Abstract
About 27% of patients with diabetes also suffer from depression, and the presence of co-morbid depression could increase the cost of care for diabetes by up to 100%. Several randomized clinical trials have demonstrated that physical and mental health are more likely to improve for diabetes patients suffering from depression when regular treatment for depression is provided in a primary care setting (called Collaborative Care). However, Collaborative Care requires additional resource utilization costs and a separate reimbursement model. When managing Collaborative Care, clinics must balance patient health outcomes with the program’s financial sustainability. Important operational levers in Collaborative Care are allocating care managers’ time to patients based on their requirements and the care managers’ staffing level. This staffing and workload allocation influences the revenue, costs, and patient health outcomes. We present a novel Markov Dynamic Programing model that, unlike existing approaches, jointly optimizes both staffing levels and treatment policies for Collaborative Care programs and quantifies the costs and benefits of collaborative care. Mathematically, we model Collaborative Care management at the clinical level as an infinite-horizon Markov Dynamic Program. The objective is a weighted sum of total patient quality-adjusted life years (QALYs) and the clinic profits. The model incorporates insurance payment, resource utilization costs, and disease progression of co-morbid diabetes and depression. We derive structural properties for the joint optimization of the staffing level and allocating care managers’ time to different patient categories. Using these structural properties, we develop a practical and easy-to-implement policy for staffing level and care managers’ time allocation that performs close to the optimal solution. We calibrate the model with data from a large academic medical center and show that our solutions can improve total QALYs and clinic profits compared to current practices. Our analysis also reveals key insights into payment models’ effects on Collaborative Care. Profit under the fixed-fee model responds nonmonotonically to payment rate increases, highlighting complex financial dynamics. Fixed-fee models show a threshold behavior, with high-intensity treatments becoming optimal only above certain payment rates. This threshold varies based on the profit-QALY weight balance, and this threshold is lower under joint-optimization than treatment-only optimization.
Introduction
Healthcare spending in the U.S. has reached $4.9 trillion in 2023, with approximately 75% of the costs incurred by patients with multiple chronic conditions. 1 , 2 Mental health disorders, like depression, are a common co-morbid condition with chronic physical conditions such as cancer, congestive heart failure, diabetes mellitus, and Parkinson’s disease. About one-third of patients with severe chronic illnesses also suffer from depression. 3
Patients with multiple chronic conditions, especially mental health issues, often receive fragmented care from different providers without central coordination. This fragmentation frequently causes poor communication, leading to worse outcomes, more emergency visits and hospitalizations, and higher costs (Kern et al., 2018). Patients with co-morbid depression and diabetes exhibit behaviors like reduced physical activity, overeating, inadequate self-care, and medication nonadherence. Consequently, they have poorer health, higher mortality, and cost 50–100% more than patients with only diabetes (Unützer et al., 2009). Despite strong evidence that treating depression improves diabetes outcomes and costs, psychiatric access is limited due to mental health provider shortages (Dampier, 2018), lack of insurance coverage (Bishop et al., 2014), and social stigma (Clement et al., 2015).
To address this, Collaborative Care was designed at the University of Washington (Unützer et al., 2002) to integrate mental and physical health treatment. Collaborative Care is a systematic treatment program for improving access to mental health resources for patients with co-morbid physical and mental health conditions. Under this program, mental health services are provided within the Primary Care Provider’s (PCP’s) office by a care manager who is typically a health professional such as a Licensed Social Care Worker. The care manager regularly consults with the PCP and often with a psychiatric consultant to provide regular follow-up and timely modification to the patient’s care plan, behavioral health therapies, and changes or adjustments to medication.
Over the past 20 years, over 80 randomized trials have demonstrated Collaborative Care’s efficacy for depression, including the most extensive study to date with over 1,800 patients showing a doubling of depression remission rates (Unützer et al., 2002). Consequently, the U.S. Surgeon General and the President’s Commission have endorsed it as a “best practice” (Unützer and Ratzliff, 2015).
Despite evidence of collaborative care’s effectiveness, adoption has been slow due to high maintenance costs and a lack of clear payer reimbursement pathways to cover implementation costs, resulting in uncertainty about the program’s financial sustainability. While randomized trials have typically focused on the clinical outcomes, the financial ramifications of the Collaborative Care program in practical implementation have not been adequately studied.
We partner with the Departments of Psychiatry and Family Medicine at a large academic medical center in the US to develop a model-driven approach to aid decision-making for managing the Collaborative Care program. We develop and test the Collaborative Care program for patients suffering from depression and diabetes. While Collaborative Care can be delivered to patients for any combination of mental health conditions, such as Generalized Anxiety Disorder, and physical conditions, such as hypertension, we select depression and diabetes as this combination constitutes the largest population of patients with co-morbid physical and mental health conditions and contributes a significant cost to the health system. Our model can easily adapt to other conditions with appropriate parameter value changes.
There are two primary levers for managing resources for Collaborative Care. The first lever is allocating care managers’ time to patients based on their needs. More complex patients will require more time from the care manager, increasing the workload for care managers and, in a fee-for-service payment model, the revenue per patient. The second operational lever for the clinic is the staffing level of care managers. Clinics often meet workload requirements with regular staffing levels and more expensive overtime hours. Lower staffing levels will lead to higher overtime costs for the same policy for allocating care managers’ time to patients. On the other hand, higher staffing levels will lead to higher wages for regular employees. Therefore, operating Collaborative Care efficiently requires the joint optimization of the policy of care managers’ time allocation based on patient needs and the staffing level of care managers. In this paper, we model the Collaborative Care program as an infinite-horizon Markov Dynamic Program to maximize the dual objectives of the clinic: Patient health outcomes and financial profits. The model’s objective reflects the clinic’s two goals, patient health outcome measured in quality-adjusted life years (QALYs) gained and clinic financial profits. The decision variables are the care manager’s time allocated to each patient type and the number of care managers required to manage the collaborative care program. Markovian dynamics is a standard approach in healthcare modeling, supported by prior literature on both diabetes and depression. This assumption is widely adopted due to its analytical tractability and acceptance in the field (Denton, 2018). Subsequently, we derive structural results for the joint optimization of the allocation of care managers’ time based on patient needs and determining the number of care managers. Using these structural properties of the optimal policy, we develop a computationally efficient and easy-to-implement model-based policy to determine the optimal allocation of care managers’ time for treatment and the staffing level. We validate our model and solution approach by testing on data from a large academic medical center. Our data consists of clinical lab tests and depression treatment over five years for 17,152 unique patients suffering from co-morbid depression and diabetes. We use a prediction-based method to estimate the model parameters from this historical patient data.
Finally, we perform a comprehensive set of numerical simulations to show that the policy recommended by the model can improve patient health and clinic profits compared to current practices and other heuristic approaches. We also derive several managerial insights on the impact of different aspects of collaborative care under different payment policies. These insights can guide clinic managers in managing collaborative care and help policy and insurance firms design payment policies for programs like collaborative care.
In this research, we do not consider granular decisions such as prescribing personalized depression treatment for each patient and scheduling individual appointments with the care managers. While optimizing such decisions could be an interesting problem to consider, it is beyond the scope of the current paper, where we consider the issue from a tactical perspective to set policy on care manager time for treatment across patient categories and overall staff levels.
The main contributions of this work are as follows. Our research is the first to study optimal resource planning and allocation for collaborative care management. We develop properties for optimal treatment plans, considering both the diabetes and depression state of a patient. Using real data and a practical policy, we provide insights on improvements that could be gained by a healthcare facility where they optimize treatment and staffing plans in collaborative care. Finally, we provide managerial insights on the impact of arrival rates, insurance plans, and weightage on financial and quality outcomes.
The remainder of the paper is as follows. In Section 2, we discuss related literature to our work. Section 3 describes the care management process in detail. In Section 4, we provide a detailed mathematical formulation of our model. In Section 5, we describe the theoretical results. In Section 6, we describe the estimation of the model parameters. In Section 7, we provide details of our computational analysis, and we follow that up with managerial and policy insights in Section 8. Finally, we conclude with future directions of research in Section 9.
Literature Review
Our work is primarily related to capacity management in healthcare operations. Related literature on chronic care management can be divided into four groups.
The first group of literature pertains to managing screening and treatment for a patient or a population of patients. Representative papers in this group are Alagoz et al. (2007), Shechter et al. (2008), and Ayer et al. (2012), which determine optimal patient screening or treatment decisions. The system state in these models is typically the health state of a single patient. Deo et al. (2013), Ayer et al. (2019), Lee et al. (2019), and Adams et al. (2023) model the sequential screening or treatment decision for a population of patients under resource constraints. The system state is typically the vector of the health status of each patient in the system. In these models, the decision-maker chooses which patients to treat or screen each period. An alternate approach is to formulate the system state as a compartmental model. In this formulation, the system state is the vector of the number of patients in each health state. Natarajan and Swaminathan (2017) uses this approach to allocate inventory procured through donor funding to patients in different health states. Ho et al. (2019) also uses the compartmental model for resource allocation for population disease management.
The second group of papers relates to capacity management for healthcare with patient disease dynamics. In the context of primary and chronic care, Liu et al. (2024) studies capacity allocation for the patient’s first appointment in coordinated care networks, where patients must visit multiple providers. Bavafa et al. (2019) investigates the impact of integrating nonphysician providers into primary care, analyzing their effects on patient health dynamics, physician decision-making, and overall healthcare outcomes. They also investigate the impact of physician payment schemes. Diamant (2021) investigates the scheduling practices of multistage outpatient health programs. In the context of behavioral health, Baek et al. (2023) investigates the problem of optimizing personalized patient interventions. In the context of inpatient workload, Liu et al. (2019) and Kamalahmadi et al. (2023) investigate capacity management to deliver care to patients in a hospital. Ahuja et al. (2020) shows empirical evidence that co-location and continuity in care improve health outcomes, and the continuity of care is more critical for more complex patients. They find that maintaining continuity of care provides significant gains for patients suffering from mental health conditions.
Our work is also related to articles such as Simon et al. (2001), Liu et al. (2003), and Yildirim et al. (2021). These papers evaluate the cost-effectiveness of Collaborative Care and depression screening programs through observational or simulation analysis. These papers typically do not optimize resource allocation decisions, and existing literature in this stream has not compared different payment policies.
More broadly, our study corresponds with the broader body of literature advocating for the use of data and analytics to enhance healthcare operations. Dai and Tayur (2020) offers an extensive examination of recent research in healthcare operations, while de Véricourt and Perakis (2020) and Mišić and Perakis (2020) concentrate on data analytics techniques for enhancing operational effectiveness in various organizations.
While there are many interesting papers in the fields as listed above, our paper is the first to study capacity and care management for the Collaborative Care model with physical (diabetes) and mental (depression) disease progression. The CMS has identified chronic care management of patients with multiple conditions as a critical component of primary care. 4 Our modeling approach is general enough to be adapted to other chronic care management programs with the appropriate model parameter values.
Second, right-sizing care management staffing levels and allocating care managers’ time based on patient severity are essential drivers for improving patient health outcomes and ensuring clinic financial sustainability. We contribute to the literature on chronic care management by explicitly capturing this trade-off. We derive structural properties for the joint optimization of staffing levels and treatment policies that determine the time allocated to patients for behavioral health services based on patient needs.
Third, we derive a computationally efficient and easy-to-implement model-based policy for simultaneous optimization of the treatment policy and staffing levels. Lastly, we derive relevant managerial insights on the different aspects of collaborative care under different payment models.
Collaborative Care for Depression and Diabetes in Primary Care
In the Usual Care (Unützer et al., 2002) model of treating diabetes patients with co-morbid depression, the PCP treating the patient for the physical health condition refers the patient to a psychiatrist if they think that the patient also requires psychiatric care. The patient is then responsible for setting up an appointment and subsequent follow-up with the psychiatrist. However, as described in Section 1, this Usual Care model has several challenges: Long wait times for appointments due to a shortage of available psychiatrists, failure of patient compliance, lack of regular patient follow-up, and difficulties with insurance coverage.
The Collaborative Care delivery model helps alleviate several of these issues. The principal change from the Usual Care model is the addition of a care manager to the PCP’s office. The care manager provides mental health services to the patient from the PCP’s office in consultation with the PCP and a psychiatric consultant. The primary activities of the care manager include—developing a detailed longitudinal care plan for each patient with benchmarks and goals for improvement; providing psychotherapy if required; counseling using evidence-based techniques such as motivational interviewing, behavioral activation, and problem-solving treatment; performing proactive follow-up; medication management; managing patient registries for depression; participating in monthly case reviews with the PCP and submitting monthly time and billing statements for approved reimbursement.
The care manager performs the above actions in person, via computer, or by telephone. While some activities can be automated, those related to interactions with the patient are in person. The above set of activities provides the patient with timely, regular, and systematic care for their depression. This care is often offered at the same location where they get treatment for their physical health condition. Insurance providers such as Medicare, Aetna, Blue Cross Blue Shield, and others have instituted billing codes that reimburse care manager activities as services received within primary care. 5 Thus, the Collaborative Care program mitigates the challenges related to timely access to professional mental health services, unnecessary use of one-on-one visits with a psychiatrist, insurance coverage, and stigma associated with visiting a psychiatrist.
The most common implementation of collaborative care is the Improving Mood Promoting Access to Collaborative Treatment Trial (IMPACT) model, which was implemented by the University of Washington’s AIMS Center.
6
We base our modeling assumptions on the IMPACT model. The main steps of the IMPACT model are as follows:
The stepped-care plan specifies the time the care manager spends on each patient. Consequently, it determines the future patient health progression, the resource utilization costs, and the fee-for-service revenue. Therefore, managing the stepped-care plan is the primary resource allocation decision of the Collaborative Care model. In the next section, we mathematically model the above characteristics of the Collaborative Care program.
Model
We model Collaborative Care for depression and diabetes as a discounted infinite-horizon MDP. A period in our model is a month. There are two reasons for having a month as a period. First, insurance firms pay clinics monthly under existing payment models for Collaborative Care. Second, the Collaborative Care program specifies monthly monitoring, follow-up, and adjustment of stepped care for patients.
Next, we define the state variables, transition probabilities, decision variables, and the objective function of the MDP model. We provide references and estimation results for model parameters in Section 6. We provide a schematic diagram of the steps in the Collaborative Care program in the Electronic Companion in EC.1, and a table of mathematical notation used in the model in section EC.2.
Patient Health State and System State
The health state of an enrolled patient is defined by a combination of their physical and mental health state. A Patient Health Questionnaire-9 (
Patient Health Questionnaire-9(
) and depression severity.
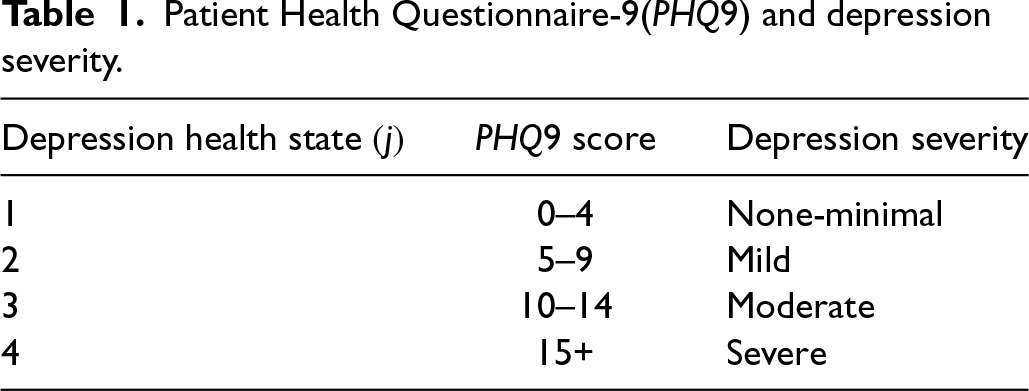
Patient Health Questionnaire-9(

The patient’s diabetes severity defines the patient’s physical health state. Consistent with standard clinical practices of monitoring diabetes, we use HbA1c to measure diabetes severity. HbA1c measures the blood sugar level, typically expressed as a percentage. For diabetes patients, the American Diabetes Association defines an
We define the physical health state of the patient by the level of
We have two additional possible states for a patient:
Then,
We first describe the state transition probability with one patient in the system. We use the notation
Each patient has four types of state transitions: Enrollment, treatment, discharge, and drop-out. These transitions correspond to the steps of the Collaborative Care program described in Section 3.
Enrollment
Estimates of the proportion of patients in each depression
Each clinic implementing Collaborative Care typically has predetermined enrollment criteria such that only patients in certain health states are eligible for enrollment in the program. Let this set of eligible states be 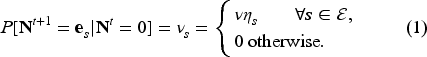
In modeling patient enrollment, we assume a common acceptance probability
In our model, the enrollment probability is not a function of the current number of patients in the system. This reflects how Collaborative Care is typically implemented: Clinics maintain a registry of patients diagnosed with depression and have visibility into the broader panel of patients with diabetes in primary care, using that information to enroll patients. It is indeed possible that the intensity of enrollment efforts may sometimes depend on the existing number of patients in Collaborative Care at a facility. Such a model of optimal admission control could be an interesting research problem. However, our focus in this paper is to develop a tactical planning model that provides insights into optimal treatment policy and the staffing levels required to serve the projected number of enrolled patients. As a result, we assume that enrollment probability is independent of the existing number of patients.
Further, the proportions
In line with standard practice, our model does not allow for re-enrollment of patients who have previously been discharged from Collaborative Care. Collaborative Care evaluations have shown that patients who reach remission tend to maintain symptom improvement over time (Gilbody et al., 2006), supporting the assumption that re-enrollment is rare.
Discharge is a deterministic process. Certain health states for enrolled patients are determined to be eligible for discharge. We call these sets of states
Therefore, 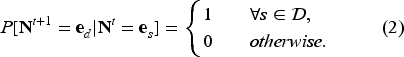
We define the discharge eligible set
In our numerical experiments, we adopt the discharge rule from the IMPACT model described in Section 3. Under this protocol, patients are discharged from Collaborative Care after they achieve remission from depression, defined as
Several patients undergoing treatment leave the program even if they have not achieved the discharge criteria. We assume a patient can drop out each month with a probability 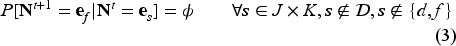
While we assume a common attrition probability
Each month, the care manager administers a
For all enrolled patients, i.e.,
We model the two levels of treatment as
An enrolled patient, who is not in a discharge eligible state, or in discharged state (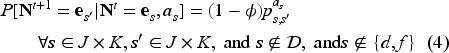
Enrolled patients could also transition to either the discharged state
Patients in the attrition state continue to stay in the attrition state, and patients in the discharge state continue to stay in the discharge state. Therefore,
Combining equations (2)–(4), we define the transition probabilities of a patient as:
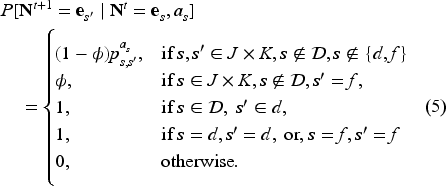
The above expressions are the system state when one patient is in the system. We assume
After enrollment, each patient undergoes monitoring, treatment, drop-out, and discharge as described above. We also assume that all patients in a given health state in a given time period are treated identically and their transition probabilities to the new state are identical.
A closed-form representation of the system state transition probability as a function of the individual patient transition probability does not exist. However, given the individual transition probabilities described above, we can simulate the system transition.
Clinics face the dual challenge of improving patient health outcomes and ensuring financial profitability. The success and longevity of clinical programs depend not only on delivering quality healthcare services but also on achieving positive financial outcomes. Although clinics are not solely profit-driven entities, financial considerations play a crucial role in sustaining their operations. Effective financial management and favorable financial outcomes are essential for supporting and expanding clinical programs, improving patient access, and fostering innovation in healthcare delivery. In formulating the objective function for the Collaborative Care program, it is necessary to capture the intricate relationship between clinical programs and their financial outcomes. The objective function should reflect the multifaceted considerations that contribute to sustainable operations and strike a balance between fiscal responsibility and the provision of quality patient care (Dai and Tayur, 2020).
Models for chronic care management come in various formulations that balance both financial and QALY factors. For instance, Deo et al. (2013) proposes a model that maximizes patient QALYs under a fixed budget constraint, which is a common approach in cost-utility analysis. In contrast, Lee et al. (2008) discusses three different formulations in the context of dialysis therapy: A weighted sum of QALY and cost, a purely QALY-focused objective with cost as a constraint, and a cost-minimizing approach. Meanwhile, several studies, such as Chen et al. (2018), adopt a net monetary benefit model. This framework calculates net benefit by multiplying QALYs by the predetermined “willingness to pay” for each QALY—effectively translating health outcomes into financial terms. This approach is commonly used to assess societal cost-effectiveness, as it combines the financial and health benefits into a single metric for evaluating health interventions from a societal perspective.
However, in the context of a specific clinic that needs to balance patient health outcomes and clinic financial performance, the net monetary benefit model, which relies on societal willingness-to-pay values, may not be suitable. The focus here is not on maximizing social welfare but on optimizing resource allocation within the clinic itself. To capture the clinic-specific trade-offs, we propose using a weighted, normalized sum of QALYs gained and clinic profits. This formulation gives the clinic flexibility to balance patient outcomes with financial sustainability. It does so without relying on societal willingness-to-pay parameters, which are typically used in models that focus on social welfare.
We formulate the per-period reward for a given system state at time 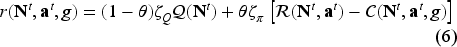
We define
We adopt this weighted-sum formulation because constrained MDPs are significantly challenging to solve. The current approach provides computational tractability in solving an MDP with a large state space. This efficiency allows us to systematically explore solutions across the spectrum of clinic preferences and to characterize how care intensity and staffing recommendations shift with different emphases on health versus profit. In contrast, an alternative formulation could have been to maximize QALY subject to a financial constraint. Such a constraint-based approach would not only be computationally challenging but also limit our ability to derive managerial insights based on relative preference for QALY in comparison to profits at clinics.
We note that the two parts of the objective function—QALYs and clinic profits are quantities of different scales. Therefore, the two parts of the objective function would need to be centered and normalized. We follow the standard normalization technique of multiobjective optimization (Mausser, 2006). In this method, each objective is centered and scaled. For example, the scaled and centered objective function for QALYs would be
Given the requirements specific to our context, we have adopted a multiobjective formulation as our modeling approach. This formulation provides a comprehensive framework for addressing the dual objectives of patient health and clinic financial outcomes, ultimately supporting effective decision-making in healthcare management.
Next, we expand the expressions for QALY, revenue, and costs to characterize the reward function.
For a patient in health state
To compare different policies, we compute the QALY gain, which is the difference between the total QALY of enrolled patients and the QALY for the no-treatment policy
The revenue will be determined by whether the Collaborative Care program follows a fee-for-service payment model (
In a per-member-per-month payment model, the insurance firm pays the healthcare facility a fixed payment (
Each care manager is available for
The total available hours for care managers in a month is
The cost of operating the Collaborative Care program is based on the salary and overhead costs for
Patients under acute treatment with more frequent follow-ups require
Patients enrolled in the Collaborative Care program will receive treatment according to the treatment plan in every period. Patients are never turned away for lack of available care manager time. The clinic manages this by acquiring additional care manager hours at an overtime rate when required. The overtime cost of exceeding
Thus, the per period cost is given by
Substituting the values of 
The optimality equation for the MDP corresponding to the Collaborative Care treatment is then given by:
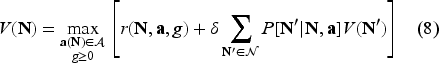
Since we perform steady state analysis, we drop the index
The optimal treatment policy is state-dependent, which means it depends on the number of patients in each health state. This is because the workload faced by the clinic, and hence the total overtime cost, which is convex in the total workload, depends on the distribution of patients across health states. As a result, the treatment decision for an individual patient may optimally depend on the current system state
The reward function
To characterize the optimal solution to equation (8), we restrict the model parameters to satisfy the following conditions:
Let,
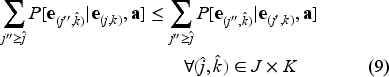
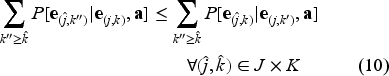
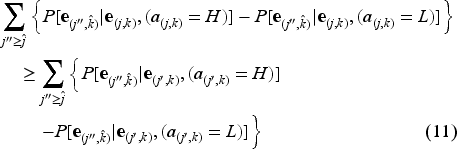
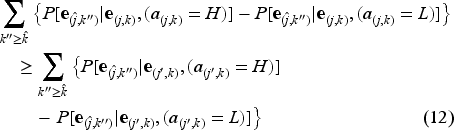
The term
Equation (11) in Condition 3 implies that the marginal difference in probability of improved health after one time period from
This observation is supported by clinical studies, justifying Condition 3. Prior research, such as Fournier et al. (2010), highlights that the marginal effectiveness of intensive interventions (e.g., Cognitive Behavioral Therapy (CBT) or antidepressants) increases with depression severity. Specifically, patients with mild or moderate depression experience negligible additional benefits from intensive therapies compared to less intensive interventions. Conversely, patients with severe depression demonstrate substantial improvements with higher-intensity interventions, aligning with stepped-care protocols in Collaborative Care models. Furthermore, Schueller et al. (2015) has established PHQ-9 cutoffs that guide the initiation of higher-intensity CBT therapy, indicating that patients with lower PHQ-9 scores derive limited benefit from such treatments, thereby supporting the rationale for allocating higher-intensity interventions to more severe cases.
Additionally, Kirsch et al. (2008), using both unpublished and published data submitted to the FDA for four new-generation antidepressants, found that the overall difference between the drug and placebo was clinically meaningful only in cases of very severe depression. Stepped-care approaches, widely utilized in Collaborative Care models, recommend allocating lower-intensity interventions to patients with milder symptoms to optimize resource use. These findings collectively support the notion that higher-intensity treatments are more beneficial for patients with greater initial severity, thereby validating Condition 3 within the framework of Collaborative Care models.
Next, given these conditions on parameter values, we obtain some structural properties of the model. These properties will help us arrive at a computationally efficient heuristic. All proofs of the propositions are provided in Section EC.8 in the Electronic Companion.
Let 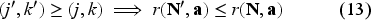
The above proposition implies that under Condition 1, the per-period reward is greater if an additional patient is in a healthier health state. In other words, the per-period objective increases when the health of enrolled patients improves.
Additionally, Proposition 1 establishes that, conditional on a given number of patients in the system, the clinic’s reward function is increasing in patient health. The proposition reflects the fact that healthier patients contribute more to QALY gains, and under Condition 1, lead to an increase in the objective function. Importantly, this result holds conditional on a nonempty system and should not be interpreted as suggesting that the clinic’s utility would continue to increase if the patient population vanished. Indeed, in the absence of any patients, both clinical outcomes and profits would be zero. Our model assumes a steady inflow of patients based on a given arrival rate, and the structural results in Proposition 1 are derived within this context.
If
The above proposition implies that the value function is greater when an additional patient is healthier.
These propositions lead us to the following result, which characterizes the optimal solution:
Given Conditions (1)–(3), the following two statements are true:
There exists an optimal treatment policy
Proposition 33 shows that for each diabetes state
Proposition 33 shows that this switching curve is nonincreasing in the physical health state. In other words, if the patient does not have glycemic control, the higher intensive treatment with more frequent follow-up should be performed at a (weakly) lower
Transition Probabilities
We use a data-based approach to estimate
Data Description
To estimate the transition probabilities, we use longitudinal EHR (Electronic Health Record) data of patients from a large academic medical center. This data was obtained from the electronic health record system, which contains details on patient visits, services received, and laboratory results such as the
The data consists of the following fields: Patient identifier, Date of PHQ9 test, PHQ9 score (PHQ9), Date of HbA1c test, HbA1c score (HbA1c), Date of depression therapy treatment, Current Procedural Terminology (CPT) code of depression therapy services provided, ED Visit Date, Psychiatry Visit date, Primary care visit date.
We categorize the treatment variable into the following three categories:
We transform the above data to extract the following additional features for each
We note several challenges with estimating the required transition probabilities from this data.
First, the measurements for depression and diabetes are not observed every month since PHQ9 and HbA1c for patients may be measured at irregular intervals under the usual care treatment. Therefore, there may be months in which only one of the depression or diabetes measurements may be recorded. Second, several months may pass between periods when depression therapy is provided to patients, and therefore, the transition to the next health state in the next period is not observed. To mitigate these challenges, we need two prediction models. First, we need to impute the missing health dimension for periods where only one dimension of the health state is observed. Second, we need a model-based method to predict the transition probability of patients when they are monitored and treated in every period. We discuss these two analyses below.
Imputing Missing Health States
Several approaches have been used to input missing longitudinal data. The most common approach is linear or nonlinear interpolation. Shechter et al. (2008) use a spline-based interpolation method and Lee et al. (2019) use a linear interpolation method. In our case, we could not use these interpolation-based methods because of the two-dimensional nature of the health state. Also, since we have other characteristics, such as the level of treatment, a data-driven method for predicting the missing data would be more appropriate. For this prediction method, we use a regression-based method to predict the missing data. Regression-based methods to predict missing data are a common strategy for missing data in clinical longitudinal studies (Zhang, 2016).
We create the following two linear regression models for 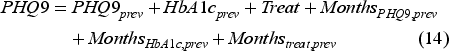
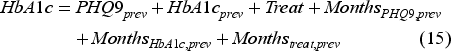
Then, using this data, for months when the
After completing the missing data, we will use this data to create a model to estimate the state transition probability.
First, we categorized the variables 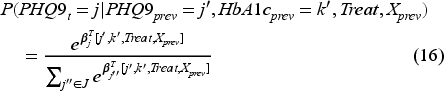
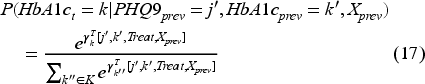
The above multinomial logistic regression models for state transitions incorporate both HbA1c and PHQ9 levels as predictors, meaning that the transitions for both health dimensions HbA1c and PHQ9 are interdependent, capturing the joint evolution of these states in response to the clinic’s Collaborative Care program. Under the Collaborative Care model, the depression-focused interventions are designed to complement the standard of care already provided to patients for diabetes management. In practice, this means that the transitions for HbA1c implicitly reflect the effects of diabetes-related treatments, which are provided independently of the Collaborative Care interventions. For example, a patient receiving insulin for glycemic control continues this treatment regardless of the Collaborative Care program, and our model incorporates these dynamics within the estimated transition probabilities. In light of this, the actions in the model focus on interventions related to depression, as this is the primary addition under the Collaborative Care framework. Including HbA1c in the state space ensures that our model captures its influence on the overall patient health trajectory and its interaction with PHQ9 levels. This approach aligns with real-world practices, where PCPs manage glycemic control through a parallel care process.
We obtain the estimates
We validate the estimates model in two ways. First, we report the Likelihood Ratio (LR) test for each variable, demonstrating that the variables improve the fit and are significant. Second, we obtain predictions from the logit model for the remaining 20% of the data, and we report the Mean Absolute Error of the model compared to the actual transitions observed in the data. We note that the model has a low Mean Absolute Error, and the LR test shows that the variables are significant and improve the model’s fit. We present these results in the Electronic Companion in Section EC.6.1.
After we obtain the estimates 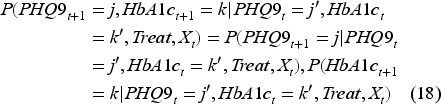
Other methods for estimating transition probabilities with covariates, such as the approach in Barratt and Boyd (2022), are more computationally demanding. To ensure robustness, we also applied the method from Barratt and Boyd (2022) in our analysis and observed no significant differences in the final recommendations for staffing levels and treatment policies.
The transition probability parameters are those for enrollment and attrition. We use
To estimate the proportion of patients in each
Distribution of patients with co-morbid diabetes and depression.
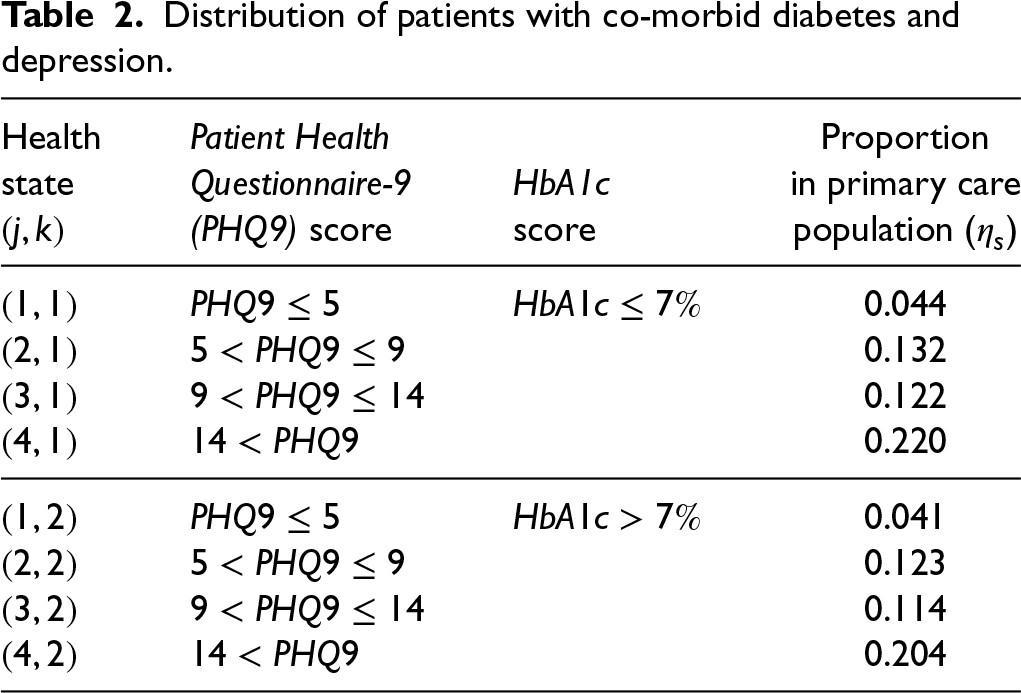
Distribution of patients with co-morbid diabetes and depression.

We model the patient population as being drawn from a distribution of starting health states, which is detailed in Table 2. This aggregate perspective aligns with the operational focus of the study, where the goal is to manage care delivery at the clinic level rather than to provide individualized recommendations for each patient. While we recognize that patient heterogeneity is important for personalized medicine, incorporating such factors into the model would significantly increase its complexity and fall outside the tactical planning scope of our work.
Secondly, our data consists of patients who were enrolled in the population and, therefore, presents a biased sample. Consequently, we have utilized the demographic data from published sources for the incoming distribution of patients rather than relying on our sample.
We have also performed a sensitivity analysis to assess the impact of increasing the percentage of patients in severely depressed states (
We use data from Liu et al. (2007), who have conducted a detailed time-motion analysis of the activities of care managers working in Collaborative Care for depression in three Veterans Health Administration clinics. The Collaborative Care process in their study was similar to the standard implementation of Collaborative Care described in Section 3. Care managers conducted follow-ups every two weeks for patients requiring more intensive intervention, and for less intensive intervention, care managers conducted follow-ups every four weeks. Each week, the care manager conducted a case review with a psychiatrist. Each follow-up resulted in a total care manager time of 60 minutes. This time consists of precall preparation, completed calls, postcall documentation, communication with providers, and all the time spent on unsuccessful call attempts. We describe the corresponding parameters in our model in Table 3.
Parameters for time spent by care managers.

Parameters for time spent by care managers.

Under a fee-for-service model, the insurance firm pays the clinic every month. The amount paid depends on the minutes of work done by the care manager. We use payment rates published by CMS for Collaborative Care for the fee-for-service payment. The payment has a tiered structure. Time spent up to the tier limit is reimbursed at a particular amount. There is an additional payment fee for enrolling patients. This is the most common implementation of fee-for-service in Collaborative Care. Similar models are in place at other large insurance firms such as Aetna and Blue Cross Blue Shield. 7
Since fee-for-service models have been identified as a potential implementation hurdle for Collaborative Care (Katon et al., 2010), health insurance providers and researchers have conducted large trials with per-member-per-month payment models. The fee-for-service is the most common payment model for Collaborative Care in practice (Carlo et al., 2018), including the payment for Collaborative Care under Medicare. It is expected that in the future, per-member-per-month models for mental health services will be more prevalent than they are today. Per-member-per-month payment models fall under the broad category of capitated payment models, where payment rates for a specific fixed duration are set prospectively. There have been some instances of the use of such models in the delivery of mental health services (Bachman et al., 2006). For the per-member-per-month payment model, we use the rates established by the New York State Department of Health for a Collaborative Care initiative conducted at 32 primary care clinics in 2012 (Moise et al., 2018). PCPs were given $150 per-member-per-month supplemental payment for adult patients receiving depression treatment under the Collaborative Care model. We use an inflation-adjusted rate of $165 for 2024.
We summarize the revenue parameters in Table 4.
Parameters for fee-for-service revenue model.
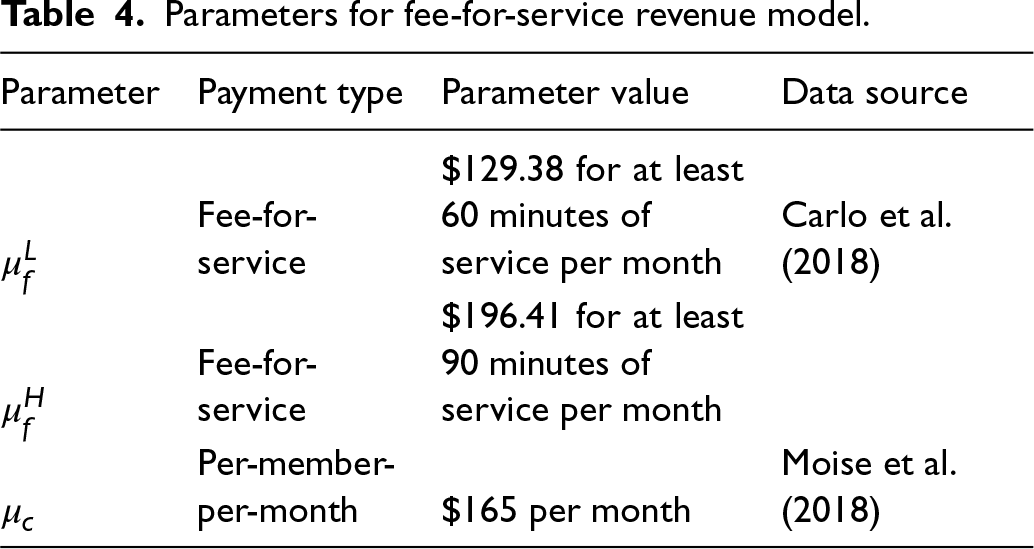
Parameters for fee-for-service revenue model.

The program’s primary cost is the care manager’s salary and overtime. We use cost analysis by Katon et al. (2012), which estimates the cost of care managers’ time towards delivering services required for Collaborative Care. This cost includes the cost of overhead, benefits, and supervision of care managers.
The clinic can meet additional demand by allocating extra care manager time as required to satisfy workload requirements. This extra time will cost the clinic an overtime rate of 1.5 times the regular cost (Table 5).
Parameters for cost and available time.

Parameters for cost and available time.

QALYs for all patients are determined by the summation of the utility value or QoL score for each patient. In the case of co-morbid conditions where multiple health conditions determine the QoL, several approaches are suggested for computing the QoL score. One of the most common recommendations is to multiply the QoL score of the individual health conditions (Ara and Brazier, 2017). We use this multiplicative model to compute patients’ QoL scores for a particular health state. The simple multiplicative model for combining health utility scores in patients with co-morbid conditions has received empirical support in several studies. One prominent example is the work by Flanagan et al. (2006), who tested this approach and found it to be robust for deriving utilities from individual condition scores. Although they acknowledged that further research is necessary across a broader range of conditions and populations, their findings strongly endorse the multiplicative method as both practical and reasonably accurate for handling comorbidities.
We present the QoL scores corresponding to the individual conditions in Tables 6 and 7. The composite QoL score computed by multiplying the QoL scores of the individual health scores is presented in Table 8. We use a QoL score of 0 for patients who drop out of the program, as these patients do not contribute to the QALYs of the Collaborative Care program.
Parameters for QoL scores for depression.
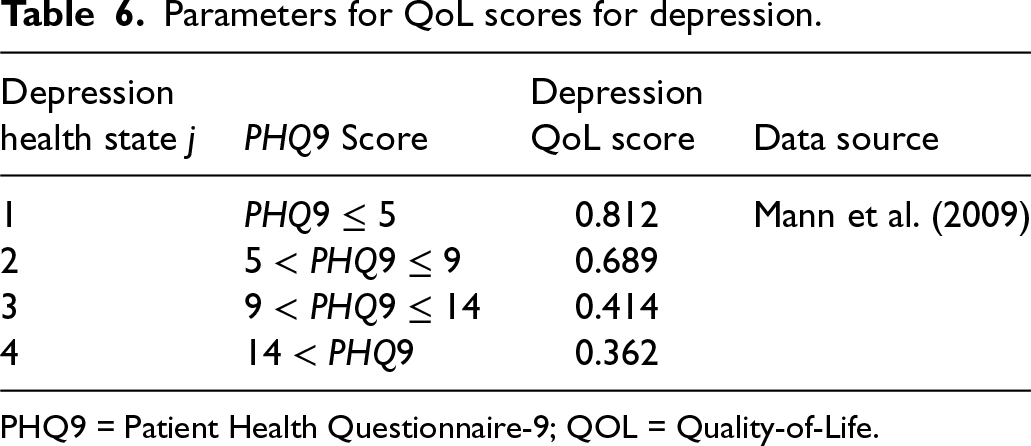
Parameters for QoL scores for depression.

PHQ9 = Patient Health Questionnaire-9; QOL = Quality-of-Life.
Parameters for quality-of-life (QoL) scores for diabetes.
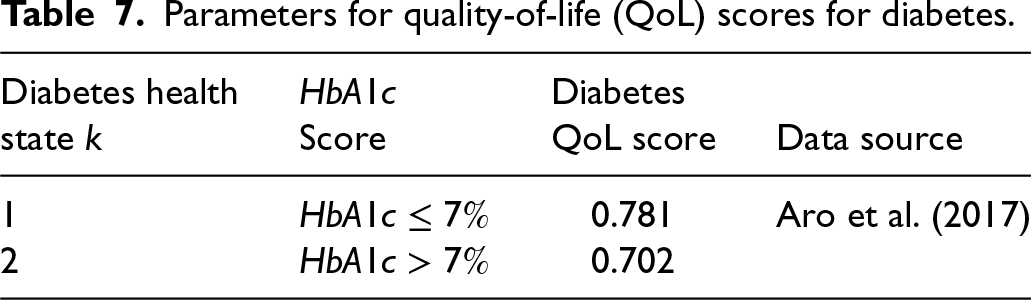
Parameters for QoL scores for patients with comorbid diabetes and depression.
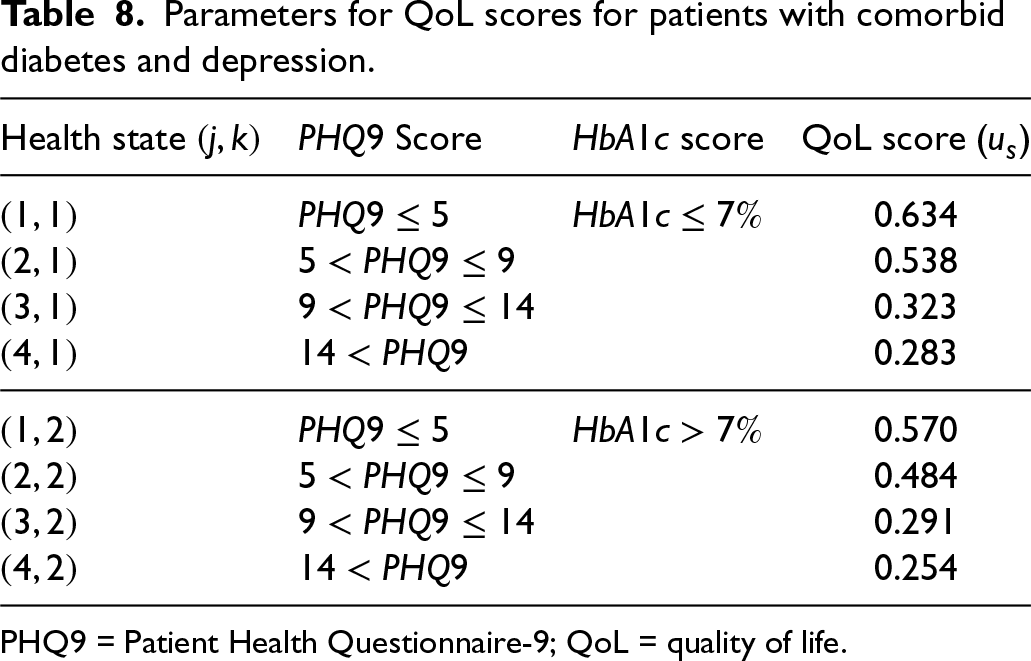
PHQ9 = Patient Health Questionnaire-9; QoL = quality of life.
The other remaining parameter is the rate of monthly attrition. We use the attrition rate from the IMPACT study randomized control trial (Unützer et al., 2002). Utilizing this study, we have an average monthly attrition rate of
As discussed in Section 4.2.1 and Section 4.2.2, we assume that the attrition probability and acceptance probability are independent of the health state. However, to demonstrate that our results are robust to this assumption, we perform a sensitivity analysis in Section EC.11.3 of the Electronic Companion, where we vary the attrition and acceptance probabilities based on the health state. We find that there is minimal impact on the overall model outcomes, indicating robustness to these assumptions.
In Section EC.4, we show that Conditions 1 to 3 are applicable for the above parameter values, verifying that the structural properties and, therefore, the solution methodology are appropriate for our problem.
Model Validation
To validate the model parameters, we conduct three sets of computations.
First, as discussed in Section 6.1, we perform an out-of-sample evaluation of the transition probability estimates. We report these results in the Electronic Companion in Section EC.6.1. The multinomial logit model for depression transition has a class prediction accuracy of 61% and is correct within one class 81% of the time. The depression transition model has a class prediction accuracy of 90.8%. Therefore, the transition probability estimates accurately predict the transition of patients in the data set.
Second, to demonstrate that the transition prediction model can accurately model patients’ transitions under collaborative care, we simulate a hypothetical cohort of 1,000 patients with the treatment policy as used in current practice. We then compare the fraction of patients discharged at six months to the prior study conducted by Angstman et al. (2012). We report this comparison in the Electronic Companion in Section EC.6.1. We find that the percentage of patients discharged at six months based on their starting depression level is close to the model-predicted rates of discharge at six months. This demonstrates that the model can accurately predict the transition of patients under Collaborative Care.
Finally, to test the results’ robustness to the estimation procedure, we perform the analysis with the approach outlined in Barratt and Boyd (2022) and show that the final recommendations of staffing levels and treatment policy do not show any significant difference for a wide range of model parameters. We report the results of this analysis in the Electronic Companion in Section EC.6.1.
Solution Methodology and Numerical Analyses
In this section, we first develop a computationally efficient solution methodology that utilizes the structural properties of the model to optimize the treatment and staffing levels jointly. Depending on the organization, the joint optimization of staffing and treatment may or may not be organizationally feasible to implement. Some individual clinics that are part of larger healthcare systems may not have the discretion to change the treatment policy. Alternatively, clinics may face hiring shortages and be unable to adjust staffing levels. We, therefore, compare the results of the joint optimization with individual optimization of treatment and staffing, where treatment and staffing are maintained at the levels in current practice at our partner organization. This also allows us to quantify the value of joint optimization treatment and staffing levels. We also compare the results of our model-based policy with the usual care process, where patients receive their depression treatment at ad hoc intervals as observed in the usual care without a Collaborative Care program, which we call the baseline policy.
Baseline Policy: Usual Care
Under the baseline policy, patients receive their depression treatment at ad-hoc intervals. We model the time between mental health treatment based on random interarrival visits for mental health treatment. We model this interarrival time as an exponential distribution with the mean interarrival rate obtained from our data. We compute the clinic profits and patient QALYs by simulating a system where the random treatment intervals are drawn from the above-mentioned exponential distribution. Then, we compute the QALYs gained and the clinic profits if patients were to arrive in the system according to the exponential distribution.
Current Practice for Collaborative Care
Under the current practice of Collaborative Care, the clinic follows a treatment policy of assigning
Model-Based Joint Optimization of Treatment and Staffing
Proposition 3 shows that, for a given number of care managers,
We take inspiration from the idea that the optimal policy has a threshold-type structure for each
We use an infinitesimal perturbation analysis (IPA) approach to search for the threshold
Next, we note that the objective function is concave in the number of care managers,
Since we have solved a state-independent policy for the threshold, this objective would be lower than a state-dependent policy. To demonstrate the performance bounds of the state-independent policy, we also solve for an upper bound of the objective function. We find that the gap of the upper bound is less than 7.4% for all the scenarios we consider. We describe the upper bound computation in Section 7.6.
We also solve for two restricted versions of the joint-optimization problem. First, we solve for the model-based policy for fixed staffing levels. We use the same algorithm described in Section 7.3, except we do not solve the optimization for staffing and keep it fixed at the current practice. Second, we solve for the optimized staffing by fixing the treatment level to the current practice described in Section 7.2, then solve for the optimal
We solve for these restricted versions of the joint optimization because, depending on the organizational restrictions, joint optimization may not be feasible to implement. Even in such a situation, we show that the model-based policy for treatment and staffing levels is superior to the current practice.
Policy Comparison
To compare the policies described above, we present results comparing the objective values in Table 9 and the differences in policy recommendations in Table 10 below. We present the results for a monthly arrival rate of 30 and 50 patients and for two values of
Policy objective value comparison.
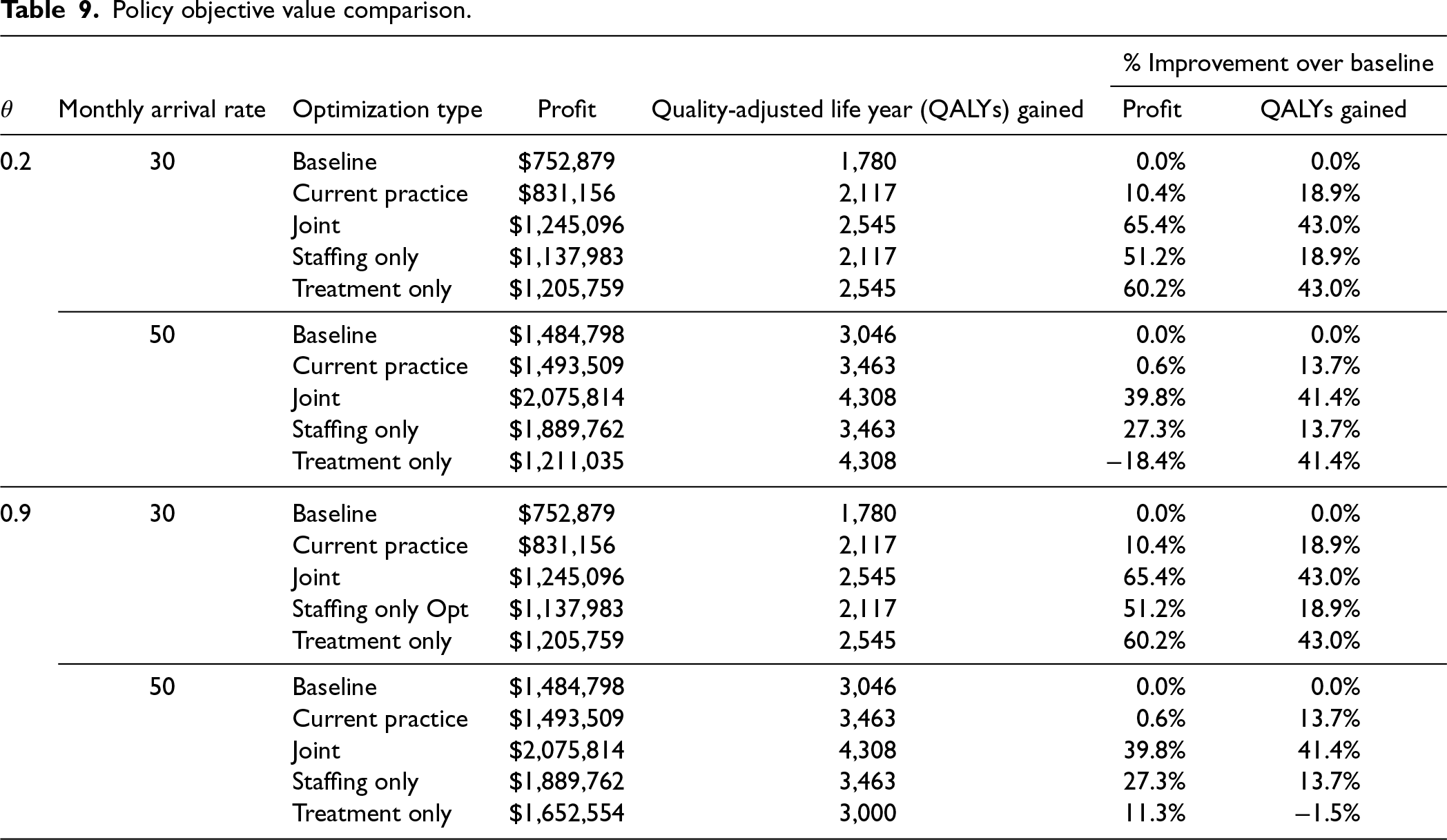
Policy objective value comparison.

Policy recommendation comparison.
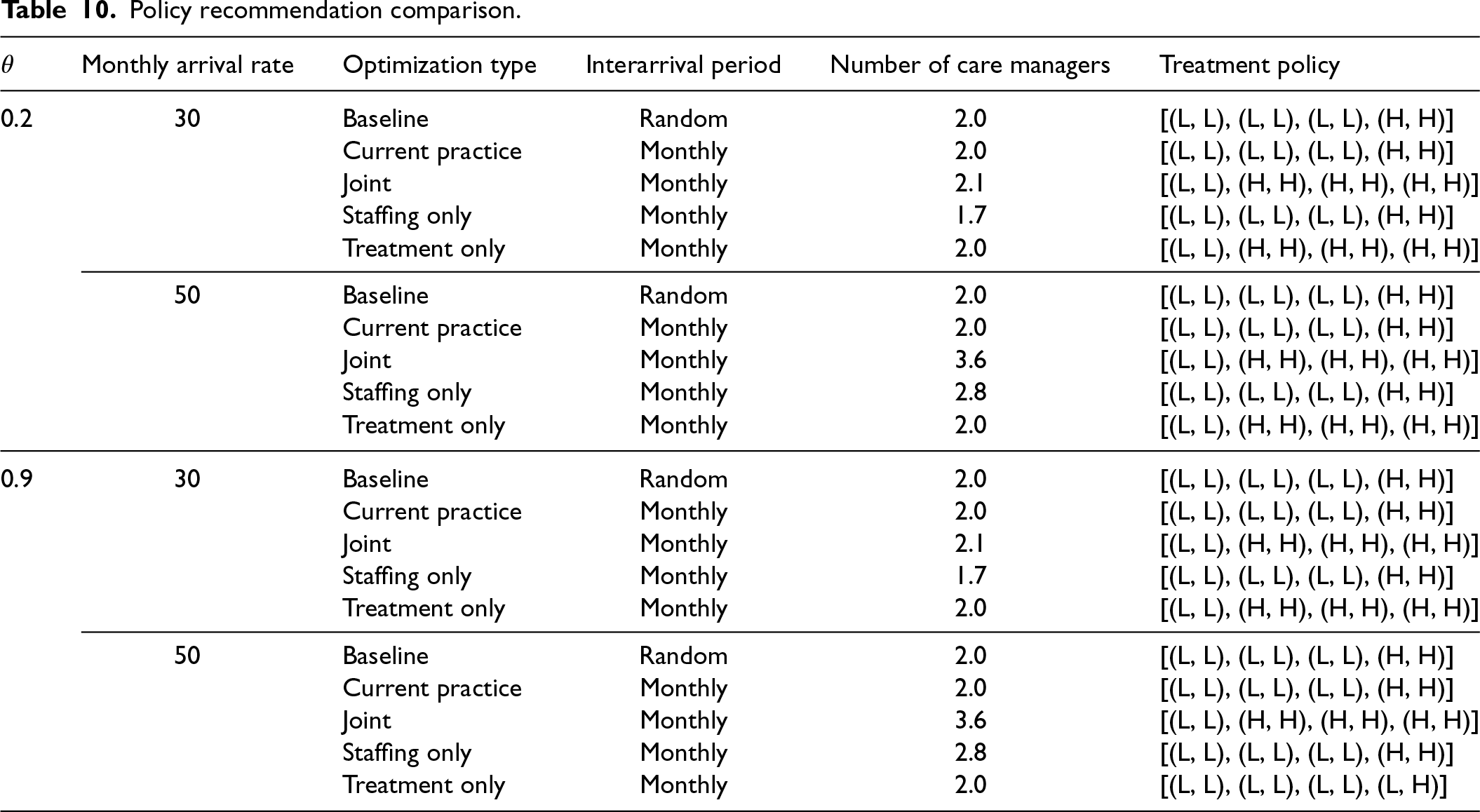
To compute the parameters
For low
The joint optimization policy leads to the highest improvement in QALYs and profits for both low and high
Next, we analyze some other characteristics of the model-based policy in Section 7.5.
We provide two analyses to give intuition on how the model-based solution provides benefits. First, we show the path from enrollment to discharge for one representative patient in Table 11. Next, in Table 12, we compare the revenue and care manager workload for the model-based policy and current practice when a larger number of patients are enrolled.
Comparison of total discounted quality-adjusted life years (QALYs) and revenue for a single representative patient.
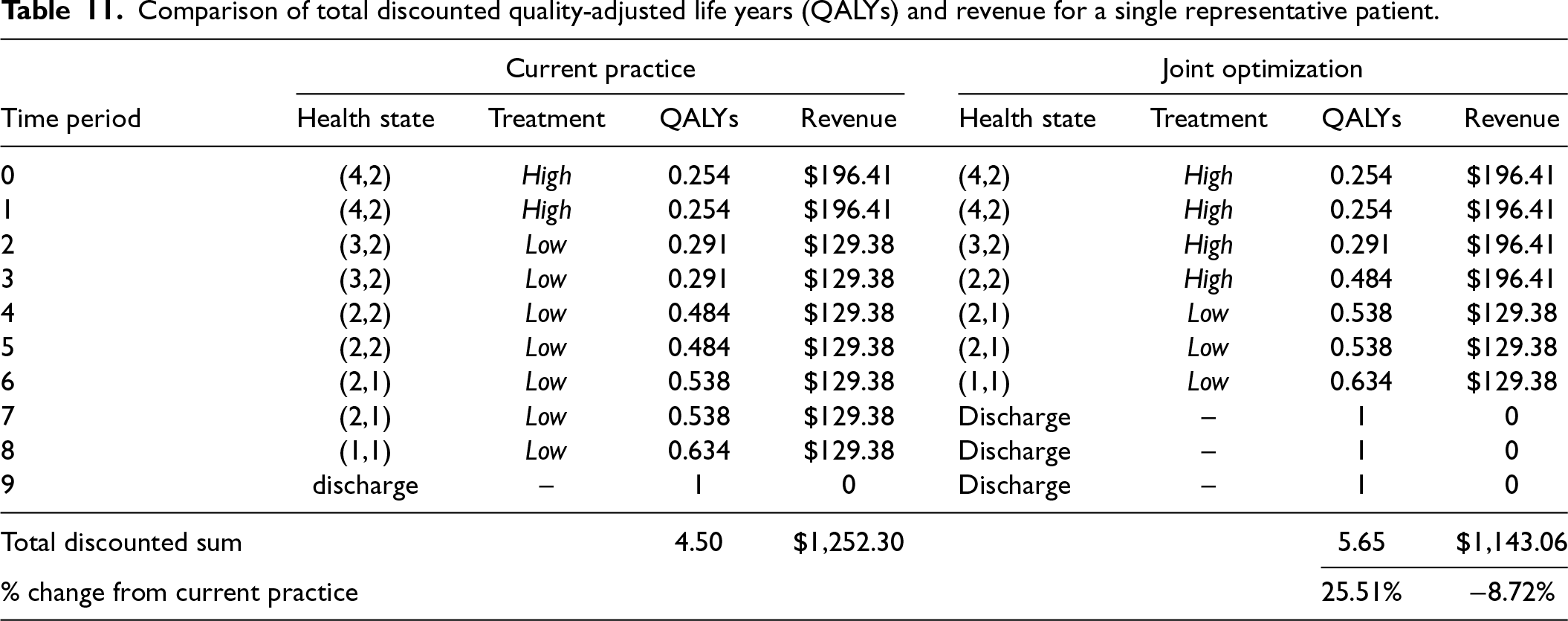
Comparison of total discounted quality-adjusted life years (QALYs) and revenue for a single representative patient.

Comparison of objective components
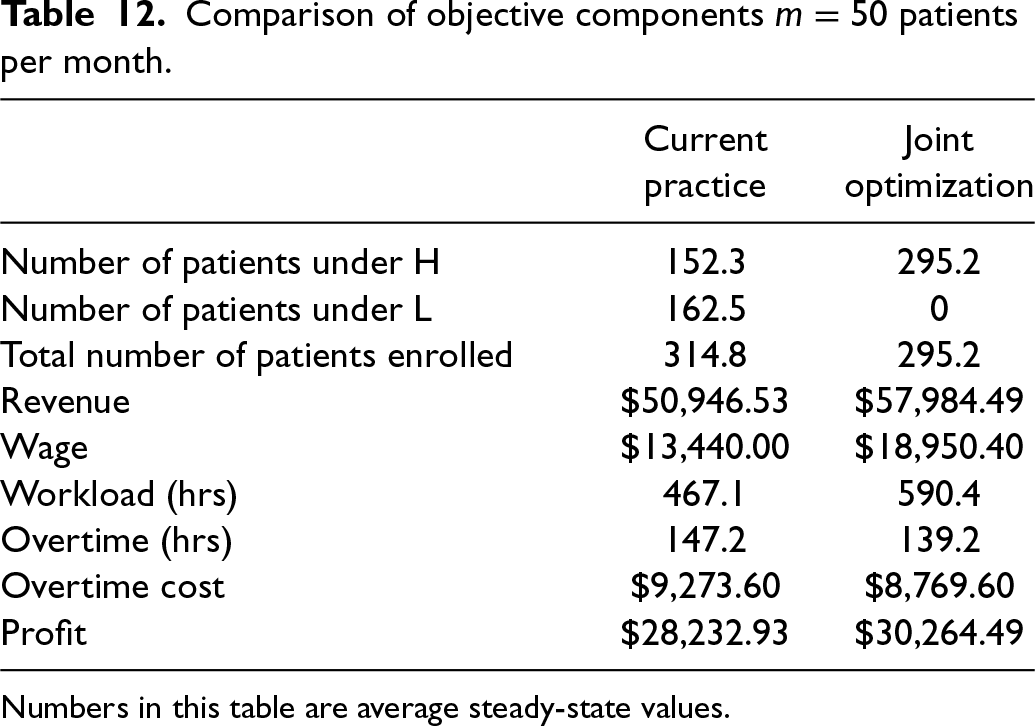
Numbers in this table are average steady-state values.
Table 11 shows transitions from health state
In Table 12, we present the average steady-state values for the per-period number of patients in each treatment level, the total number of patients enrolled, revenue, wage, workload, overtime hours, overtime costs, and profit. We present average steady-state values for ease of interpretation. We can observe that under Joint Optimization, all health states are given
One might think that there might be caps on the total number of
There are two items we would like to highlight here. First, for the Collaborative Care program, the clinic should adopt a system perspective to determine the optimal allocation of care managers’ time because the trade-offs are different for a single patient compared to a population of enrolled patients. Second, the assignment of treatment levels to patient health states determines the number of patients and their distribution under
To test the efficacy of our heuristic solution for the joint optimization, we compare the solution to an upper bound.
The following linear program is an upper bound to the MDP given in equation (8).
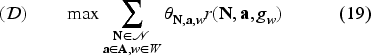
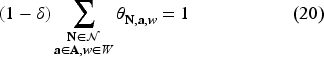
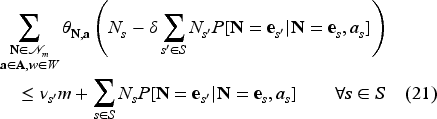
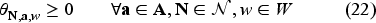
We provide the proof of Proposition 4 in the Electronic Companion EC.8.2. The above program is linear in many variables; we solve this by the column generation method. We describe the column generation method in the Electronic Companion EC.9.
Next, we present the result of the model upper bound in Table 13. We present the results for a monthly arrival rate of 30 and 50 patients and two values of
Model gap to upper bound.
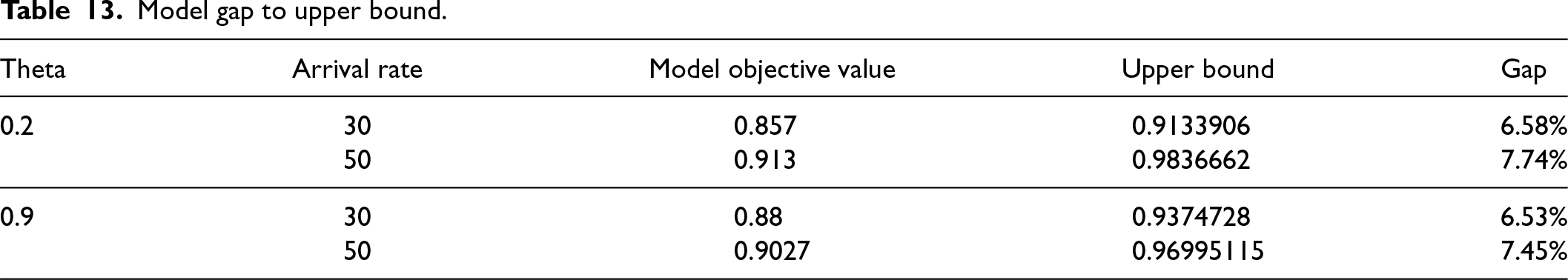
We observe from the results of Table 13 that the gap to the upper bound is less than 7.4% for all the scenarios we consider. This shows that the model-based policy is close to the upper bound.
In this section, we discuss sensitivity to changes in model parameters and provide managerial insights. First, we look at how the model solution changes for different values of
Sensitivity to Arrival Rate
We perform a sensitivity analysis on the arrival rate of patients. We present the results for a range of arrival rates for two values of
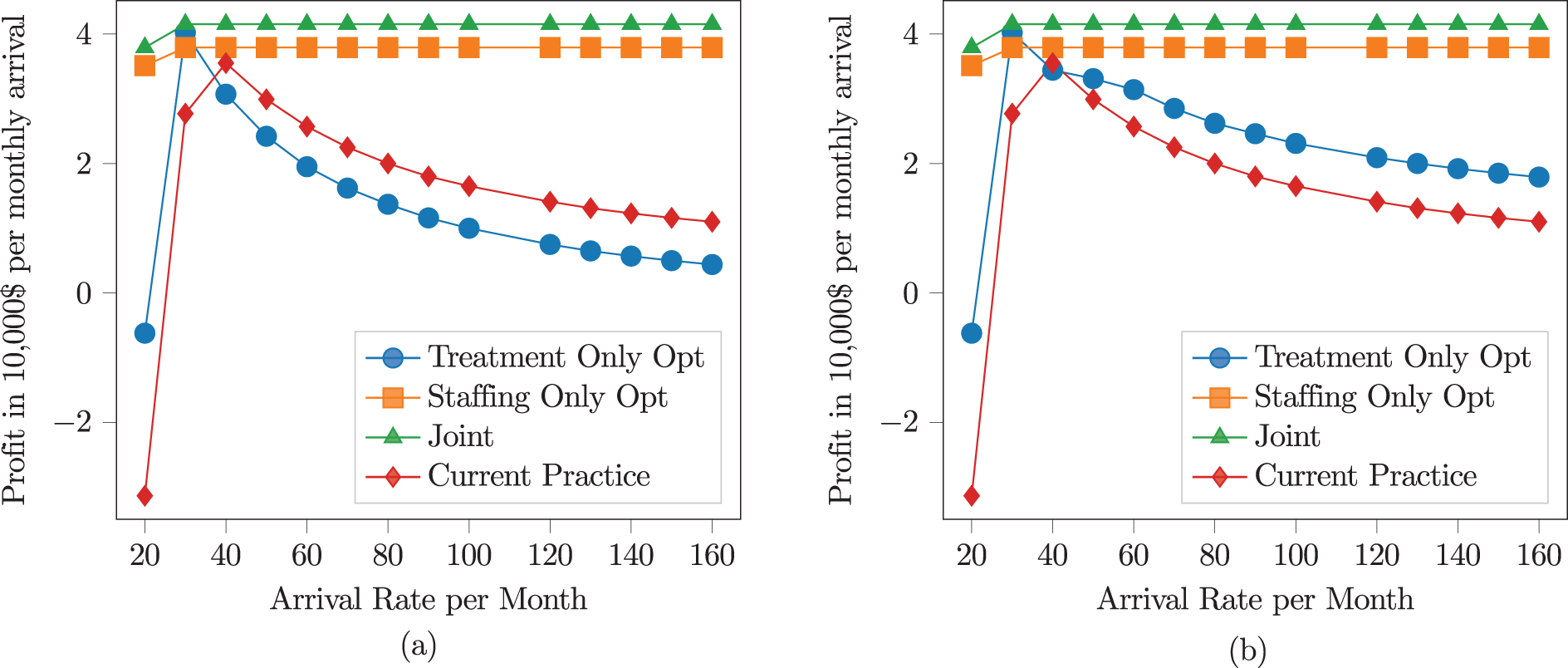
Profit sensitivity to arrival rate for
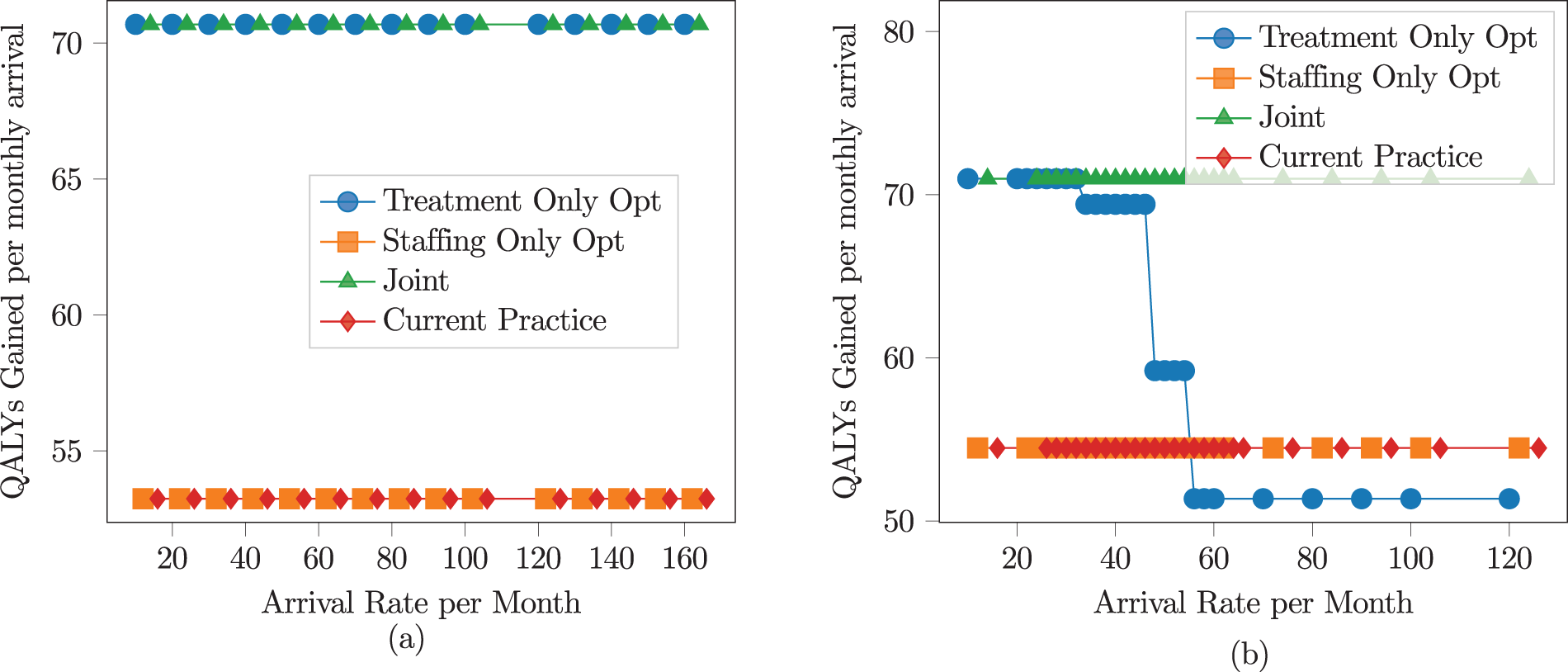
Quality-adjusted life year (QALY) gained sensitivity to arrival rate for
We can observe from Figure 1 that profit with staffing and joint optimization is not sensitive to an increase in arrival rate. This is because, with staffing optimization and joint optimization, the staffing level can be optimally decided to maximize the profit for any arrival rate. However, under treatment-only optimization and current practice, the profit first increases and then decreases. This is because when the arrival rate is low, increasing the arrival rate increases revenue, but there is no overtime. As the arrival rate crosses a particular threshold, any additional increase in arrival rate leads to overtime costs and, therefore, lowers profits.
We can also observe that for low
For high
These results show that different optimization types are uniquely sensitive to the arrival rate. Staffing-only optimization and joint optimization behave similarly, with joint optimization slightly better than staffing-only optimization in terms of profits. The response of treatment-only optimization and whether it performs better than current practice depends on the value of
Since there is no optimization under Current Practice, the response to the arrival rate does not change for different values of
Alongside the fee-for-service model developed by CMS, some locations have also implemented a fixed-fee payment model, known as the per-member-per-month model Moise et al. (2018). In this section, we assess the sensitivity of various optimization models to changes in the fixed-fee payment rate.
For the fixed fee payment, in our mathematical model, we set
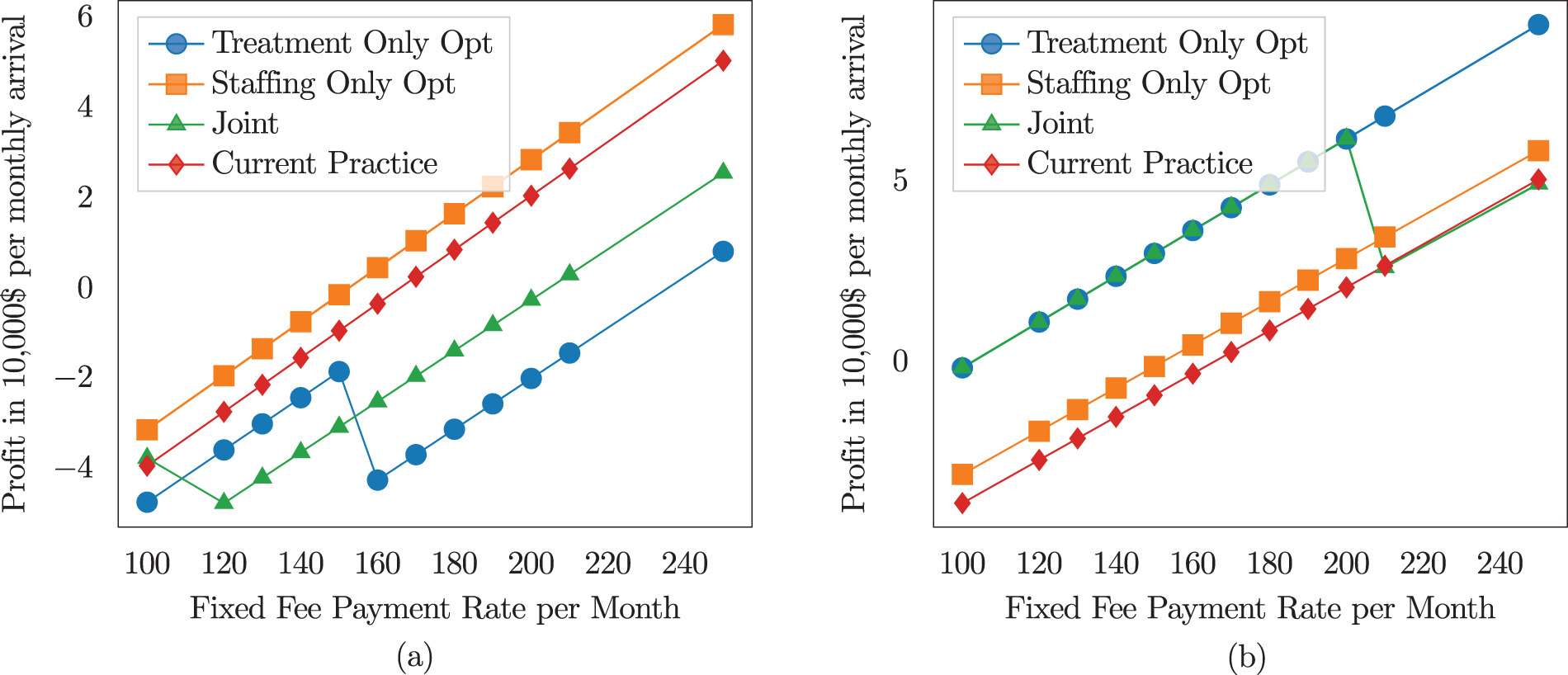
Profit sensitivity to fixed fee payment for
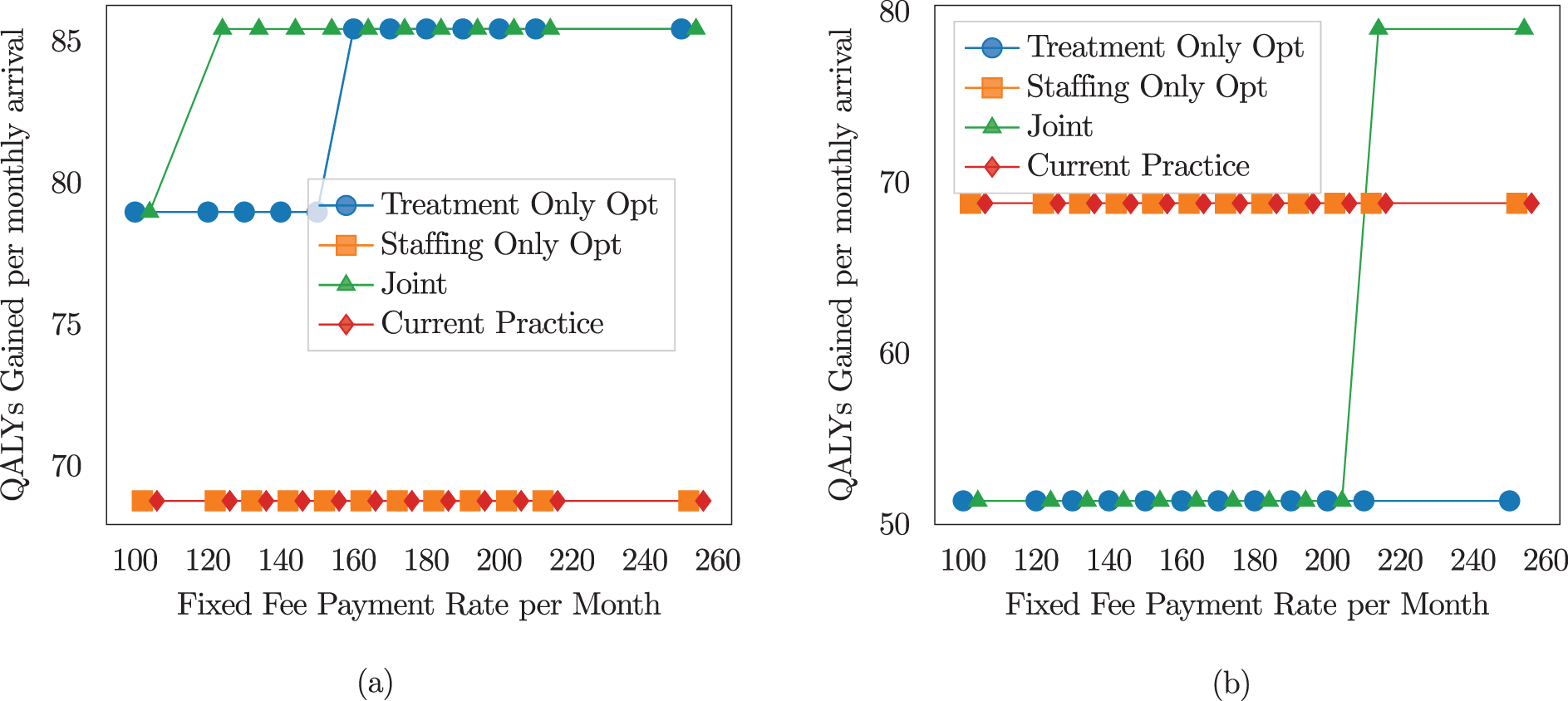
Quality-adjusted life year (QALY) gained sensitivity to fixed-fee payment for
In Figure 3(a) and (b), profit increases linearly with the payment rate for the current practice and staffing-only optimization models. This linearity arises because the treatment level is fixed in these models. Since the treatment level determines the discharge rate, it also determines the number of patients in the system at a steady state. Therefore, the number of patients in the system remains constant regardless of the payment rate. Since each patient generates revenue proportional to the payment rate, profits linearly increase as the payment rate increases.
In contrast, we observe a nonmonotonic profit response in the joint optimization and treatment-only models. For payment rates below a certain threshold, revenues are insufficient to support higher treatment levels, resulting in low treatment levels across more health states. In this range, profit increases linearly as the payment rate rises. However, once the payment rate exceeds the threshold, the optimization model increases the treatment level, initially decreasing profits. Higher treatment levels lead to faster patient discharges, reducing the number of patients in the system at a steady state and, consequently, lowering revenue. As the payment rate continues to rise beyond the threshold, treatment levels stabilize at a higher level, and profits again increase linearly with the payment rate. Notably, the threshold for this switch is lower when
The managerial implication is that the sensitivity to QALY and Profit is nonmonotonic. For the clinics to improve treatment levels, a fixed fee payment model requires a payment rate above a certain threshold. This threshold depends on the clinic’s objective weight on QALY and profits, as well as the type of optimization performed. A higher weight on profit will result in the switch to higher treatment occurring at higher payment rates, whereas joint staffing and treatment optimization will lead to the switch at a lower payment rate.
Access to mental health has been increasingly challenging. The need for improved access to mental health services is particularly high for patients suffering from chronic physical conditions. Treating both the mental and physical health of patients has been shown to increase the likelihood of improving patients’ health outcomes. The Collaborative Care program is a crucial way to increase access to mental health services. However, as we mentioned in Section 1, the adoption of Collaborative Care programs has been slow due to several challenges, such as managing care manager resources and a lack of visibility in insurance payment models.
One of the primary operational levers in managing population-based chronic care programs, such as Collaborative Care, is allocating the time of the care managers to patients depending on their requirements. Based on the patient’s disease severity, they may need more time from the care manager for therapy and treatment. From standard clinical practice for collaborative care, patients can receive two levels of depression care from their care manager: A higher intensity, more frequent follow-up, or a lower intensity, less frequent follow-up. Insurance revenue payments are typically tied to the time spent by the care managers. Therefore, allocating care managers’ time to patients determines patient health outcomes and clinic profits.
In this paper, we develop a mathematical model for Collaborative Care to optimize the allocation of care managers’ time and offer managerial insights for effective program management. Our model-based approach has a population health perspective, where total revenue, costs, and patient outcomes for all enrolled patients are incorporated. We calibrate our model parameters using patient transition data from a large academic medical center.
From our model, we derive a theoretical result that the optimal allocation of the care managers’ time is a threshold-type policy in the two-dimensional patient health state of diabetes and depression severity. Patients with high diabetes severity should receive more intensive treatment with more frequent follow-up at lower depression severity. This threshold-type policy will lead to improvement in both QALYs and profits. This threshold depends on the distribution of the enrolled patients in the different health states.
The computation of the optimal state-dependent threshold policy is intractable. Additionally, the optimal threshold policy would lead to patients receiving a level of care that depends on the health status of other patients, which is impractical from an implementation perspective. Therefore, we derive a computationally efficient state-independent policy, implementing a static threshold that does not change based on the distribution of enrolled patients. We show that this policy is close to optimal through an upper bound.
Subsequently, we conduct numerical experiments to show that the model-based heuristic policy can dominate current practice for both QALYs and profits. The choice of treatment levels determines how long it takes patients to be discharged. This determines the number of enrolled patients and, consequently, the total system revenue and the total system workload. The model-based heuristic policy determines the treatment levels to balance QALYs, total revenue, and total workload to improve system performance.
Next, we show that for fixed-fee payment models, profit increases linearly with the payment rate in the current practice and staffing-only models due to fixed treatment levels, stabilizing the patient count and revenue. However, joint optimization and treatment-only models show a nonmonotonic profit response, with profits initially rising, then decreasing as treatment levels increase, and rising again once payment rates surpass a threshold. This threshold is lower for models prioritizing QALY and joint optimization, which balances staffing and treatment. Similarly, QALY gains improve with higher treatment levels at lower payment rates. Clinics need payment rates above this threshold, which varies based on the clinic’s focus on profit or QALY, and joint optimization leads to earlier treatment improvements at lower payment rates. Prior literature on fixed-fee payment models has identified that providers may deliver lower than optimal treatment, which is called skimping (Folland et al., 2016). However, we find that in the management of programs such as Collaborative Care, skimping may be avoided if the fixed-fee rate is high enough and by using joint optimization of staffing and treatment levels.
There have been several studies analyzing the relative impact of different payment models. Our study contributes by showing the effect of different payment models in a Collaborative Care context, with chronic care of patients suffering from multiple conditions.
There are several opportunities for future research. First, in our study, we have considered financial and health outcomes observed within primary care. Patients with improved access to mental health services can also be expected to show lower healthcare utilization outside of primary care. Patients under Collaborative Care could also show lower visits to emergency rooms and fewer inpatient admissions. Therefore, one extension of the model could be incorporating the impact of Collaborative Care on the broader healthcare system outside primary care.
We did not adopt a Partially Observable Markov Decision Process approach due to its computational complexity, interpretability challenges, and difficulty in determining the model’s structural properties. However, we perform a sensitivity analysis in the Electronic Companion (EC.12) to assess how policies derived under fully observable states perform when health states are subjected to observation errors. The joint-optimization continues to perform the best for both QALYs and profits under observation errors. However, the sensitivity to the observation errors depends on whether the errors lead to false positive errors or false negative errors.
Finally, this approach does not include patients with other severe mental health conditions such as schizophrenia, anxiety, bipolar disorder, or substance abuse. We chose depression because it is one of the most common co-morbid conditions with other physical conditions. Secondly, the
Supplemental Material
sj-pdf-1-pao-10.1177_10591478251392328 - Supplemental material for Optimal Staffing and Treatment for Collaborative Care of Diabetes and Depression
Supplemental material, sj-pdf-1-pao-10.1177_10591478251392328 for Optimal Staffing and Treatment for Collaborative Care of Diabetes and Depression by Sandeep Rath, Jayashankar M Swaminathan and Charles A Coleman, Jr. in Production and Operations Management
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Notes
How to cite this article
Rath S, Swaminathan JM and Coleman CA Jr (2025) Optimal Staffing and Treatment for Collaborative Care of Diabetes and Depression. Production and Operations Management XX(X): 1–24.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
