic medical center in St. Louis, MO. between January 1, 2010 to December 31, 2017 was conducted. The study was approved by the Washington University in St. Louis institutional review board (IRB ID: 20191009).
Participants, Variables, and Definitions
Patients were identified using a pharmacy informatics database query. Eligible patients were 18 years or older, diagnosed with septic shock, received vasopressin and norepinephrine for treatment of septic shock, and were admitted to the medical or surgical intensive care unit (ICU) within the study timeframe. To minimize potential confounding, patients were excluded if they received any other vasoactive or inotropic agent during their hospital stay. In addition, patients with a history of cardiac arrhythmias or admission from an outside hospital were excluded. Consistent with prior literature, patients were included in the early vasopressin group if vasopressin was initiated within 6 hours of the onset of septic shock and were included in the late vasopressin group if vasopressin was initiated more than 6 hours after the onset of septic shock. 24 Only the first episode of septic shock that met all inclusion criteria was included in the analysis. At the time of this study, there were no standardized protocols for the addition of vasopressin at our institution; however, the use of sepsis care bundles and order sets to encourage compliance with guideline-directed therapy were present throughout the study period.
Data were collected from the electronic medical record via an informatics data query and manual chart review. Information collected included patient demographics, modified Acute Pathophysiology and Chronic Health Evaluation II (mAPACHE II) score, hospital and ICU LOS, and hemodynamic and laboratory values. 30 Additional data included, but were not limited to, antibiotic administration, receipt of corticosteroids, receipt of renal replacement therapy (RRT), infection source, laboratory data surrounding NOA, NOA treatments, and time to NOA.
All study definitions were determined prospectively. Septic shock was defined as the presence of ICD-9 or 10 diagnosis code(s) consistent with septic shock (see Appendix A) and suspected or proven infection as evidenced by the presence of microbiologic cultures and receipt of empiric antimicrobial medications within 24 hours of septic shock onset. Onset of septic shock was defined as the date and time of norepinephrine initiation. Septic shock resolution was defined as the time point when both norepinephrine and vasopressin had been off for 24 consecutive hours. Other vasoactive or inotropic agents were defined as epinephrine, dopamine, phenylephrine, dobutamine, and milrinone. Culture-positive patients were considered those with positive bacterial culture results obtained within 72 hours of the onset of septic shock. Appropriate empiric antibiotics were defined as receipt of an antimicrobial(s) active against the organism(s) identified on the positive culture based on susceptibility testing performed in the institution’s microbiology laboratory and interpreted by microbiology technicians.
History of cardiac arrhythmia required patients to meet 1 of 2 conditions. First, identification of any ICD-9 or 10 diagnosis code(s) consistent with cardiac arrhythmia(s) in the 12 months prior to the hospital admission. To identify patients meeting this condition, all medical records within 12 months of admission were electronically queried and verified for the presence of prespecified ICD-9 and/or 10 diagnosis codes (see Appendix A). Second, patients administered an anti-arrhythmic or rate control agent before the onset of septic shock were considered to have a history of cardiac arrhythmia. Anti-arrhythmic or rate control agents/drug classes included adenosine, amiodarone, diltiazem, digoxin, dofetilide, esmolol, flecainide, intravenous (IV) lidocaine, metoprolol, mexiletine, procainamide, and propafenone. To be considered for NOA, patients needed to satisfy 2 criteria. First, receipt of an anti-arrhythmic or rate control agent(s) during or after septic shock onset or resolution was required. This criterion was utilized to identify clinically relevant NOAs warranting treatment. Second, any clinically relevant NOA required confirmation by a board-certified cardiologist (author B.D.G.). Confirmation was determined via interpretation of pertinent electrocardiograms (EKG), telemetry, and clinical data surrounding receipt of the anti-arrhythmic/rate control agent(s). All NOAs were classified as either supraventricular tachycardia (SVT) or ventricular tachycardia (VT). Supraventricular arrhythmias included SVT, atrial fibrillation and atrial flutter. Ventricular tachycardia included ventricular fibrillation in addition to sustained (≥30 seconds) and nonsustained VT (<30 seconds).
Study Outcomes
The primary outcome of the study was the incidence of NOA. Secondary outcomes included incidence of arrhythmia type, total number of arrhythmias, time to NOA, septic shock duration, length of ICU and hospital stay, need for new RRT, total exposure and maximum doses of norepinephrine and vasopressin, and in-hospital mortality.
Statistical Analyses
Continuous data were presented as median and interquartile range. Nonparametric testing via the Mann-Whitney U test was performed on all continuous variables. Furthermore, the Hodges-Lehmann method was utilized to estimate the median difference between groups and associated 95% confidence intervals (CI). Categorical data were expressed as n (%) and analyzed using chi-square or Fisher’s exact test. Furthermore, the absolute difference between each group was reported with associated 95% CI determined via a z test. To control for potential confounding, a multivariable logistic regression model was constructed considering covariates’ clinical plausibility, univariate analyses, prior literature, and co-linearity diagnostics to determine the odds ratio (OR) and 95% CI of predictor variables’ effects on the primary outcome. Time-to-event analyses were expressed using the Kaplan-Meier method with differences addressed via log-rank test. Missing data were not imputed. However, only variables without missing data were eligible for inclusion in the multivariable logistic regression model. A two-sided P value less than 0.05 represented statistical significance. Data were analyzed using IBM SPSS Statistics 25 for Windows (SPSS Inc., Chicago, IL).
Results
Participants
We identified 2115 eligible patients during the study period. After applying our exclusion criteria, 436 patients were included in the analysis: 220 patients in the early vasopressin group, and 216 in the late vasopressin group. The most common reason for exclusion was receipt of other vasopressors or inotropes during admission (Figure 1).
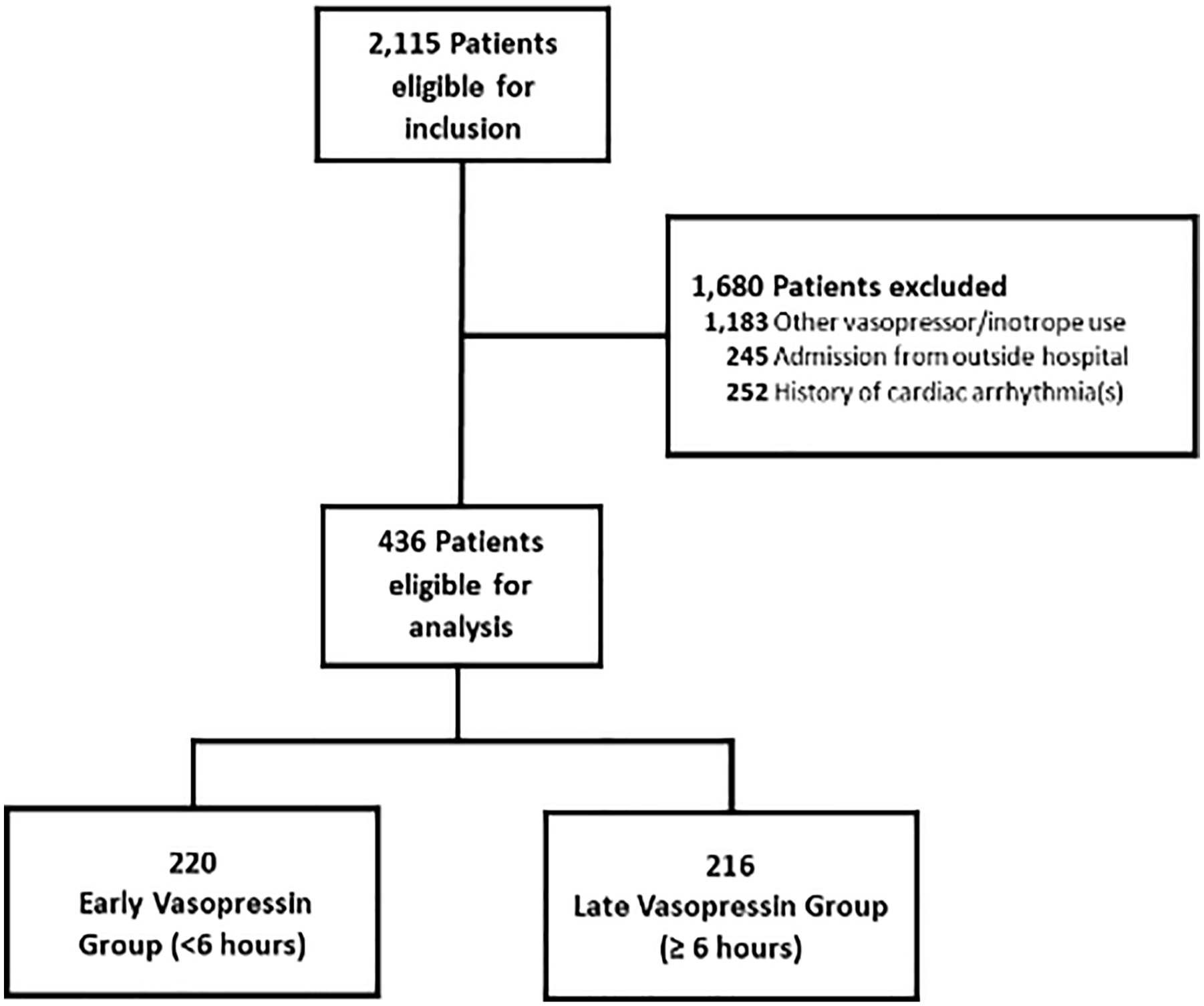
Consort diagram for cohort inclusion.
Descriptive Data
Baseline characteristics are presented in Table 1. Groups were observed to be well-matched with no significant differences observed in race, Charlson comorbidity index, mAPACHE II score, or receipt of mechanical ventilation. In addition, septic shock treatment was similar between groups with no differences observed in net fluid balance on day of shock, appropriateness of empiric antimicrobial treatment, or adjunctive corticosteroid use. No difference was observed in hemodynamic parameters at septic shock onset. Median time to vasopressin initiation was significantly shorter in the early vasopressin group compared with the late vasopressin group (2 vs 17 hours; median absolute difference [95% CI]: 15 [14, 18], P < 0.001).
Baseline Characteristics.
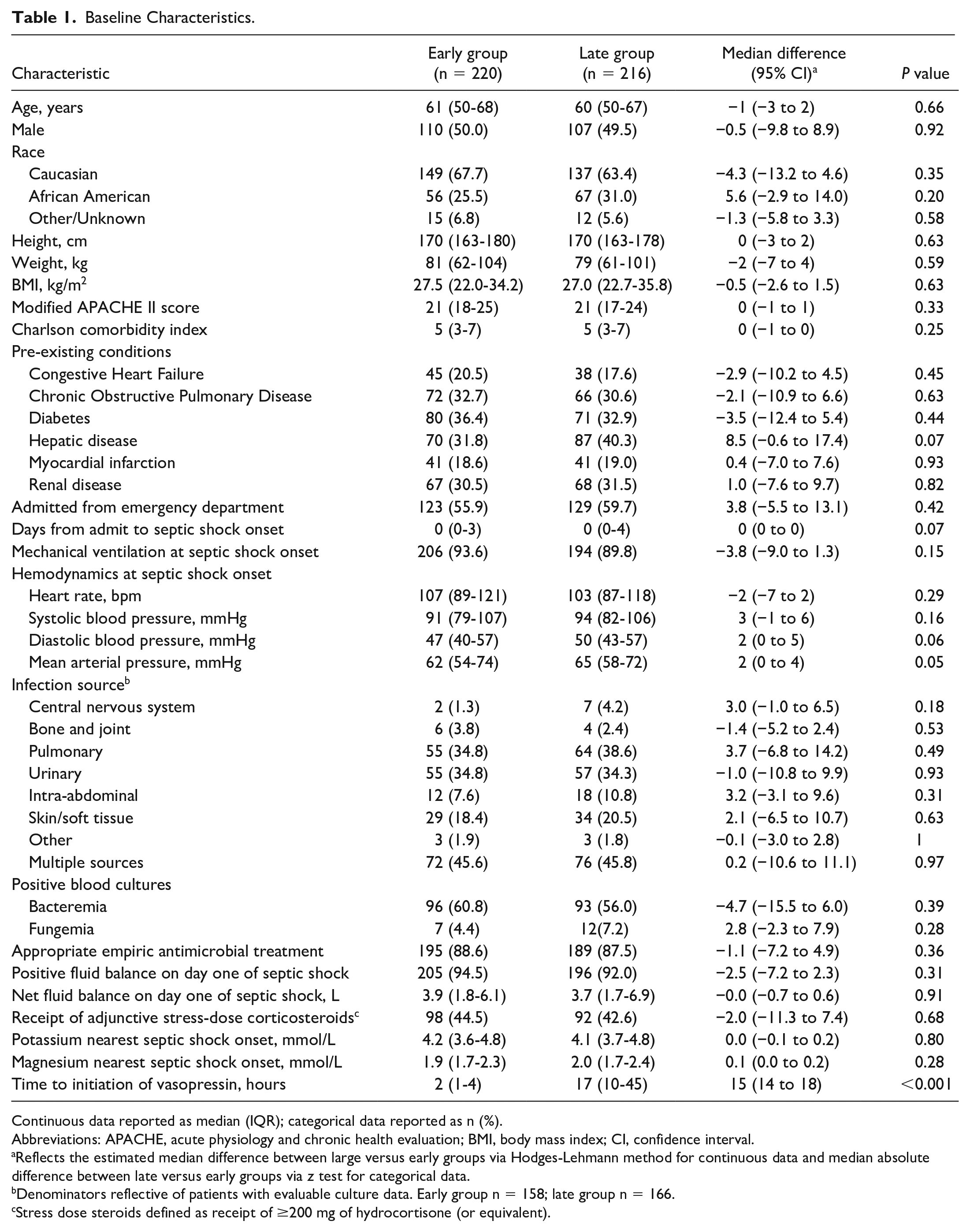
Continuous data reported as median (IQR); categorical data reported as n (%).
Abbreviations: APACHE, acute physiology and chronic health evaluation; BMI, body mass index; CI, confidence interval.
Reflects the estimated median difference between large versus early groups via Hodges-Lehmann method for continuous data and median absolute difference between late versus early groups via z test for categorical data.
Denominators reflective of patients with evaluable culture data. Early group n = 158; late group n = 166.
Stress dose steroids defined as receipt of ≥200 mg of hydrocortisone (or equivalent).
Eighty-eight patients received an anti-arrhythmic and/or rate control agent during or after septic shock onset and were reviewed by a board-certified cardiologist for confirmation and/or classification of NOA. Fifty-three patients were deemed to not have experienced an NOA following this review. Reasons for not being included were use of an anti-arrhythmic/rate-control agent(s) for non-arrhythmia-related indications (eg, hypertension, sinus tachycardia, or sympathetic storm) (n = 51) and misinterpretation of EKG (n = 4).
Outcome Data
No difference was observed in the primary outcome of incidence of NOA between the early vasopressin and late vasopressin group (9.1% vs 6.9%; median absolute difference [95% CI]: −2.1 [−7.2, 3.0], P = 0.41, Table 2). Similarly, no difference was observed when comparing the incidence of individual arrhythmia types between groups. Significant differences were not observed between groups when comparing maximum doses of norepinephrine or vasopressin received; however, median total hours of norepinephrine exposure was significantly lower in the early vasopressin group compared with the late vasopressin group (42 vs 81 hours; median absolute difference [95% CI]: 34 [24, 44], P < 0.001). Similarly, the early vasopressin group had a significantly shorter median duration of septic shock (2 vs 4 days; median absolute difference [95% CI]: 2 [1, 2], P < 0.001), shorter ICU LOS (6 vs 7 days; median absolute difference [95% CI]: 1 [0, 2], P = 0.02), lower incidence of new RRT (10.9% vs 21.3%, median absolute difference [95% CI]: 10.4 [3.5, 17.3], P < 0.001), and lower incidence of in-hospital mortality (40.9% vs 59.3%; median absolute difference [95% CI]: 18.4 [9.0, 27.7], P < 0.001). Time to NOA from septic shock onset did not differ significantly in the early versus late vasopressin group (1 vs 1 day, median absolute difference [95% CI]: 0 [−2, 1], P = 0.64). A Kaplan-Meier analysis revealed no significant difference in time to NOA from septic shock onset (Figure 2).
Primary and Secondary Outcomes.
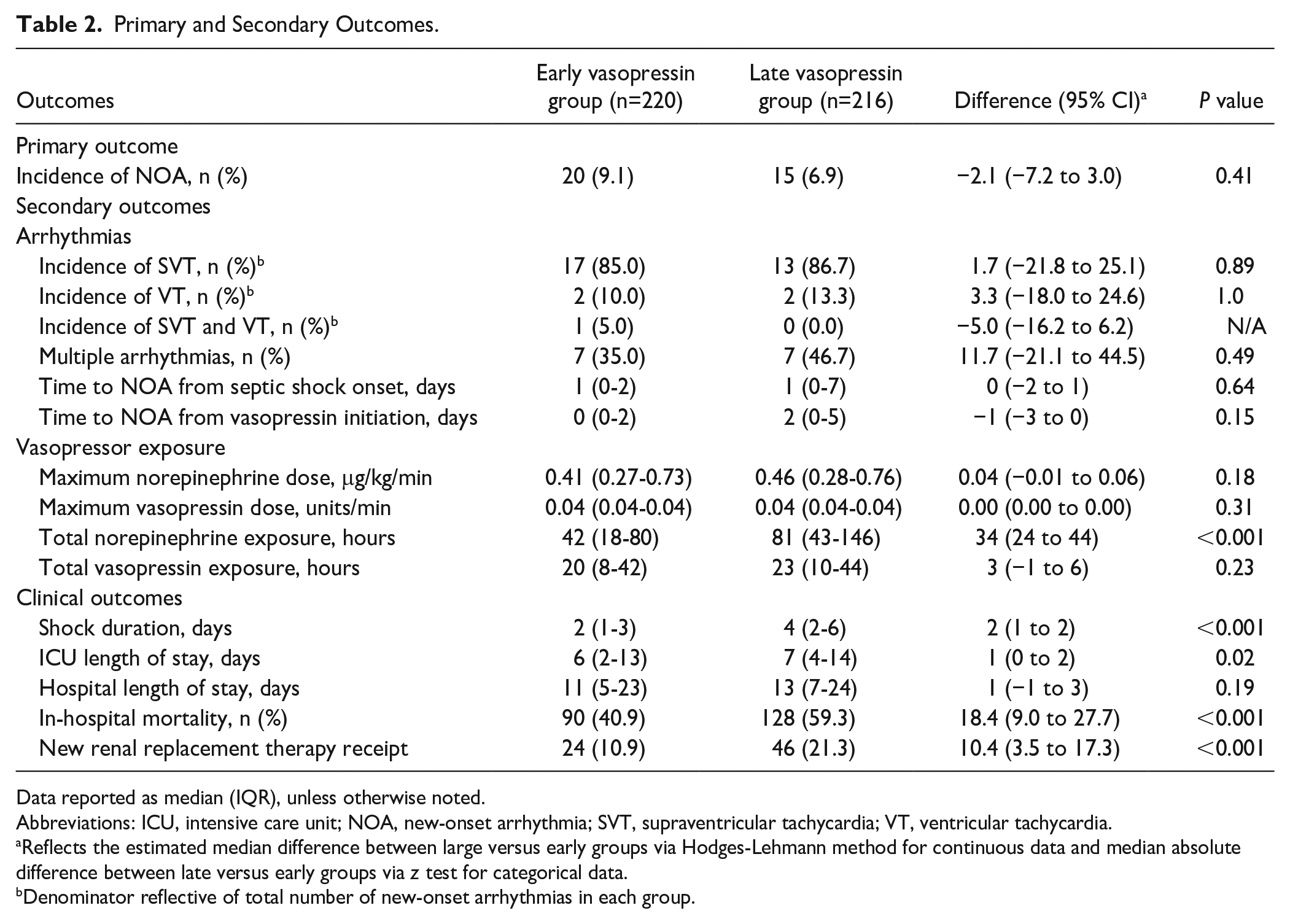
Data reported as median (IQR), unless otherwise noted.
Abbreviations: ICU, intensive care unit; NOA, new-onset arrhythmia; SVT, supraventricular tachycardia; VT, ventricular tachycardia.
Reflects the estimated median difference between large versus early groups via Hodges-Lehmann method for continuous data and median absolute difference between late versus early groups via z test for categorical data.
Denominator reflective of total number of new-onset arrhythmias in each group.
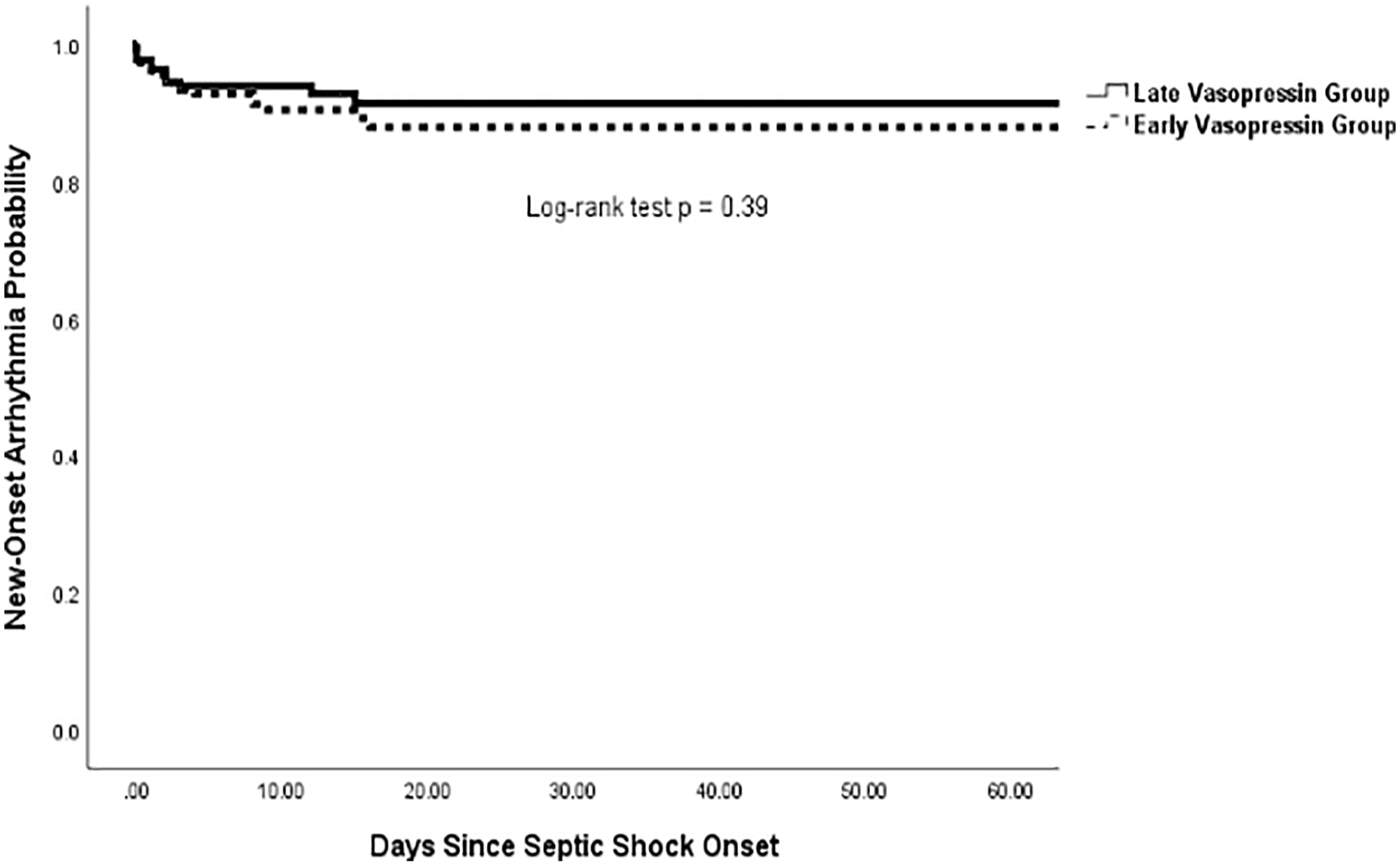
Kaplan-Meier of time to new-onset arrhythmia from septic shock onset.
A multivariable logistic regression model assessed the following covariates as potential risk factors for NOA: time to vasopressin initiation, mAPACHE II score, maximum norepinephrine dose, adjunctive corticosteroid receipt, mean arterial pressure (MAP) at the time of septic shock onset, and total hours of norepinephrine exposure. None of the included covariates were significantly associated with the development of NOA (Table 3).
Multivariable Logistic Regression Analysis for New-Onset Arrhythmias.
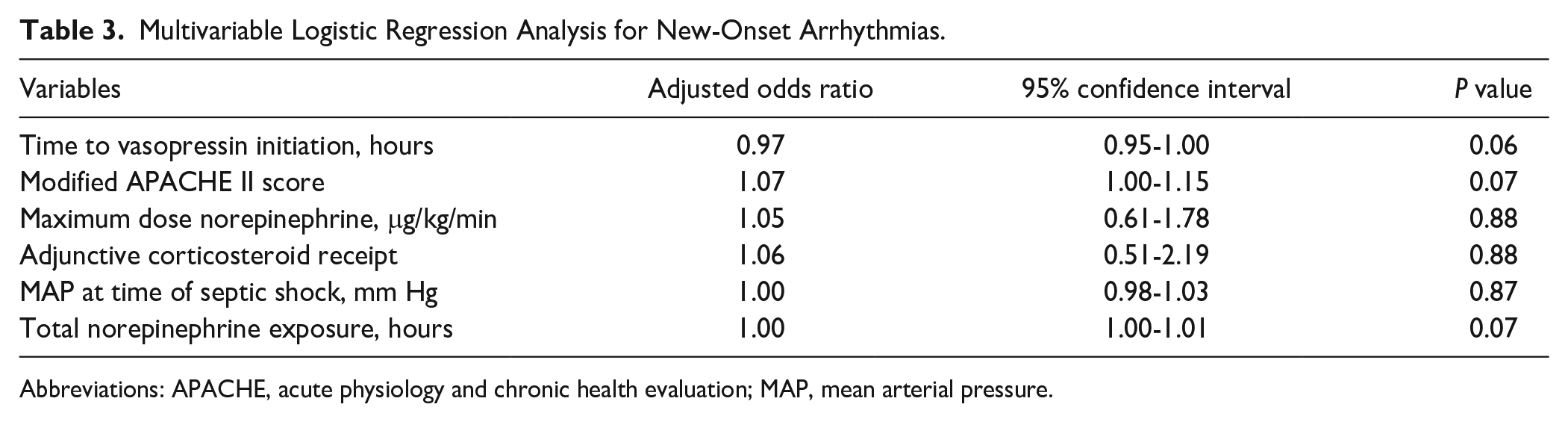
Abbreviations: APACHE, acute physiology and chronic health evaluation; MAP, mean arterial pressure.
Discussion
The frequency and potential sequelae of NOA and potential hemodynamic benefits of vasopressin in septic shock patients are well described. However, evidence describing the relationship between vasopressin’s catecholamine-sparing effects and the incidence of NOA are scarce. This retrospective cohort study compared the incidence of NOA between early and late vasopressin initiation in septic shock patients with no prior history of cardiac arrhythmias that required norepinephrine and vasopressin for hemodynamic support. The results suggest early vasopressin initiation does not reduce the incidence of NOA.
Results of this study were consistent with prior data observing use of vasopressin alone or in conjunction with other catecholamines in septic shock results in no difference in the incidence of arrhythmias, with observed rates of 9.1% and 6.9% in the early and late vasopressin groups, respectively. 23 A subgroup analysis of sepsis randomized controlled trials observed no difference in the relative risk of atrial fibrillation in patients receiving catecholamines and vasopressin analogues compared with catecholamines alone (10% vs 15%; RR 0.76; 95% CI: 0.55, 1.05; P = 0.09). 23 Similarly, the subgroup of only vasopressin randomized controlled trials observed no difference in the relative risk of ventricular arrhythmias (8% vs 9%; RR: 0.88; 95% CI: 0.56, 1.38; P = 0.57). The slightly lower observed incidence of NOA in our study may be explained by the inclusion of patients with no history of cardiac arrhythmias and/or no defined NOA requiring clinical intervention. Related, exclusion of patients requiring additional/alternative vasoactive agents in the present study likely reduces the potential for confounding factors. McIntyre et al acknowledged limitations surrounding catecholamine and vasopressin timing, dosing, and titration strategies, which the present study described. The variable dosing regimens (titrated vs fixed, physiologic vs pharmacologic, etc.) observed between included studies of the McIntyre et al meta-analysis and the current study are noteworthy when interpreting results. Our median and maximum dose of vasopressin of 0.04 units/min is consistent with the fixed-, physiologic dosing strategy done at our institution. Data evaluating differences in vasopressin dosing strategies are limited by their observational design, however, do not initially suggest an obvious association of improved clinical or hemodynamic outcomes or catecholamine exposure.31-33 These intricacies of vasopressin utilization are prudent considerations as research in the arena of vasopressin optimization continues.
In contrast, a retrospective cohort study by Reardon et al demonstrated initiation of vasopressin within 6 hours of septic shock onset was associated with a significantly lower incidence of NOA compared with later initiation (37 vs 63%; P < 0.01). 23 The stark difference in the incidence of NOA between Reardon et al and our study may be secondary to differences in patient population and definitions of history of arrhythmias and/or NOA. Higher APACHE II scores and rates of alternative vasoactive agent use, as well as exclusion of patients receiving vasopressin >48 hours after catecholamine initiation, highlight important differences between Reardon et al and our study. Of note, both the present and Reardon et al study observed use of fixed 0.04 units/min vasopressin. Reardon et al provided minimal detail surrounding identification of patients’ history of cardiac arrhythmias. The present study first utilized historical ICD information to exclude patients with a history of cardiac arrhythmias. Furthermore, inclusion of pertinent medication administration data before septic shock onset allowed exclusion of patients not identified via ICD review or who developed an NOA secondary to an unrelated cause. Finally, while Reardon et al similarly classified NOA, our study required EKG or telemetry evidence and was reviewed and confirmed by a board-certified cardiologist in addition to administration of an anti-arrhythmic or rate control agent. This was done to ensure a focus on clinically relevant NOA and minimize potential charting or administrative errors. Notably, variable incidence of NOA has been observed across prior critical care and sepsis literature due to heterogeneity of study design, patient populations, disease severity, and arrhythmia identification strategies. 7
We conducted a multivariable logistic regression analysis to control for confounders and identify potential predictors of NOA in septic shock patients with no history of cardiac arrhythmias. This analysis failed to reveal a significant association between time to vasopressin initiation and the incidence of NOA. To our knowledge, this is the first study to incorporate time to vasopressin initiation in a predictive model for NOA. In a recent meta-analysis, Bosch et al observed vasopressor use was associated with a greater than 50% change in odds of NOAF. 7 Several other sepsis-related risk factors were identified as potential targets for NOAF prevention. Our study’s inclusion of vasopressin initiation and timing built upon their findings and should serve as a basis for future study.
Although our study was not powered to detect a mortality difference, we observed a statistically significant association between early vasopressin initiation and reduced in-hospital mortality. Prospective and retrospective evaluations of norepinephrine with or without adjunctive vasopressin as well as norepinephrine versus vasopressin monotherapy, which are not directly applicable to our study, have yielded conflicting results related to mortality. Reardon et al reported a higher overall incidence of in-hospital mortality, but no significant difference between early versus late vasopressin administration (89% vs 89%, P = 1.0). 23 These results contradict our study’s findings but are likely driven by prior described differences in severity of illness and/or vasopressor prescribing practices. The observed lower incidence of need for new RRT in the early vasopressin group is also of interest, given contrasting data in this realm.20,21,34,35 While study design precludes the ability to determine causality in secondary outcomes, these findings are hypothesis-generating and highlight the need for continued study of vasopressin timing in septic shock.
The current study had several strengths. To date, it was the largest study to assess the effect of early administration of vasopressin on NOA in septic shock patients with no history of cardiac arrhythmias treated with only norepinephrine and vasopressin. It attempted to minimize and control for factors known or likely to contribute to NOA development in sepsis/septic shock via stringent inclusion and exclusion criteria and the use of a multivariable logistic regression analysis inclusive of novel details surrounding vasopressin timing. This study’s internal validity was strengthened by the novel approach employed to ensure NOA events were both clinically relevant and truly new onset. The use of a historical ICD-9/10 code query combined with medication administration, clinical, EKG, and/or telemetry correlation by a board-certified cardiologist reflects methodology not described previously.
Limitations of this study include first the observed low incidence of the primary outcome compared with some prior literature, which limited the scope of our statistical analysis. While investigators attempted to limit the number of included, clinically relevant covariates, it is still possible the regression model was overfit, and many other factors contribute to NOA in septic shock patients. Second, the utilized definitions for history of cardiac arrhythmias and NOA may still have allowed for inclusion of patients inappropriately and/or failure to capture NOA events because patients did not have prior documented ICD codes within the 14-hospital health system, did not require clinical intervention, and/or lacked telemetry, EKG, or other clinical support on manual chart review. Third, the study spanned 8 years in which temporal changes in vasopressin prescribing and clinical outcomes were possible. While an expected trend toward reduced mortality was observed during the study timeframe, no temporal patterns related to vasopressin timing, utilization, or dosing or incidence of NOA were observed. Other limitations of this study remained inherent to its retrospective, single-center design. These included limited generalizability to patients less severely ill, admitted to nonmedical or surgical ICUs or requiring additional/alternative vasopressor or inotropic agents. Related, our exclusion of patients with a history of arrhythmias and low incidence of cardiac comorbidities highlights populations who may benefit from earlier use of vasopressin. Next, it is possible patients in the late group received vasopressin as salvage therapy; however, the exclusion of patients receiving other vasoactive agents and the median time to initiation of 14 hours may limit this confounder. Finally, the study design precluded the ability to control for all confounding and/or biases that may occur within an institution that lacked any formal restrictions surrounding vasopressin administration.
Conclusions and Relevance
Among septic shock patients treated with vasopressin and norepinephrine, initiation of vasopressin within 6 hours of septic shock onset was not associated with a decreased incidence of NOA, despite controlling for potential confounders. Observed lower incidences of need for new RRT and in-hospital mortality, as well as shorter ICU and hospital LOS in patients receiving early vasopressin, highlight the need for continued study of the effect of vasopressin timing on NOA and other clinical outcomes.
Footnotes
Appendix A
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.D.B. discloses being a member of La Jolla Pharmaceutical Company’s speaker’s bureau. M.H.K.’s research efforts are supported by the Barnes-Jewish Hospital Foundation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
