Abstract
Background:
False penicillin allergies lead to increased antimicrobial resistance, adverse effects, and health care costs by promoting the use of broad-spectrum antibiotics. The Infectious Diseases Society of America recommends the implementation of allergy testing.
Objectives:
The primary objective of this research was to estimate the number of pharmacist full-time equivalents (FTEs) required for an intervention aimed at determining penicillin allergy in hospitalized patients. Acceptance of pharmacists’ suggestions on antibiotic therapy are described.
Methods:
A quasi-experimental study was conducted in a 712-bed university hospital involving hospitalized patients with a suspected penicillin allergy and an infection treatable with penicillin. The time required for the intervention, which included a questionnaire, penicillin allergy testing (skin-prick test, intradermal injection, and oral provocation test), and recommendations on antibiotic therapy were measured to calculate the number of pharmacist FTEs.
Results:
A total of 55 patients were included. Scarification allergy testing was performed on 37, intradermal allergy test on 33, and oral provocation test on 26 patients. The intervention ruled out penicillin allergy in 26 patients, with no serious adverse effects. The intervention was associated with a median weekly pharmacist FTE of 0.15 (interquartile range = 0.12-0.25). The acceptance of pharmacists’ suggestions was high and led to 9 patients being switched to an antibiotic with a narrower spectrum of activity.
Conclusions and Relevance:
This study describes penicillin allergy testing and the number of median weekly hospital pharmacist FTEs required, which was approximately 0.15. These data may aid in the implementation of this safe intervention that promotes narrower-spectrum antibiotherapy.
Introduction
About 10% of the general population has a history of penicillin allergy documented in their medical records; less than 10% of them will have an allergic reaction when reexposed to these antibiotics. 1 False allergies to penicillin encourage the use of broader-spectrum, second-line antimicrobial agents. It also increases the length of hospital stay, adverse effects, health-related costs, risks of intrahospital mortality, and microbial resistance.2-12 In fact, the prevalence of Clostridioides difficile, vancomycin-resistant enterococci, and methicillin-resistant Staphylococcus aureus infections are higher in patients allergic to penicillins.3,13-15 The Infectious Diseases Society of America (IDSA) recommends adding diagnostic tests for penicillin allergies to antimicrobial stewardship programs to exclude false allergies. 16 In our institution and in previous studies, these tests are carried out by immunology, allergology, or infectious diseases (ID) departments, thereby limiting their accessibility.17,18 A 2017 multicenter, prospective study at 3 Toronto hospitals assessed the feasibility of diagnostic β-lactam allergy testing among hospital patients. 19 This intervention was spearheaded entirely by pharmacists from the antimicrobial stewardship program and led to a 4.5 times increase in β-lactam use over a 15-month period (P < 0.0001). 19 They concluded that it was possible to perform diagnostic allergy tests at the bedside of hospitalized patients. A New Zealand study published in 2019 assessed the implementation of a penicillin-allergy review service coordinated by hospital pharmacists. The service included a questionnaire, followed by an oral challenge test or referral to an allergy clinic, when relevant. About 80% of patients had their allergies withdrawn. 20
No study appears to report the time required for a pharmacist to perform allergy tests during a patient’s hospitalization. Furthermore, an intervention similar to those described above has not been evaluated within the Québec health care context (Canada), where practices differ. The main objective of this study was to assess the number of pharmacist full-time equivalents (FTEs) needed to implement an intervention aimed at confirming or ruling out penicillin allergies in patients hospitalized in a tertiary care center receiving antibiotics for an infection treatable with a penicillin.
Methods
Study Design and Setting
This was a prospective, transversal, quasi-experimental study with a posttest analysis, conducted in a single intervention group. The intervention studied was carried out at 2 sites of a tertiary care facility with a total of 712 beds (Centre intégré universitaire de santé et services sociaux de l’Estrie, Centre Hospitalier Universitaire de Sherbrooke, Hôtel-Dieu and Fleurimont sites). An antimicrobial stewardship program has been in place since 2005 and includes 1 pharmacist FTE and collaborating ID physicians. The recruitment period lasted 27 weeks, running from February 12 to September 20, 2019. Four pharmacists were involved in collecting and analyzing data. The work was conducted in compliance with the requirements of the study site’s Institutional Review Board/Human Subjects Research Committee. The pharmacy department provided the materials and equipment needed to carry out the intervention. The pharmacy management team were not involved in the collection, analysis, and interpretation of data or the drafting of this article.
Patient Selection
Eligible patients were screened and selected by the pharmacist on a daily basis, with the collaboration of the ID physician, when relevant. Patients had to be at least 18 years old, hospitalized, and receiving antibiotic treatment. In addition, they must have had an allergy to ampicillin, amoxicillin, amoxicillin/clavulanic acid, penicillin (V, G, or unspecified), or piperacillin documented in their electronic medical record and have an infection that could be treated with a penicillin according to local or IDSA guidelines. Patients were excluded if admitted to the palliative care unit or the psychiatric ward, if they were unable to give consent (eg, dementia), when the intervention was contraindicated (eg, cutaneous allergic reaction during admission precluding interpretation of the allergy test), if they had cystic fibrosis, or if they had had an allergy skin test in the past to determine penicillin allergy. Detailed inclusion and exclusion criteria are available on request. The first step of patient selection was performed with Antimicrobial Prescription Surveillance System (APSS) software (Lumed) to identify patients aged 18 years or older who were hospitalized and on antibiotic therapy and had a documented penicillin allergy. This software is also used by the pharmacist on a daily basis for his regular activities at the antimicrobial stewardship program. Screening for potential inclusion was completed by consulting the medical records.
Study Outcomes
The primary objective was to estimate the number of FTEs required for the pharmacist of the antimicrobial stewardship program to complete the intervention. Secondary objectives were to describe (1) the number of penicillin allergies ruled out, (2) the number of pharmacist suggestions (about antibiotic therapy) approved by treating physicians, and (3) the number of suggestions that led to the use of an antibiotic with a narrower spectrum of activity, based on a clinical tool developed by a hospital pharmacist association (Association des pharmaciens des établissement de santé du Québec 21 ).
Description of the Intervention
The intervention was designed in accordance with the steps that would normally be performed by the antimicrobial stewardship team. The first step of the intervention was the identification of eligible patients. The following step consisted of a pharmacist filling out a questionnaire during an interview with the patient and consultation of their medical record. The questionnaire was adapted by the antimicrobial stewardship pharmacists from a public health document 1 (available on request). This questionnaire was used during a 3-month period in the year preceding this project and aimed at clarifying any mention of penicillin allergy in medical records of patients at our institution. If the penicillin allergy could be confirmed or ruled out on the basis of the questionnaire, no further allergy testing was planned. Otherwise, patients were offered skin tests to rule out penicillin allergy. Treating physician’s approval was solicited. First, a skin-prick test was performed by a pharmacist. It consisted of putting a 0.03-mL drop of each reagent on the patient’s forearm and then scratching the skin with a scarifier. The 5 reagents used were benzylpenicilloyl polylysine (PRE-PEN), sodium penicillin G (10 000 U/L), ampicillin (10 mg/mL), a positive control test with histamine (6 mg/mL), and a negative control test with NaCl 0.9%. All the products, except the histamine, were diluted and/or withdrawn in a syringe by a pharmacy technician as part of their usual daily workload. The test was considered positive if, 15 minutes after scarification, the diameter of induration associated with the drop of allergen and histamine was 3 mm larger than that of the saline solution. If the skin-prick test was negative, an intradermal skin injection was performed by ID physicians involved in the antimicrobial stewardship program. According to provincial regulations, pharmacists were not allowed to perform intradermal injections. This procedure involved an injection of 0.02 mL of the particular allergen and a control product into the dermis with a 25-G needle. In this study, the PRE-PEN, penicillin G 10 000 U/mL, ampicillin 10 mg/mL, and NaCl 0.9% injections were used. The reading of the intradermal skin test was done by the pharmacist. When the intradermal test result was negative, an oral challenge test was planned. With their concurrence, the test was proposed to the patient. It consisted in administering liquid amoxicillin orally at a dose of 50 mg (50 mg/mL, 10% of the usual dose). If the patient exhibited no allergic reaction within 30 minutes of administration, a 450-mg dose was given (50 mg/mL, 90% of the usual dose). If the patient showed no allergic reaction in the hour following the 450-mg dose, an immediate penicillin allergy was ruled out. Patient follow-up was also performed at 48 hours to check for nonimmediate hypersensitivity reaction. Overall, the pharmacist interpreted the allergy skin test results and monitored allergy testing at the patient’s bedside.
The allergist associated with the project trained the pharmacists to perform the skin-prick test and the ID physician to perform the intradermal skin injection. The pharmacists were also trained to read and interpret the skin tests by a nurse from the allergy clinic. The training period totaled approximately 1 hour for the ID physician and 2 hours for the pharmacists. A decision algorithm was created by pharmacists (including pharmacists from the antimicrobial stewardship program) and collaborating physicians (available on request). It was based on current practice at the allergy clinic of our institution.
Finally, when the penicillin allergy was excluded by questionnaire or allergy skin tests, the pharmacists suggested to the treating physician a modification to the current antibiotic therapy, when relevant, based on the antimicrobial stewardship pharmacist’s recommendation. When an oral challenge test was done, the recommendation was made at least 1 hour after the 450-mg dose. When the allergy tests were negative, a sticker was affixed to the patient’s hospital card to advise the removal of penicillin allergy from the patient’s medical record. Final results of the allergy testing were documented in the patient’s medical record and sent to their family physician and community pharmacist.
Statistical Analyses
For the primary outcome, the median pharmacist FTEs with an associated interquartile range were calculated by adding the time allowed for each of the following steps to complete the intervention: (1) identify eligible patients; (2) review the medical records and contact the community pharmacists, if required, to enquire about the allergy; (3) explain allergy tests to the patients; (4) computerize the prescriptions for skin allergy tests; (5) prepare the material for the allergy tests; (6) perform the allergy tests by the pharmacist, including the waiting times between the different doses planned in the allergy skin tests; (7) make suggestions about the antibiotic therapy to the physician; (8) send allergy tests results to the family physician and community pharmacist; (9) document the allergy test results in the medical record; and (10) explain the results of the allergy tests to the patient. The median weekly time to complete the intervention on all included patients was estimated, and total FTEs were calculated by dividing this time by 36.25 hours (number of weekly hours usually worked by a full-time hospital pharmacist). Pharmacy technician time for reagents preparation and ID physician time for intradermal injections were estimated by an open-ended question at the end of the study but were not included in the calculation of the pharmacist FTEs. To determine if the intervention is feasible, we consulted the pharmacy management team. They determined that a maximum of 0.5 FTEs could be added to the resource currently allocated for the antimicrobial stewardship program. Therefore, if the FTE estimated in our study was 0.5 or lower, the intervention was considered feasible.
To describe the patients’ characteristics, means (with standard variations) and proportions were estimated and reported for 3 groups: all patients, patients who refused part of the intervention, and patients who agreed to all steps of the intervention. Because this study was designed to estimate the time required to implement the service, no formal sample size calculation is reported. Statistical analyses were conducted with the Statistical Package for the Social Sciences (SPSS).
Of note, a preliminary project in the summer of 2017 was aimed at evaluating penicillin allergies using the same questionnaire, but no allergy testing was carried out. A total of 129 patients were questioned by a pharmacy student under the supervision of the antimicrobial stewardship pharmacist. This earlier project provided for ruling out penicillin allergies for several patients and reduced the number of potentially eligible patients.
Results
A total of 406 patients were assessed for eligibility (average of 15 patients per week), and 55 were included (average of 2 patients per week). Figure 1 presents the reasons for exclusion and allergy test results. Of the 55 retained patients, penicillin allergies were excluded for 26 of them subsequent to the intervention (45.5%). Of this number, 2 allergies were excluded based on the questionnaire, and 24 allergies were removed after allergy testing. As the result of excluding patients who refused allergy testing (n = 7), those who had an invalid allergy test result (n = 2), and those who had their treating physician refuse the intervention (n = 4), 61.9% (ie, 26/42 instead of 26/55) of the allergies were withdrawn from the patients’ medical records. Table 1 provides the baseline characteristics of the retained patients. The most common type of infection affected the pulmonary system; a mucocutaneous reaction was the most frequent type of past allergy documented.
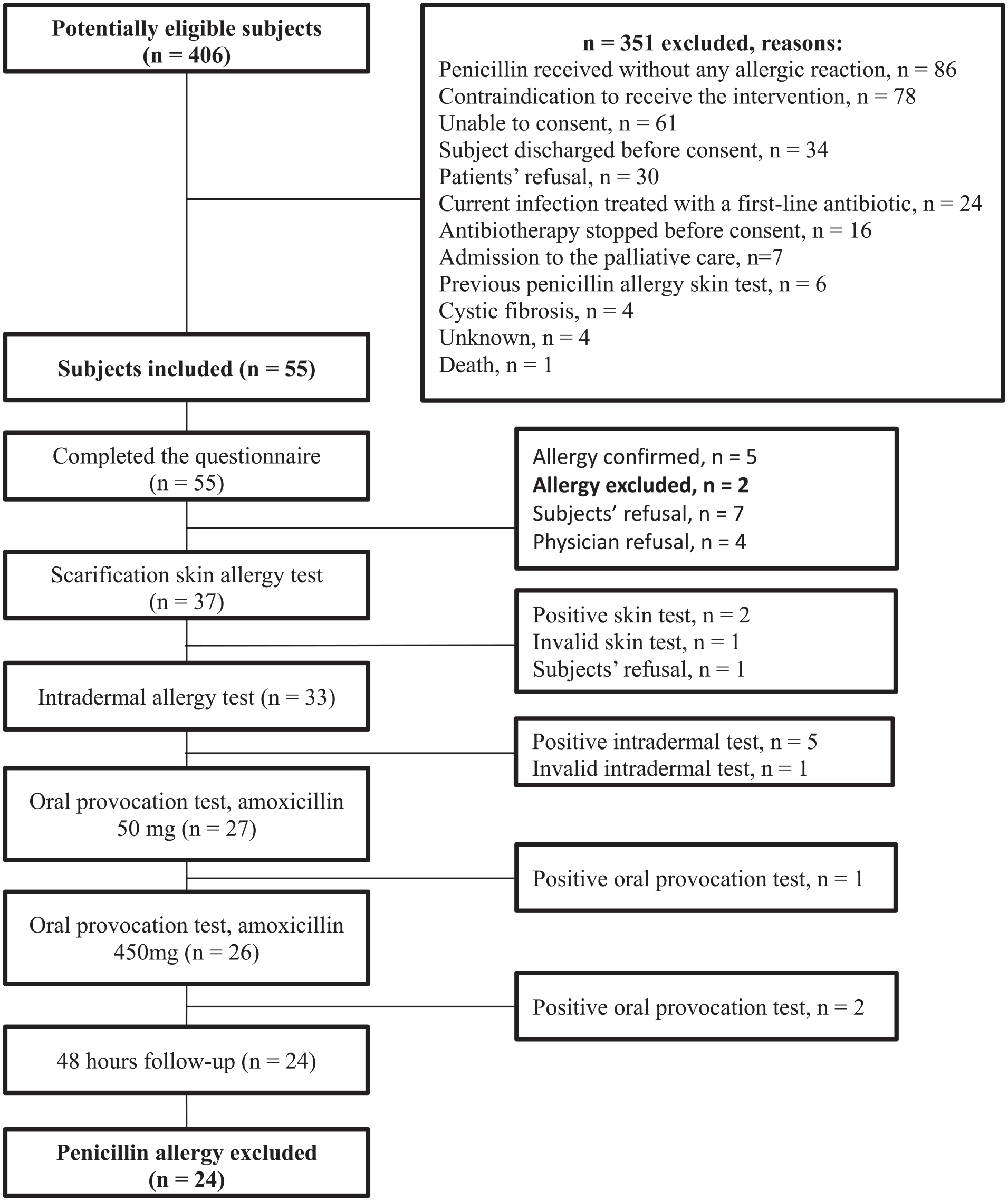
Study flowchart.
Baseline Characteristics of Patients Included (n = 55).
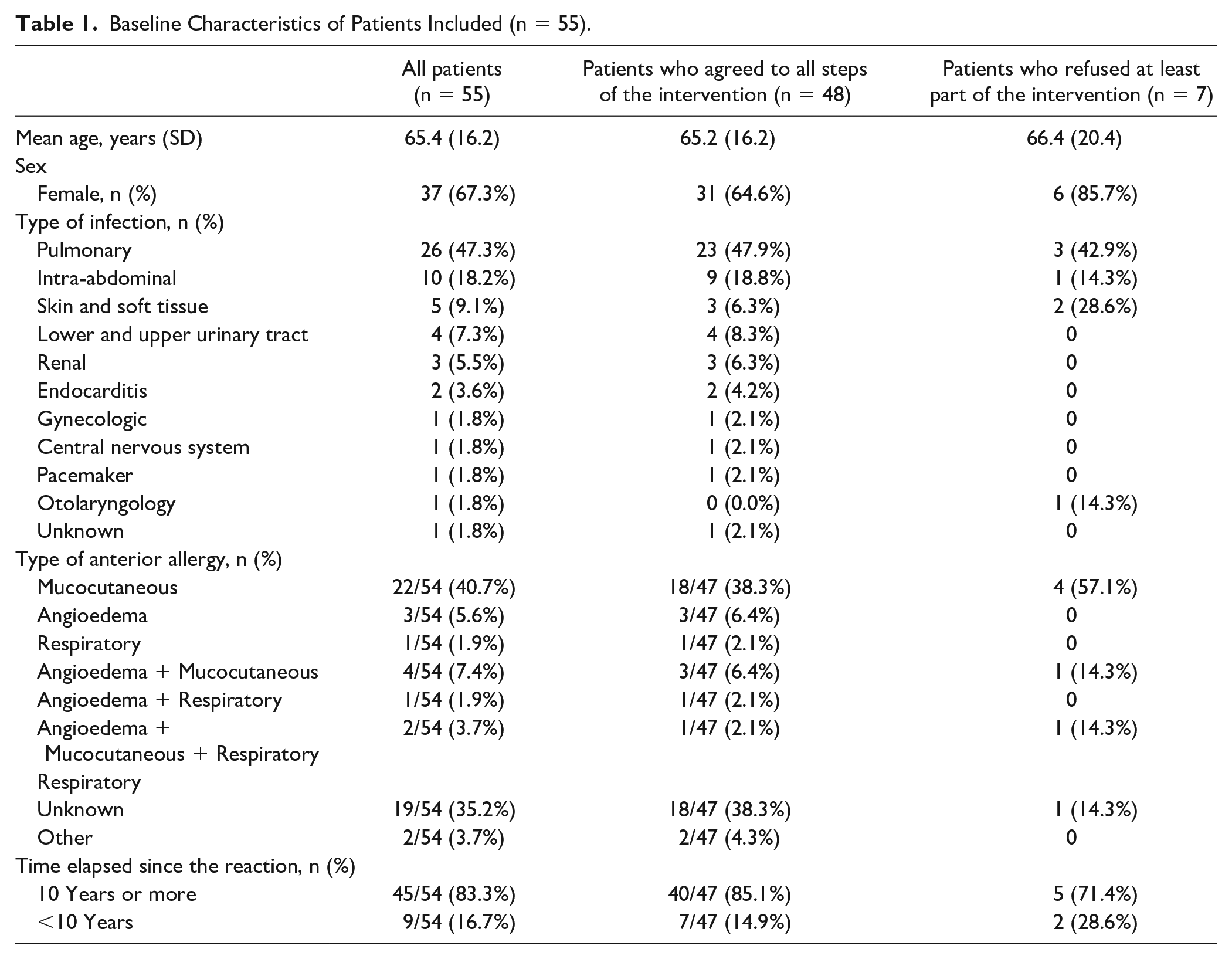
Seven patients refused allergy testing (see Table 1 for the characteristics). Three declined on the basis of their poor current clinical condition, 2 out of fear of another allergic reaction, 1 reported a lack of motivation, and 1 refused because of a fear of needles. Four treating physicians refused allergy testing. Two of the refusals were a result of the imminent discontinuation of the antibiotic therapy: 1 as a result of the patient’s imminent discharge and the last 1 because of the patient’s poor clinical state. Three of the 4 cases had no ID physician involved in the care of those patients.
The pharmacist intervention involved an estimated workload of 0.15 FTEs (Interquartile range (IR): 0.12-0.25) for a hospital pharmacist working 36.25 h/wk (times required for several steps are detailed in Table 2). Pharmacy technician time was estimated to be 30 min/wk for the preparation of multiple syringes of the reagents, and ID physicians spent 15 minutes per patient for the intradermal injection, when completed.
Steps Included in the Intervention and Estimated Times Associated.
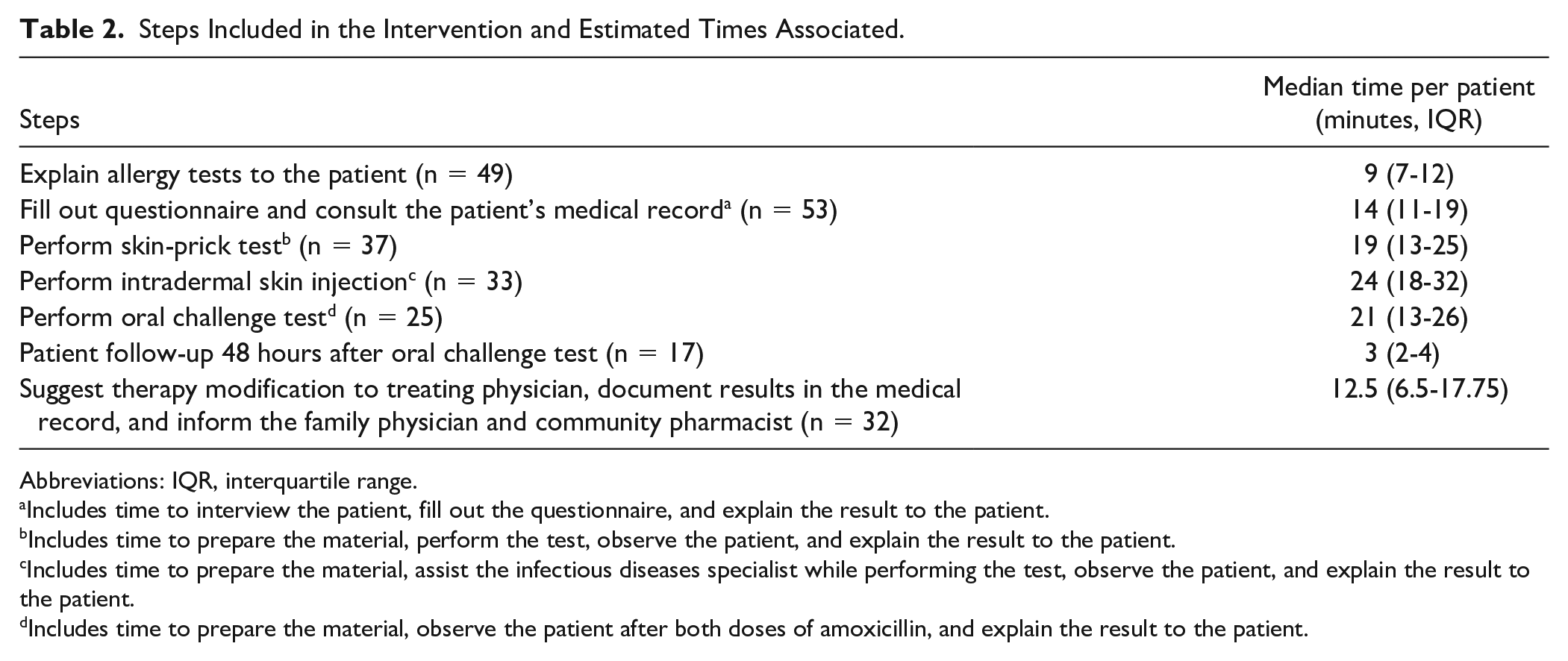
Abbreviations: IQR, interquartile range.
Includes time to interview the patient, fill out the questionnaire, and explain the result to the patient.
Includes time to prepare the material, perform the test, observe the patient, and explain the result to the patient.
Includes time to prepare the material, assist the infectious diseases specialist while performing the test, observe the patient, and explain the result to the patient.
Includes time to prepare the material, observe the patient after both doses of amoxicillin, and explain the result to the patient.
Of the 26 patients for whom allergy was ruled out, 16 suggestions were made to change the antibiotic therapy (61.5%); 13 were accepted by the treating physician (81.3%). Reasons for the 3 refusals were imminent discontinuation of the antibiotic therapy and patient discharge; 1 refusal was without explanation. Of the 13 suggestions to change the antibiotic therapy that were accepted, 9 led to the use of an antibiotic with a narrower spectrum of activity (69.2%).
Discussion
Based on our findings, a median weekly equivalent of 0.15 FTEs is required to clarify penicillin allergies in a tertiary care hospital. The FTE estimated is lower than 0.5, which supports our definition for feasibility of the intervention. The number of patients recruited per week was small, which could have been influenced by an earlier project using the same questionnaire to rule out allergies and because only patients receiving an antibiotic for which penicillin was indicated were included. In addition, implementing the intervention in a research project might have underestimated the number of patients who would be eligible for the intervention as part of usual care. Furthermore, the length of time needed to obtain the treating physician’s agreement, have the pharmacy technician prepare the reagents, and have the ID physician perform the intradermal injection and monitor the intervention at the patient’s bedside as well as the time required for the pharmacist to travel from one hospital site to another were not included in our estimation and constitute limiting factors. We suggest considering a daytime loss of productivity that, according to Guillotte and Charbonneau 22 represents 2 to 3 h/wk with an 8-hour workday. Empirically, adding about 2.5 hours of productivity lost daily per week would yield a total of 0.21 FTEs. Given our estimation, it would take a total of 1.15 to 1.21 pharmacist FTEs at our institution to integrate the intervention into the current workload. Time could be saved if part of the bedside monitoring were performed by nurses in the course of usual care. Therefore, the FTEs calculated are an empirical estimation that proves that the intervention requires little additional pharmacist resources and can be included in the regular pharmacy technician workload.
Overall, allergies were ruled out for 45.5% of the patients, which is lower than what was previously estimated by Guillotte and Charbonneau, 22 who determined that this type of intervention could rule out about 90% of false penicillin allergies. This could be explained by the fact that 85 patients were excluded from our study because they had taken a penicillin in the past with no allergic reaction. These patients were not accounted for in our estimation, although they were included in the estimation reported by Guillotte and Charbonneau. They represent a significant proportion of patients who would not have an allergic reaction during allergy testing. If they had been included in our study, we would have ruled out penicillin allergy in 86.6% of patients.
The 2017 multicenter, prospective study of Leis et al 19 conducted in Toronto confirms the feasibility of allergy skin tests at the bedside of hospitalized patients performed by a pharmacist from the antimicrobial stewardship program. That study, however, reported no specific information about the number of FTEs required. Unlike in our study, all the skin tests were performed by pharmacists who had a full day of training beforehand. Leis et al reported a negative rate of 94% for the skin allergy test, compared with 73% (27/37) in our study. Differences in study design such as the criteria for patient selection could explain this difference. Furthermore, the lack of experience of the pharmacists performing the skin-prick test and ID physicians conducting the intradermal injection might have influenced test results. In fact, appropriate procedures are necessary for adequate skin indurations and valid interpretation of the test results.
This study has several limitations. The sample size was small, and the number of patients refusing to participate was high. The small sample size might be a result of the study involving a single center and the exclusion of several patients. This would limit generalization of the results and any statistical analysis. The exclusion criteria, however, remain appropriate in a context of usual practice. For instance, the exclusion of patients who tolerated penicillin in the past, even if they still might have had this allergy status, influenced our results but would be more rational in the context of allergy testing.
The involvement of 4 different pharmacists in the intervention and data collection might have influenced the results. Moreover, no interevaluator reliability tests were performed. This situation, however, represents usual practice. In fact, several pharmacists are involved in the antimicrobial stewardship program and would likely alternate in providing the service. The questionnaire used for the intervention has not been validated, but it was adapted from a public health document published by a group of experts. Intradermal testing was done by an ID physician, which made the intervention more complex. This was intentional because pharmacists in Québec are not authorized to perform intradermal injections. This constraint does not necessarily apply to other jurisdictions in Canada or elsewhere. Although legislative changes are in the works, they will not allow pharmacists in Québec to give intradermal injections. The time required for this part of the intervention was not measured but was integrated in the antimicrobial stewardship activities of the ID physician. Skin and oral allergy testing procedures were slightly different between our study and the previous studies consulted. The actual workload and FTE calculation should be interpreted with caution and applied to a similar intervention. Our definition of feasibility should be evaluated according to the resources available in different institutions seeking to implement the intervention. Finally, switching to narrower-spectrum antibiotics that are less expensive could finance part of the costs of the intervention, but no specific analysis was done to explore this.
The strengths of the study include that there were no patients lost to follow-up. An effort to communicate the results of the allergy test to the community pharmacist and the primary care physician was made in order to remove all potential sources of incorrect information regarding the patient’s penicillin allergy. The results provide additional data on the resources required to implement the intervention. The intervention appears safe, with no serious reactions, and it promotes the use of narrower-spectrum antibiotics. It stands out as one of the first studies on the feasibility and relevance of an intervention involving penicillin allergy testing by pharmacists from the antimicrobial stewardship program, thereby extending the accessibility of the test to a wider proportion of the population. Finally, the study is an excellent example of interprofessional collaboration.
Conclusion and Relevance
To the best of our knowledge, this is the first study that provides an estimation of the time required for pharmacists from an antimicrobial stewardship program to conduct an intervention to assess penicillin allergy at the bedside of hospitalized patients. About 0.15 to 0.21 additional pharmacist FTEs were required to implement the intervention, which was safe and enabled the use of narrower-spectrum antibiotics. Patients benefited from this intervention because it eliminated the mention of penicillin allergy from their medical record and promoted rational use of antimicrobials. Despite the limitations of the study, it serves as an example for hospital administrations from tertiary-care centers of about 700 beds in planning this important antibiotic stewardship activity. Future studies could investigate the resources needed for allergy testing on a broader range of antibiotics.
Footnotes
Authors’ Note
Presentation of the research: Poster presentation at the Faculté de pharmacie, Université de Montréal, December 5, 2019.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funds were granted by the pharmacy department at the Centre intégré universitaire de santé et de services sociaux de l’Estrie, Centre hospitalier universitaire de Sherbrooke (CIUSSSE–CHUS) to conduct this study.
