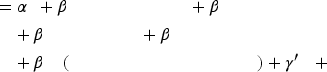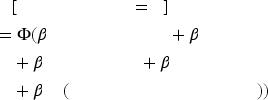Abstract
In the life sciences industry, pharmaceutical firms often collaborate with biotechnology firms to access technological expertise and R&D capabilities, enabling them to accelerate innovation and bring products to market more effectively. In this study, we examine the collaboration experiences of biotechnology startups and pharmaceutical firms in 287 biotech–pharma partnerships and analyze how firms’ experience impacts the likelihood of product commercialization. Experience is defined as the number of times each firm has participated in supply-based activities in biotech–pharma partnerships. We also conjecture that products developed through biotech–pharma partnerships are more likely to progress toward commercialization if both firms have similar levels of experience. However, when the partnering firms’ experience gap increases and they collaborate to develop the product jointly, we expect progress toward commercialization to become less likely. Our results show that both the biotechnology startup's experience and the pharmaceutical firm's experience increase the likelihood of progression toward commercialization. Our results also confirm that co-development moderates the relationship between experience gaps and the progression toward commercialization. Based on our findings, partnering experience and experience gaps are critical considerations as pharmaceutical firms engage in co-development projects with biotechnology startups. By focusing on partnering experience, our study contributes to the technology management literature and research on interfirm partnerships, specifically, in the biotechnology and pharmaceutical industries.
Keywords
Introduction
In knowledge-intensive industries such as the life sciences, product development processes are characterized by lengthy, costly, uncertain, and resource-intensive drug development cycles (Drakeman et al., 2022; Subramanian et al., 2020). At the same time, the life sciences industry relies on partnerships between pharmaceutical (hereafter also “pharma”) firms and biotechnology (hereafter also “biotech”) firms. In this study, we build upon existing work on partnering experience in the life sciences industry by focusing specifically on supply-based partnering experience and experience gaps between biotech startups 1 as suppliers and pharmaceutical firms as buyers. These partnerships are motivated by the benefits of combining complementary technologies or resources, as well as by the desire to boost the pharmaceutical pipeline (Nature Reviews Drug Discovery, 2008). Partnerships between pharmaceutical firms and biotech firms potentially enable moving drugs through stages of the discovery and development process, including clinical trials, regulatory approvals, and commercialization (Arora et al., 2009; Hora and Dutta, 2013). Given a broad product base, large pharmaceutical firms frequently seek scientific knowledge and experience that provide the foundation for research that biotech startups generally possess. Biotech startups, due to their small and informal structures, offer closer communication, greater flexibility, and faster decision-making compared to mature companies. However, they often lack the expertise and capital to take the drugs through the arduous process of obtaining approval from regulatory authorities to commercially market the drug. Established pharmaceutical firms, on the other hand, can offer access to financial, human, and operational resources, which are critical assets for their biotech partners’ survival, and can help them to enter commercial markets (Hitt et al., 2001; Katila and Shane, 2005).
According to existing research, prior partnering experience fosters value creation through partnerships (Anand and Khanna, 2000; Dyer and Singh, 1998). This experience enables companies to develop relational capabilities, such as the ability to select, govern, and manage partners. These capabilities can then be leveraged for value creation in future partnerships. For the purposes of this study, we define experience as the number of times each firm has participated in supply-based activities in biotech–pharma partnerships. Furthermore, the partnering experience of both firms may differ, which can place them in different phases of organizational learning (March, 1991). This can impact value creation and the outcome of the partnership.
We are interested in the progression of drug candidates toward commercialization in such partnerships. Given the complexity of the drug development process and the length of the drug development cycle (i.e., 10–14 years) (Danzon et al., 2005), we define progression toward commercialization as the advancement of a drug through the drug development process, including FDA approval. Progression toward commercialization occurs when a product receives FDA approval and becomes ready for market launch, or advances from one stage in the drug development process to the next, reflecting progress toward approval (Arora et al., 2009; Hora and Dutta, 2013; Lerner et al., 2003; Subramanian et al., 2022). 2
Our main research question related to experience and experience gaps in biotech–pharma partnerships is therefore: How is progression toward commercialization in supply-based biotech–pharma partnerships related to (i) partners’ supply-based partnering experience and (ii) the experience gap between the two partners? We hypothesize that supply-based experience positively and experience gaps negatively affect the likelihood of progression toward commercialization in biotech–pharma partnerships.
Drawing on relational governance mechanisms (Cao and Lumineau, 2015; Poppo and Zenger, 2002), we recognize that biotech–pharma partnerships can also involve technology co-development. In such arrangements, both partners agree to devote financial, physical, and human capital for jointly developing the product, in our case, the drug (Savva and Scholtes, 2014; Tian et al., 2021). This investment requires the partners to share know-how and costs to develop technology, learn from one another, detect asset deficiencies, and improve their capabilities, which are essential elements for turning innovative projects into commercial success (Hagedoorn, 2002). For example, in July 2004, mondoBIOTECH, a biotech startup firm, and Bachem, a well-established peptide manufacturing company, announced a partnership to support the development of mondoBIOTECH's lead compound, Aviptadil. Under the agreement, mondoBIOTECH manufactured and supplied Aviptadil for clinical trials, while Bachem contributed manufacturing and process-development capabilities. The collaboration involved coordinated development activities related to the compound (Bachem, 2004). Another example is the 2009 collaboration between the biotech firm, Biocon and Amylin Pharmaceuticals to co-develop, commercialize, and manufacture a novel peptide-hybrid therapeutic for diabetes, under which Biocon manufactures and supplies the compound for the collaboration (Biocon, 2009). Given the prominence and benefits of co-development, we therefore pose the following additional research question: How does technology co-development in supply-based biotech–pharma partnerships affect the relationship between experience gaps and progression toward commercialization? We focus on co-development within the supply-based partnerships. 3 We conjecture that products developed through supply-based partnerships with minimal experience gaps are more likely to progress toward commercialization when both partners collaborate on the product's development.
We test our hypothesized model using dyadic data on 287 partnerships between biotech startups as suppliers and established pharmaceutical firms as buyers announced between 2004 and 2015. The merged data for the analysis was obtained from five different sources: Recap IQ, Compustat, Orbis, Patstat, and individual company websites. The findings show that supply-based partnering experience is critical in partnerships where biotech startups are suppliers to pharma firms, together with the need to assess the asymmetries between a firm's own partnership history and that of the potential partner (Cuypers et al., 2017). To further interpret the empirical findings, we also conducted a series of post-hoc interviews with managers and executives from biotech startups and pharma firms.
Background: Biotech–Pharma Partnerships
Biotech–pharma partnerships are collaborations between often younger and typically smaller biotechnology firms (e.g., Biocon or BioNTech) and larger pharmaceutical firms (e.g., Amylin Pharmaceuticals or Pfizer). Over the past years, the prevalent form of collaboration between biotech and pharma firms has been through co-development, corporate venture capital, accelerators, incubators, licensing, joint ventures, or acquisitions (Brower, 2005; Chakma et al., 2013; Higgins and Rodriguez, 2006; Shan et al., 1994). Despite good intentions and often substantial investments, many biotech–pharma partnerships fail to achieve their intended objectives (Baum et al., 2000; Kehrel et al., 2016).
Younger biotech firms are prone to the liabilities of newness and smallness (Asgari et al., 2025; Stinchcombe, 1965). They often lack the resources needed for later-stage development, and for commercializing and bringing new therapies and drugs to market. One way for them to cope with the liabilities of newness and smallness, and to increase the likelihood of successful drug discovery and development, is by establishing exchange relationships with established and larger pharmaceutical firms. With their innovative early-stage research, they aim to join forces with larger pharmaceutical firms. Finding a partner can be challenging for biotech startups since they have several disadvantages compared to more mature firms. They are characterized by limited financial and organizational resources (Katila and Shane, 2005), a high level of quality uncertainty (Hannah and Freeman, 1984), lower levels of social approval due to no or limited prior exchange partners (Boeker, 1989), and rudimentary operational and management capabilities (Terjesen et al., 2011).
From an inter-organizational perspective, biotech startups and pharma firms can offer substantial mutual opportunities due to the complementarity of their assets. Biotech firms can gain support in the form of tangible resources, such as human capital, technical resources, and financial resources, as well as intangible resources, such as specific market and regulatory knowledge. Pharmaceutical firms that gain access to new technologies can benefit from the technological and scientific expertise of biotech startups in two ways. First, they can use the acquired knowledge to stay competitive in the market and even position themselves as leaders in innovative and novel drug development (Kennedy et al., 2023; Khanna, 2012). As Pan et al. (2010, 463) stated, the “biotechnology industry is establishing itself as the discovery arm of the pharmaceutical industry.” Similarly, Kennedy et al. (2023, 1) state “that innovative drug discovery is now largely driven by small biotech companies and academic labs, and that large pharma companies rely on buying or partnering with small startups in order to sustain their portfolios.”
Second, biotech–pharma partnerships can lead to an array of complex learning opportunities from the partnership. Established pharma firms can develop their capabilities by collaborating with biotech firms, which will assist them in the identification, selection, and management of biotech startups (Baum et al., 2000; Lacetera, 2001). Biotech startups can also develop their partnering, managing, and manufacturing capabilities through feedback from the established pharma firm (Wiederrecht et al., 2006). Perhaps more importantly, these partnerships enable biotech startups to develop market legitimacy, which can open opportunities for additional partnerships with other customers or alliance partners in the industry.
While research shows that both parties benefit from joining complementary assets in biotech–pharma partnerships, not all partnerships achieve their intended goals. Additional experiences and capabilities are necessary for biotech–pharma partnerships to be successful (Cabello-Medina et al., 2020). In the following section, we draw on the literature of absorptive capacity and organizational learning to shed light on several factors that impact a product's progression toward commercialization in a biotech–pharma partnership.
Theory and Hypotheses
The concept of absorptive capacity helps in theorizing about the impact of partnering experience on commercial outcomes of collaborative activities in biotech–pharma partnerships. In their seminal paper, Cohen and Levinthal (1990) defined a firm's absorptive capacity as its ability to recognize, assimilate, and apply new knowledge for commercial purposes. This definition has later been extended by Zahra and George (2002), who argue that absorptive capacity should also encompass a firm's ability to transform knowledge, which they note is essential for problem solving, and thus, for commercial exploitation.
Prior research suggests that partnering experience contributes to a firm's absorptive capacity by improving its ability to acquire, assimilate, and exploit external knowledge (Cohen and Levinthal, 1990; Zahra and George, 2002). Experience gained through repeated interactions with partners enhances firms’ internal processes for understanding and interpreting new information that may be contextually relevant. Pharma firms partner with biotech firms to obtain and transform knowledge residing at these firms. Conversely, biotech firms partnering with pharma firms gain knowledge on how to obtain and transform knowledge residing at these firms.
Two distinct dimensions of absorptive capacity are present in a biotech–pharma partnership: (1) The function-specific absorptive capacity of a firm in working with a partner in a specific functional domain (in our case, supply-based partnership), and (2) the gap in function-specific absorptive capacity, which we define as the difference between the biotech firm's and the pharma firm's function-specific absorptive capacities. Both dimensions influence the problem-solving ability, joint problem solving and hence value creation in biotech–pharma partnerships (Brown and Eisenhardt, 1995; Takeishi, 2001).
Building on the considerations of function-specific absorptive capacity and a gap in absorptive capacity, the resulting conceptual framework shows how partnering experience in partnerships between the biotech startups and pharma firms is associated with progression toward commercialization (Figure 1). The next sub-sections present the four hypotheses that are part of the model.
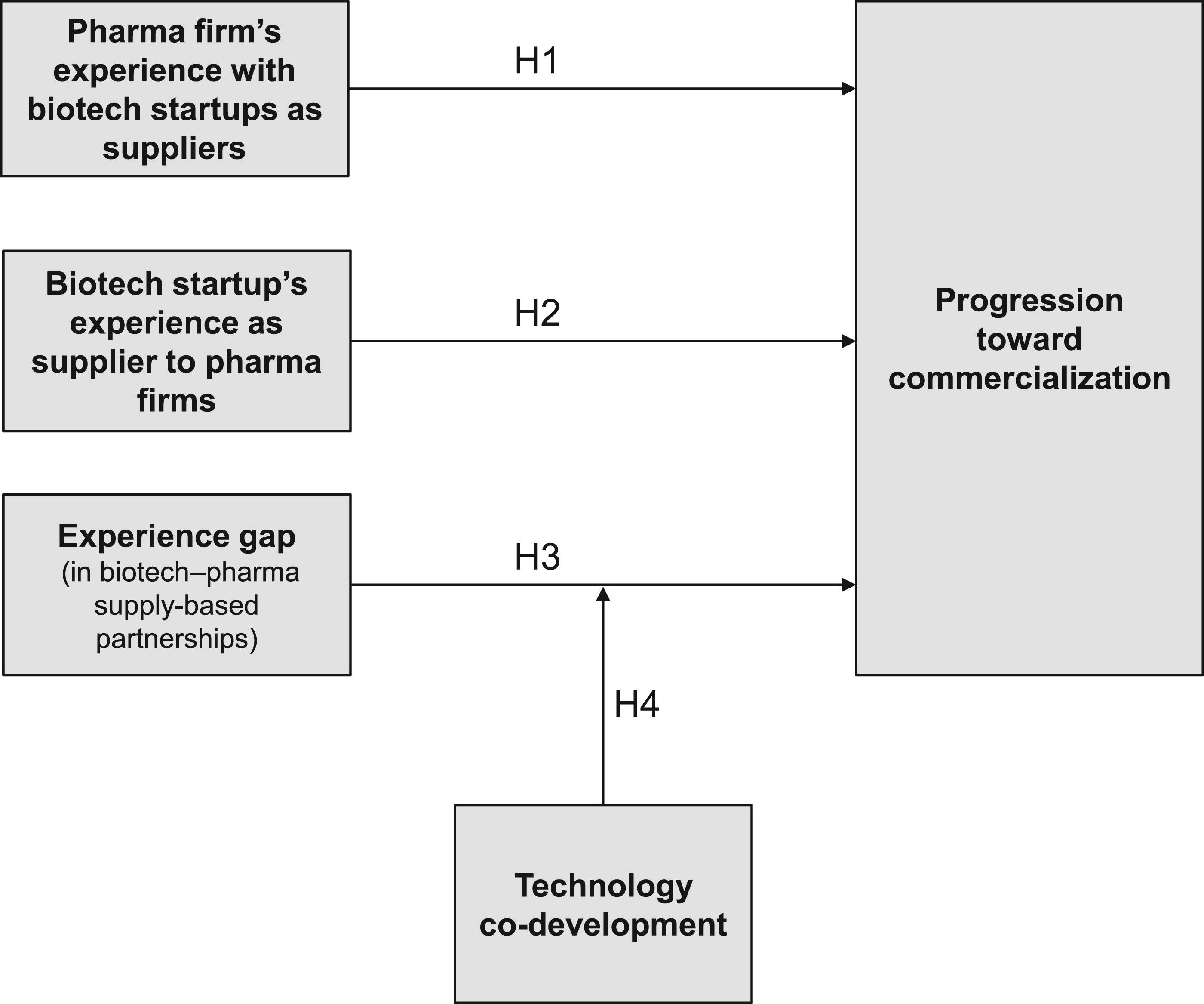
Conceptual framework.
Prior literature has explored several sources of experience that can contribute to value creation in partnerships: General partnering experience (e.g., Gulati et al., 2009; Hoang and Rothaermel, 2005), partner-specific experience (e.g., Findikoglu and Lavie, 2018), and function-specific experience (e.g., Yang et al., 2015). The existing literature on different types of partnering, including the industry of the empirical study, the outcome variable, and the overarching finding of the tested association(s), is summarized in Table EC-1 in the e-companion. These findings have led to varying explanations for the effects of different experiences, including research setting (e.g., industry, type of partnership) and the operationalization of relational rents (e.g., individual firm's performance measures or partnership-related performance measures). With the intent to address some of the mixed findings, our study focuses on a specific and highly relevant industry (the life sciences/biotech–pharma industry), a specific and emergent type of partnership (supply-based partnerships), and the potential of such partnerships to increase the likelihood of progression toward commercialization.
Partnering experience—defined as the overall number of prior partnerships formed—can improve a firm's ability to learn from and acquire valuable knowledge from partners (Cohen and Levinthal, 1990; Zahra and George, 2002). When firms repeatedly participate in similar engagements, they stimulate the process of organizational learning, draw inferences from prior activities, encode them in organizational routines, and retrieve knowledge for future similar activities (Levitt and March, 1988). As Lane et al. (2001, 1143) observe, “an organization's capacity to learn is not absolute but rather varies with the learning context,” which in our study is a relationship between a biotech startup and a pharma firm. The pharma firm must exhibit an ability to recognize, assimilate, transform, and exploit knowledge from the biotech startup in a supply-based collaboration (i.e., the pharma firm's function-specific absorptive capacity as a buyer). The supplier, in turn, must also exhibit a similar ability to recognize, assimilate, transform, and exploit knowledge from the pharma firm (i.e., the biotech firm's function-specific absorptive capacity as a supplier). In our industry study, the established pharma firm needs to be able and have the experience to absorb and integrate knowledge from biotech startups, and vice versa. And these abilities are generally learned through prior function-specific, that is, supply-based, collaborations.
Partnering capabilities obtained from prior engagements are embedded in mechanisms such as tools, processes, and dedicated organizational members (Kale et al., 2002). While these mechanisms are appropriate for existing or established firms, they are less appropriate for startup firms, such as the biotech startups in our study. Collaboration with biotech startups requires more time and effort than with mature firms, and the processes set up for mature firms cannot be applied to startup firms without modification (Kurpjuweit et al., 2021). Biotech–pharma collaborations typically necessitate flexible governance and alliance management capabilities on the part of the pharmaceutical firm (Pisano, 1990). As biotech startups often lack organizational experience or resources, the pharma firm must take on a mentoring or guiding role, for example, by helping with clinical trial design, regulatory filings, and scaling up. While the power imbalance is greater, so too is the need for trust and learning mechanisms.
Higher experience in partnerships between biotech startups and pharma firms in supply-based collaborations allows the pharma firms to improve their information-rich position in their supply network. First, it improves their ability to acquire information about potential partners. This allows them to codify the knowledge from prior partnerships and facilitate intra- and inter-organizational coordination. Second, it enables them to leverage their established reputation and brand themselves as an attractive partner for biotech startups (Pulles et al., 2016).
In summary, based on the above arguments on function-specific absorptive capacity, learning, and partnering experience, we hypothesize that:
H1: A positive relationship exists between pharma firms’ partnering experience with biotech startups in supply-based partnerships and progression toward commercialization.
Because the life sciences industry is characterized by a highly complex, tacit, and interdisciplinary knowledge base, absorptive capacity is crucial for biotech startups. It enables them to leverage external knowledge for innovation and commercial purposes. Cope (2005) emphasizes that in addition to the process dimension of entrepreneurial learning, the success of startup firms depends on the content dimension and their ability to engage in context-specific learning. By partnering with pharma firms, biotech firms can nurture the capability to learn from and internalize critical knowledge from the pharma firm. Through accumulated experience in partnerships, biotech startups establish legitimacy and improve their visibility in the market, enabling them to gain access to dedicated partnership programs and be more competitive (Baum et al., 2000).
The biotech firm's function-specific absorptive capacity is equally important in the drug development process within biotech–pharma partnerships. Absorptive capacity gained through function-specific, supply-based partnering facilitates speed and agility in relationships between biotech firms as suppliers and pharma firms as buyers. With accumulated experience in supply-based partnerships, biotech firms build their partnership management competencies, and pharmaceutical buying firms value the previous partnering experience of biotech firms as suppliers. More experience has been shown to represent higher productivity and innovation (Adegbesan and Higgins, 2011). Furthermore, while for buyers, exploring market opportunities with startup firms is an option, for startups with negligible experience, engaging in partnerships is essential for survival (Gulati and Higgins, 2003; Schoonhoven et al., 1990). Therefore, through recurring collaborations, startup firms improve their capabilities to identify and pursue marketplace opportunities (Kazanjian et al., 2002), thereby developing customer-specific relational capabilities.
In summary, absorptive capacity and learning enabled through partnering experience in supply-based biotech–pharma partnerships allows firms to acquire knowledge useful for the development process towards drug commercialization. Consequently, we hypothesize that:
H2: A positive relationship exists between biotech startups’ partnering experience with pharma firms in supply-based partnerships and progression toward commercialization.
Experience Gap in Biotech–Pharma Partnerships
Gap and fit (or asymmetries and symmetries) between relational variables have been studied in the context of supply chain (e.g., Wagner et al., 2012) and interfirm relationships (e.g., Griffith and Myers, 2005). Thomas and Esper (2010, 476) define relational asymmetry as a “lack of dyadic balance or proportionality of relationship attributes and/or behaviors.” Such disequilibrium in relationship attributes and behaviors can affect collaboration, communication quality, relationship leadership, and conflicts. The general conclusion from supply chain literature is that relational gaps, such as trust asymmetry (e.g., Michalski et al., 2019), power asymmetry (e.g., Nyaga et al., 2013), dependency asymmetry (e.g., Gulati and Sytch, 2007), and relational capital asymmetries (e.g., Villena and Craighead, 2017) negatively affect the operational or financial performance.
Building on the notion of the “absorptive capacity gap” introduced by Lascaux (2019, 5) as the “discrepancy between the two organizations’ ability to acquire, internalize, and utilize the external knowledge” where “one organization surpasses another in detecting, assimilating, and exploiting new knowledge,” we argue that a biotech supplier's absorptive capacity gained through prior partnerships with established pharma firms ideally should be similar to that of the pharma buying firm's absorptive capacity gained through prior partnerships with biotech suppliers. Function-specific absorptive capacity can be balanced where the buyer has a comparable level of function-specific absorptive capacity obtained through prior partnering in supply-based collaborations with biotech firms, as the biotech firm has as a supplier to pharma firms. Conversely, an unbalanced level of absorptive capacity can be detrimental.
As discussed in the preceding section a firm's prior partnership experience affects not only the way firms interact with each other but also the structures and tools that they form to communicate and collaborate in interfirm partnerships. Although the management literature recognizes that the experience gap among the partners can be a source of asymmetries in alliances (Yang et al., 2015), less is known of such asymmetries in biotech–pharma partnerships. In the context of our study, we define the experience gap between pharma firms and biotech startups as the difference in their partnering experience. We measure this as the difference in the number of supply-based biotech–pharma partnerships formed by the pharma firm and the biotech firm.
Consistent with the findings from other asymmetries among supply partners, we postulate for biotech–pharma partnerships that the experience gap attenuates the likelihood of progression toward commercialization. This attenuating effect can result from imbalances arising from the existence or non-existence of structures and the mismatch of expectations. For example, a gap in experience might indicate that while one firm already has specialized organizational departments or experienced personnel, the other is just starting to assign responsibilities to the employees to identify potential partners. Such an imbalance in experience can cause communication problems, as one partner does not have clear interaction channels (Mohr and Spekman, 1994). Furthermore, contrary to the less experienced partner, the more experienced one is expected to have dedicated resources and stronger support from the leadership. Suboptimal resource allocation and asynchronous decision-making processes between partners could lead to conflicts, which can affect the performance of the partnership (Terjesen et al., 2011). It is also probable that the two partners are in different phases of organizational learning (Levitt and March, 1988). While one firm could be exploring options without a clear understanding of the necessary commitment in this type of partnership, the other could be in an exploitative phase, whereby it applies the knowledge gained through prior experiences, codified in processes, organizational structures, or tools. In other words, the more experienced firm is “playing safe” by leveraging existing capabilities, that is, implementing and refining existing knowledge. In contrast, the less experienced firm is experimenting with possibilities and taking risks (March, 1991). As a result, there might be a mismatch of both dedication and expectations, which could jeopardize the success of the partnership. Therefore, we hypothesize that:
H3: A negative relationship exists between the experience gap in biotech–pharma supply-based partnerships and progression toward commercialization.
Technology Co-Development in Biotech–Pharma Partnerships
In interfirm relationships, the partners can choose between two forms of governance mechanisms: Contractual or relational (Cao and Lumineau, 2015; Wagner and Bode, 2014). In the first, formal contracts define the terms and conditions (Poppo and Zenger, 2002) to prevent opportunistic behavior and establish legal coordination of partners’ expectations and actions (Malhotra and Lumineau, 2011). In biotech–pharma partnerships, among others, contracts define the collaboration activities (e.g., supply, co-development, co-marketing etc.), intellectual property, financial arrangements (e.g., milestone payments, royalties), and control rights. In the second, relational governance is typically focused on collaborative continuity, where partners integrate their strengths and share knowledge. A common instrument of this type of governance is joint investments in relationship-specific assets, that is, assets tailored to the particular needs of a partnership (Dyer and Singh, 1998).
One type of relationship-specific investment often present in partnerships focusing on new products is technology co-development. Partners agree to jointly develop the product by sharing knowledge and costs (Hagedoorn, 2002). However, an imbalance in the experience of the two partners can affect the gains that they aim to achieve through this relationship-specific investment. Central to this argument is that firms that lack experience in comparison to very experienced firms might perceive the effort for technology co-development differently. A less experienced biotech startup is interested in increasing the number of exchange relationships to improve its legitimacy and reputation and is likely more inclined to take risks (Andriopoulos and Lewis, 2009). In other words, startup firms are keen on accepting new opportunities, irrespective of the availability of resources, and might rely on the experienced firms’ commitment to new product development. Likewise, a less experienced pharmaceutical buying firm with biotech firms as suppliers, which is exploring options for gaining new ideas externally, might expect the experienced biotech firm to lead the development process. Non-alignment of goals and responsibilities and a mismatch of the partners’ expectations can affect the partnership's performance (Cuypers et al., 2017), which can be especially critical in innovation processes (Kumar et al., 2020; Michalski et al., 2019). Hence, we hypothesize:
H4: The negative relationship between the experience gap and progression toward commercialization is stronger (i.e., more negative) for partnerships with technology co-development than for those without co-development.
Methodology
Sample
To investigate the role of specific types of experience in partnerships between biotech startups as suppliers and pharma firms as buyers, experience gaps, and technology co-development on the progression toward commercialization, we examine alliance partnership data between biotech firms and pharma firms from 2004 to 2015.
The data collection process followed several steps to create measures as the data was obtained from five different sources: Recap IQ, Compustat, Orbis, Patstat, and individual company websites. Initially, we obtained data about partnerships between biotech and pharma firms over 12 years (2004–2015) from Recap IQ. Recap IQ is a database that gathers information from news outlets about contracts (i.e., for partnerships among entities) in the biotech and pharmaceutical sector. It scans media coverage for pharmaceutical and biotechnology firms. The database includes a variety of alliances in the biopharma industry (for example, licensing agreements, mergers and acquisitions, etc.). Recap IQ also includes clinical trials data and clinical pipeline information. As Figure 2 shows, we retrieved the information on supply-based dyadic partnership agreements, which includes specific details such as the name of firms involved in a partnership, date of the alliance, contract specifics including types of alliance, stage of product development lifecycle, equity investment, technology, partnership scope, etc. Prior studies validate the credibility of this database by comparing competing databases of alliances and finding no significant difference in the data (Hoang and Rothaermel, 2005). Similar to other research studies that have used this data source (Hoang and Rothaermel, 2005; Robinson and Stuart, 2007), we retrieved 1201 supply-based dyadic partnership agreements (i.e., where one partner acts as a supplier and provides an ingredient or intermediate product, which the other partner uses to manufacture its mainstream product). Figure 2 depicts the data sources and the selection process, including the eligibility criteria at each step and the operationalization of the relevant variables.
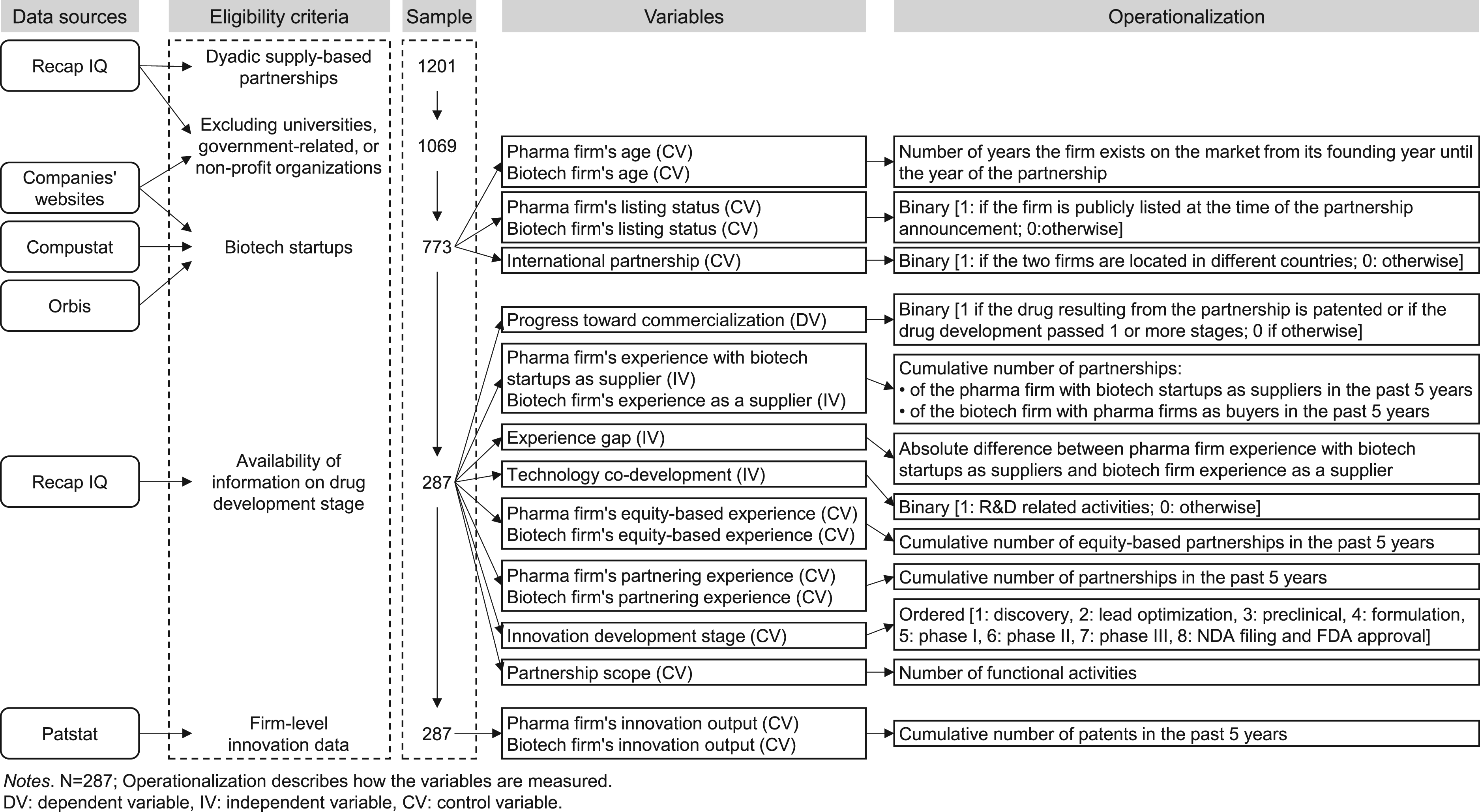
Description of the data collection process and the operationalization of the variables.
We started by selecting all dyadic supply-based partnerships, that is, all partnerships where “supply” is an activity of collaboration of the partnership agreement. Then, we ensured that we only considered partnerships between biotech suppliers and pharmaceutical buyers. We excluded all dyads where one partner was a university, public research institution, hospital, governmental organization (e.g., ministry of health or department of defense), or non-profit organization (e.g., MedicAlert Foundation). This first exclusion reduced our sample to 1069 partnership agreements. For each of these partnerships, from the Recap IQ database, we obtained the following details: The name of the biotech firm, the name of the pharma firm, the year of the partnership establishment, and the type of partnership.
The purpose of our second exclusion criterion was to ensure that the biotech firms in our sample are startup firms. Startup firms are typically young, innovative companies operating in knowledge-intensive industries with rapid technological development (Asgari et al., 2025; Shan et al., 1994; Stuart et al., 1999). Undoubtedly, the life sciences industry operates under hyper-competition, whereby being innovative is imperative for biotech entry and survival (Jaakson et al., 2011). Although, in the existing literature, the most commonly used criterion for determining startup firms is the age of the firm, there is no consensus about the cut-off point. Some studies consider startup firms until their 15th year of existence (Anyadike-Danes and Hart, 2018), while others are more rigorous, considering firms that operate for eight years (Vaznyte and Andries, 2019). This discrepancy is often ascribed to the variability in the duration of new product development cycles across products and the variability across the age of startups and its stage of life cycle development (Agarwal and Gort, 2002). For example, Anyadike-Danes and Hart (2018) follow about 240,000 firms in the United Kingdom that were founded in 1998 and find that two-thirds of firms did not survive the first five years, and although survival chances improve after age 5, only 10% of the cohort survive to age 15. Considering the length of a drug development cycle (i.e., 10–14 years on average) (Danzon et al., 2005), and consistent with other studies conducted in the same industry (Hora and Dutta, 2013), we identified the higher end, that is, 12 years, to be more appropriate as a threshold age for biotech startups (Moustakbal, 2014). To apply this selection criterion, we obtained the founding years of the biotech firms and calculated their ages. We initially retrieved this information from the Compustat and Orbis databases. However, as most biotech firms are small and privately owned, they were not publicly listed. We extracted the founding year for those firms by manually checking their websites. We excluded 126 partnerships for which we could not retrieve the founding year of the biotech firm. Finally, as Figure 2 shows, the startup cut-off age reduced our sample to 773 dyads.
The third exclusion was shaped by the availability of information necessary to estimate our dependent variable, progression toward commercialization. Knowing when a drug transitions from one stage of development to the next was a critical element in assessing progression. We excluded observations without this information. As a result of this exclusion criterion, our sample decreased to 287 dyads. Finally, we control for prior patenting activity, which is a key indicator of innovative activity (Kumar et al., 2020). We obtained the number of patents held by each pharma and biotech firm from the Patstat database (Wagner and Goossen, 2018). Patstat is a web-based interface developed and maintained by the European Patent Office that provides a comprehensive and detailed overview of firms’ patenting activity worldwide (EPO, 2021). To match the company names listed in Recap IQ with those in Patstat, we developed a customized Scala algorithm.
Dependent Variable
Progression toward commercialization (PROG.COMM). Despite the length of the entire cycle, there is a possibility of failure at any stage, which makes the success of drug development highly uncertain. If a drug candidate shows promise in the preclinical testing stage of the nonclinical trials, the drug developer files an Investigational New Drug application with the relevant regulatory authority, such as the FDA. This application provides detailed information about the drug's chemical composition, manufacturing process, preclinical data, and proposed clinical trial design.
Thus, the drug development process (FDA, 2018) comprises exploratory research, drug discovery (including nonclinical activities such as basic research, target identification, lead discovery, and preclinical development and testing) and clinical development (including phase I, phase II, and phase III clinical trials, as well as submission and FDA review of a New Drug Application [NDA]). For the purpose of this study, progression toward commercialization is assessed within the drug development process, which includes (1) discovery or lead identification, (2) lead optimization, (3) preclinical development, (4) formulation development, (5) phase I clinical trials, (6) phase II clinical trials, (7) phase III clinical trials, and (8) NDA filing and FDA approval. If approved, activities can include manufacturing, supplying, and marketing.
Arora et al. (2009) argue that compounds with a higher expected probability of FDA approval are more likely to advance into clinical development. Lerner et al. (2003) examine the success of biotech–pharma alliances and, for their empirical analyses, term “success” as alliances transition into the next “major milestone in the [FDA] approval process” (p. 437). Similarly, Hora and Dutta (2013) and Dutta and Hora (2017) conceptually and empirically employ movement along the drug development process in the biotech–pharma alliances as commercialization success. The data from Recap IQ provides information for every partnership between a biotech firm and a pharma firm, whether the drug transitions through the stages and final approval. Progression toward commercialization is coded as 1 if, after the partnership, the drug candidate progressed to the next stage of development or was approved, and 0 otherwise. 4 This measure and its operationalization capture the progression toward commercialization rather than commercialization in the strict sense of a market launch (Arora et al., 2009; Hora and Dutta, 2013).
Independent Variables
Pharma Firm's Experience With Biotech Startups as Suppliers in Supply-Based Partnerships (
Biotech Firm's Experience as Supplier to Pharma Firms in Supply-Based Partnerships (
Experience Gap (
Technology Co-Development (
Control Variables
To isolate the effect of alternative factors that can influence the progression toward commercialization of a biotech–pharma dyad, we control for buyer-level, supplier-level, and dyad-level variables.
Pharma Firm Controls. Following Hora and Dutta (2013), we control for a range of partner-specific variables, including buyer's age, listing status, prior innovation output, prior equity-based experience, and prior general partnering experience. We measure age as the number of years that a firm has been in existence as of the date of the partnership's announcement. The listing status is a binary variable, where “1” indicates that a firm is publicly listed, and “0” otherwise. The next three variables represent the cumulative number of patents (sourced from Patstat), cumulative number of equity-based partnerships (sourced from Recap IQ), and cumulative number of partnerships (excluding the number of equity-based partnerships), respectively, within the past five years prior to the partnership announcement, consistent with prior research (e.g., Ahuja, 2000; Hoang and Rothaermel, 2005; Lee et al., 2015).
Biotech Firm Controls. In addition to Pharma firm controls, we also control for partner-specific variables for the supplier, that is, supplier's age, listing status (public vs. non-public), prior innovation output, prior equity-based experience, and prior general partnering experience. We measure these variables following the operationalization pattern of the buyer-level variables.
Dyad-Level Controls. Following Lee et al. (2015), we include a set of dyad-level controls: International partnership, innovation development stage, and partnership scope. International partnership is a dichotomous variable, where “1” is assigned to partnerships if the locations of partners are in different countries and “0” otherwise. Due to the differences in cultural, legal, and political environments, international partnerships are prone to more risks (Ghemawat, 2001), potentially affecting partnership success. The development stage of the innovation process is an ordinal variable where the ranking is assigned in the following order: (1) Discovery, (2) lead optimization, (3) preclinical, (4) formulation, (5) phase I, (6) phase II, (7) phase III, and (8) NDA filing and FDA approval. Since the probability of failure decreases as the drug moves from one stage to another (Giovannetti and Morrison, 2000; Lerner et al., 2003), it is expected that drugs in later stages of development at the time of partnership establishment are more likely to be commercialized. Partnership scope reflects the number of functional domains of partnering (e.g., research, development, manufacturing, and marketing). Broader partnership scope implies greater diversity of cooperative activities, stronger resource commitment, increased possibilities for exchanging ideas, and diversified access to resources (Jiang and Li, 2009). Thus, we expect a positive relationship between partnership scope and progression toward commercialization. We also control for the year of the partnership's announcement to account for any time effects.
Methods and Results
Model Testing
To test our hypotheses, we use a probit regression predicting the probability of a binary outcome (e.g., progression toward commercialization) based on a set of estimates in equations (1) and (2):
Table 1 reports the descriptive statistics and correlation matrix. The average age of the biotech firms is about 7 years (variable #7), whereas the average age of the pharma firms is about 37 years (variable #2). About 29% of the biotech firms (variable #8) are publicly listed, while the percentage of publicly listed pharma firms is higher, that is, about 60% (variable #3). A biotech firm has on average 0.82 supply-based partnerships (variable #17) with pharma firms, while a pharma firm has on average 1.60 supply-based partnerships (variable #16) with biotech firms in the five years prior to the partnership of interest. Interestingly, the average pharma firm experience is 9.28 partnerships captured as general partnering experience (variable #6). In contrast, general partnering experience for the biotech firms is 1.87 partnerships (variable #11). However, it is not surprising that the average general partnering experience of pharma firms is much higher than that of biotech firms. The average experience gap of the firms in the sample is 1.74 (variable #18). In about 43% of the partnerships, the firms perform technology co-development (variable #19) in the drug development process. Progress in the drug development process towards clinical trials or a commercial product is observed in 39% of the partnerships.
Descriptive statistics and correlation matrix.
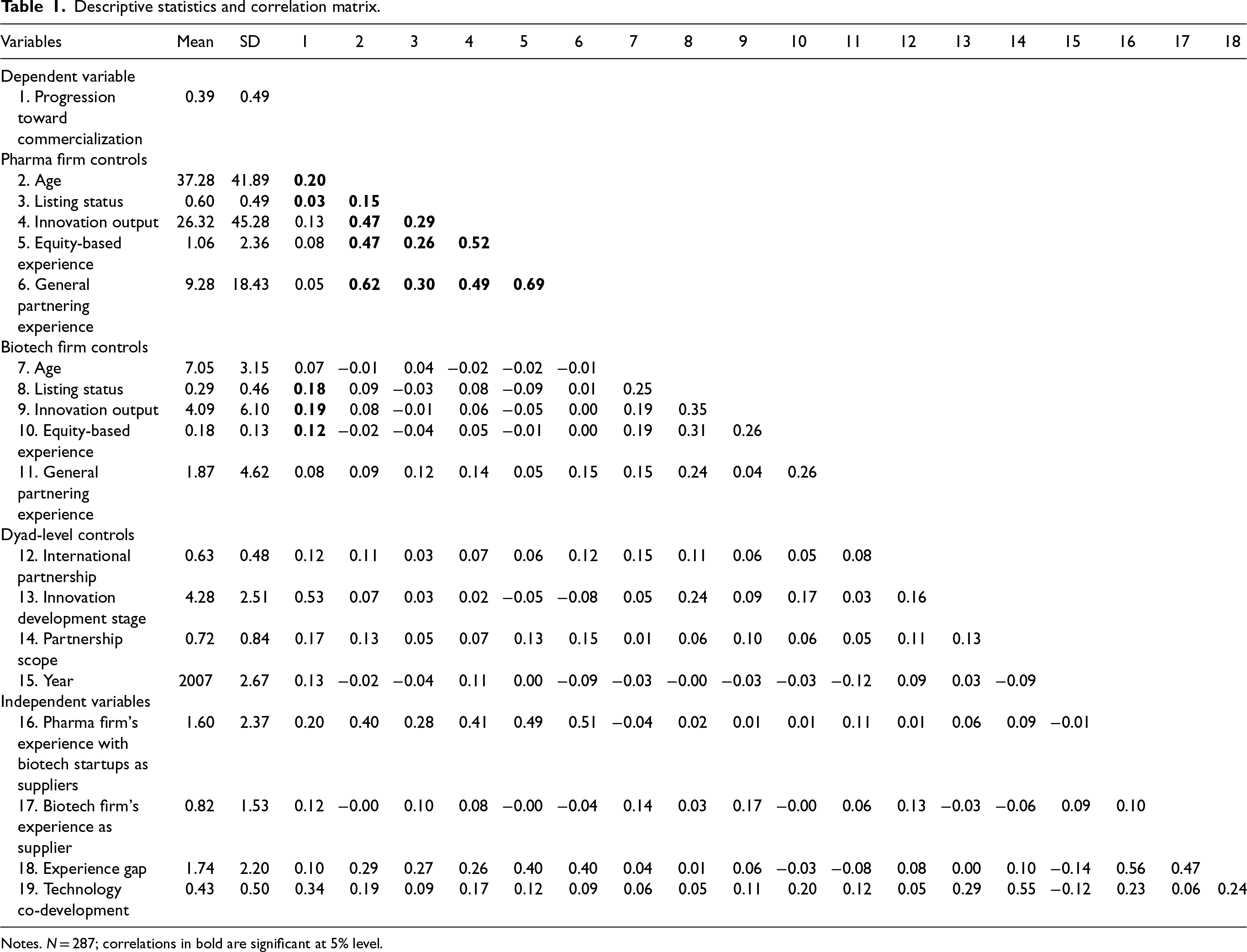
Notes. N = 287; correlations in bold are significant at 5% level.
To assess multicollinearity, we obtain variance inflation factors (VIFs) associated with each variable in the full model. Overall VIF values are 2.22, and VIF values for variables are below six, keeping VIFs within the prescribed limit of 10 (Neter et al., 1996). 5
Table 2 shows the results from the regression analysis. We employ hierarchical regression, that is, a multi-model step-by-step regression approach, where in each step we extend the model by adding additional regressors. Model 1 shows the impact of biotech-related, pharma-related, and dyad control variables on progression toward commercialization. Model 2 extends Model 1, considering the pharma firm's and the biotech firm's experiences in supply-based partnerships, and the experience gap. In Model 3, we add the binary variable for technology co-development, while Model 4 includes the interaction term between this variable and the experience gap.
Probit regression results for likelihood of progression toward commercialization.
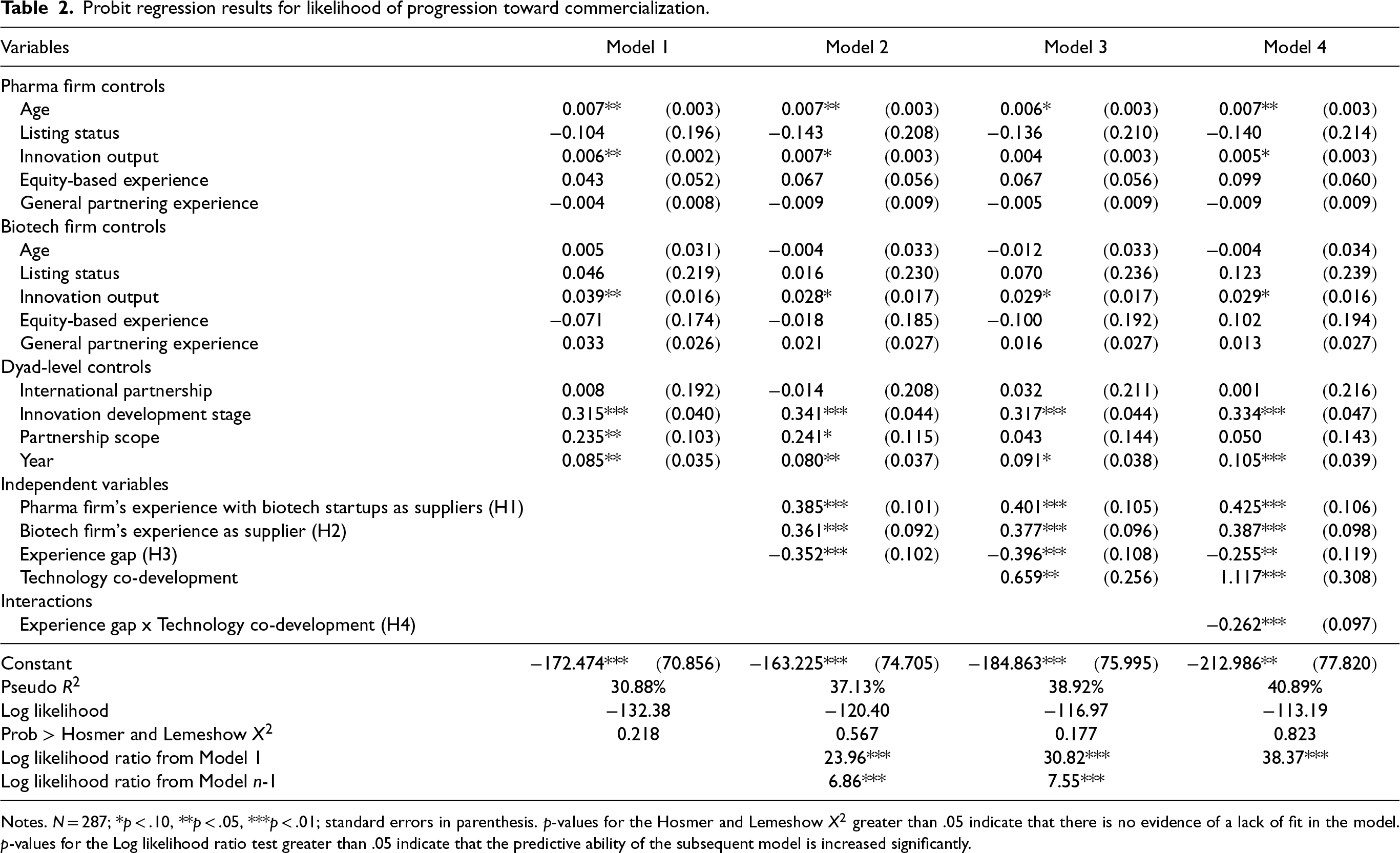
Notes. N = 287; *p < .10, **p < .05, ***p < .01; standard errors in parenthesis. p-values for the Hosmer and Lemeshow
To assess how well the model fits the data, we conducted the goodness-of-fit test by Lemeshow and Hosmer (1982). Based on the obtained results (
Moving from Model 1 to Model 4, we observe a gradual reduction of the log likelihood value (that is, from −132.38 to −113.19), indicating that the added variables at each step contribute to improvement of the overall model fit. We used multiple log-likelihood tests (Buse, 1982) to check whether the overall fit of the models improves compared to the baseline model and gradually in each step, that is, compared to the prior model. The test results (as shown in Table 2) are significant, indicating that the predictive ability of each successive model (i.e., model n) is better than both, the baseline model (i.e., Model 1), and the prior hierarchical model (i.e., Model n-1). The Pseudo R2 increases from 30.88% to 40.89%.
As shown in Model 4, the results support H1 and H2. As hypothesized, the findings show that there is a significant positive relationship between a pharma firm's experience with biotech firms as suppliers and the likelihood of progression toward commercialization (β=0.425; p < .01). Furthermore, the likelihood of progression toward commercialization increases significantly as the experience of the biotech firm in supply-based partnerships increases (β=0.387; p < .01). Furthermore, the results provide support for H3, that is, there is a negative and significant relationship between the experience gap and the progression toward commercialization (β=−0.255; p < .05). Furthermore, the analysis shows that this relationship is moderated by technology co-development, thus supporting H4 (β=−0.262; p < .01).
To enhance the robustness of our results and address potential concerns, we conducted a set of robustness checks. First, we modified the specifications and ran two alternative estimation models. We run logit regressions using the same specifications as the main models as an alternative estimation method (Model A in Table EC-2 in the e-companion). Both the probit and the logit regressions provide similar results. Another concern in regression arises when the dependent variable has fewer ones (successes) than zeros (non-successes) in the total number of cases. In our model, 39% of the observations are coded as “success” however, the number of total observations is relatively low, that is, 287. To address possible small-sample bias and validate the results, we run Firth logit regression (Firth, 1993), a type of regression estimation used in which small samples and moderately negatively skewed distributions of the dependent variable are typically observed. The results from the Firth logit regression (Model B in Table EC-2 in the e-companion) provided similar results.
Second, for robustness, we use alternative and additional measures for some independent and control variables. We start with an alternative measure for evaluating prior experience. Instead of evaluating prior experience through two different measures, that is, equity-based experience and general partnering experience (excluding equity-based experience) as control variables, in Model A in Table EC-3 in the e-companion, we used one combined measure, composed of the sum of the two. We label this variable as overall partnering experience and estimate it for both the pharma firm and the biotech firm, and find consistent results. With this approach, we show that the hypothesized result is not dependent on the base model specification, and also eliminate the regressors with the highest observed correlation (the highest correlation (r = .69, p < .01, that is detected between buyer's general partnering experience and buyer's equity-based partnering experience). In our main model, we consider the pharma and biotech firms’ supply-based partnering experience over the past five years. For robustness, we ran the same analysis, using cumulative supply-based partnering experience since their founding until the partnership of interest was announced. The results are similar to the main analysis and are presented in Model B in Table EC-3 in the e-companion. Partnerships can be formed for developing various product technologies. At a dyad-level, there is a possibility that the biotech firm and the pharma firm forming an alliance have had prior partnerships. This prior partner-specific experience can influence progression toward the commercialization of an alliance. Thus, we create a variable for this partner-specific experience and include it as a control variable for a robustness check. The results are presented in Model C in Table EC-3 in the e-companion where partner-specific experience is termed as common experience under dyad-level controls. As expected, the relationship between prior partner experience and progression toward commercialization is positive and significant. The results of the hypothesized relationships remain similar to the main analysis.
Our sample has 17 different product technologies. 6 In Model D in Table EC-3 in the e-companion, we extend the main model by adding a variable controlling for product technologies with 16 dummy variables, each representing a different type of technology that the partners were developing. The direction and the significance of the independent variables remain the same, indicating that the type of technology does not significantly affect the main results. Prior studies (e.g., Hora and Dutta, 2013) controlled for the number of employees as a proxy for firm size. Incorporating firm size as a control in our model would reduce the sample size to 59 dyads and lead to a loss of explanatory power. We did however, run two separate models, where we only consider the size of the pharma firm, with a sample size of 157 dyads and where we consider the size of the biotech firm with a sample size of 109 dyads. The results remain unchanged in both these models.
Third, although the VIF in our model is within the allowed values, we addressed the highly correlated independent variables to enhance confidence in our results. Specifically, in model E in Table EC-3 in the e-companion, we use orthogonal values for the pharma firm's experience with biotech firms, biotech firm's experience and experience gap (Golub and Van Loan, 2013). Our results from the estimation model are consistent with the base model. To account for the firms with multiple observations in our sample, we also rerun the model with clustered standard errors on both pharma firm level and biotech firm level (Cameron and Miller, 2015). The results are shown in Table EC-3 in the e-companion and are consistent with our base model.
Furthermore, we considered endogeneity issues that might arise in our model. Among the four explanatory factors, technology co-development is an endogenous variable because whether the firm engages in a joint collaboration or co-development as part of the supply contract is a strategic decision. Therefore, as recommended by Ketokivi and McIntosh (2017), we considered multiple instrumental variables: The pharma firm's prior technology co-development partnerships (
In addition, we also run a control function approach, which essentially is a two-stage estimation process (Petrin and Train, 2010). The control function approach in the second-stage equation, besides the instrumented technology co-development, adds the error term from the first-stage equations. Model B in Table EC-4 in the e-companion shows the results. The results indicated that the regression coefficients for the residuals were significant, suggesting that technology co-development is endogenous.
The robustness checks provide similar results to the base model. The only difference is observed on the impact of experience gap in Model A and Model B in Table EC-4 in the e-companion. Contrary to our expectations, the effect of the experience gap on progression toward commercialization is not direct, but rather conditional on whether the company invests in relationship-specific assets, in particular, whether the two firms devote resources for co-development of the product.
Post hoc Analysis
Our model suggests that there is a negative relationship between experience gap and progression toward commercialization which is moderated by technology co-development, as an activity which the two parties might decide to pursue or not as part of the partnership scope. While our results support our hypotheses, we further explore the direction of the experience gap. For this purpose, we modified the experience gap measure into a categorical variable, where “−1” is assigned to partnerships where the biotech firm has more experience than the pharma firm, “1” is assigned to partnerships where the pharma firm has more experience than the biotech firm and “0” is assigned to partnerships where both the pharmaceutical and biotech firms have equal experience. The results of the interaction effect of the model in Table EC-5 are presented in Figure EC-1 in the e-companion. The results show that the direction of the gap does not make a difference to the progression toward commercialization, that is, irrespective of whether the pharma or the biotech firm has more experience, the likelihood for progression toward commercialization remains at a similar level. We also ran robustness checks excluding partnering scope from the specification; the results remained consistent with our main findings.
Discussion
Discussion of the Empirical Findings
Economic performance is increasingly reliant on entrepreneurial activities and the creation and application of new knowledge (e.g., Nonaka and Takeuchi, 1995). Firms, especially those operating in knowledge-intensive industries, are subject to rapid technological changes (Ben-Jebara and Modi, 2021) and constant pressure to commercialize new products to market faster than their competitors (Parikh, 2001). To cope with these demands, firms search for additional sources of innovation, which makes networking and collaboration among firms on both vertical and horizontal levels increasingly important (Park et al., 2018).
In this study, we examine factors that may influence the likelihood of progression toward commercialization of products for partners with complementary resources. Specifically, we focus on partnerships under certain types of experiences, that is, the experience of the biotech firm being a supplier to a pharma firm, and the experience of the established pharma firm, and the association with progression toward commercialization. We also study the association between experience gap and progression toward commercialization and the moderating role of technology co-development on this relationship. Progression toward commercialization is defined as progression to the next stage of development or FDA approval. Thus, our 39% success rate reflects stage transitions as well as approvals, which explains its higher value compared to industry-wide FDA approval benchmarks.
In line with the absorptive capacity perspective (Lascaux, 2019; Zahra and George, 2002), the results show that the relationship between the biotech firm's experience and the pharma firm's experience in partnerships affects the progression toward commercialization. For a pharma firm, repeated partnerships with biotech startups in supply-based collaborations are critical for developing the necessary partnering capabilities and mechanisms to meet the requirements of this type of partnership. Furthermore, it ensures greater access to information and enables firms to advance their knowledge about collaboration opportunities with biotech startups. Similarly, as the experience of biotech firms in supply-based partnerships increases, they build capabilities for managing such partnerships. The improved partnering capabilities and the advanced understanding of the market opportunities of each partner through prior experience in supply-based biotech–pharma partnerships lead to a higher likelihood of relational rents in future partnerships.
We find support for H4. The results depict that for partnerships with technology co-development, the negative relationship between experience gap and progression toward commercialization is stronger when compared to partnerships with no technology co-development. Figure EC-2 in the e-companion shows the interaction effect. We plotted the predictive probability for progression toward commercialization for partnerships with and without technology co-development from low experience gap to high experience gap. 7 The figure highlights that when the experience gap is small, partnerships involving co-development exhibit higher predicted probabilities of progression compared to partnerships without co-development, reflecting the positive main effect of co-development. However, as the experience gap widens, the probability of progression declines more steeply for co-development partnerships. In other words, technology co-development amplifies the detrimental effect of experience gap. Next, to estimate the value of experience gap up to which partnerships with technology co-development are performing significantly better than partnerships without technology co-development, we estimated average marginal returns. The results show that partnerships with experience gap below 2.17 have a significantly higher predicted likelihood of progression toward commercialization for partnerships with technology co-development (Figure EC-3 in the e-companion). Thus, when the difference in the number of prior partnerships between pharmaceutical and biotech firms is three or higher, the experience gap between the partners outweighs the potential effect of technology co-development.
Post hoc Interviews
To substantiate and further interpret our empirical findings, we conducted a series of post hoc interviews with managers and executives from biotechnology firms and established pharma companies. Six interviews were conducted, comprising three executives from pharma companies and three from biotech firms. These interviewees had an average of 25 years of experience in the life sciences industry and held positions of Vice President or higher within their respective firms. Different general guiding questions were employed for these interviews, tailored to the two different perspectives of executives from biotech firms and established pharma firms. In discussing the experience of biotech firms, interviewees from pharma firms, as buyers, confirmed the significance of a biotech firm's experience as a supplier when assessing potential partnership agreements. They emphasized the importance of a biotech firm's technical knowledge and expertise within a specific therapeutic area or technology. Conversely, two interviewees from the biotech firms suggested that although pharma firms, in general, have extensive experience with alliances, they may not primarily consider the specific number of prior partnerships. Instead, they focus on whether the pharma company has a strong understanding of the therapeutic area, the ability to effectively integrate the biotech firm's technology and expertise, and a compatible corporate culture.
Results pertaining to H3, which indicate a negative relationship between experience gap and progression toward commercialization, were not that surprising to the executives of pharma firms when compared to the executives of biotech firms. One executive from a pharma firm suggested that when experience gaps exist, open communication and a focus on building trust are essential. Thus, in instances where buyer experience is much higher than the biotech firm's experience as a supplier, pharma firms generally pay more attention to the expertise and experience of the leadership team of the biotech firm. Regarding H4, that is why co-development may benefit under conditions of low experience gap, the interviewees from both biotech firms and pharma firms highlighted that the goal becomes broader than just developing a drug. In many instances, the partnership focuses and continues to develop a platform technology with applications in multiple therapeutic areas, leveraging the expertise of both partners. Conversely, a significant experience gap can create challenges during co-development, as it can lead to misaligned expectations, communication breakdowns, or difficulties in coordinating resource allocation.
Contributions, Implications, and Limitations
Scholarly Contributions
First, we contribute to the literature in operations and supply chain management, elaborating on supply-based partnerships between biotech firms and pharma firms and empirically addressing the significance of pharma firms’ capabilities to liaise with and manage biotech startups as suppliers and biotech firms’ capabilities to partner with pharma firms in supply-based partnerships (Joglekar and Lévesque, 2013). While literature has examined such capabilities in general, we focus on the life sciences industry. Given the peculiarity of the biotech–pharma alliances in the life sciences industry, examining the relationship between experience and progression toward commercialization adds to the existing literature, specifically industry studies (Joglekar et al., 2016; Terwiesch et al., 2020). Second, drawing on absorptive capacity, we contribute to the literature on partnering experience by theorizing and empirically testing that under certain circumstances, function-specific partnering experience is more important for the creation of relational rents relative to the general partnering experience. Third, we conceptualize the experience gap, arguing that the asymmetry in experiences can pose problems between partners in communication, collaboration, and a mismatch of expectations. By examining experience gap, we extend research on relational asymmetries in interfirm relationships. Fourth, we contribute to the growing literature on sourcing critical and novel knowledge. Sourcing or having access to innovation outside the firm boundaries by collaborating with external organizations has become increasingly important (e.g., Argote and Hora, 2017; Fritsch and Lukas, 2001). The supply base of a firm can be a source for critical and novel knowledge (e.g., Primo and Amundson, 2002; Yan et al., 2020) and can contribute to better financial performance (e.g., Dong et al., 2020). We demonstrate that forming partnerships between small biotech startups as suppliers and large and established pharma firms—with the purpose of combining know-how and developing innovations—also contributes to progression toward commercialization, thus adding to the literature on innovation success in such partnerships (e.g., Deng et al., 2022; Joglekar and Lévesque, 2013; Wagner and Bode, 2014).
Finally, we extend prior research that has considered co-development as one of many value chain activities in biotech–pharma alliances (e.g., Hora and Dutta, 2013) or predominantly as a matter of financial resources (e.g., Savva and Scholtes, 2014; Tian et al., 2021). Our study underlines that technology co-development on its own contributes to progression toward commercialization. Moreover, when experience gaps are low, co-development enhances progression toward commercialization; however, when gaps are large, co-development exacerbates misalignments and reduces success likelihood.
Managerial Implications
The prevalent forms of collaboration between startup firms and established firms have been corporate venture capital, accelerators, and incubators (Brower, 2005). Despite massive investments, many partnerships fail to achieve innovation objectives, which leads to other forms of collaboration, including supplier contracts. As a relatively unexplored phenomenon, it raises questions about the timing and specific conditions under which such partnerships are likely to succeed. In this study, we provide insights into these partnerships that can reinforce the choices of decision-makers. From the perspective of the established buying firm, managers should not be discouraged by early unsuccessful partnerships with biotech startups but rather perceive this as a process through which they develop the necessary capabilities to partner with biotech startups, which will allow them to harness the benefits at a later stage.
However, managers should recognize that young and resource-scarce startups are a distinctive category, and they should ensure the provision of sufficient resources and organizational support to leverage their potential. Since shared efforts by biotech startups and established pharma firms in developing the products can be beneficial for progression toward commercialization, managers of established pharma firms should also consider and deliberate technological collaboration as a component of the contract. This form of collaboration is especially effective given similar levels of experience of both partners. As an executive of a pharma company mentioned, during the post-hoc interviews, that co-developing a new drug with a biotech firm has inherent risks, and with low experience gap, risk sharing becomes less challenging due to trust in each other's capabilities.
Limitations and Future Research
This study has several limitations. First, when interpreting the findings, it is important to recognize that the study is based on the life sciences/biotech–pharma industry. Selecting one sector limits the generalizability of the findings. This industry is characterized by lengthy, costly, and uncertain product development cycles that are resource-intensive, which creates a need for collaboration with external sources (Rothaermel and Deeds, 2004). It is also an environment with an abundance of new biotech firms and scientific university entrepreneurs and a long history of collaboration between established pharma firms and biotech startups (Henderson et al., 1999). Nevertheless, it would be valuable to replicate the analysis and attempt to verify the findings within the same industry but with different types of drugs (Xu et al., 2023) and in other industries, particularly those with shorter and less risky new product development cycles. In industries where product development outcomes are highly uncertain (e.g., aerospace and defense, cutting-edge medical devices), experience and experience gaps in partnerships may have a similar impact. Conversely, when product development outcomes are less uncertain (e.g., fast-moving consumer goods and household appliances), considering experience gaps in partnerships might be less critical. Moreover, the dataset covers partnerships announced till 2015. As future research, replication with more recent partnerships would strengthen external validity.
Second, while we find a positive relationship between the individual experience of partners in biotech–pharma partnerships and the likelihood of progression toward commercialization, there may be other factors that explain success. Future research can examine factors affecting the innovation potential of each partner separately, such as R&D intensity, financial performance, partnership-related indicators, such as relationship length and partnership investment, and environmental factors, such as industry competitiveness. Moreover, future research could expand the scope of analysis from life science firms to individual managers, as managers have a significant impact on firms’ absorptive capacity (Volberda et al., 2010).
Third, we rely on a count of experiences of the two partners for explaining the potential for partnerships on the progression toward commercialization. The number of times the firms participated in partnerships is essential for the partners to develop specialized knowledge and capabilities. However, the ability to learn is a more complex process, and as such, it is not uniformly spread among firms, but it rather depends on a set of micro-level and macro-level factors. For example, in all our post hoc interviews, the interviewees highlighted that not only experience of biotech firms was crucial in forming partnerships, but so was the experience of the individuals in senior positions. Thus, while the biotech firms may be young, the leadership team of such firms may have highly experienced individuals. Future research can examine the effect of top management team experience on progression toward commercialization. Moreover, Fiol and Lyles (1985) summarize four contextual factors that affect the likelihood of learning: Corporate culture, organizational strategy, organizational structure, and the environment. Other studies have shown that firms vary in understanding and interpreting events (Daft and Weick, 1984), perceiving experiences and drawing lessons (Rose, 1991), developing an organizational memory (Walsh and Ungson, 1991), and utilizing their learning (Brown and Duguid, 2001). Understanding the differences in learning capacities and learning styles between the biotech startup and the established pharma firm could be rather informative and help in refining developed theories.
Conclusion
This study advances the research on relational rents in partnerships with complementary resources by revealing the benefits of experience in specific types of partnerships. The results show that, in the life sciences industry, function-specific partnering experience, rather than the commonly accepted general partnering experience, can be more relevant for success under certain circumstances. Further, our findings confirm that the relationship-specific investments in the form of technology co-development increase the likelihood of progression toward commercialization. Moreover, when the two partners have similar levels of supply-based partnering experience, technology co-development can enhance the likelihood of progression toward commercialization. Thus, the study encourages firms to explore opportunities through supply-based biotech–pharma partnerships while at the same time assessing the asymmetries between their own partnership history and that of the potential partners.
Supplemental Material
sj-pdf-1-pao-10.1177_10591478261419268 - Supplemental material for Experiences, Experience Gaps, and the Moderating Role of Technology Co-Development in Biotech–Pharma Partnerships
Supplemental material, sj-pdf-1-pao-10.1177_10591478261419268 for Experiences, Experience Gaps, and the Moderating Role of Technology Co-Development in Biotech–Pharma Partnerships by Stephan M Wagner, Manpreet S Hora and Ivana Mateska in Production and Operations Management
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Notes
How to cite this article
Wagner SM, Hora MS and Mateska I (2026) Experiences, Experience Gaps, and the Moderating Role of Technology Co-Development in Biotech–Pharma Partnerships. Production and Operations Management xx(x): 1–20.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.