Abstract
Objective:
To describe interventions that target patient, provider, and system barriers to sedative-hypnotic (SH) deprescribing in the community and suggest strategies for healthcare teams.
Data Sources:
Ovid MEDLINE ALL and EMBASE Classic + EMBASE (March 10, 2021).
Study Selection and Data Extraction:
English-language studies in primary care settings.
Data Synthesis:
20 studies were themed as patient-related and prescriber inertia, physician skills and awareness, and health system constraints. Patient education strategies reduced SH dose for 10% to 62% of participants, leading to discontinuation in 13% to 80% of participants. Policy interventions reduced targeted medication use by 10% to 50%.
Relevance to Patient Care and Clinical Practice:
Patient engagement and empowerment successfully convince patients to deprescribe chronic SHs. Quality improvement strategies should also consider interventions directed at prescribers, including education and training, drug utilization reviews, or computer alerts indicating a potentially inappropriate prescription by medication, age, dose, or disease. Educational interventions were effective when they facilitated patient engagement and provided information on the harms and limited evidence supporting chronic use as well as the effectiveness of alternatives. Decision support tools were less effective than prescriber education with patient engagement, although they can be readily incorporated in the workflow through prescribing software.
Conclusions:
Several strategies with demonstrated efficacy in reducing SH use in community practice were identified. Education regarding SH risks, how to taper, and potential alternatives are essential details to provide to clinicians, patients, and families. The strategies presented can guide community healthcare teams toward reducing the community burden of SH use.
Introduction
In community practice, sedatives and hypnotics (SH), including certain benzodiazepines and nonbenzodiazepine GABA-receptor agonists, as well as drugs with sedating adverse effects, are frequently prescribed, especially for insomnia.1-4 Systematic reviews and clinical practice guidelines for insomnia suggest that these prescriptions should primarily be of short duration because of insufficient evidence to support extended use, evidence of harm, and availability of safe and effective nonpharmacological alternatives (eg, cognitive behavioral therapy for insomnia [CBTi]).5-10 However, chronic SH utilization in the community is high, with reported rates between 3% and 17%.1-3,11-13
SH exposure is associated with serious adverse events, including falls, cognitive impairment, and increased mortality, and extended use of some classes may be associated with drug dependence (eg, benzodiazepines).14-17 Professional societies include SH on their list of potentially inappropriate or high-risk medications for insomnia management for older adults and endorse reduction of SH use to minimize the risk of adverse events.18-20 Recent observational data suggest that prescribing practices have shifted from traditional benzodiazepines to newer Z-drugs and other sedating medication classes (eg, off-label use of quetiapine and trazodone) because these newer medications are falsely perceived as safer alternatives.1,2,21-25
Intervention studies and systematic reviews have shown the plausibility of reducing SH utilization with strategies such as patient education, prescribing effective nonpharmacological alternatives, and tapering or deprescribing in various patient care settings. There are specific patient- and health care provider–related barriers to applying these interventions in the outpatient setting.26-28 Patient-related barriers include being unaware of medication-related harms, developing psychological dependence, and being unable to find effective alternatives. 26 The prescriber-patient relationship can also create barriers such as when prescribers perceive that patients are expectant of a SH prescription and ambivalent or reluctant to taper or stop. 27 Prescriber inertia is often a result of health care system constraints, such as difficulty finding the time to assess the appropriateness of medications not directly related to the presenting complaint.27,28 Furthermore, health care systems and regulatory policies may permit multiple months to be dispensed at once, thus limiting the opportunity to review SH prescription use. Prescriber self-efficacy can be a barrier if prescribers are unaware of the evidence or clinical tools to guide tapering, deprescribing, and substitution with nondrug therapy. 27 Finally, access to effective nonpharmacological therapies such as CBTi may be limited depending on the resources available to the patient or provider.27,28 The primary objective of this narrative review is to describe interventions that target patient, health care provider, and system barriers to reduce SH use for adults in primary care. Our secondary objectives are to describe the impact of these identified interventions on reported outcomes and provide strategies for quality improvement (QI) teams to use these interventions to reduce SH use in primary care.
Methods
Data Sources
With the assistance of a Medical Information Specialist, we conducted an electronic search of Ovid MEDLINE ALL, EMBASE Classic + EMBASE (inception to June 15, 2021) to identify studies that investigated interventions to reduce SH use.
Study Selection and Data Extraction
The search strategy was previously published; articles were sorted into those that applied to the inpatient setting and those for the outpatient setting (the focus of this article). 29 We sought studies that tested an intervention in adult participants to limit SH use (either reduced initiation of new prescriptions, drug tapering, or deprescribing) in the community, outpatient, or primary care setting. We included studies that also addressed other drugs classes (eg, proton pump inhibitors, antihypertensives) if SH results were individually reported. We excluded studies that (1) were set specifically in a hospital, long-term care, or psychiatric setting; (2) focused on pediatric populations; (3) focused specifically on alcohol withdrawal or seizure management; or (4) were not published in English. We extracted (TM, CT, HJC, EG, FK, AV) the details of the studies using a standardized case report form. Findings were summarized (LB, JT) as 3 broad themes based on the barriers addressed: (1) patient-related barriers and prescriber inertia; (2) physician skills and awareness; and (3) health system constraints.
Results
We (LB, CS) screened the 5286 citations identified from the electronic search, of which 22 citations met inclusion criteria: 20 studies and 2 citations were long-term follow-up of the included studies (Appendix A, available online).
Data Synthesis
Included studies were themed and are presented as patient-related and prescriber inertia (Table 1),30-40 physician skills and awareness (Table 2),41-47 and health-system constraints (Table 3).48-52 Among the identified studies, we noted considerable heterogeneity in study design, sample size, population assessed, and outcomes reported. Of the 20 studies, 12 (60%) were randomized controlled or cluster randomized trials. The majority of studies focused specifically on benzodiazepine use; however, more recent studies included a broad range of SH drugs or targeted multiple drug classes, including SH. Although many of the interventions were shown to successfully reduce SH use in the community setting, the majority of interventions (1) were tested in a single study or with a limited sample size or duration of exposure, (2) were studied in a specific setting or demographic that may limit generalizability of findings, (3) did not examine clinical outcomes to allow understanding of the full impact of the intervention, and (4) may have included other indications for SH beyond insomnia.
Studies Evaluating Patient Education to Overcome Patient Barriers to Reduce SH Use in Community-Dwelling Adults.

Abbreviations: BZD, benzodiazepine; CBT, cognitive behavioral therapy; DDD, defined daily dose; GP, general practitioner; N/A, not applicable; NNT, number needed to treat; MOS, medical outcome study; NSAIDs, nonsteroidal anti-inflammatory drugs; OR, odds ratio; PPD, prescribed daily dose; RCT, randomized controlled trial; RR, risk ratio; RRR, relative risk ratio; SF-12, Short-Form 12; SH, sedative-hypnotic.
Studies Evaluating Prescriber-Focused Interventions to Overcome Prescriber Barriers to Reduce SH Use in Community-Dwelling Adults.
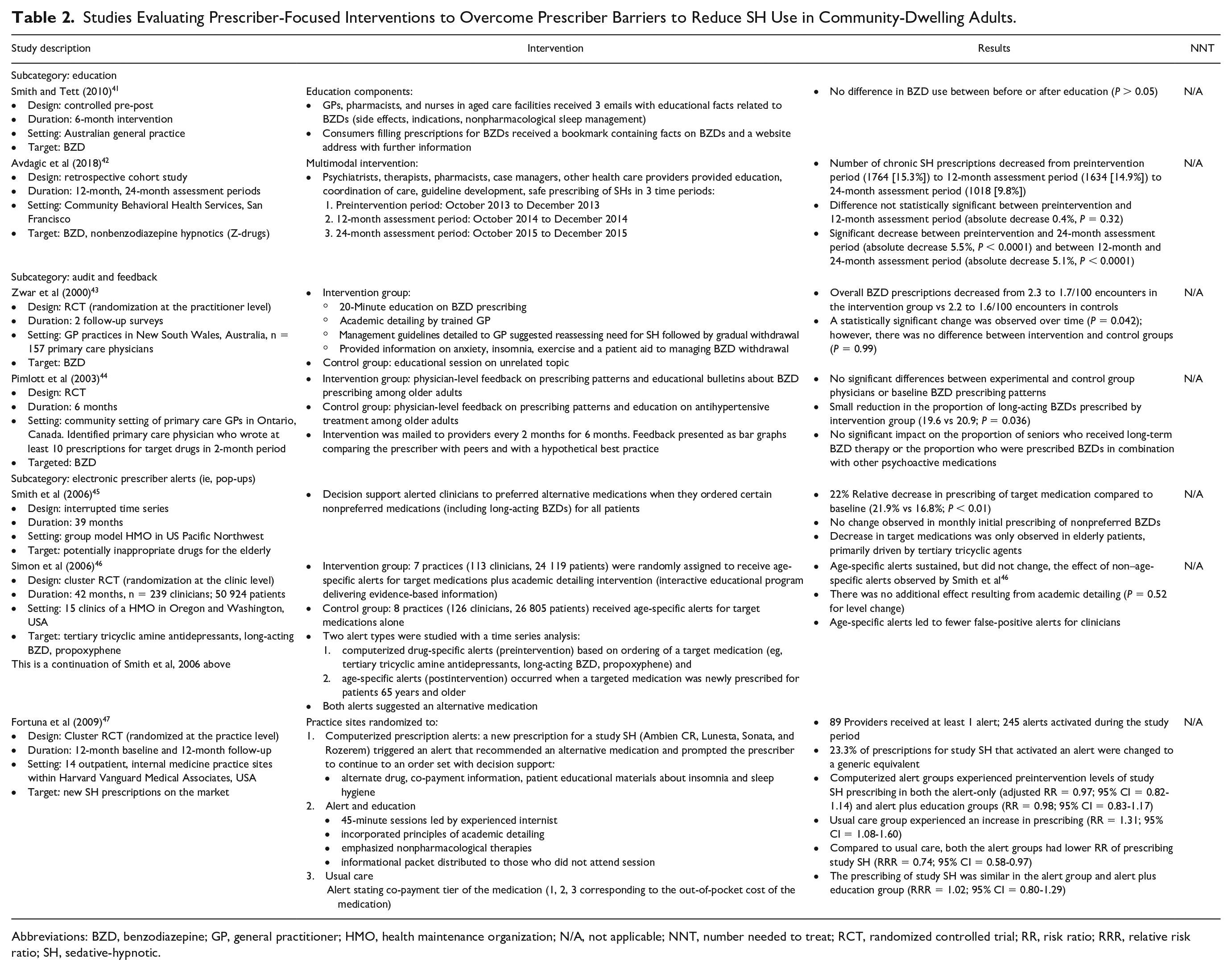
Abbreviations: BZD, benzodiazepine; GP, general practitioner; HMO, health maintenance organization; N/A, not applicable; NNT, number needed to treat; RCT, randomized controlled trial; RR, risk ratio; RRR, relative risk ratio; SH, sedative-hypnotic.
Studies Evaluating Interventions to Overcome Health System Constraints to Reduce SH Use by Community-Dwelling Adults.
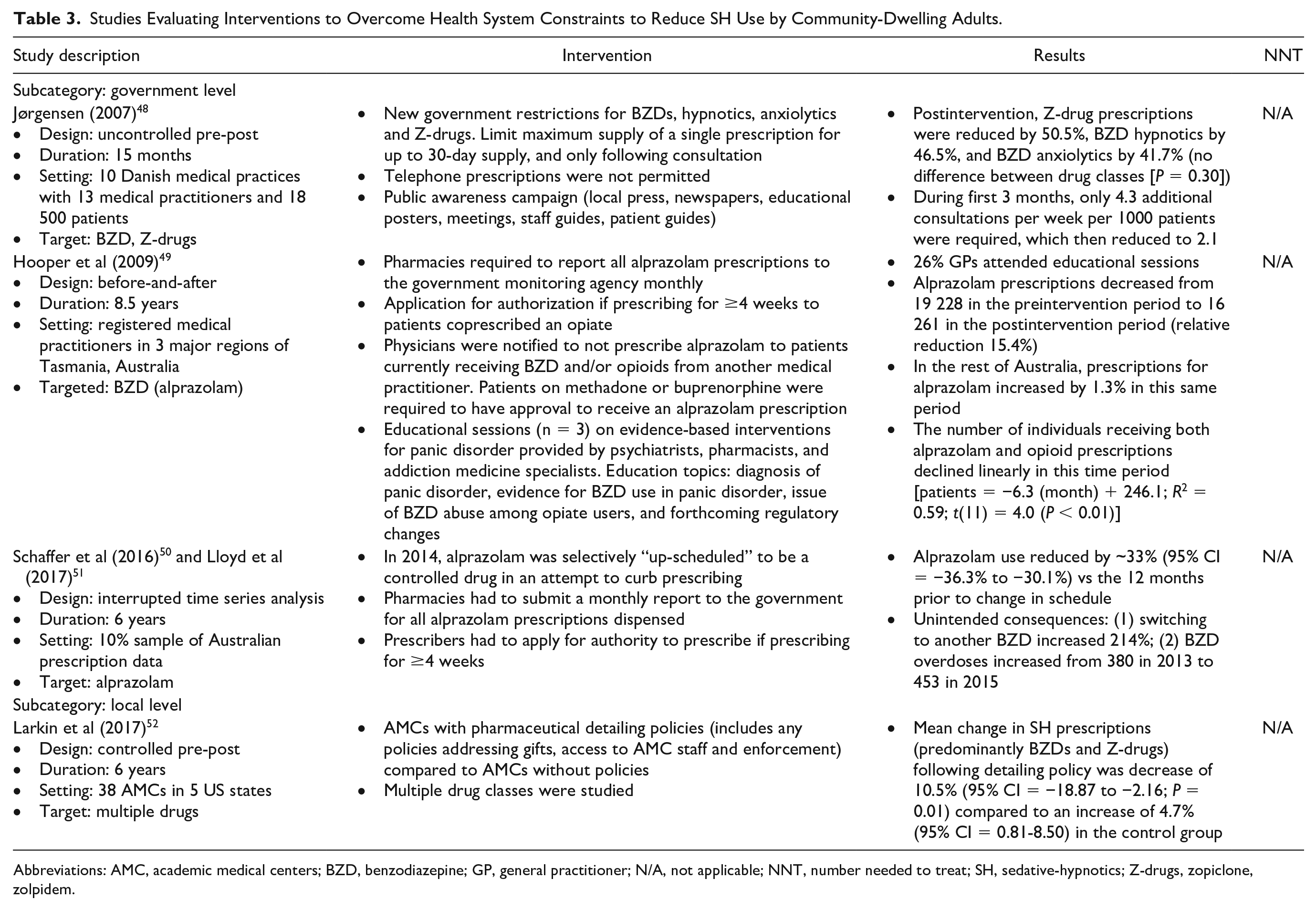
Abbreviations: AMC, academic medical centers; BZD, benzodiazepine; GP, general practitioner; N/A, not applicable; NNT, number needed to treat; SH, sedative-hypnotics; Z-drugs, zopiclone, zolpidem.
Evidence Identified
Studies Overcoming Patient-Related Barriers and Prescriber Inertia
Patients need to be informed of the risks associated with chronic SH use in order to be engaged in shared decision-making. Identified studies showed that patient education reduced the SH dose for 10% to 62% of participants and led to SH discontinuation in 13% to 80% of participants (Table 1).30-40 All identified trials examined strategies to reduce use, taper, or discontinue existing SH use rather than deter SH initiation. Patient education interventions included a doctor’s letter or educational brochure explaining adverse consequences of chronic SH therapy and encouraging the patient to gradually taper their dose (with complete cessation if possible) on their own and an invitation to see their doctor for further discussion or CBT to address insomnia. Empowering patients to engage in conversations with their prescriber about SH use can overcome prescriber inertia by reducing patient expectations for prescribing and reviewing the necessity of the medication and how it could be stopped.40,53
Gorgels et al 33 conducted a prospective controlled intervention at 30 Dutch primary care sites with 3833 participants. Benzodiazepine users received a letter from their primary care doctor with advice to gradually discontinue therapy followed by an invitation at 3 months for consultation, whereas the control group received usual care. At 21 months, 26% of the intervention group reduced their benzodiazepine consumption versus 9% in the usual care group (prescribed daily dosages difference: 12.5%; 95% CI = 8.2%-16.8%). The authors demonstrated a sustained effect of their intervention in a subset of patients at 10 years. 34
Vicens et al 35 evaluated the efficacy of 2 structured interventions that enabled discontinuation of long-term benzodiazepines. In this multicenter, 3-arm cluster-controlled study with randomization at the physician level, general practitioners were allocated (1:1:1) to usual care, or a patient education intervention with or without follow-up visits. The intervention consisted of structured educational patient interviews, where physicians provided information about risks of SH use, a self-help leaflet, and a written tailored gradual dose reduction plan. In the arm including follow-up, physicians scheduled appointments with the patient every 2 to 3 weeks. At 12 months, 45% of the patients in both intervention groups had discontinued benzodiazepine compared with 15% in the control group (RR = 3.01, 95% CI = 2.03-4.45, for intervention with writing and RR = 3.0, 95% CI = 2.04-4.40, for intervention with follow-up visits). The results were sustained at 36 months. The most frequently reported withdrawal symptoms were insomnia, anxiety, and irritability. Sleep satisfaction was reported as significantly higher in the intervention group with follow-up.
Nurses and pharmacists can also provide patient education and empowerment regarding deprescribing. Lopez-Peig et al 38 evaluated a nurse-led benzodiazepine withdrawal program using a before-after study design in 2 primary care centers (n = 51 participants). Nurses provided patient education, and SH doses were reduced every 2 to 4 weeks by approximately 25%. Alternative drugs valerian or hydroxyzine were permitted for sleep to support benzodiazepine tapering. At 6 months, 80% of the patients had discontinued benzodiazepine use and 64% maintained discontinuation at 1 year. Those who successfully discontinued benzodiazepine therapy reported improvements in depression and anxiety without reduction in sleep quality. A pharmacy-based patient education program was examined in 2 randomized controlled trials.39,40 Tannenbaum et al 39 conducted a cluster randomized trial to compare the effect of a direct-to-consumer education tool versus usual care via 30 community pharmacies in Québec. Pharmacies were randomly allocated to the intervention, which involved mailing an 8-page brochure to chronic benzodiazepine users aged 65 years and older either immediately (intervention) or after a 6-month wait period (control). The brochure included education about harms, peer support stories, a tapering protocol, and alternatives. At 6 months, 27% of the intervention group had discontinued therapy versus 5% of the control group (risk difference = 23%; 95% CI = 14%-32%), and 11% had reduced their dose (95% CI = 6%-16%). Lack of physician support was identified as a deterrent to deprescribing. To address this barrier, a second randomized trial that added an evidence-based pharmaceutical opinion sent to physicians, at the same time as pharmacists mailed the education brochure to patients, sought to better foster communication around deprescribing. 40 The study enrolled 489 participants from 69 community pharmacies who were prescribed 1 of 4 Beers Criteria medications (SH, nonsteroidal anti-inflammatories, glyburide, or first-generation antihistamine). SH discontinuation was 43.2% for the intervention group versus 9.0% for the control group (risk difference = 34%; 95% CI = 25%-43%); 38% of individuals who attempted to taper SH reported withdrawal symptoms.
CBT has been investigated as an add-on to patient education to improve deprescribing rates and sustainability of discontinuing SH use. Voshaar et al 37 conducted a trial to evaluate the efficacy and feasibility of tapering off long-term benzodiazepine use in patients who did not stop benzodiazepines within 3 months of receiving a letter. This study randomized 180 participants from 30 Dutch general practices to either medication tapering with weekly follow-up visits, tapering with weekly visits plus CBT, or usual care. The CBT delivered included relaxation training and cognitive restructuring aimed at reducing impact of any perceived withdrawal, without stimulus control, sleep restriction, or other modalities aimed specifically at insomnia treatment. Both intervention groups showed similar reductions in the proportion of patients who successfully discontinued benzodiazepines at the 3-month evaluation. Benzodiazepine dosage was the only independent predictor of successful discontinuation (OR = 4.5, 95% CI = 2.0-10.2, for daily doses < 10 mg diazepam equivalent). Although delivering CBT for insomnia may be limited in some outpatient settings, research supports the use of digital CBT for insomnia, which some patients may find suitable and easy to access. 54
Studies Increasing Prescriber Awareness
Electronic alerts and audit-and-feedback (ie, process of measuring and reporting individual performance to desired standard or peers) have also been studied to increase prescriber awareness to reduce SH use (Table 2).41-47 One RCT explored audit-and-feedback on benzodiazepine prescriptions for 374 primary care physicians. Physicians were randomized to the intervention group that received physician-level feedback on prescribing patterns and education, whereas the control group received similar information on antihypertensives. The study found a 0.7% decrease from baseline for long-acting benzodiazepines in the intervention group and a 1.1% increase in the control group (P = 0.036). 44
Clinical decision support aids, such as guidelines, recommendations, or indications/contraindications, can be incorporated into electronic prescribing systems as computerized alerts or pop-ups. Smith et al 45 examined the effect of clinical decision support on use of potentially contraindicated medications in older adults. In this interrupted time series analysis, there was a 22% relative decrease in the prescribing of target medications (21.9% vs 16.8%; P < 0.01) following alert implementation, with reduction primarily driven by reduced tricyclic antidepressant use. In a follow-up cluster-randomized study, the investigators examined the effect of replacing medication-specific prescribing alerts with age-specific alerts compared to alerts with academic detailing. 46 The addition of academic detailing did not enhance the effect of the alerts.
Fortuna et al 47 demonstrated the greatest impact of alerts on prescribing in a cluster-randomized controlled trial in 14 outpatient internal medicine sites. The objective of this study was to reduce prescribing of heavily marketed new SHs (zolpidem, eszopiclone, zaleplon, or ramelteon). The sites were randomized to receive computer alerts with and without prescriber education or usual care for new prescriptions only. Targeted SH prescriptions dropped in the alert group (relative risk reduction [RRR] = 0.74; 95% CI = 0.57-0.96) and the alert plus education group (RRR = 0.74; 95% CI = 0.58-0.97) compared to the usual care group. Overall, clinicians reported that the alerts did not interfere with their workflow and that the alerts provided information perceived to be beneficial. The impact on patients or clinical outcomes were not examined.
Studies Investigating Health System Policy Interventions
Policy interventions to reduce SH prescribing can be implemented at the local, state, or national health systems level (Table 3). Policy interventions we identified involved prescribing restrictions such as reducing the number of days supplied, rescheduling, prohibiting telephone orders, and local policies that restricted detailing visits by pharmaceutical company representatives to prescribers. Some policies were supported with prescriber and public education. Implementation of these policies produced a 10% to 50% reduction in targeted medication use.48-52 The greatest reduction in SH use was reported by Jørgensen 48 who conducted a pre-post study following new government restrictions that broadly targeted multiple SH classes (benzodiazepines, hypnotics, anxiolytics, and Z-drugs) to limit prescribing to a 1-month supply at a time following consultation. The new restrictions were publicly advertised, targeting both patients and staff. The 15-month pre-post study involved 10 medical practices, targeting 18 500 patients. Postintervention Z-drug use was reduced by 50.5%, benzodiazepine-hypnotics by 46.5%, and benzodiazepine-anxiolytics by 41.7%, with no difference between the various SH classes. Hooper et al 49 conducted an 8.5-year before-and-after regional study specifically targeting alprazolam. Physicians were notified to not prescribe alprazolam to patients who were receiving benzodiazepines and/or opioids from other medical practitioners. Pharmacists were required to submit a monthly report of alprazolam prescriptions dispensed to a government monitoring agency. Physicians required authorization to prescribe for more than 4 weeks to patients also prescribed an opioid. Supporting education for general practitioners was provided of whom 26% attended the educational sessions. There was a 15.4% relative reduction in alprazolam prescriptions following the intervention; in regions without the policy, alprazolam use increased by 1.3%. In 2014, the alprazolam intervention was upscaled nationally, and alprazolam was rescheduled from a prescription medication to a controlled drug. Schaffer et al 50 found that alprazolam use reduced by 33.3% (95% CI = 36.3%-30.1%) compared to 12 months prior to the policy change, but alternative benzodiazepine use increased by 214%. Poison control center data after the policy change showed that benzodiazepine overdoses also increased. 51 These studies suggest that balancing measures should be factored into policy interventions and safer pharmacological and nonpharmacological alternatives should be considered.
Relevance to Patient Care and Clinical Practice
QI initiatives need to address patient and physician barriers while considering the context and environment in which the intervention will be applied as well as the resources available (see Table 4 for specific resources). The outpatient setting is expansive, and local ecology factors as well as province/state rules or policy should be considered. We emphasize an approach that can be applied within a microsystem (eg, individual prescriber or primary care clinic) and upscaled over time (Figure 1). Regardless of the scale of the intervention(s) applied, it is clear that patient engagement is valuable. A common thread between the most effective interventions was facilitation of communication/relationship development between patients and health care providers. Based on our interpretation of the literature reviewed and the harm associated with chronic SH use, we propose the following implementation strategies to guide QI initiatives in primary care settings.
Patient and Health Care Provider Resources to Facilitate Sedative-Hypnotic Reduction.


Context-specific interventions to reduce SH use in community practice.
Strategy 1: Interventions to Reduce Patient Barriers
Patient engagement and empowerment by the QI team will be crucial to convincing patients to reduce or discontinue chronic SH use because patients or caregivers are ultimately in control of their medication (eg, continue to fill repeats or seek out an alternative prescriber). Patients’ attitudes, beliefs, and experiences will influence their willingness to deprescribe.55,56 Primary care clinics can send information to their patients via letters or brochures and invite them to participate in a medication review process with members of their health care team. A possible explanation for the observed success of education targeting patients to reduce SH is that these interventions simultaneously addressed multiple barriers: patients received the information directly from a known influencer; patients were encouraged to discuss their SH use with their prescriber, thus overcoming prescribing inertia; and both patients and prescribers increased their knowledge of the benefit to harm ratio associated with SH use and the potential alternatives for managing insomnia. 57 Community pharmacists are readily accessible to the public, uniquely positioned to easily identify chronic SH use, and are poised to easily distribute patient and provider educational materials.
Strategy 2: Interventions to Overcome Prescriber Barriers
The QI teams’ strategy should also consider interventions directed at prescribers given the demonstrated effectiveness reported in the literature. These can include education and training, drug utilization reviews, and computer alerts indicating a potentially inappropriate prescription by medication, age, dose, or disease. Educational interventions were effective when they facilitated patient engagement and provided information on the limited evidence supporting chronic SH use and the associated harms, as well as the effectiveness of alternatives. Despite the ease with which they can be incorporated into prescribing software, decision support tools were less effective than prescriber education with patient engagement. Monitoring and tracking practice-wide rates of SH use provides benchmarks and enables goal setting. Most electronic health record software permit this function. We also note that an interdisciplinary team approach involving pharmacists and nurses can support drug utilization reviews and share the responsibility of educating patients and prescribers about SH harms and alternative therapies. Depending on the local context of the community practice setting and resources available, some or all of these interventions can be implemented.
Strategy 3: Interventions to Overcome Health System Barriers
Although policy interventions such as prescribing restrictions can have a large immediate impact on SH use, these interventions require tremendous state/province, community, and stakeholder engagement to be successfully implemented. These interventions are beyond the scope of QI teams, but policy interventions can be successfully implemented at a local level. In particular, enabling a multidisciplinary approach and implementing restrictive pharmaceutical detailing policies can effectively reduce SH use.
Conclusions
It is imperative to overcome barriers to reducing both initiation and continued SH use in the community to reduce unnecessary harm. We identified several intervention strategies with demonstrated efficacy in reducing SH use in community practice: engaging and educating prescribers may be effective; however, better results are achieved by providing patients with educational materials that highlight SH risks, how to taper therapy, and potential alternatives. More substantial reductions were observed when providing prescribers and patients with educational materials with or without engaging patients in CBT for insomnia. The strategies presented can guide multidisciplinary QI teams toward reducing the community burden of SH use.
Supplemental Material
sj-pdf-1-aop-10.1177_10600280211033022 – Supplemental material for Addressing Barriers to Reducing Prescribing and Implementing Deprescribing of Sedative-Hypnotics in Primary Care
Supplemental material, sj-pdf-1-aop-10.1177_10600280211033022 for Addressing Barriers to Reducing Prescribing and Implementing Deprescribing of Sedative-Hypnotics in Primary Care by Lisa Burry, Justin Turner, Timothy Morgenthaler, Cara Tannenbaum, Hyung J. Cho, Evelyn Gathecha, Flora Kisuule, Abi Vijenthira and Christine Soong in Annals of Pharmacotherapy
Footnotes
Acknowledgements
CS, LB, HJC, EG, FK, AV, and TM are members of the High Value Practice Academic Alliance. The High Value Practice Academic Alliance is a consortium of academic medical centers in the United States and Canada working to advance high value health care through collaborative quality improvement, research, and education. For additional information, please visit ![]() . We thank Patrician J. Erwin for her assistance in the development of the literature search.
. We thank Patrician J. Erwin for her assistance in the development of the literature search.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
