Abstract
Background
Lenalidomide is an immunomodulatory drug used to treat multiple myeloma that requires renal dosing adjustment based on Cockcroft-Gault (CG). Various equations to estimate kidney function exist and pose a potential issue with lenalidomide dosing.
Objective
The objective of this analysis was to evaluate the impact of estimating kidney function in newly diagnosed multiple myeloma patients with CG, Modification of Diet in Renal Disease (MDRD), and Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) and their potential impact on lenalidomide dosing.
Methods
Data from 1121 multiple myeloma patients at the time of diagnosis acquired from the Mayo Clinic were used to calculate creatinine clearance (CrCl) using Cockcroft-Gault with actual body weight (CGABW), ideal body weight (CGIBW), or adjusted body weight (CGAdjBW); MDRD; and CKD-EPI for each subject. Discordances in dosing were then analyzed, and lenalidomide exposure was calculated for each subject to assess impact on pharmacokinetics of lenalidomide for patients who received discordant doses.
Results
Overall, approximately 16% of patients received a discordant dose when using MDRD or CKD-EPI instead of CGABW. The most common dose discordance was the decrease of a full dose of lenalidomide 25 mg when using CGABW down to 10 mg and when using MDRD or CKD-EPI with 53.8% to 55.6% of all discordances in this category. When assessing different body weights, the most common discordance was a decrease from 25 to 10 mg when using CGIBW instead of CGABW; the same trend was observed when using CGAdjBW instead as well. Patients were also at risk of over- or underexposure based on area under the concentration versus time curve (AUC) for discordant dosing.
Conclusion and Relevance
A significant proportion of patients are at risk of under- or overdose of lenalidomide if CKD-EPI or MDRD are used instead of CGABW. Physicians should use CGABW when estimating renal function to dose lenalidomide.
Introduction
Renal impairment is common in patients with cancer and may be a result of older age, prior anti-cancer therapies, or the cancer itself. 1 Renal impairment often necessitates dose adjustments of cancer therapeutics and other drugs that are primarily excreted by the kidneys. Historically, the Cockcroft-Gault (CG) equation has been used to estimate creatinine clearance (CrCl) to guide renal dose adjustments in clinical practice. This equation incorporates weight, age, sex, and serum creatinine. The Modification of Diet in Renal Disease (MDRD) equation is also used for clinical purposes, including for staging of chronic kidney disease (CKD) by the estimated glomerular filtration rate (eGFR) and uses 6 variables: age, sex, ethnicity, serum creatinine, urea, and albumin with a simplified 4-variable version (MDRD-4) that does not use urea and albumin.2-4 The MDRD-4 equation was used in this analysis. Another equation used to calculate eGFR, the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI), was established to address the issue of underestimating GFR at higher GFR. 5 The CKD-EPI equation incorporates 4 variables as well, taking into account age, sex, ethnicity, and serum creatinine like the simplified MDRD-4 equation. Discrepancies between the CG, MDRD, and CKD-EPI equations introduce risks for dosing errors, which are further compounded by the prominent display of the eGFR in electronic health records (EHRs). Another factor that may affect the calculation of renal function is handling overweight patients by using adjusted body weight (AdjBW) instead of actual body weight (ABW) that can lead to overestimation of renal function for heavier patients. Dosing errors for renally excreted anti-cancer therapies are of particular concern as many of these drugs have a narrow therapeutic window and minor changes in drug exposure may increase toxicity or decrease efficacy.
Lenalidomide, a derivative of thalidomide, is an immunomodulatory imide drug (IMiD) that is primarily renally excreted and has steep dose reductions for declining renal function based on calculated CrCl.6-8 The immunomodulatory, anti-angiogenic, anti-inflammatory, and anti-neoplastic activity, as well as the favorable toxicity profile of lenalidomide led to its Food and Drug Administration (FDA) approval in combination with dexamethasone alone or with other anti-myeloma classes of drugs such as proteasome inhibitors or anti-CD38 monoclonal antibodies for relapsed/refractory multiple myeloma (MM), newly diagnosed MM, and as monotherapy for post-autologous hematopoietic cell transplant (HCT) maintenance in 2006, 2015, and 2017, respectively.6,9 Lenalidomide is administered orally, rapidly absorbed, highly bioavailable, and moderately distributed into tissues.7,8 Lenalidomide is predominantly eliminated unchanged in urine (85%) and decreased kidney function results in an increase in lenalidomide plasma concentrations, exposure, and toxicity.7,8 Recommended dose adjustments of lenalidomide for patients with a CrCl of less than 60 mL/min involve a reduction of the induction dose from 25 mg daily to 10 mg daily for 21 days of a 28-day cycle.7,8 This steep dose reduction may be especially problematic when disparate equations for renal function are utilized in error and shift the lenalidomide regimen from one dose to another. The objective of this study was to evaluate the impact of estimating kidney function in newly diagnosed MM patients with the CG, MDRD, and CKD-EPI on lenalidomide dosing, including pharmacokinetic outcomes.
Methods
Demographic data and clinical characteristics from 1121 MM patients at the time of diagnosis were used (Table 1). The data were obtained via a data use agreement (DUA) with the Mayo Clinic in Rochester, Minnesota. The senior author was added to the Institutional Review Board (IRB) for this study at Mayo Clinic. For each individual subject, CrCl (milliliter per minute) was calculated with the Cockcroft-Gault equation using either actual body weight (CGABW), ideal body weight (CGIBW), or adjusted body weight (CGAdjBW); eGFR (mL/min/1.73 m2) was also estimated using both MDRD and CKD-EPI for each subject. For each estimate of renal function, a lenalidomide dose was assigned based on FDA-approved drug labeling.9,10 For the purposes of this analysis, the difference in units between CrCl and eGFR was not considered in determining lenalidomide dosing (mL/min vs mL/min/1.73 m2) to reflect (1) how renal function estimates are displayed within EHRs and (2) clinicians do not typically calculate an absolute eGFR in milliliter per minute.
Table of Subject Demographics.

Abbreviations: BMI, body mass index; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CrCl, creatinine clearance; IQR, interquartile range; MDRD, Modification of Diet in Renal Disease.
Lenalidomide exposure for each subject was estimated using a linear regression model described previously. 7 Apparent clearance (CL/F) was estimated using CrCl calculated with ABW, and the area under the concentration versus time curve (AUC) was then calculated as Dose/(CL/F). AUCs were calculated for each subject based on the different doses assigned for each renal function calculation. Discordance in dosing was assessed between CGABW versus MDRD, CGABW versus CKD-EPI, CGABW versus CGIBW, and CGABW versus CGAdjBW.
For subjects assigned different doses when the renal function calculation switched from CGABW to MDRD or CKD-EPI, the AUCs at the 50th, 75th, 90th, 95th, and 99th percentile cutoffs were compared with the AUCs of patients who received the full 25-mg dose (ie, those patients with CrCl >60 mL/min) to assess differences in lenalidomide exposure.
Results
Assessment of the concordance between CGABW and MDRD demonstrated that 16.6% of patients with newly diagnosed MM would have received a different lenalidomide dose if renal function were estimated by MDRD instead of CG (Figure 1). Of the subjects assigned a different dose using MDRD versus CG, the most common discordance was decreasing the full dose (25 mg) using CGABW to 10 mg using MDRD (53.8%). The next most common discordance was an increase from a dose of 10 mg using CGABW to a full dose of 25 mg using MDRD (28%). The other discordances of 10 mg using CGABW to 7.5 mg (ie, 15 mg every other day) using MDRD were 13.4% of those in the discordant category, while the converse of 7.5 mg using CGABW to 10 mg using MDRD was 4.3%. The least common discordance with only 1 subject (0.5%) was decreased from 25 mg using CGABW to 7.5 mg using MDRD.
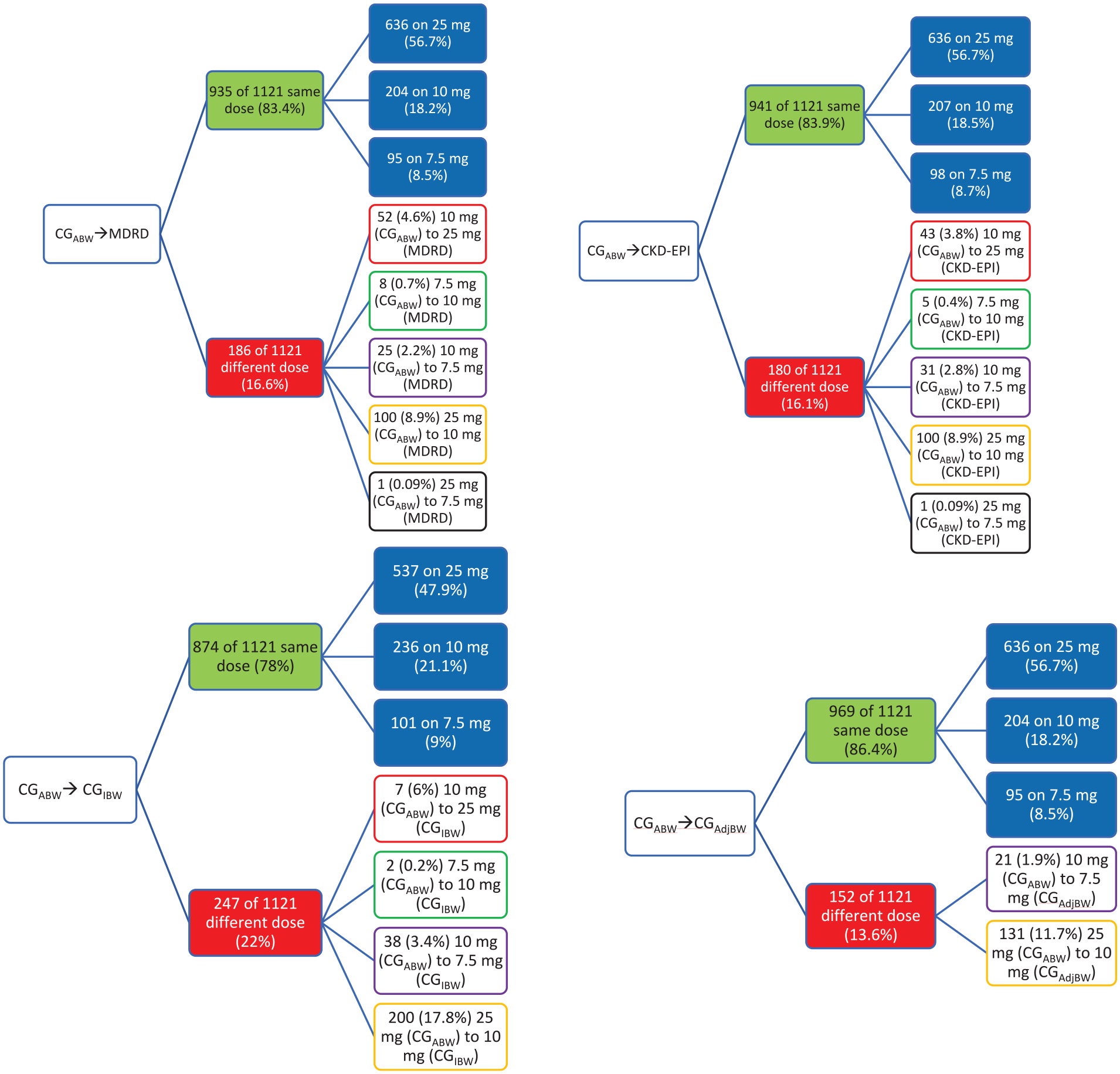
Chart summarizing the 4 comparisons analyzed and the statistical breakdown of all dose discordances found.
Comparing CGABW with CKD-EPI revealed similar trends, with 16.1% of subjects receiving a discordant dose (Figure 1). Like the CGABW versus MDRD comparison, the most common discordance was decreasing from the full 25-mg dose using CGABW to 10 mg using CKD-EPI, with 55.6% of subjects in the discordant category demonstrating this dose decrease. The next most common discordance was 10 mg using CGABW to 25 mg using CKD-EPI, with 23.9% of subjects in the discordant category experiencing this dose increase. Those subjects in the discordant group whose dose decreased from 10 mg using CGABW down to 7.5 mg using CKD-EPI accounted for 17.2% of subjects, with only 2.8% increasing from 7.5 mg using CGABW to 10 mg with CKD-EPI. Again, the least common discordance was decreasing from 25 mg with CGABW down to 7.5 mg with CKD-EPI with only 1 subject (0.56%).
Next, the use of the CG equation with different body weights was compared. When using CGIBW instead of the standard CGABW, 22% of all subjects showed discordant doses. Again, the most common discordance was a decrease from 25 mg using CGABW to 10 mg using CGIBW, with 81% of all the discordances falling into this category. Fewer dose modifications occurred in the categories of 10 to 7.5 mg (15.4%), 10 to 25 mg (2.8%), and 7.5 to 10 mg (0.8%). When comparing CGABW with CGAdjBW, the discordance was less drastic with only 13.6% of subjects receiving a discordant dose. Of those, the majority (86.2%) once again decreased from the full dose of 25 mg using CGABW to 10 mg using CGAdjBW; the only other discordance was a decrease from 10 mg using CGABW to 7.5 mg using CGAdjBW (13.8%).
For each comparison, the estimated AUCs in each discordance group were determined (Figure 2). The trends seen in AUC change are consistent based on the expected dose change; those dropping from a 25-mg dose using CGABW to 10 mg using MDRD had a 60% decrease in dose, with a similar decrease observed in AUC. Conversely, subjects assigned an increased dose using MDRD similarly experienced a relatively proportional increase in AUC. Last, for the subjects assigned a different dose when their renal function was estimated using MDRD or CKD-EPI, the AUCs in each group were plotted and compared with the AUCs of all subjects who received the full dose (25 mg) using CGABW (ie, CGABW > 60 mL/min). As shown in Figure 3, the subjects who switched from a dose of 10 mg using CGABW to 25 mg using CKD-EPI or MDRD instead had significantly higher AUCs, whereas the remaining dose changes remained within the range of values seen for subjects who received the full dose.
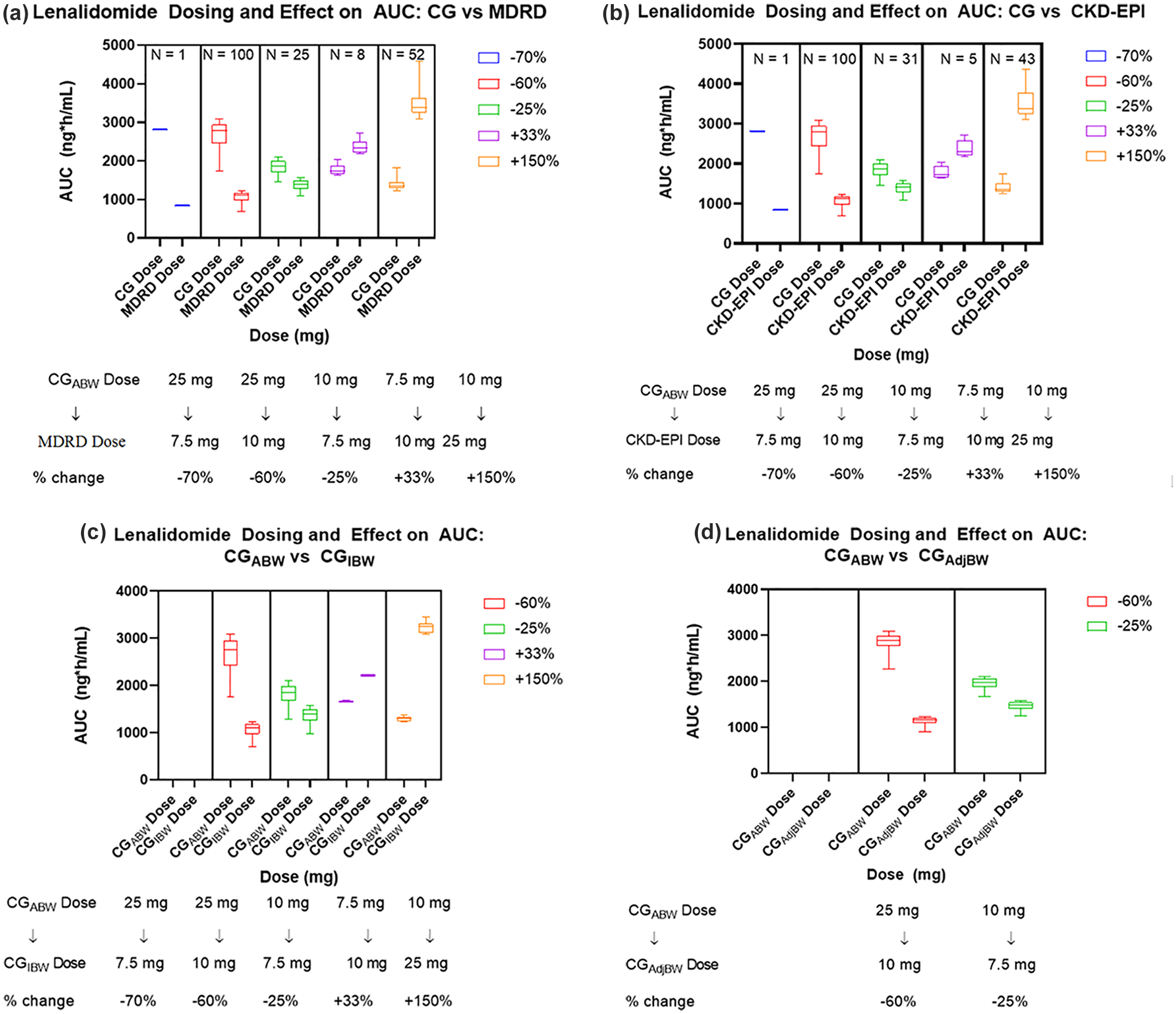
Boxplot demonstrating the AUC differences seen in subjects with discordances between (a) CG and MDRD renal function calculations, (b) CG and CKD-EPI, (c) CG using actual body weight and CG using ideal body weight, and (d) CG using actual body weight and CG using adjusted body weight.
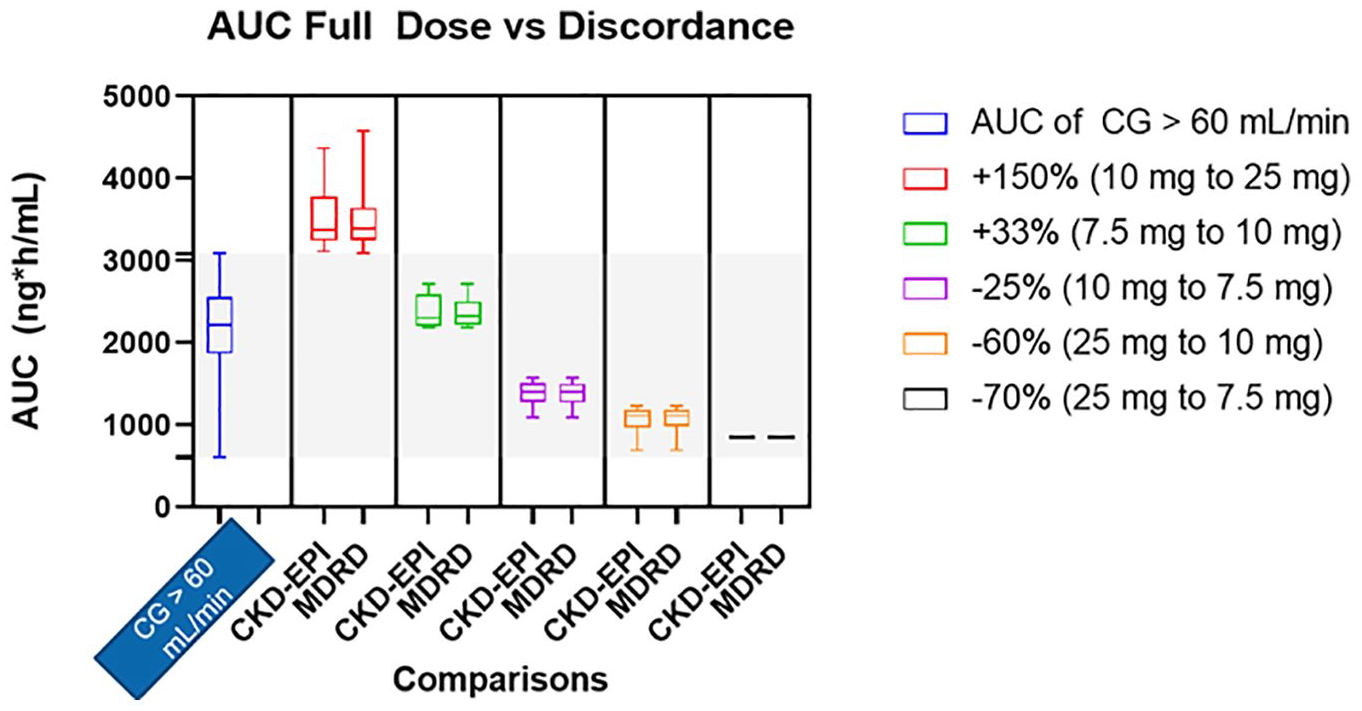
Boxplot demonstrating the AUC differences seen in subjects with discordances between CKD-EPI and MDRD compared with subjects with a renal function of 60 mL/min using Cockcroft-Gault.
Discussion
The purpose of this analysis was to assess discordance between CG, MDRD, and CKD-EPI on lenalidomide dosing and exposure in newly diagnosed MM patients. Across each comparison, subjects with a CrCl above 60 mL/min (ie, received a full lenalidomide dose of 25 mg) were less at risk of underexposure compared to subjects with moderate or severe renal impairment who were at risk for overexposure (ie, received a higher dose than they should). The current renal dosing recommendations for lenalidomide based on the package insert are such that minor changes in kidney function estimates can result in large changes in dose. 10 For example, if an EHR displays an eGFR derived via MDRD as 59 mL/min/1.73 m2 but the CG CrCl is above 60 mL/min, the patient may be prescribed 10 mg instead of 25 mg. Indeed, this was the most common discordance seen in this analysis and has implications on potential efficacy of therapy. Previous analyses recommend using the MDRD study equation for drug dose adjustments; however, our analysis demonstrates that for lenalidomide, major dose and exposure changes may result and therefore would not be advised. 11 Similar calls from the International Myeloma Working Group (IMWG) to use CKD-EPI or MDRD equation for renal function calculations also complicate the picture and are not supported by this analysis. 12 Based on the results presented here, the CKD-EPI equation and the MDRD equation led to significant discordance that most often leads to underdosing (prescribing 10 mg instead of 25 mg) for subjects with borderline CrCl around 60 mL/min using CG (8.9% of the time when converting from CGABW to MDRD or CKD-EPI; Figure 1). Conversely, using MDRD or CKD-EPI also led to overdose (subjects prescribed 25 mg instead of 10 mg) in 4.6% of subjects who had their renal function recalculated using MDRD from CGABW or 3.8% if recalculating using CKD-EPI (Figure 1). The package insert renal dosing recommendations for lenalidomide are based on CG; based on this analysis, health care providers should use ABW in the renal function calculation when determining the dose of this drug.
CG can be calculated using a variety of different body weights, including IBW (ideal body weight), ABW, or AdjBW. AdjBW is calculated such that if the subject’s ABW is less than their IBW, their ABW is used; if their ABW is more than 1.3 times their IBW, AdjBW is calculated using the equation, AdjBW = IBW + 0.4 × (ABW – IBW). Thus, the only subjects who will have a different AdjBW from their ABW are subjects with relatively higher body mass indices (BMIs), and their AdjBW will be lower than their ABW. Clarity and expert consensus regarding which body weight (actual, ideal, or adjusted) to utilize in the CG formula is lacking and has the potential to result in inaccurate CrCl results and subsequent dosing errors. 4 Many robust studies have sought and failed to resolve the controversy surrounding the appropriate body weight to utilize for the CG formula. 4
As CG is highly depending on weight, the discordance seen in our study with the majority of subjects in the discordant category comparing CGABW with CGAdjBW was a decrease in lenalidomide dose from 25 to 10 mg; the only other discordance was decreasing from 10 to 7.5 mg, meaning that all subjects who experienced discordance when re-estimating their renal function using CGAdjBW received a lower dose. Similarly, using IBW instead of ABW often underestimates subject’s weight and subsequently their renal function; as a result, the majority of the discordance was again seen in decreasing 25 to 10 mg. Thus, this analysis supports using ABW instead of AdjBW or IBW.
One of the challenges of assessing lenalidomide exposure among subjects receiving discordant doses is that there are no well-established exposure-response relationships for efficacy or toxicity. As such, there is an unclear threshold to avoid toxicity and an unclear minimum exposure for efficacy. This guided the decision to instead compare predicted lenalidomide exposure with the group of subjects who received the full dose. Based on our findings, theoretically, the group at greatest risk of potential toxicity are the subjects with CGABW slightly below 60 mL/min who then are increased to above 60 mL/min/1.73 m2 using MDRD or CKD-EPI; this particular subpopulation demonstrated AUCs all above the 99th percentile of AUCs for those receiving the full 25-mg dose. Therefore, caution is warranted for subjects with borderline CrCl calculations.
The dosing approach for lenalidomide in renal impairment could potentially be improved by integrating these findings into pharmacokinetic-pharmacodynamic (PK-PD) models, but the lack of an exposure-response relationship is a barrier. Drug developers should consider potential discordance between estimating equations and put forward meaningful recommendations that are relevant to the clinical setting. In sum, this analysis elucidates the potential problems with using different renal function calculations that lead to dose discordance and presents a strong argument for the standardization of renal function calculations used in clinical studies that match those that are used in clinical practice.
The main limitation of this study is the lack of ethnic diversity seen in the subject pool; in our analysis, only 2% of subjects were black or African American; however, African Americans can make up over 24% of the total newly diagnosed cases and MM is the second most common blood cancer in people of African descent. 13 Therefore, the conclusions of this analysis are not generalizable to centers with larger proportions of African American patients with MM. In addition, the conclusions may have been different if a larger proportion of the subjects were African American as MDRD and CKD-EPI both incorporate ethnicity in the calculation.
Conclusion and Relevance
This analysis highlights the importance of dosing lenalidomide for patients with impaired renal function using CGABW. Using MDRD or CKD-EPI instead of CG or using AdjBW or IBW instead of ABW increases the risk of an inappropriate dose for the patient. An interdisciplinary clinical team of oncology pharmacists and nurses in collaboration with the oncologist can work together to ensure the correct apparent clearance (CL/F) and body weight is used to ensure the correct dose of lenalidomide is selected for a patient.14,15
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
