Abstract
Objective
To determine whether tensor veli palatini muscle (mTVP) tenopexy performed during furlow palatoplasty (FP) reduces the occurrence of otitis media with effusion (OME) in children with cleft palate.
Design
Parent- and assessor-blinded randomized controlled trial.
Settings
Tertiary clinical care institution.
Patients
In total, 81 nonsyndromic children with cleft palate Veau I–IV were randomized to undergo traditional FP or FP with mTVP tenopexy (modified FP [MFP]). Middle ear status was evaluated longitudinally between 2 and 7 years of age.
Interventions
FP with or without mTVP tenopexy.
Main Outcome Measures
Presence of OME after extrusion of ventilation tubes (VTs).
Results
The mean age at palatoplasty was similar between groups (FP: 1.05 ± 0.17 years; MFP: 1.07 ± 0.18 years; p = .585). Baseline characteristics, including sex, race, preoperative OME, and Veau classification were comparable. Rates of additional surgical interventions (cheiloplasty, revision palatoplasty, alveolar bone graft, adenoidectomy, VT reinsertion, and tympanoplasty) did not differ between groups. The frequency of OME from ages 2 to 7 years was similar when analyzed by child (p = .09–.90), by ear (p = .145–.96), or using cumulative incidence (χ2 = 0.324; p = .569).
Conclusions
In this first randomized, blinded clinical trial evaluating mTVP tenopexy during FP, no improvement in the frequency or cumulative incidence of OME was observed. Given the absence of benefit and the additional operative time and cost, mTVP tenopexy is not recommended.
ClinicalTrials.gov ID
NCT01535131
Introduction
Orofacial clefts including cleft palate (CP) and cleft lip (CL) are the most common congenital abnormalities with a prevalence of approximately 1.6/1000 live births. 1 The dysmorphologies associated with CP+/–CL cause abnormal craniofacial development that affects dental development and the muscles associated with Eustachian tube (ET) opening that are essential for normal middle ear (ME) function,2,3 resulting in ET dysfunction (ETD), otitis media with effusion (OME) and hearing loss.
While progressive improvement in hearing with advancing age was reported, the prevalences of conductive and sensorineural hearing losses in CP+/-CL patients are greater than those of age-matched controls at all ages. 4 As with the prevalence of hearing loss, that of OME decreases with advancing age.
There is good evidence that the functional efficiency of the ET is constrained by its anatomy.5–9 Two muscles are intimately related to the cartilaginous ET, the levator veli palatini muscle (mLVP) and the tensor veli palatini muscle (mTVP).5,10–12 From early on, the potential for applying this anatomic knowledge to palatoplasty techniques was recognized as a means to enhance ET function (ETF) and reduce the incidence of OME and hearing loss. 13 However, it is crucial that the design of any new procedures aimed at decreasing OME prevalence also considers the essential objectives of CL and CP surgery, which include achieving an aesthetically pleasing outcome, ensuring improved velopharyngeal competence, and facilitating normal speech development.14–16
In the past two decades, palatoplasty techniques like the furlow palatoplasty (FP) that lengthen the palate while reconstructing the mLVP sling were shown to establish more “normal” mLVP vector orientations and, consequently, to improve speech and velopharyngeal competence.17,18 However, those procedures usually transect the mTVP tendons to provide more surgical freedom for mLVP repositioning, and this has unknown but expected adverse consequences to mTVP function and ETF. A few studies describe technical modifications designed to reduce the prevalence of the otologic consequences of the CP condition. These included suturing the opposing mTVP tendons to create a prehamular sling 19 or tensor tenopexy,20,21 a procedure modification that creates a fixed bony insertion for those tendons. An imaging study with MRI after CP repair concluded that the integrity of the hamulus and mTVP during CP surgery, for example, not breaking the hamulus and not sectioning the mTVP, is crucial for ETF and prevention of otitis media (OM). 22 From functional–anatomic considerations, these modifications are expected to improve ETF and, by consequence, decrease the prevalence of OME and hearing loss. However, these preliminary studies were not robust enough (or did not have a control group, had a small sample size, or were not randomized) to provide evidence of the benefits of this new technique. In this study, we used a randomized controlled trial to test if mTVP tenopexy reduces the recurrence of OME in cleft children undergoing FP.
Methods
The study was conducted from 2012 to 2019 to compare the effect of tensor tenopexy during FP on ETF and OM rate. An institutional Data and Safety Monitoring Board (DSMB) was formed, and the Institutional Review Board (IRB) application was approved. After forming the DSMB and IRB approval, enrollment began. The DSMB met every 6 months to review the relevant aspects of study progress, particularly recruitment and adverse event logs. CP+/–CL patients potentially available for study recruitment were those who were nonsyndromic, classified as Veau types I through IV, 23 initially presenting to the Craniofacial/Cleft Palate Clinic at up to 15 months of age, assigned to one of the four study surgeons and whose parents could read and write English. Children were excluded if they had already undergone any type of palatoplasty or had genetic abnormalities. As was usual for CP patients followed at the Craniofacial/Cleft Palate Clinic, a CL, if present, was repaired at approximately 3–6 months of age using a technique chosen by the surgeon and, for all children, bilateral ventilation tubes (VTs) were inserted by a pediatric otolaryngologist concurrent with the CL repair, but this was deferred in some until the time of palate repair at the discretion of the surgeon and parents based on the history and findings,
Between 9–15 months of age, children underwent CP closure. After informed consent by parents, children were randomly assigned to either undergo traditional FP or FP with mTVP tenopexy (modified FP or MFP). The randomization allocation was 1:1, with stratification by surgeon (total of four plastic surgeons who were all trained to perform a standardized MFP) and by Veau type. An envelope with the randomized assignment was opened in the operating room at the time of surgery. All subjects received a new set of VTs at the time of palatoplasty.
Bilateral tensor tenopexy was performed during FP in the following fashion. Through a lateral relaxing incision made between the retromolar trigone of the mandible and the maxillary tuberosity, the pterygoid hamulus and mTVP tendon were exposed. A resorbable suture of 3.0 Polydioxanone (PDS) was placed through the mTVP tendon and around the hamulus to anchor the mTVP tendon to the hamulus. Then the tendon of the mTVP was sectioned just medial to the hamulus (Figure 1).
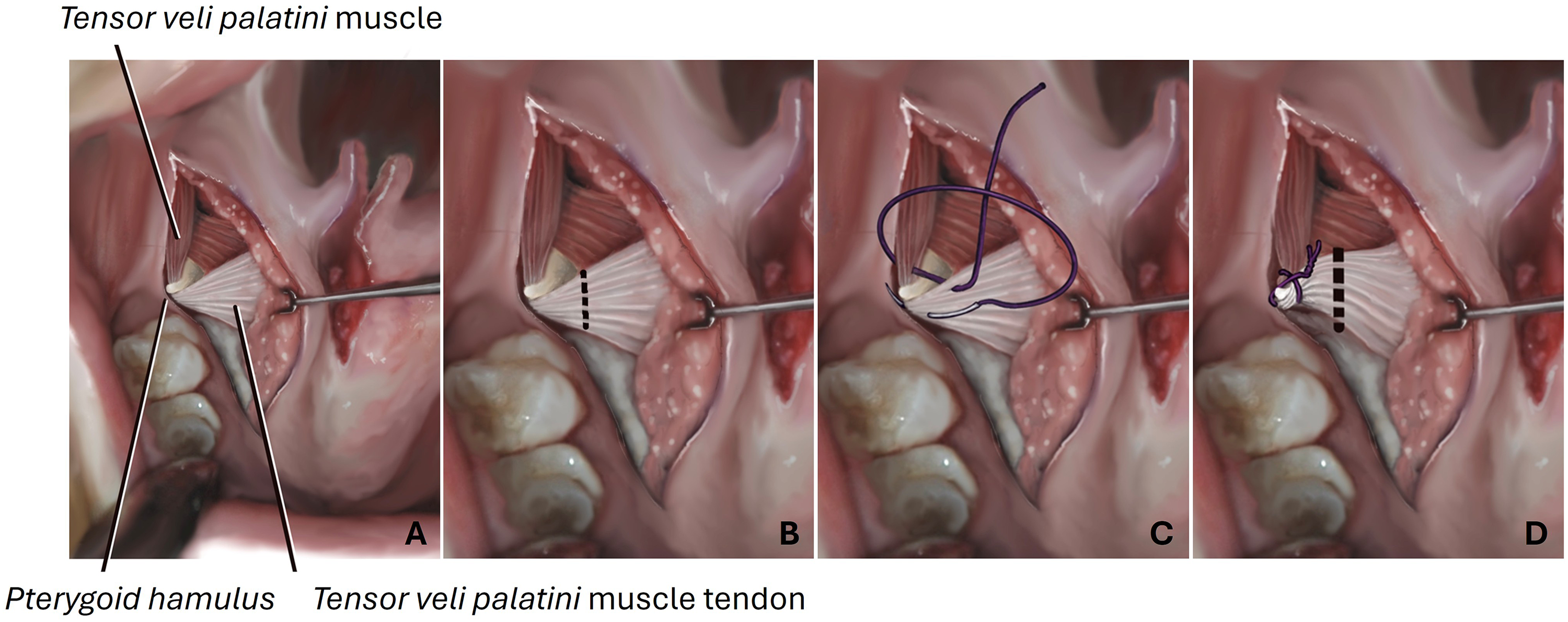
(A) Anatomy exposed with lateral relaxing incision; (B) Dotted line representing the sectioning of the mTVP tendon as it courses medially around the hamulus, performed in routine furlow palatoplasty; (C) Figure of eight suture tenopexy anchoring mTVP tendon to the hamulus; (D) Dotted line representing the sectioned mTVP tendon as it courses medially around the hamulus following tenopexy.
After palatoplasty, subjects were followed for their usual care at the Craniofacial/Cleft Palate Clinic. In addition, they were seen in the ENT clinic at approximately 3-month intervals by a study physician or nurse practitioner blinded to the study group assignment for bilateral pneumatic otoscopy, tympanometry, ENT examination, and interval history. These assessments were used to determine the presence/absence of acute OM, OME or otorrhea at that visit and to determine VT patency. These visits and assessments were planned to be continued until the child's “graduation” from the study at the end of his/her 7th year of age. When possible, scheduled visits were coordinated with a scheduled visit to the Craniofacial/Cleft Palate Clinic for primary care. If the subject and parent were unable to come to the research clinic (e.g., due to parent's work, scheduling, etc), a “home visit” by the nurse practitioner and/or study physician was arranged for the interim history, pneumatic otoscopy and tympanometry. If an examination either at the clinic or at home was not possible (e.g., due to weather, illness, etc), an interim history was obtained from the parent by phone and if the subject had been examined by an outside health-care provider since the last study examination, the records for the visit(s) were requested from the service provider and the information included in the database.
During the follow-up visits when a VT was found to be no longer functional, either due to having been extruded or occluded, management of the subject was as per clinical standards. Occluded tubes received ear drops to attempt to restore tube function. In an ear without a patent and functioning tube, the ME was assessed by pneumatic otoscopy and tympanometry. Ears without ME effusion were followed to assess if the aerated ME status was sustained. Ears that developed OM were treated according to clinical standards, and if OME developed, they were followed to assess the duration of effusion and determine if the ears (subjects) would need reinsertion of tube(s). Bilateral VTs were reinserted if unilateral OME was diagnosed on three consecutive visits (6 months with unilateral OME), bilateral OME was diagnosed on two consecutive visits (3 months with bilateral OME) or if two or more episodes of acute OM were diagnosed over three consecutive visits. All VTs used during the study were designed for short-term (∼12 months) retention.
Primary outcome: the prevalence of OME between 2 and 7 years of age was assessed by otoscopy and tympanometry. We hypothesized that OME would be less prevalent in the MFP group when compared to the FP group. The effect of ETF on the middle-ear status cannot be assessed when there is a VT or a perforation after extrusion of a tube. The effect of the modification in the FP procedure on ETF manifests itself as the presence or absence of OME, after extrusion of a VT and healing of the tympanic membrane (TM), that is, an intact TM. To evaluate the outcomes, we conducted repeated assessments of the ears following VT extrusion and the healing of the TMs. Ears that met this criterion were deemed eligible for analysis. To have better confidence in the effect of the modified procedure on ME status of the child, the primary outcome analysis was on subjects who had bilateral qualifying ears.
Statistical Methods
Sample size calculation: A two group Fisher's exact test with a 0.05 two-sided alpha error was found to have 80% power to detect a between-group difference in OM proportions of .80 and .50 with equal sample sizes of 45 subjects or between .80 and .55 for equal sample sizes of 60 (assuming an analysis in year 6 based on the 120 enrolled subjects in years 1–4). A prospective study of 128 CP children who had undergone FP showed that 55% had unilateral or bilateral evidence of OM by tympanometry at 5 years of age, which, taking into account the age-related decrease in OM prevalence, supports our estimate for OM prevalence in the FP group. 24 The retrospective study by Flores and colleagues 20 reported a 26% difference in ears of 4-year-old children that did not require VT insertion in the previous year (diagnosis of OM at age 3.5 years) favoring those who had palatoplasties with tensor tenopexy when compared to those who had a variety of palatoplasties with tensor transection alone.
Data entered for each age time-point was defined as data from appointments within the child's date of birth ±6 months. For dichotomous measures of primary and secondary outcomes at each corresponding age, a Chi-square or Fisher's exact test was used to test for statistical significance in the frequency of OME between palatoplasty groups. Children who had VT reinsertion and extrusion were reassessed in a subsequent year if the TM had healed. To assess the cumulative incidence of OME occurrence between FP and MFP groups, a Kaplan–Meier time-to-event analysis was performed. Time to event was defined as time from palatoplasty until the date of appointment where OME was detected in one or both ears. The between-groups assessment was calculated using the Mantel–Cox Log Rank test. For Chi-square analysis by age, patients could re-enter the data set in the subsequent year if their ears qualified. On the other hand, Kaplan–Meier was censored, meaning that once they developed OME, they were kicked out of the model. Enrollment was stopped before reaching the originally planned sample size due to loss of external funding. Analysis was conducted utilizing the available data of the enrolled patients.
Results
Throughout the enrollment period, a total of 195 subjects with CP were screened. Sixty-seven were found to be ineligible due to various reasons (Figure 2). Of the 128 eligible, 26 did not consent, and 19 were awaiting consenting at the time of closing the enrollment. A total of 83 subjects were enrolled in the clinical trial by 15 months of age. Of these consented subjects, two were disqualified by their surgeon before randomization due to anatomical reasons. This resulted in 81 subjects who were randomly assigned, stratified by surgeon and Veau type: 42 subjects in the FP group and 39 subjects in the MFP group. After randomization on the day of surgery, a total of two subjects were disqualified, one in the MFP group with the diagnosis of submucous cleft and the other in the FP group due to excessive width of the CP that impacted surgical repair. Two subjects were lost to follow-up immediately after surgery, one in each group, with no further study data available. Four subjects had no visits after 2 years of age, three in the FP and one in the MFP group. A total of 37 subjects with FP and 36 subjects with MFP were included in this analysis.
Average age at the time of palatoplasty was 1.05+/–0.174 in the FP and 1.07+/–0.184 in the MFP groups (p: .585). A total of 26 females and 47 males were enrolled (Table 1), with no significant differences between the FP and MFP groups (p: .931). There were only two nonwhite subjects, one in each group. Over 94% of subjects in both groups had the diagnosis of OME prior to palatoplasty. Secured by the randomization method, Veau distribution between the FP and MFP groups was not statistically different (p: .848). History of cheiloplasty was also similar between the two groups (p-value = .677)
Baseline Characteristics of Children Undergoing FP Versus MFP at the Time of Cleft Palate Repair.
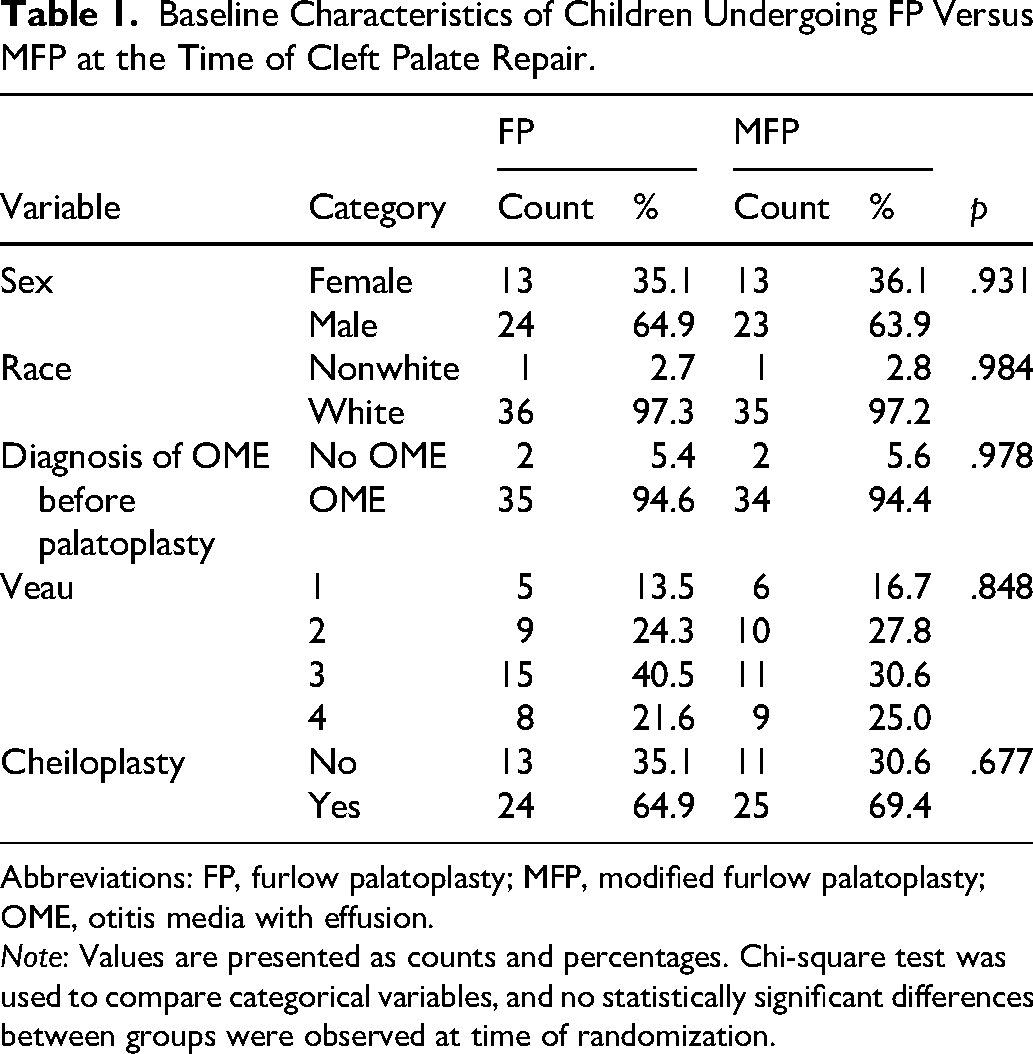
Abbreviations: FP, furlow palatoplasty; MFP, modified furlow palatoplasty; OME, otitis media with effusion.
Note: Values are presented as counts and percentages. Chi-square test was used to compare categorical variables, and no statistically significant differences between groups were observed at time of randomization.
As the primary outcome measure, presence and absence of OME was analyzed between the groups for each year, including only the subjects that had bilateral qualifying ears, that is, both ears with intact TMs after extrusion of the VT (Table 2). A total of 16 (eight FP, eight MFP), 30 (16 FP, 14 MFP), 34 (19 FP, 15 MFP), 31 (18 FP, 13 MFP), 15 (five FP, 10 MFP), and nine (four FP, five MFP) subjects had bilateral ears qualifying for statistical analysis for the primary outcome for years 2, 3, 4, 5, 6, and 7, respectively. There were no differences between the FP and MFP groups over years 2–7.
Frequency of OME by Age among Children with Bilateral Intact Tympanic Membranes after Ventilation Tube Extrusion.
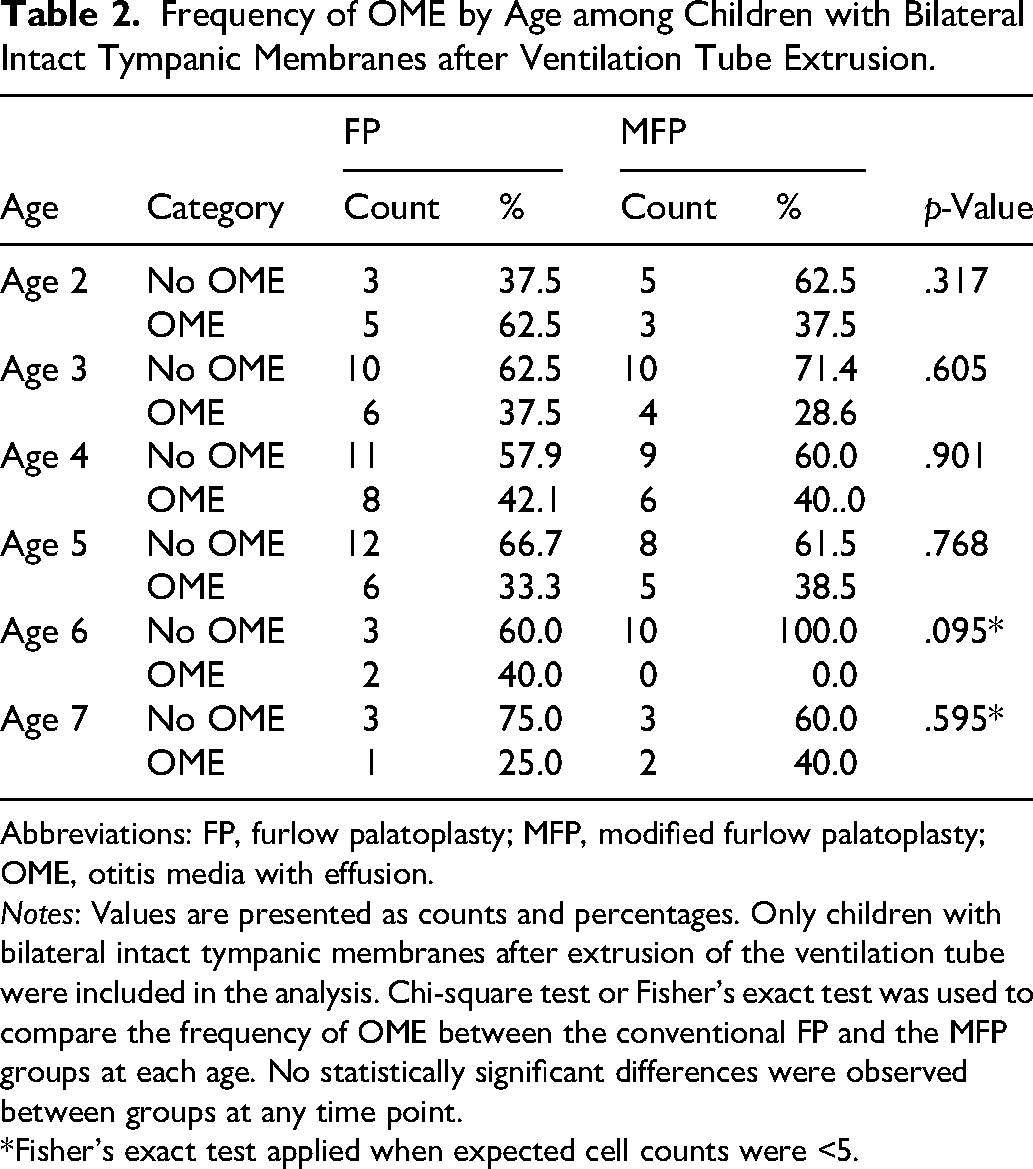
Abbreviations: FP, furlow palatoplasty; MFP, modified furlow palatoplasty; OME, otitis media with effusion.
Notes: Values are presented as counts and percentages. Only children with bilateral intact tympanic membranes after extrusion of the ventilation tube were included in the analysis. Chi-square test or Fisher's exact test was used to compare the frequency of OME between the conventional FP and the MFP groups at each age. No statistically significant differences were observed between groups at any time point.
*Fisher's exact test applied when expected cell counts were <5.
Although not predefined as the primary outcome measure, an additional analysis of the presence and absence of OME was conducted between the groups including those with at least one qualifying ear irrespective of the condition of the other ear (Table 3). A total of 47 (25 FP, 22 MFP), 83 (45 FP, 38 MFP), 82 (45 FP, 37 MFP), 68 (38 FP, 30 MFP), 35 (14 FP, 21 MFP), and 21 (nine FP, 12 MFP) subjects had at least one qualifying ear for statistical analysis for the secondary analysis of the primary outcome for years 2, 3, 4, 5, 6, and 7, respectively. There were no differences between the FP and MFP groups over years 2–7 (p-values for each year varied between .145 and .963).
Frequency of OME by Age among All Ears with Intact Tympanic Membranes after Ventilation Tube Extrusion.
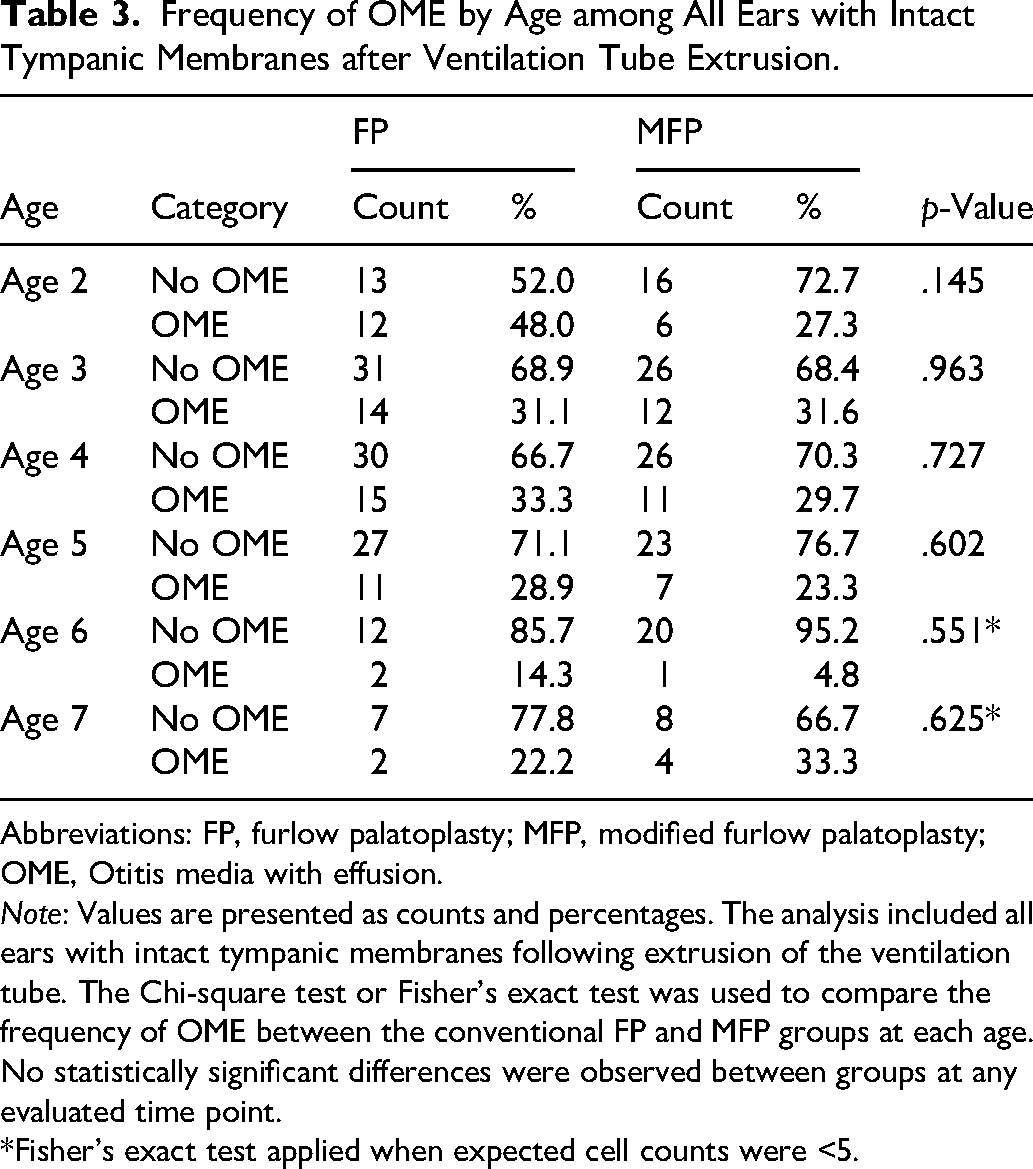
Abbreviations: FP, furlow palatoplasty; MFP, modified furlow palatoplasty; OME, Otitis media with effusion.
Note: Values are presented as counts and percentages. The analysis included all ears with intact tympanic membranes following extrusion of the ventilation tube. The Chi-square test or Fisher's exact test was used to compare the frequency of OME between the conventional FP and MFP groups at each age. No statistically significant differences were observed between groups at any evaluated time point.
*Fisher's exact test applied when expected cell counts were <5.
A Kaplan–Meier time-to-event analysis was conducted for time to first episode of OME, displayed as cumulative incidence in Figure 3. There was no difference between the FP and MFP groups regarding the cumulative incidence of OME as per available follow-up data of the subjects.
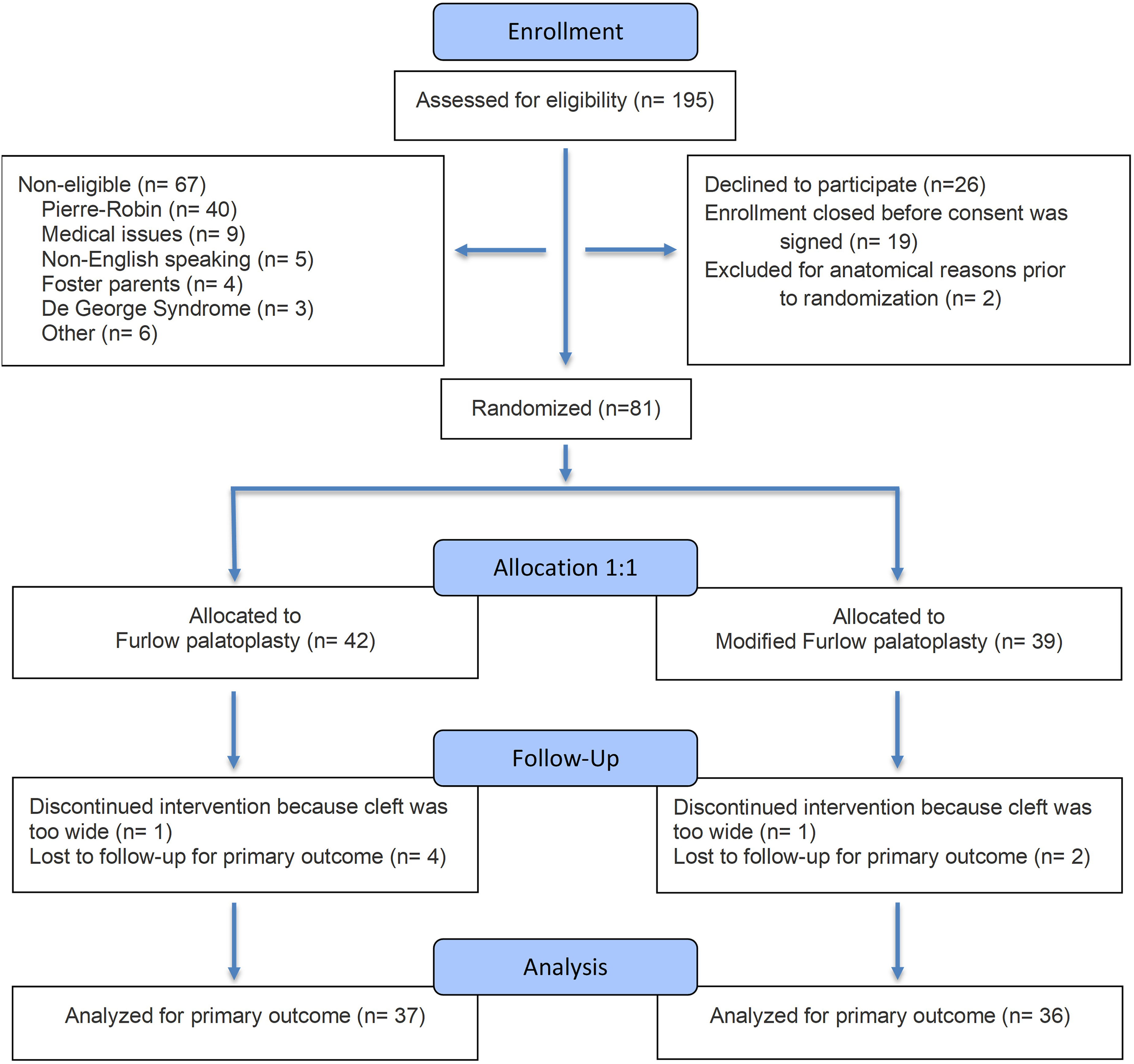
Flowchart demonstrating number of patients assessed for eligibility, enrollment, randomization, exclusion, and those included in the analysis.
Secondary analyses were performed comparing the FP and MFP groups regarding other palatoplasty and otologic outcomes (Table 4). Comparison of need for Furlow revision or alveolar graft showed no statistical difference. Similarly, need for adenoidectomy, VT reinsertion and tympanoplasty on right or left ears did not show any statistical difference.
Surgical and Otologic Procedures Performed During Follow-up in Children Undergoing FP and MFP.
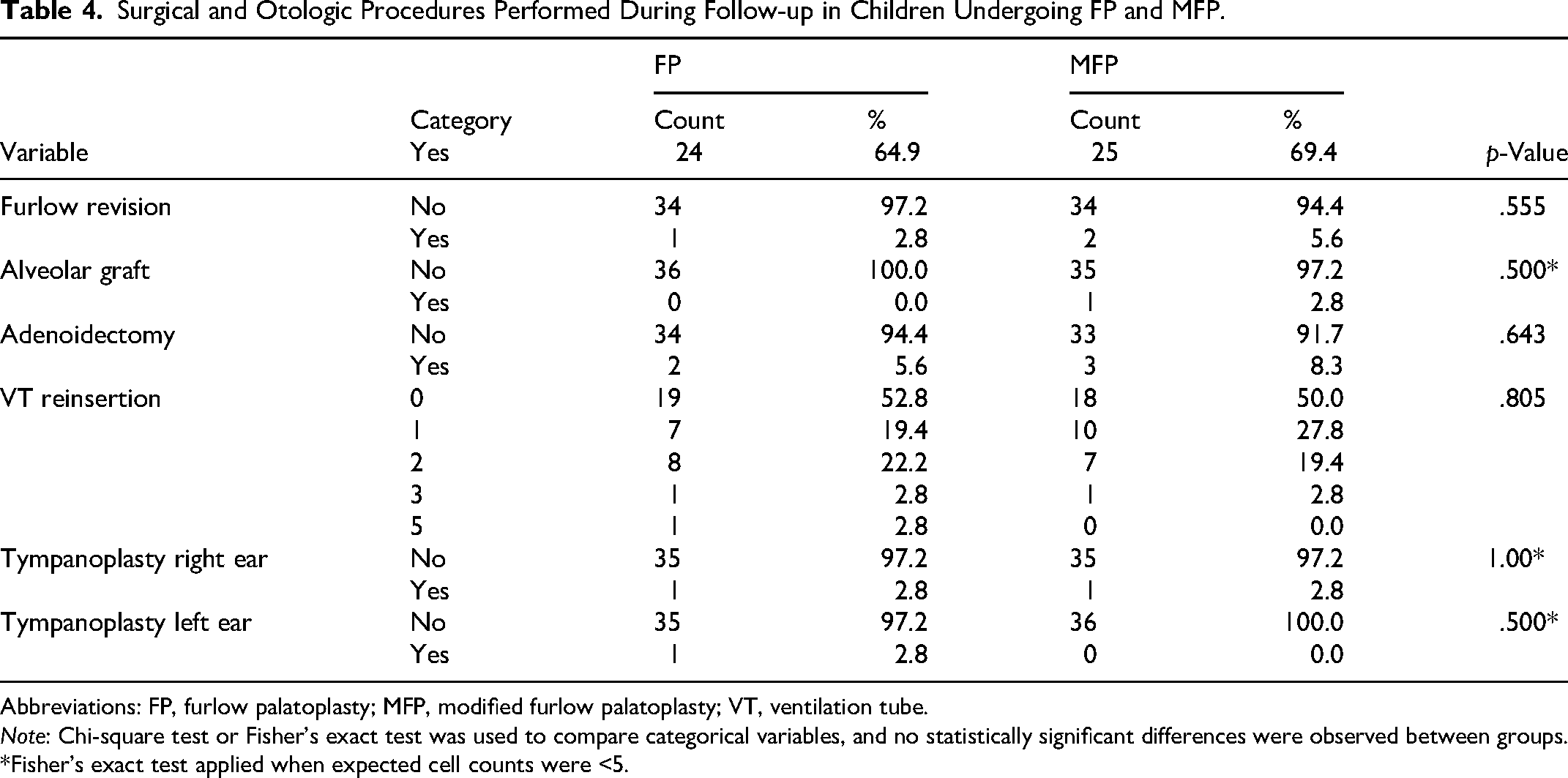
Abbreviations: FP, furlow palatoplasty; MFP, modified furlow palatoplasty; VT, ventilation tube.
Note: Chi-square test or Fisher's exact test was used to compare categorical variables, and no statistically significant differences were observed between groups.
*Fisher's exact test applied when expected cell counts were <5.
After the extrusion of those VTs, reinsertion was considered as per the tube reinsertion criteria in the ENT practice.
All subjects underwent routine audiologic testing at the first postoperative visit with the otolaryngology clinic. Although tests were often conducted in the sound field, there was no evidence of abnormal hearing in any subject. At the 3- and/or 4-year time-points, 52 of the 71 subjects (73.1%) had an audiologic evaluation. Of the 52, 80.8% had normal hearing, and 17.3% had mild conductive hearing loss, consistent with their ME status. There was no difference between the study groups with respect to the hearing results.
DSMB Reports
There were nine adverse events reported to and reviewed at the DSMB meetings. None of the adverse events required breaking the randomization code. All but one event were considered nonserious in the MFP group with chronic otorrhea possibly related to the study procedure, requiring a peripherally inserted central catheter (PIC-line) insertion and intravenous antibiotic treatment, resulting in resolution. Four of the adverse events were assessed as unrelated to the study procedure, including respiratory distress after palatoplasty in two children in the MFP group that required Intensive Care Unit (ICU) care after which the condition resolved. In the FP group, unrelated events included a VT displacement into the ME that was recovered and a patch myringoplasty was performed and one other subject had brief postoperative bleeding from the repaired soft palate that resolved (which was the only unexpected event among all nine adverse events). Four other adverse events were possibly related to the study procedure: oro-nasal fistula in three subjects, two in the MFP group (one required repair, the other resolved while being observed); and one in the FP (required repair). One subject in the MFP group required revision palatoplasty with pharyngeal flap.
Discussion
The prevalence of OME in children undergoing palatoplasty decreases gradually with age. In a prospective study of 150 CP+/–CL children enrolled between 2 and 18 months of age, Robinson et al. reported a 92% prevalence of OME before palate repair, no short-term change after palate repair, but a reduced prevalence of 70% at 4 years of age. 25 Almost identical results were reported by Duroux and colleagues. 26 Moller and colleagues described a universal prevalence of OME at <3 years of age, a decreasing prevalence between 3 and 5 years and a stable prevalence from 6 years into adolescence. 27 The prevalence of OME was estimated by others to be between 10 and 30%. 28 The results of a previous longitudinal study of 52 CP+/–CL subjects who had FP and were prospectively followed from approximately 6 months to 6 years of age showed a linear decrease in the prevalence of OME over that age range, with no evidence of modification by palate repair. 29 Specifically, the cumulative percent OME resolution for ears/children at ages <1, 1, 2, 3, 4, and 5 years of age was 4.1/4.4, 14.3/10.9, 31.6/21.7, 45.9/37.0, 56.1/50.0, and 70.4/60.9% with OME resolution slopes of 13.5% (r2 = 0.99) and 11.9% (r2 = 0.99) resolutions per year for ears and children, respectively. 29
The prevalence of OME is not affected by the standard procedures for palatal repair25,30,31 or by modifications in the stepped timing of those procedures15,16,32,33 but may17,18 or may not24,34 be affected by procedures that change the orientation of the mLVP.
Previous observations suggest that OME in CP+/–CL patients is a consequence of ETD. Not only is the functional anatomy of the ME system abnormal but comparative histological study of specimens collected from fetuses, newborns, and young children with CP+/–CL35–37 reported other anatomical differences from “control” specimens. These include a greater medial to lateral compression of the ET lumen, a narrower angle between the mTVP and the cartilaginous ET lumen, a lesser area of mTVP origin, a wider angle between the lateral and medial cartilage and a lesser cartilage volume in the CP+/–CL specimens.38–42 Many of these anatomical features for the CP+/–CL ET system have expected adverse effects on ETF.
While only a few studies described the effect of palatoplasty on ETF, 43 a number of studies examined the effects of those procedures on OME prevalence at different ages. Most palatoplasty methods other than the FP do not re-establish a physiologic realignment of the mLVP while closing the cleft. Unlike other surgical techniques that close the palatal defect at midline without attention to muscle fiber direction, FP, a double z-plasty technique, actually results in redirecting the mLVP muscles from each side, overlapping in parallel orientation at the midline, and results in better outcomes with respect to establishing VP competence. 44 However, like some other palatoplasty methods, FP technique requires cutting the mTVP attachments to the hard palate shelf just medial to the hamulus to release the tethered mLVP, reposition it at the middle and elongate it to achieve better VP closure, and better swallowing and speech outcomes. The abnormal attachment of the mTVP to the hard palate shelf already impairs stiffness of the lateral mTVP muscle bundle necessary for the medial bundle to dilate the ET lumen. The loose mTVP results in unpredictable re-establishment of a new insertion point as it heals with scarring within the soft tissues of the soft palate resulting in potentially weak and variable stiffness of the main mTVP muscle bundle. This variability in attachment site and angle may account for the continued and unpredictable ETF after palatoplasty. This expected negative effect brought a potential to improve ETF further, if the negative impact of cutting the mTVP could be prevented. Tensor tenopexy, placement of a stitch securing the mTVP to the hamulus prior to cutting its attachment to the hard palate was proposed to maintain mTVP stiffness and improve the ETD outcomes.
An earlier study that investigated the effect on ME disease of unilateral fracture of the pterygoid hamulus during soft palate closure did not show a significant difference. 22 Two uncontrolled, nonrandomized, retrospective reports that initially described modifications to the FP noted decreased OME prevalence in CP+/–CL children who had the modified palatoplasties when compared to more standard palatoplasties. However, both reports had significant design limitations that preclude uncritical acceptance of those findings. The first study published by Butow and colleagues 19 compared OME prevalence after intravelar veloplasties with mTVP tendon transection for groups that had palatoplasty with (prospective) and without (retrospective) the modification to create an mTVP sling. The authors’ analysis of the otologic data suggested a better postoperative ME status in patients who had the modified palatoplasty procedure (mTVP sling), but this improvement was not carefully documented and the group sample sizes (n = 20 and 19, respectively) were small. Also, ears rather than persons were treated as the experimental unit; surgeries for the two groups were performed at different ages (6–13 months and 24–36 months, respectively) and by different surgeons; the follow-up period was relatively short and not the same for the two groups (1, 3, and 9 months postpalatoplasty and immediately postpalatoplasty, respectively), and the postpalatoplasty assessment of ME status was limited to tympanometry. Notably, while the procedure description and its purported benefits for ME status were first described in a 1991 publication, no further reports (either positive or negative) have been published, which suggests that the procedure has been largely ignored by CP surgeons.
The second study by Flores and colleagues published in 2010 20 is a retrospective, uncontrolled, nonrandomized study that compared “at least” three palatoplasty procedures with respect to OME resolution; the two-flap Bardach or the von Langenbeck procedures, both without mTVP tendon transection, and performed at one institution by the on-site surgeons (Group Ia,b, retrospective). Levator sling palatoplasty with mTVP transection was done at a second institution by a different group of surgeons (Group II, retrospective), and levator sling palatoplasty with tensor tenopexy was carried out by the surgeon who developed that modification (Group III). Patients were followed infrequently (every 6 months) to age 7 years by otoscopy and tympanometry with the primary endpoint being a 1-year period without the need for VT reinsertion. Pair-wise comparisons between groups suggested that Group III had fewer VT insertions than the other two groups at the various assessment ages. A general critique of the study includes the facts that: it is not known if the cleft types were balanced for the three/four groups; the Bardach and von Langenbeck procedures were not analyzed as separate subgroups; the inclusion of different surgeons and institutions for each group introduces biases that may have influenced the results consequent to differences in the timing of the palatoplasties; the skill level of the surgeons, the otoscopic skill of the evaluators, and, as pointed out by the authors, the methods of evaluation, the criteria for VT insertions and the type of VTs used by the different surgeons at the various institutions may have impacted the result. Also, the failure to randomize subjects to the different surgical groups introduces the possibility of patient preselection based on undefined/unspecified, preferential surgeon-defined assignment to a given group. Other problems with the study design include: the small number of subjects studied (N = 32, 16, and 26 for Groups Ia,b, II, and III, respectively); the failure to control for the many multiple comparisons done and to use continuity corrected statistical methods which artificially increases the significance levels for the repeated pair-wise comparisons; the lack of a description of adverse events associated with the procedures and the apparent inconsistency of the results for the Group Ia,b–Group III comparison where differences in otologic outcome were noted despite the fact that both retained or established a fixed mTVP insertion. A third study, by Tiwari and colleagues published in 2013, compared tensor tenotomy (the release of the tensor for improved speech outcomes) and tensor tenotomy with tensor tenopexy (placement of a stitch around the hamulus, as it is performed in our study) in a total of 17 subjects (on eight and nine subjects, respectively). 21 Subjects did not receive VTs concurrent with CP repair and were followed for only 1 year with otoscopy, tympanometry, and brainstem evoked response audiometry. There were no significant differences between ME pressures and compliance between groups; however, the authors found worse otoscopic findings in the tensor tenopexy group and concluded that this modification does not improve ETF and can sometimes hamper ET function, resulting in OM and hearing loss in the short term. The main weakness of this study was the short-term follow-up and the small number of subjects in each group. In both the latter studies, the need for better controlled, appropriately powered, long-term studies was emphasized, and a review of that procedure modification concluded that its potential impact on clinical practice will be minimal, at best, until (and if) the positive results are validated in a prospective, randomized, blinded study. 45 A more recent publication described the tensor stitch placement during CP closure, claiming that this modification improves ETF and prevents OM, without any data presentation. 46 It is not clear currently the proportion of surgeons who are routinely performing tensor tenopexy during CP repair, based on those publications that claim benefit.
We conducted this study to eliminate the limitations of the previously described studies to assess the effect of tensor tenopexy on the otologic outcomes after palatoplasty. The randomized study design with stratification as per Veau type and surgeon, and study outcomes assessors blinded to the study group assignment were to eliminate bias. While long-term outcomes could not be assessed due to early termination of the study, a near complete dataset is available only for ages 3 and 4. Because enrollment ended before the target sample size was achieved, the final sample was smaller than originally planned and therefore reduced the statistical power to detect smaller treatment effects.
The interpretation of otologic outcomes of the current study has limitations due to the short follow-up. However, comparison of the FP and MFP groups at the 2–7-year-old time-points does not demonstrate any benefit from tensor tenopexy. Similarly, the VT reinsertion and tympanoplasty rates between the groups did not demonstrate a statistically significant difference between the groups. This study is not able to demonstrate any harm in performing this modification but the lack of benefit in these results in the well-controlled study should be considered while making decisions in palatoplasty method for CP repair. On the other hand, the number of subjects included in the analysis being below our power calculations may have weakened the ability of this study to capture possible benefits of the tensor tenopexy. Although possible, looking at the current analysis, particularly the Kaplan–Meier cumulative incidence (Figure 3), this seems unlikely. Assuming that this modification to the FP does require additional operative time with associated operative cost and lack of demonstrable benefit, it is advisable to not perform tensor tenopexy during the FP procedure.
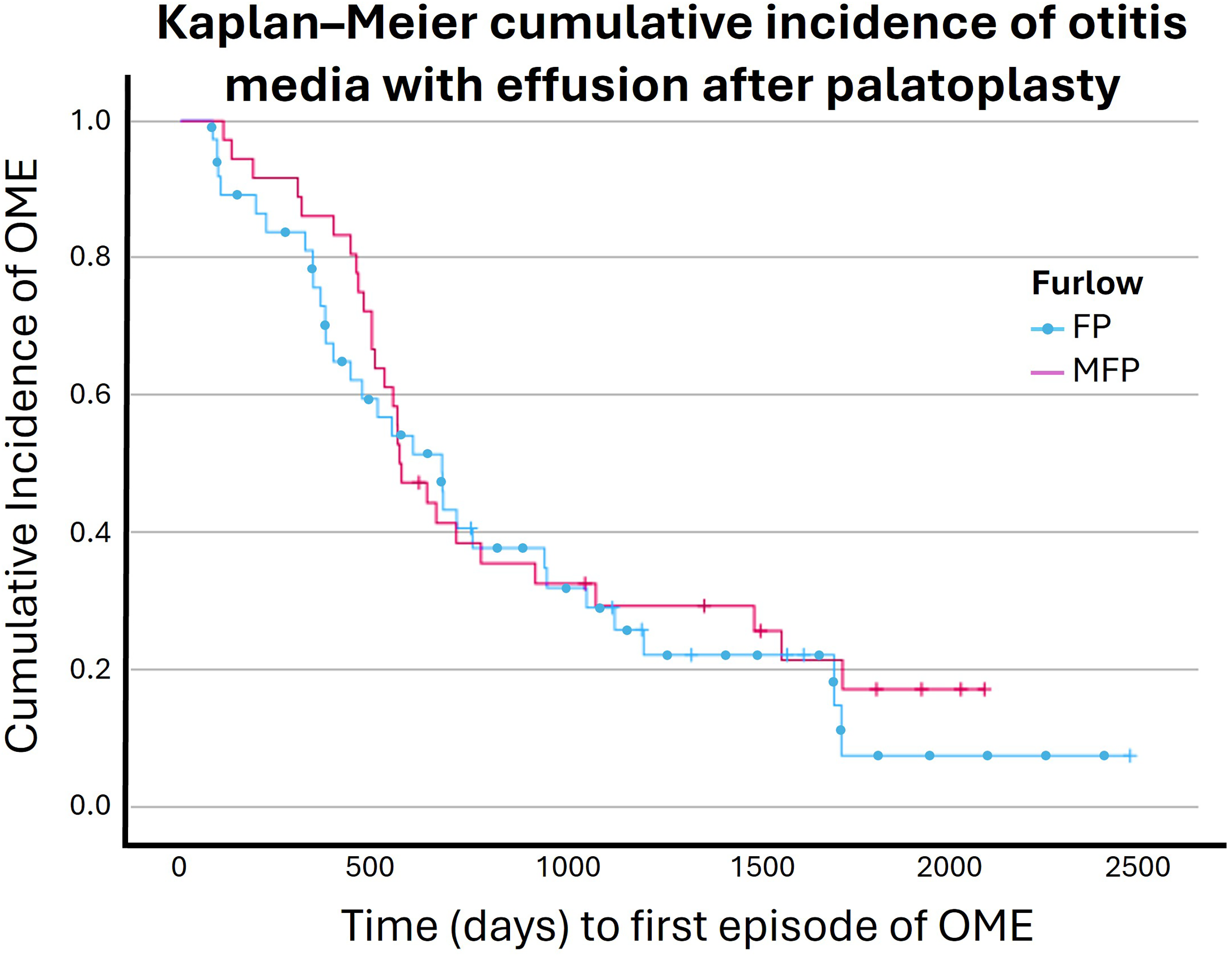
It shows the Kaplan–Meier time to event analysis for both FP and MFP groups. The Mantel–Cox log rank test demonstrated that there was no significant difference between groups related to the occurrence of OME over time (χ2 = 0.324 p = .569).
Footnotes
Acknowledgments
This study was supported in part by a grant from the National Institutes of Health, R01DC011524. The authors thank Dr William J Doyle, PhD, for his help in the study concept, initial data analysis, and interpretation; Thomas Wasser, PhD, for his help in the statistical analysis; and Beverly C. Richert CRNP, PhD, James Seroky, MA, and Julianne Banks, BS, for data collection and entry. Illustration by David Low, MD, Professor Surgery, University of Pennsylvania (services for hire).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Deafness and Other Communication Disorders (grant number R01DC011524).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional Review Board Approval Number and Approval Date
PRO11060248, 7/17/2012
