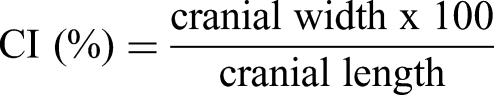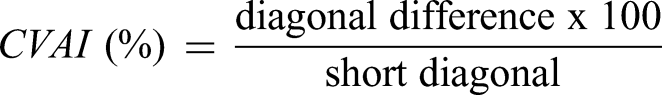Abstract
Objective
This study aimed to investigate the impact of helmet therapy on changes in cranial asymmetry of infants with positional cranial deformities and to identify clinical and morphometric factors associated with treatment outcome.
Design
This retrospective monocentric cohort study included 455 infants treated between 2011 and 2021.
Setting
Data were collected using manual measurements in a tertiary care setting within the outpatient clinic of a maxillofacial surgery department in Germany.
Patients
Infants with positional cranial deformities, including plagiocephaly, brachycephaly, and combined types, were included.
Intervention
All patients were treated with a helmet orthosis, and data were analyzed to assess changes in cranial measurements and associated factors.
Main Outcome Measures
Predefined outcome thresholds based on cranial asymmetry reduction and prognostic factors.
Results
Plagiocephaly was the most common deformity in 332 infants. Increased ear shift (Exp(B) = 0.199; 95% CI: 0.087–0.451; p ≤ .001) and large initial diagonal difference (Exp(B) = 0.518; 95% CI: 0.334–0.802; p = .003) were correlated to unfavorable outcome. Delayed treatment was associated with lower reduction in diagonal difference (rs = –0.229; p ≤ .001), and recognized as a negative prognostic factor (Exp(B) = 0.993; 95% CI: 0.989–0.997; p ≤ .001). In contrast, a higher baseline cranial index increased the probability of success (Exp(B) = 1.057; 95% CI: 1.030–1.084; p ≤ .001).
Conclusion
Early diagnosis and intervention are crucial for effective treatment of cranial deformities with helmet orthosis, especially in cases with ear shift or severe initial deformation.
Introduction
Deformational cranial deformities, including deformational plagiocephaly (DP) and brachycephaly, are prevalent during infancy and can be evident at birth or become obvious during the initial weeks of life.1–6 A notable upward trend has been observed since 1992, when the “Back to Sleep Campaign” was launched in April by the American Academy of Pediatrics (AAP). The campaign recommended a supine sleeping position to reduce sudden infant death syndrome (SIDS). Although this campaign has been successful in reducing SIDS, it has contributed to a rise in the incidence of positional skull deformities.2,7,8
Unilateral occipital flattening is the typical clinical manifestation of DP in infants. In moderate-to-severe cases, this may be accompanied by anterior displacement of the ipsilateral auricle and prominence of the ipsilateral frontal region, culminating in a parallelogram-like cranial contour.9,10 The deformity is the consequence of constant external pressure (in a supine position) on the newborn's incompletely ossified and pliable cranial vault. 11 The etiology of DP is multifactorial, encompassing mechanical influences such as the preference for a supine position, intrauterine restrictions, primiparity, assisted deliveries, multiple births, premature births, and muscular torticollis.1,2,4,12–15 The mother’s age (>35 years) and male sex can also be cited as risk factors. 16
The diagnosis is primarily based on clinical and anthropometric examinations.13,17 Three-dimensional (3D) imaging is recommended because it is less dependent on compliance and the experience of the examiner. 18
Although a causal relationship between skull deformities and abnormal brain development in infants and the occurrence of neurophysiological problems has not been established, the prevention of these deformities is of great importance to avoid aesthetic and functional consequences, such as impaired jaw growth, malpositioning of the eyes and ears, otolaryngological dysfunction, and asymmetries of the facial skeleton and skull base.11,19,20–22 Early identification and intervention have been described in the literature as important for reducing the likelihood of long-term complications.5,23,24 Various sources exist regarding the age at the start of therapy, which is commonly reported to occur between the fourth and twelfth month of life. 25 Several authors have suggested that helmet therapy should ideally be initiated between four to six months of age. One explanation is that the growing brain, as a stimulator, provides the foundation for neurocranial development, and as brain growth slows significantly after the age of 12 months, correction of existing asymmetries becomes more difficult as infants get older.21,24–27
Prevention options include correct vitamin D and folic acid levels during pregnancy, using a passive sleep mattress or education of parents.6,19 Furthermore, prone positioning (“tummy time”) of at least 30 min per day under adult supervision has been frequently described in the literature as a preventive measure associated with a reduced risk of occipital flattening. Unlike physiotherapy or helmet therapy, the positioning of the child can easily be delivered without specialist staff or long journeys.15,19,28
Therapeutic interventions of cranial deformities may be observation, physiotherapy, external orthotics, parental education on active counter positioning and in cases of craniosynostosis surgical procedures.14,29–33 The primary treatment options include conservative approaches such as positioning therapy for the infant using positioning aids and regular changes of position, physiotherapy and osteopathy at an early age. 4 In moderate to severe cases, helmet therapy (cranial orthotic therapy) has been described as an additional treatment option. 34 The objective of helmet therapy is to redistribute external pressure and guide the skull into a more symmetric shape. 13
Despite its efficacy, helmet therapy is not without controversy. Challenges include high treatment costs, potential adverse effects such as skin irritation,25,35 and the psychosocial burden for parents. Moreover, there is a concern that a significant proportion of cases may resolve spontaneously as the child grows, given the difficulty in measuring the direct impact of helmet therapy due to the influence of numerous co-factors.4,17 Nevertheless, helmet therapy remains the preferred treatment option for more severe deformities, particularly in cases where conservative measures prove ineffective.11,13,21
The current evidence base indicates that the therapeutic outcome of non-synostotic cranial deformities is influenced by a range of factors, including severity of the deformity, age at initiation, treatment compliance, health insurance and potential muscular or genetic comorbidities.5,11,17,25,26,36
Our retrospective observational study aimed to investigate the impact of helmet orthosis therapy on changes of cranial asymmetry parameters in infants with cranial deformities. Parameters potentially influencing therapy success like age at treatment initiation and severity of baseline deformity were focused.
The null hypothesis (H₀) posited that there is no significant association between treatment timing or deformity severity and therapy success. The alternative hypothesis (H₁) assumed that earlier treatment initiation and greater initial severity are associated with differences in treatment outcomes.
Materials and Methods
Patient Selection and Data Collection
This retrospective, monocentric study was conducted at the Department of Cranio- and Maxillofacial Surgery. Between 2011 and 2021, a total of 455 infants with positional cranial deformities who were treated with a helmet orthosis due to a cranial deformity were included in the study. Patient selection was based on the diagnosis of a cranial deformity, and all infants who were treated with a helmet orthosis for plagiocephaly (n = 332), brachycephaly (n = 14), a combination of plagiocephaly and brachycephaly (n = 108), or a combination of plagiocephaly, brachycephaly, and dolichocephaly (n = 1) were included. This study was conducted in accordance with the Declaration of Helsinki and approved by the institutional ethics committee. As this was a retrospective, anonymized analysis of routine clinical data, informed consent was not required, except for the infant whose photograph is included. In this case, parental consent was obtained. Inclusion criteria consisted of infants who underwent helmet orthosis therapy as part of routine clinical care for positional cranial deformities. Exclusion criteria were incomplete medical documentation, absence of a confirmed positional cranial deformity, or cranial deformities caused by craniosynostosis. Data collection was carried out using conventional patient records. The relevant information was manually gathered from the paper-based records, and then compiled into an Excel spreadsheet (Microsoft Excel 2024, Microsoft Corporation, Redmond, WA, USA) for further analysis. The collected data included demographic, clinical, and morphometric variables, such as sex, age therapy initiation and completion, treatment duration, diagnosis, ear shift, referral pathway, and type of birth. All collected data were anonymized to ensure patient confidentiality.
Manual Measurement
The data were collected by residents from the Department of Cranio- and Maxillofacial Surgery as part of the routine helmet consultation. Manual cranial measurements were performed using a measuring tape and a Bertillon-type head caliper (Germany; measurement range: 0–250 mm; total length: 320 mm). Each child was measured by a single trained resident and only manual measurement data were used throughout the study to ensure methodological consistency. Since 2016, the measurement has additionally been performed using 3D digital photography utilizing a five-lens setup combined with a multi-flash lighting array (Vectra M5 3D Imaging System, Canfield Scientific, Parsippany, NJ, USA) (see Figure 1). 18 However, only manual measurement data were included in the present analysis to ensure comparability across the entire study period. The manually collected data included several cranial parameters: head circumference, head length and width; right (RD) and left (LD) diagonal head lengths, measured at a 30° angle relative to the median sagittal plane; the cranial asymmetry index derived from the difference between these diagonals; and the glabella-to-tragus distances on both sides (GTR and GTL, respectively), which served as a reference measure for ear shift. These measurements were recorded regularly during follow-up consultations to monitor changes in cranial shape over time. All data were transferred to the patient record by a dental assistant. Cranial circumference was measured with a flexible measuring tape, ensuring consistency in the placement of the tape around the largest part of the head. Diagnoal head lengths, cranial length, and cranial width were measured with the anthropometric caliper, ensuring accuracy in the angles and distances being recorded. The glabella-to-tragus distance was again measured with a flexible measuring tape. These manual measurements provided the essential baseline data for descriptive analysis and longitudinal assessment of cranial shape changes. Figure 1 shows a representative clinical image of the camera setup. Figure 2 illustrates cranial measurement points.

Configuration and calibration of the Vectra M5 360 imaging system, shown in conjunction with a toddler positioning chair and exemplary helmet.
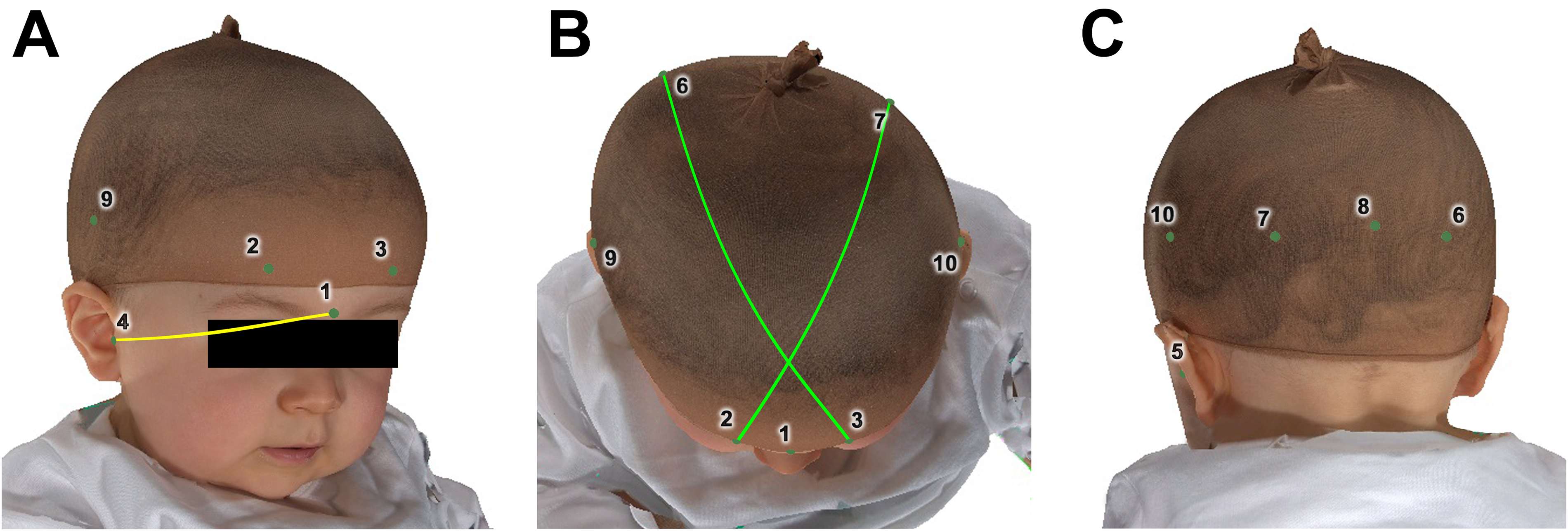
Anatomical landmarks used to measurement individual cranial parameters, illustrated from the right anterior (A), superior (B), and left posterior (C) perspectives. The yellow line represents the glabella-to-right tragus distance; green lines indicate diagonal measurements extending from the most prominent supraorbital point to the contralateral most prominent occipital point. Landmark references: (1) glabella; (2) right supraorbital prominence at a 30° angle to the median plane; (3) left supraorbital prominence at a 30° angle; (4) right tragus; (5) left tragus; (6) right occipital prominence at a 30° angle; (7) left occipital flattening at a 30° angle; (8) maximal occipital width; (9) maximal lateral width on the right; (10) maximal lateral width on the left 18 .
Data Analysis
For a more precise assessment, we used the cranial index (CI), also known as the cephalic index, which describes the relationship between the width of the skull and the length of the skull. The CI is a widely accepted anthropometric parameter used to classify head shapes and assess cranial abnormalities. It is especially valuable in evaluating conditions like plagiocephaly and brachycephaly, where disproportionate skull dimensions can be present. Calculation of the CI allows for a standardized description of cranial proportions and their changes over time, providing an objective morphometric parameter for analysis.20,37
The Cranial Vault Asymmetry Index (CVAI) was additionally calculated and derived from the length discrepancy between 2 diagonal measurements. The diagonal difference is the difference between the long diagonal (3 and 6, Figure 2) and the short diagonal (2 and 7) oriented at a 30-degree angle relative to the Y-axis, as shown in Figure 2. A CVAI exceeding 3.5% is indicative of asymmetry and classified as pathological. The CVAI provides further detail on the asymmetry of cranial shape by specifically measuring the diagonal disparities between the skull's right and left sides. This is particularly helpful in assessing complex deformities.
20
These indices were already suggested by Loveday et al. in 2001 20 and Mortenson et al. in 2006 37 forming the foundation for reliable diagnostic assessments and offering a standardized approach to evaluating cranial deformities.
Statistical Analysis
IBM SPSS Statistics version 29.0 (IBM Corp., Armonk, NY, USA) and Microsoft Excel (Microsoft Corp., Redmond, WA, USA) were used for statistical analysis and data visualization. Descriptive statistics were employed initially to describe the parameters collected and summarize the distribution of the data. Mean values provided a reference point for comparing cranial measurements, while standard deviation (SD) and standard error of the mean (SEM) were used to quantify variability and the precision of the estimates.
Subsequently, Spearman's rank correlation coefficient was used to assess associations between relevant variables. In addition, logistic and linear regression analyses were performed to examine associations between clinical or morphometric variables and predefined outcome measures. A p-value of < .05 was considered statistically significant. For analytical purposes, outcome categories were defined based on previously published criteria. According to Steinberg et al., a final diagonal difference of ≤ 0.5 cm was used as a threshold for categorizing outcomes 21. Furthermore, an initial ear shift of ≥ 1.0 cm was defined as clinically relevant for analytical stratification, while an ear shift of ≤ 0.3 cm at the end of treatment was considered indicative of a favorable morphometric outcome. These analyses were conducted to explore factors associated with changes in cranial morphology during helmet orthosis therapy. This manuscript adheres to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for reporting observational cohort studies.
Image Processing:
Adobe Photoshop Elements 2023 for Mac (Adobe Inc., San José, CA, USA) was used for the image processing of Figure 2. This software was applied to optimize contrast and visual clarity, ensuring that anatomical landmarks and measurement points were clearly identifiable.
Results
Clinicopathological Characteristics
Between 2011 and 2021, a cohort of 455 infants with positional cranial deformities was treated with a helmet orthosis for cranial deformity at our hospital and was included in this study. Of these, 300 were male and 155 were female. The primary diagnosis was plagiocephaly in 332 (73.0%) cases. Of the remaining infants, 14 (3.1%) were diagnosed with brachycephaly, 108 (23.7%) with a combination of plagiocephaly and brachycephaly and 1 (0.2%) with a combination of plagiocephaly, brachycephaly, and dolichocephaly. Helmet therapy was initiated at a mean age of 6.98 months (SD ± 1.80), with a mean duration of 162.90 (SD ± 55.31) days and a range of 21‒ 490 days. The mean age at the end of therapy was 12.41 (SD ± 2.77) month. Multiple births were recorded in 58 cases, while 397 infants were singletons. The most common mode of delivery was spontaneous vaginal birth in 232 infants (51.0%), followed by spontaneous birth with a special birth position in 4 cases (0.9%). Furthermore, 168 infants (36.9%) were born via planned cesarean section, 15 (3.3%) via emergency cesarean section, 9 (2.0%) via cesarean section with a special fetal position, and 27 (5.9%) were delivered using a vacuum extractor (suction cup). A total of 370 cases (81.3%) exhibited an ear shift of >1.0 cm and 85 cases (18.7%) showed no clinically relevant ear shift. An overview of the clinicopathological characteristics of the cohort is provided in Table 1.
Summary of Clinical and Pathological Characteristics Within the Cohort.

Note: 1 MV = mean value; SD = standard deviation.
Head Measurements
All manual measurements were performed using a flexible tape measure and an anthropometric head caliper, and were gauged in centimeters. A diagonal difference was calculated based on the cranial measurements, with a mean value of 1.42 ± 0.50 cm at baseline and 0.36 ± 0.28 cm at the final assessment. The initial ear shift measured 0.58 ± 0.43 cm and the mean cranial index (CI) at baseline was 93.37 ± 8.79%. The CVAI at baseline was 11.28 ± 4.28%. The final measurements showed a mean diagonal difference of 0.36 ± 0.28 cm, an ear shift of 0.19 ± 0.20 cm, a cranial index (CI) of 89.34 ± 6.07%, and a CVAI of 2.56 ± 2.02%. Based on predefined analytical thresholds, a final diagonal difference of ≤ 0.5 cm was observed in 353 infants (77.6%), whereas 102 infants (22.4%) exceeded this threshold. The overall reduction in diagonal difference was 1.06 ± 0.47 cm. Similarly, a final ear shift ≤ 0.3 cm was observed in 305 infants (67.0%), while 150 infants (33.0%) exceeded this value. The mean reduction in ear shift was 0.39 ± 0.41 cm. Detailed head measurement data are summarized in Table 2.
Head Measurements of the Patient Cohort (n = 455).

Note: 2 MV = mean value; SD = standard deviation; CI = cranial index CVAI = cranial vault asymmetry index.
Furthermore, to evaluate potential co-factors, the travel distance of participating families was recorded. The mean distance was 65.70 ± 42.83 km, ranging from 1.6 to 226 km.
Spearman-Correlation
The Spearman correlation coefficient was employed to quantify the strength and direction of associations because the data were not normally distributed, as shown by the Kolmogorov-Smirnov test. A statistically significant negative correlation (ρ = ‒0.094; p = .045) was observed between age at therapy initiation and final diagonal difference category. In addition, a more considerable reduction in diagonal difference was observed in children with earlier treatment (ρ = ‒0.229; p < .001). Instead, there was no correlation between age and duration of therapy (ρ = 0.033; p = .477). A strong correlation was observed between the initial CVAI measurement and the reduction of the diagonal difference (ρ = 0.811; p < .001). A significant negative correlation was identified between initial ear shift ≥1 cm and final ear shift category (ρ = ‒0.252; p < .001). Significant negative correlations were observed between age at therapy initiation and both initial CVAI (ρ = −0.190, p ≤ .001) and initial diagonal difference (ρ = −0.130, p = .005). Among infants with an initial ear shift ≥ 1 cm, negative correlations were observed between diagonal difference category (ρ = −0.109; p = .005) and final ear shift category (ρ = −0.252; p = .001). With regard to therapy duration, longer treatment periods were associated with higher final diagonal difference categories (ρ = −0.127; p = .001). The referral pathway demonstrated no statistically significant correlation with any outcome measure. These associations are illustrated in Figure 3.
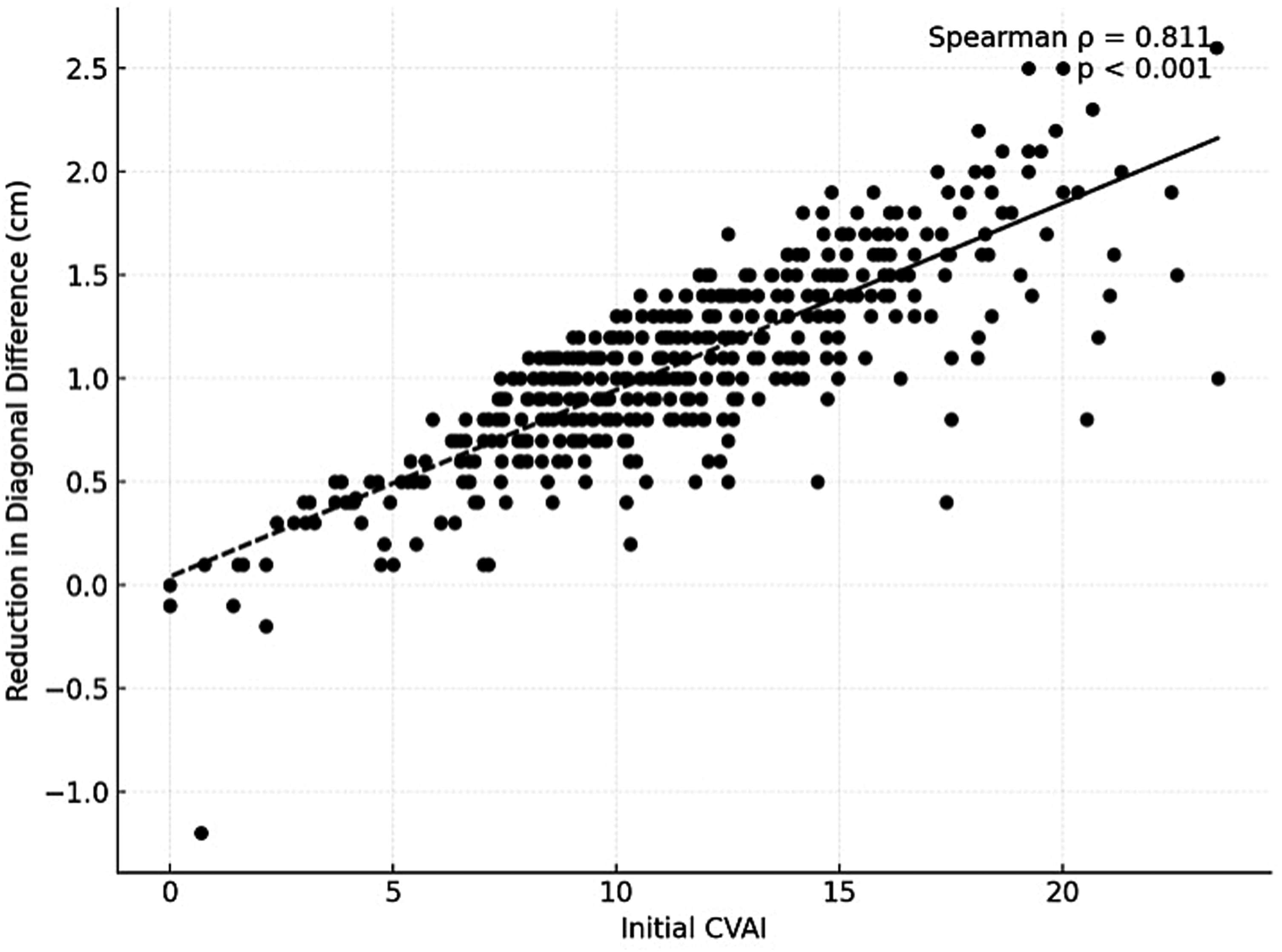
Spearman correlation: reduction in diagonal difference vs initial CVAI.
Logistic Regression Analysis
Logistic regression models were applied to investigate the relationship between dichotomous variables typically assessed in infants with positional cranial deformities and to quantify their association with predefined outcome categories regarding ear shift and diagonal difference. Overall model fit was evaluated using the Omnibus test, indicating acceptable model performance. The proportion of variance explained by the models was estimated using the Nagelkerke R2, yielding values of 0.184 for the ear shift outcome category and 0.163 for the diagonal difference outcome category, indicating that approximately 18.4% and 16.3% of the variability, respectively, were accounted for by the models. Model calibration was assessed using the Hosmer–Lemeshow test, which demonstrated adequate agreement between observed and predicted values. With regard to the ear shift outcome category (≤ 0.3 cm), an initial ear shift ≥ 1 cm was associated with lower odds of achieving this predefined outcome category (Exp(B) = 0.199; 95% CI: 0.087–0.451; p < .001). An increase in the initial diagonal difference was likewise associated with lower odds of meeting the predefined diagonal difference threshold, with an approximate 48% reduction in odds per additional centimeter (Exp(B) = 0.518; 95% CI: 0.334–0.802; p = .003). Conversely, a higher baseline cranial index was associated with higher odds of reaching the predefined outcome category (Exp(B) = 1.057; 95% CI: 1.030–1.084; p < .001). Increasing age at therapy initiation was associated with lower odds of achieving a final diagonal difference ≤ 0.5 cm, with each additional day at treatment initiation corresponding to a 0.6% decrease in odds (Exp(B) = 0.993; 95% CI: 0.989–0.997; p < .001). This analysis was based on the infant's chronological age at the initiation of helmet orthosis therapy.
Linear Regression Analysis
Linear regression analyses were conducted to examine associations between metrically scaled variables and changes in ear shift and diagonal difference. To assess overall model significance, an analysis of variance (ANOVA) was performed using an F-test. The coefficient of determination (R2) was .181 for the ear shift change model and .695 for the diagonal difference change model, indicating moderate and strong proportions of explained variance, respectively. Additionally, Durbin–Watson tests confirmed the independence of residuals, with values of 2.093 and 2.162, respectively, which are close to the ideal value of 2.0 and indicate the absence of autocorrelation. With respect to ear shift change, a statistically significant association (p < .001) was observed between the dependent variable and the included predictors, explaining 18.3% of the variance. No significant association was found between ear shift change and age at treatment initiation (p = .188) or travel distance (p = .256). The strongest association with limited ear shift change was observed for an initial ear shift ≥ 1 cm (β = 0.356; p < .001). In addition, the CVAI showed a modest positive association (β = 0.106; p = .013) with ear shift change, whereas the cranial index (CI) was negatively associated (β = −0.125; p = .005). For diagonal difference change, the model likewise demonstrated a statistically significant association (p = .001), with 69.5% of the variance explained. A strong positive association was observed for pre-treatment CVAI (β = 0.828; p < .001), indicating that greater baseline asymmetry was associated with larger observed changes. Conversely, longer therapy duration (β = −0.099; p < .001) and higher age at treatment initiation (β = −0.072; p = .007) were associated with smaller reductions in diagonal difference. No significant association was found between diagonal difference change and travel distance.
Discussion
The objective of this study was to examine clinical and morphometric parameters associated with outcomes of orthotic helmet therapy for positional cranial deformities. These findings contribute to the expanding body of literature on cranial remodeling and provide insights into factors associated with variability in morphometric outcomes. The distribution of cranial deformities in the cohort revealed that plagiocephaly was the most prevalent condition (n = 332), followed by cases of combined plagiocephaly and brachycephaly (n = 108), isolated brachycephaly (n = 14) and one instance of a combination including dolichocephaly. This is consistent with previous research showing that plagiocephaly is the most common cranial asymmetry in infants.5,13,38
Several parameters were found to be associated with outcome measures of helmet orthosis therapy. An initial ear shift ≥ 1 cm was associated with lower odds of achieving predefined ear shift outcome categories, corresponding to an approximate 80% reduction in odds. This factor was also highly associated with a reduced likelihood of achieving optimal outcomes, as evidenced by a weak negative correlation (ρ = ‒0.109, p = .005) based on the diagonal difference and a stronger negative correlation (ρ = ‒0.252; p = .001) based on the ear shift. This finding corroborates earlier studies highlighting the complexity of cranial base asymmetries and their impact on correction potential. 20 The study by Kuba et al. showed that helmet therapy has a positive influence on earshift correction especially in moderate asymmetries. This also shows that earshift is relevant in terms of diagnostics and therapy. 39
A larger initial diagonal difference was associated with lower odds of meeting predefined diagonal difference thresholds (Exp(B) = 0.518; 95% CI: 0.334–0.802, p = .003). These results support the notion that severe asymmetries are harder to correct, emphasizing once more the need for timely initiation of therapy to capitalize on the plasticity of the infant skull. 14 However, there was a greater potential for reducing the diagonal difference in the case of large asymmetries, confirming that helmet therapy is particularly effective in such cases. Chrenko et al. also observed that the best outcomes were achieved with severe and very severe non-synostotic deformities. 13
The timing of therapy initiation has been widely discussed in the literature. Several studies have reported more favorable morphometric outcomes when helmet therapy is initiated earlier in infancy. Argenta et al. observed improved outcomes when therapy began at four months of age in comparison to nine months of age. 12 Beuriat et al. and Cevik et al. recommend starting therapy before the age of 6 months,11,40 and Chrenko et al. recommend starting therapy at 4–7 months. 13 One possible explanation for this is given by Pollack et al. and is based on the fact that the cranial calvaria increases in size and thickness by approximately one year of age, the cranial base bones may exhibit structural changes due to prolonged positional forces, reducing the skull's responsiveness to repositioning strategies or orthotic intervention. 41 In the present study, older age at therapy initiation was associated with smaller reductions in diagonal difference (ρ = −0.229; p ≤ .001) and with lower odds of achieving predefined outcome categories (Exp(B) = 0.993; 95% CI: 0.989–0.997; p ≤ .001). Together, these findings indicate an association between treatment timing and morphometric change and are consistent with previously published observations describing earlier initiation of helmet therapy, often between 4 and 6 months of age.
In this context, early recognition of cranial asymmetries has been discussed in the literature as a relevant factor for clinical decision-making and referral pathways. Parental counseling and clear communication regarding the expected course of helmet therapy have likewise been described as potentially relevant contextual factors, particularly with respect to adherence and expectation management. 42 In cases with later therapy initiation or more pronounced baseline deformities, previously published studies have emphasized the importance of realistic expectation setting, as the extent of morphometric change may be limited. 43
Further, significant negative correlations were observed between age at therapy initiation and both baseline CVAI (ρ = −0.190; p ≤ .001) and initial diagonal difference (ρ = −0.130; p = .005). These findings suggest that younger infants in this cohort presented with more pronounced cranial deformities, which may reflect referral patterns or selection effects. Such factors may confound age-related comparisons and should be considered when interpreting associations between age and outcome measures.
No correlation was found between age and duration of the therapy (ρ = 0.033; p = 0477). However, higher final diagonal difference categories were observed in association with longer treatment duration (ρ = −0.127; p = .001). This observation likely reflects increased case complexity or reduced compliance in prolonged treatment courses and does not imply a direct effect of treatment duration itself. Similar findings were reported by Argenta et al., who observed decreasing wearing times over longer treatment periods due to increasing head and body control. 1 The mean treatment duration in the present cohort was 162.9 days. While no optimal treatment duration can be inferred from the present data, shorter treatment courses were more frequently observed in association with earlier therapy initiation.
A strong positive correlation was observed between baseline CVAI and reduction in diagonal difference (ρ = 0.811; p < .001), indicating that greater initial asymmetry was associated with larger absolute morphometric changes. At the same time, a larger initial diagonal difference was associated with lower odds of meeting predefined outcome thresholds (Exp(B) = 0.518; 95% CI: 0.334–0.802; p = .003). Together, these findings suggest that baseline severity is an important factor in shaping observed morphometric changes, a pattern that aligns with previous reports by Kluba et al. 27 who reported that baseline severity influences the extent of achievable correction during helmet therapy.
Conversely, an increased cranial index at baseline was positively associated with predefined outcome categories (Exp(B) = 1.057; 95% CI: 1.030–1.084; p ≤ .001). This association suggests that baseline cranial shape characteristics may contribute to variability in observed outcomes, potentially reflecting differences in growth patterns or remodeling capacity.
Prior studies, including those by Loveday et al. 20 have demonstrated that substantial reductions in CVAI may be observed during helmet therapy, particularly when treatment is initiated earlier. The present findings are consistent with these observations and support the relevance of CVAI as a descriptive parameter for monitoring morphometric change, especially in cases with pronounced baseline asymmetry. In our cohort, higher pretherapeutic CVAI values were associated with larger absolute reductions in diagonal difference, indicating that baseline severity contributes to variability in observed morphometric changes.
Similarly, the association between initial diagonal difference and subsequent reduction highlights the relevance of baseline severity for analytical stratification. Greater initial diagonal differences were associated with larger absolute changes but with lower odds of meeting predefined outcome thresholds, reflecting the complex relationship between baseline severity and categorical outcome definitions. These observations underscore the importance of considering both continuous and categorical outcome measures when interpreting treatment-associated changes.
Using a predefined threshold of final diagonal difference ≤ 0.5 cm, 353 infants (77.6%) met this analytical criterion, while 305 infants (67.0%) met the corresponding ear shift threshold. Consequently, 22.4–33.0% of infants did not meet these predefined categories. These thresholds were applied for statistical classification purposes and may not fully capture the clinical relevance of individual morphometric changes, as infants with improvements marginally above a cutoff may still experience meaningful aesthetic or functional changes. Accordingly, outcome interpretation should consider these limitations.
Taken together, these findings allow rejection of the null hypothesis and support the alternative hypothesis that treatment timing and baseline severity are associated with differences in morphometric outcomes in infants undergoing helmet orthosis therapy for positional cranial deformities.
Study limitations include the retrospective design and reliance on manual measurements. Three-dimensional photography was introduced in 2016 and evaluated in a subset of 111 infants, demonstrating good agreement with manual measurements. Study limitations include its retrospective design and reliance on manual measurements. Due to the long period of data collection, 3D photography was only introduced in 2016 and examined at our clinic in the course of the study from Nieberle et al. In this context 111 infants underwent additional comparative 3D digital photography utilizing a five-lens setup combined with a multi-flash lighting array (Vectra M5 3D Imaging System, Canfield Scientific, Parsippany, NJ, USA) (see Figure 1) to evaluate the accuracy of manual and digital measurement methods. The 3D measurement method was shown to represent a dependable alternative to manual measurements and may offer advantages for future studies focusing on cranial morphology.18 2.5,44,45 While digital methods may offer advantages in contemporary practice, the consistency of measurement techniques over time was prioritized in this study. The focus on a single orthosis manufacturer may limit generalizability, and additional factors such as socioeconomic status, insurance coverage, comorbidities, and ethnicity were not systematically captured and may influence observed outcomes. 36 Additionally, ethnicity was not systematically documented in the retrospective clinical records and may represent an unmeasured factor influencing cranial morphology. The absence of a control group represents another limitation, as it restricts the ability to differentiate treatment-associated changes from natural cranial growth and spontaneous improvement. This aspect should be considered in the design of future prospective studies.
To explore potential additional co-factors, the travel distance of participating families was analyzed as a surrogate parameter for treatment burden and access to care. As some families traveled considerable distances to attend follow-up visits, this variable was examined in relation to morphometric outcomes. No significant association was observed between travel distance and outcome measures. However, it should be noted that data on whether travel distance influenced initial presentation to the outpatient clinic were not available. Consequently, a potential pre-selection of the study population based on access to care or willingness to travel cannot be excluded. Furthermore, it remains a challenge to isolate the direct contribution of helmet therapy from spontaneous cranial remodeling, as a proportion of positional cranial deformities may improve with growth alone. This limitation is inherent to observational study designs and is further compounded by the influence of multiple interacting factors. 17 In the present analysis, only approximately 20% of the assessed variables were associated with outcome measures, underscoring the multifactorial nature of cranial shape development and treatment response.
Future research should therefore aim to incorporate additional clinical, socioeconomic, and psychological variables, as well as prospective control groups, to better characterize factors contributing to outcome variability and to further explore determinants of therapy adherence.
Conclusion
Early initiation of helmet therapy promises more pronounced morphometric changes in infants with positional cranial deformities, before compensatory growth may limit correction potential. Baseline severity parameters, including diagonal difference, CVAI, and ear shift, indicate success limitations and provide realistic clinical expectations for both the physician and the patients’ caregivers. Objective cranial measurements, including manual assessment methods, remain valuable for standardized documentation and longitudinal monitoring of cranial morphology during orthotic treatment.
Footnotes
Ethics Approval
All of the following procedures were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. The retrospective analysis was approved by the Ethics Committee of the University Regensburg (Approval No. 22-3217-104, date of approval 9 January 2023).
Informed Consent
Based on retrospective analysis, a fully anonymized set of clinical data was used, and in accordance with the Ethics Committee's decision, signing informed consent was not required. Informed consent for the usage of the patient's face in this work was obtained from the child's parents.
Authors’ Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Juergen Taxis, Felix Nieberle and Katja Himmelstoß. The first draft of the manuscript was written by Katja Himmelstoß, Felix Nieberle and Jürgen Taxis. All authors commented on previous versions of the manuscript or provided passages for the first manuscript draft. All authors read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available due to patient confidentiality but are available from the corresponding author on reasonable request.