Abstract
Objective
Palatal fistulas after palatoplasty could pose difficulties for both patients and surgeons. Numerous surgical approaches are available to treat palatal fistulas. In this manuscript, we investigate surgical treatment options for palatal fistula repair looking at the different anatomical locations, and we create a summary of surgical approaches to facilitate the decision-making process for palatal fistulae repair.
Design
In this cross-sectional survey, nine anonymized patient cases with palatal fistulae that differed in severity and anatomical location were presented to participants from the International Cleft Master Course in Amsterdam about “Palatal Fistulas”. Participants were invited to participate in this survey. A total of 141 participants reported their preferred surgical treatment options for fistula repair at different anatomical locations.
Results
We created different options for fistula treatment, catalogued by fistula location. This overview gives the surgeon possible approaches for each location. If the soft palate is involved, this overview underscores the importance of including velopharyngeal insufficiency management into the fistula repair. For hard palate involvement, our overview lists techniques available for nasal lining repair and for oral lining repair in each region.
Conclusions
We provide a comprehensive overview of potential surgical approaches to repair palatal fistulae. This inventory of techniques is grouped per location to support surgeons in their decision-making process when confronted with a palatal fistula.
Introduction
Cleft lip and palate is a common congenital malformation, with an incidence of approximately 1 in 700 live births. 1 After surgical repair, the development of palatal fistulae shows a wide range of incidence rates, varying from 4.9% to 31.3%.2,3 Additionally, surgical treatment of fistulas is complicated by a high recurrence rate of up to 33%. 4 Factors contributing to fistula formation are multifactorial and often difficult to determine but may include the width of the cleft, the ratio of cleft width to available surrounding palatal tissues for reconstruction and the surgeon's level of skills, scarring from previous surgery, surgical factors (eg, excessive tension on the suture line, injudicious use of electrocautery on the medial edges, too tight knots) and patient factors (finger sucking postoperatively, nutritional deficiency postoperatively, compliance).5,6
Patients suffering from palatal fistulae can either be asymptomatic or they experience speech problems, nasal regurgitation of fluids, and difficulty in maintaining oral hygiene.7,8 Fistulae can be classified based on anatomical description, using the Pittsburgh classification or the Pakistan Comprehensive fistula classification. 9 The most prevalent anatomical location for fistulae is at the junction of the hard and soft palate, followed by fistulae located in the hard palate (2) and fistulae in the soft palate (3). 2 Various surgical approaches are described which enable the repair of palatal fistulae, e.g., oral mucoperiosteal flap, buccal flap, facial artery musculomucosal (FAMM) flap, Furlow palatoplasty. 10 The debate continues on whether oronasal fistulae occur more frequently after a two-stage palate repair compared to the single-stage repair.11,12 This comparison remains challenging due to the substantial impact of confounders such as surgeon experience, cleft phenotype and patient characteristics. To determine the most appropriate surgical approach, it is essential to consider multiple factors, including size, shape and location of the fistula, as well as whether it is a primary or a recurrent fistula. 13 It is important to note that there is a lack of consensus and limited literature that focuses on managing fistulas following primary surgery.14,15 While categorization and the relationship of fistulas associated with primary repair requires further study, the focus of this study is on how to address a fistula once it is present.
The aim of this cross-sectional study is to summarize the clinical surgical strategies of cleft surgeons from around the world on palatal fistula repair per anatomical location. A secondary objective is to investigate the influence of a digital webinar on the preferred choice for closing palatal fistulas.
Methods
In this cross-sectional survey study, two questionnaires were sent to 242 participants from 74 different countries who attended the “Palatal Fistulas: Prevention, Classification & Treatment” Master Course hosted in Amsterdam, the Netherlands in June 2021. The complete scientific program can be found in Supplemental Data Content 1. All participants attended the webinar digitally, it was not possible to attend this master course physically. Most of the participants were either plastic, maxillofacial, ENT or pediatric surgeons. Speech language pathologists and residents in training for the previously mentioned specialties were also included. Speech language pathologists, (PhD) students and residents not in training were excluded from this questionnaire since they do not perform palatal surgery. These questionnaires were sent before and after the webinar. Nine patient cases were presented with different oronasal fistulae, each case was accompanied with a digital image, some of which were adapted from the Surgical Atlas of Cleft Palate and Palatal Fistulae by Prof. Ghulam Qadir Fayyaz (Figure 1, Supplemental Data Content 2). 16 The nine patient cases were selected from a large database by ten of the course organizers. Criteria for selection were the following: both females and males included, all three anatomical locations covered (ie, soft palate, hard palate and junction soft/hard palate), several degrees (ie, 1–5 mm fistula, 5–20 mm fistula and >20 mm fistula). These patient cases were presented in a random fashion in terms of severity and anatomical location. For readability, we rearranged the order and brought together patient cases categorized in three anatomical locations (ie, soft palate, hard palate and junction soft/hard palate). The questions in the survey were designed to ascertain the participants’ preferred surgical approach in each individual case. If the nasal lining closure negate the possibility of doing a marked out oral closure, participants were not able to fill in an answer for oral closure with that patient case. Participants received an invitation to complete the survey fourteen days prior to the webinar. A reminder was sent seven days before the event. The survey was closed at the start of the webinar.
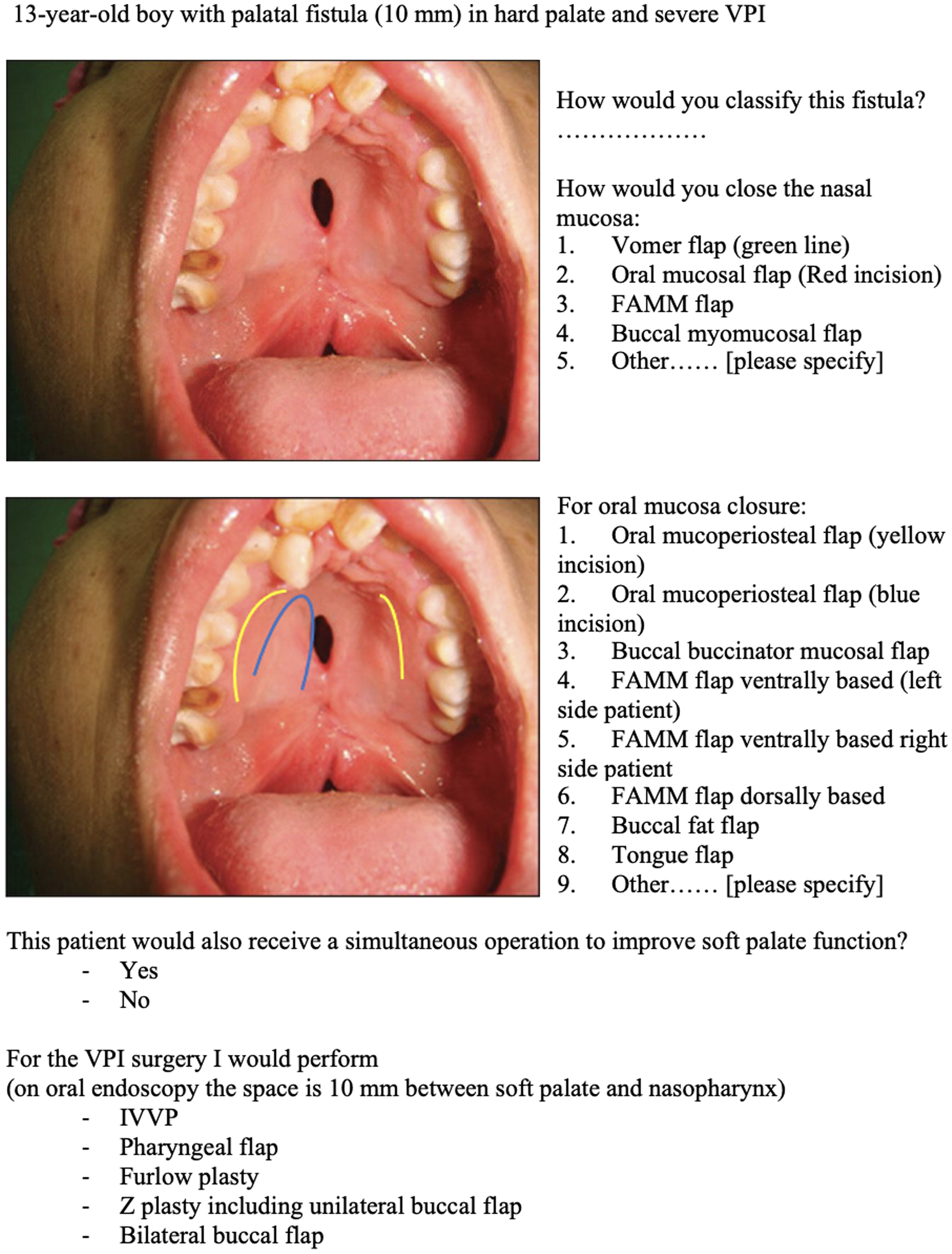
An example of a patient case from the digital survey.
The second questionnaire, which mirrored the cases and images from the first questionnaire, was sent immediately after the conclusion of the webinar. A reminder to complete the webinar was sent seven and fourteen days after the event respectively. Only fully completed questionnaires were included in the analysis. The questionnaires were created and distributed through Castor Electronic Data Capturing (EDC). The data were subsequently analyzed using Statistical Program for Social Sciences (SPSS, version 29; SPSS Inc., Chicago, IL, USA). The Pearson Chi-Square test was used to compare the distribution of answers between the two groups. The Fisher-Freeman-Halton Exact Test was used to compare the distribution of answers between groups. A p value of <0.05 was considered a significant result. Ethical approval for this study was waived by the local Ethical Committee (W22_158 #22.205) and participants provided consent for the anonymous processing of their data for scientific purposes. Reporting adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist guidelines.
Results
A total of 141 participants, representing a response rate of 58.3%, completed the questionnaires before the webinar. After the webinar, 109 participants (response rate of 45.0%) completed the survey. In the pre-webinar group, the largest group of participants consisted of plastic surgeons (n = 73; 51.8%), followed by maxillofacial surgeons (n = 47; 33.3%), Ear, Nose, and Throat (ENT) surgeons (n = 7; 5.0%), pediatric surgeons (n = 5; 3.5%). Nine participants were active in other disciplines (6.4%). Eleven participants (7.8%) did not have any experience with primary cleft palate surgeries, 18 participants (12.8%) did less than 10 primary surgeries annually. An additional thirty-one (22.0%) did between ten and twenty-five primary cleft palates per year, thirty-six (25.5%) did between twenty-five and fifty primary cleft palate surgeries per year and lastly forty-five surgeons (31.9%) treated over fifty patients with primary cleft palates per year. Respondents were categorized into two groups. The first group consisted of respondents who performed less than 25 primary palatoplasties per year (infrequent palatoplasty group; n = 49; 37.7%). The second group consisted of respondents who performed 25 or more primary palatoplasties per year (frequent palatoplasty group; n = 81; 62.3%). In general, most participants advocated a two-layer closure (n = 124; 88.0%). Some even suggested a third layer (n = 7; 5.0%). Acellular dermal matrices (ADMs) were only used by 24 respondents (17.0%). Figures 2 and 3 present a comprehensive summary of all the respondents’ mentioned surgical approaches for the presented cases.
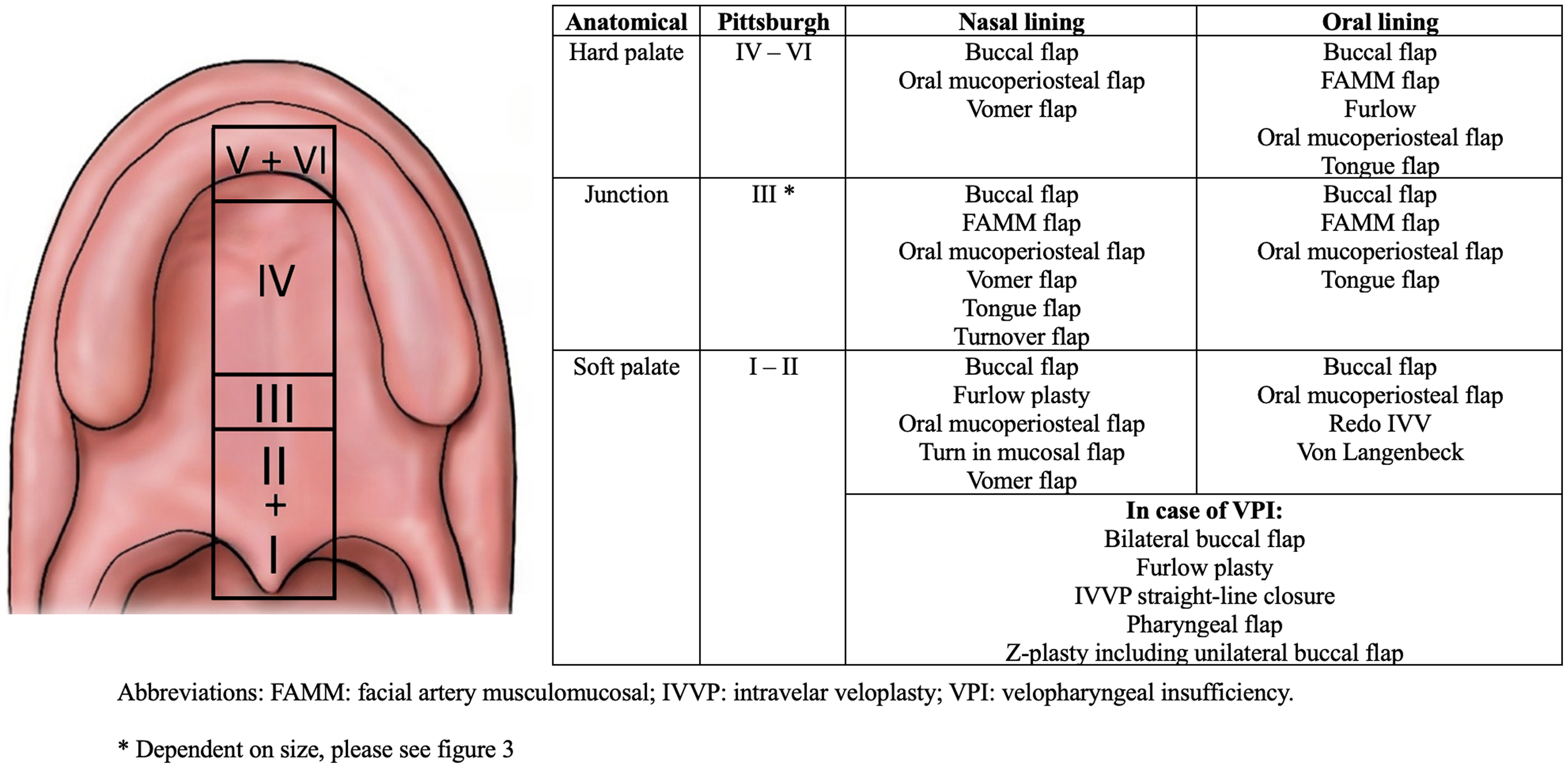
An overview of all surgical options per anatomical region.
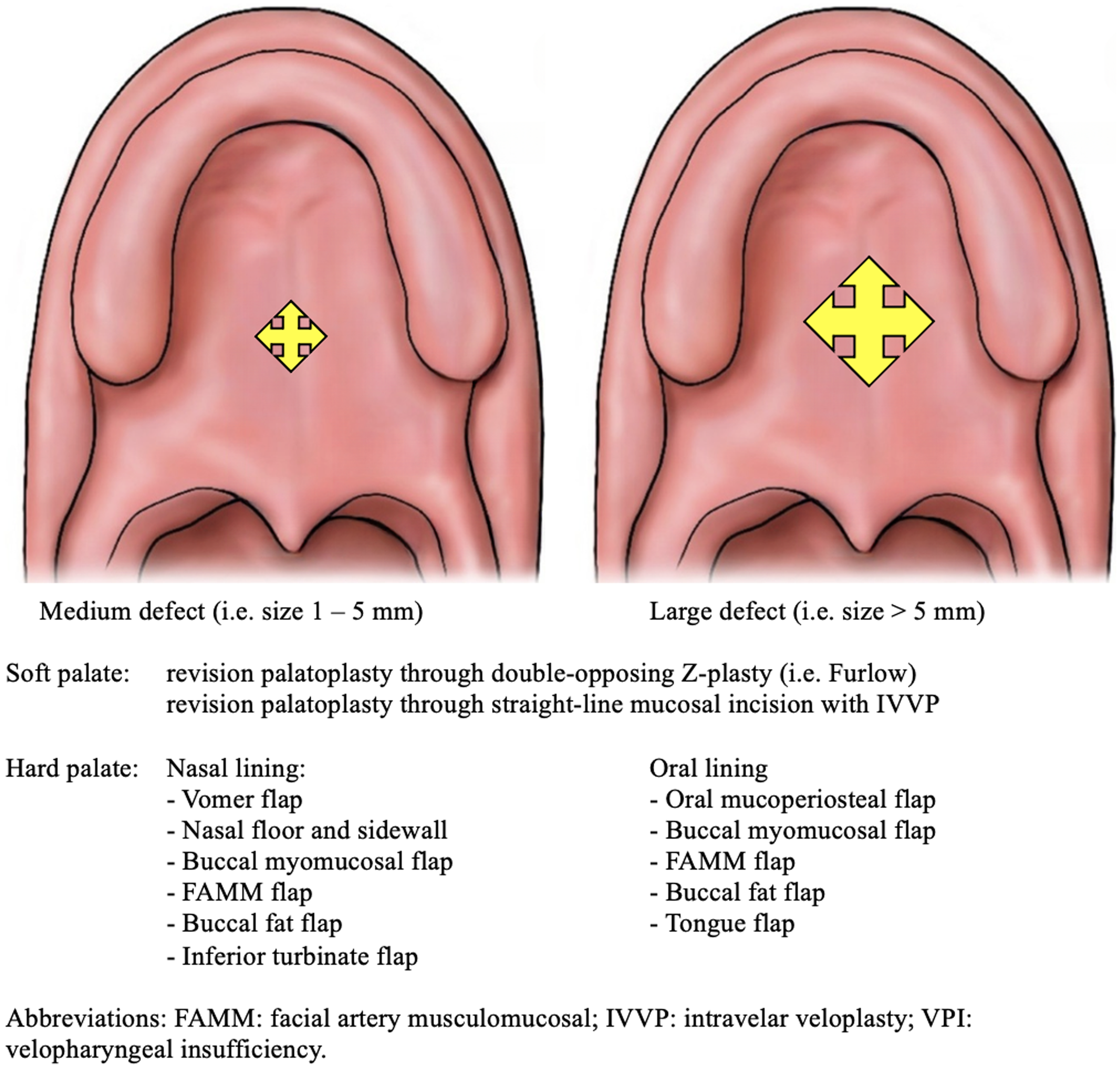
Surgical options per anatomical region dependent of size of the fistula.
Soft Palate
Case 1 presented a patient suffering from a fistula located at the soft palate and junction hard/soft palate three weeks post-surgery. The majority of respondents indicated, prior to the webinar, to wait 6 months before deciding on any surgical intervention (n = 110; 84.6%). After the course, this treatment strategy remained the most popular (n = 96; 84.2%), while a minority would prefer to close the fistula after 4 weeks (n = 16; 14.0%) and only 2 respondents (1.8%) would perform immediate surgery.
In case 2, a patient was shown with a combination of a large hard palate and small soft palate fistula requiring nasal mucosa closure. Before the webinar, the most frequently suggested option for treatment was performing a mucoperiosteal flap (n = 59; 45.4%), which was comparable after the congress (n = 44; 38.6%). Interestingly, for the closure of the oral mucosal layer, a shift was observed from performing oral mucoperiosteal flaps to the Furlow palatoplasty (n = 50; 38.5% and n = 49; 43.0% respectively).
Hard Palate
In case 3 involving a patient with a hard palate fistula, the preferred choice of treatment, prior to the meeting, was to perform orthodontic alignment before surgical closure (n = 70; 53.8%), which relatively increased after the webinar (n = 71; 62.3%). Concerning the nasal closure, prior to webinar, the overall preferred choice was the Vomer flap (green incision) (n = 51; 39.2%) whereas the oral mucosal flap (yellow incision) was more preferred post-webinar (n = 44; 38.6%). The experienced palatoplasty group preferred the Vomer flap (green incision) (n = 36; 44.4%) vs the oral mucosal flap (yellow incision) (n = 22; 44.9%) in the less experienced group. This significant difference also existed in the oral closure strategy. The frequent palatoplasty group most often opted for the oral mucoperiosteal flap (yellow incision, A) (n = 33; 40.7%) and the infrequent group most often for the oral mucoperiosteal flap (blue incision, A) (n = 12; 24.5%).
Case 4 presented a patient suffering from a hard palatal fistula with the need for nasal mucosa closure. Most participants opted for a vomer flap as treatment plan (n = 53; 40.8%). However, after the meeting, the majority of the respondents chose oral mucosal flaps (n = 48; 42.1%). Between the groups of frequent palatoplasty surgeons and infrequent palatoplasty surgeons, a significant difference (p = 0.045) was found where surgeons who perform palatoplasty more frequently preferred a Vomer flap (green line) (n = 35; 43.2%) and participants from the infrequent palatoplasty group, the oral mucosal flap was chosen (blue incision) (n = 24; 49.0%). For closure of the oral mucosal layer in this case, the oral mucoperiosteal flap (red incision [A]) remained the suggested treatment option of choice (n = 36; 27.7%) prior to the meeting versus after the webinar (n = 37; 32.5%) [Supplemental Data Content 2]).
The patient in case 5 was suffering from a hard palatal fistula. For nasal mucosal layer closure, most attendees chose to perform mucoperiosteal flaps (yellow incision) as the treatment strategy before attending the meeting (n = 89; 68.5%) as well as after the congress (n = 83; 72.8%). In contrast, the oral mucoperiosteal flaps (red incision [A]) were selected most for oral mucosal closure after the webinar (n = 75; 65.8%).
Case 6 involved a patient having a fistula located at the hard palate with severe velopharyngeal insufficiency (VPI). The most popular treatment option for nasal mucosal layer closure amongst the respondents pre-webinar was the vomer flap (n = 62; 47.7%) as well as post-webinar (n = 56; 49.1%). The oral mucoperiosteal flap (yellow incision) was the preferred method for closing the oral mucosal layer after the meeting (n = 66; 57.9%).
Junction Hard/Soft Palate
In case 7, a patient with a large fistula located at the junction of the anterior and posterior halves of the hard palate and extends to the junction of the hard and soft palate was presented. Participants indicated that they preferred to close the nasal mucosa with oral mucoperiosteal flap (n = 54; 41.5% before the meeting), as did the participants after the meeting (n = 49; 43.0%). Interestingly, for closing the oral mucosal layer, the buccal buccinator mucosal flap was preferred after the webinar (n = 43; 37.7%).
In case 8, in which a six-year-old girl with a large palatal fistula (18 mm in length) in the ventral half of the hard palate was show. The oral mucoperiosteal flap (yellow incision) was initially chosen for nasal mucosal closure (n = 81; 62.3%), while after the webinar the selection of this treatment option was more or less the same (n = 75; 65.8%). In addition, for closing the oral mucosal layer, the preference was an oral mucoperiosteal flap (green incision) both pre- and post-webinar (n = 42; 32.3% vs n = 34; 29.8%). The frequent palatoplasty group most often chose either the oral mucoperiosteal flap (green incision) or the bilateral mucoperiosteal flap (for both n = 24; 29.6%) whereas the infrequent group most often opted for the oral mucoperiosteal flap (green incision) (n = 18; 36.7%).
In case 9, a twelve-year-old girl was presented with a large cleft palate (covering almost the complete hard and soft palate). The use of a free flap was the preferred choice for oral closure prior to the webinar (n = 38; 29.2%). After the webinar, this surgical approach remained the most chosen option (n = 38; 33.3%). For nasal closure, the oral mucoperiosteal flap (red incision) was the most popular surgical approach both before (n = 35; 26.9%) and after the webinar (n = 48; 42.1%). A total of 25.4% (n = 33) would perform simultaneous surgery to improve soft palate function if VPI was present.
Figures 2 and 3 show an overview of all surgical options per anatomical region.
Discussion
Although prevention of palatal fistulae development is an important goal in primary cleft palate surgery, palatal fistulae are well known complications of palate repair. This study introduces an inventory of techniques, subsequently grouped by location to support surgeons in their decision-making process when confronted with a palatal fistula.
Palatal fistulae can be a great burden to patients, due to nasal regurgitation and speech impairment.8,17 Correlations between VPI and quality of life have been demonstrated multiple times.18,19 Adequate planning and proper treatment of fistulae is very important since surgical treatment of fistulae may have a high recurrence rate of up to 33%. 4 Adequate intervention is paramount for both patients and cleft surgeons. There appears to be no gold standard regarding regional approaches to the surgical treatment of fistulae. 20 In this large cross-sectional survey, surgeons from all over the world proposed different approaches for each patient case and high levels of consensus were reached at the end of the meeting. Carefully identification of the problem is of paramount importance, together with clear knowledge and expertise of all possible surgical approaches as per anatomical location in the form of a schematic toolbox that makes it easier to finalize and choose the best suitable technique. Results of this study could serve as a guide for colleagues that are confronted with difficult fistula cases in the future. In the literature, only a few studies have been conducted on palatal fistulae in which surgeons were asked their opinion on surgical options, often with limited participants involved.13,21–24 Although numerous surgical techniques are available, none of them have been proved to be superior. 20 In the absence of international guidelines, surgeons are most likely to choose surgical techniques based on anatomical location, width and size of the fistula, age of the patient and surgical experience with specific techniques.13,14 In this current study, most respondents indicated that they would prefer local tissue advancement in order to close both nasal and oral mucosal layers. This is in concordance with previous research, in which surgeons suggested mucosal turn-over flaps.13,14 Although Rothermel et al. only included 17 craniofacial specialists in their cross-sectional survey, results remain consistent in our study with 141 participants (agreement up to 67%), underlining the substantial preference for mucoperiosteal flaps. 25
It is important to emphasize that in concordance with Rothermel et al. this study emphasizes the assessment of the function of the palate that is very important when fistulae are treated. 13 Moreover, in soft palate involvement, relocation of the levator sling is an important part of the procedure in addition to closure of the fistula. 26 As demonstrated in the overview (Figures 2 and 3), several options including the Furlow palatoplasty, bilateral buccal flap or the unilateral buccal flap can facilitate levator muscle reconstruction.22–29
This study indicates that most respondents (88.0%; n = 124) advocate a two-layer closure. However, some (5.0%; n = 7) even suggest a third layer. ADMs can be introduced in palatal fistula repair, offering a scaffold for tensionless closure and tissue regeneration. 30 These matrices could enhance neovascularation and epithelialization, reducing the risk of fistula recurrence.31,32 There is still no consensus on superiority of closure with ADM compared to closure without use of ADM. In the current study, nearly 80% of the respondents indicated not to use ADM, while 17% indicated that they would sometimes introduce dermal matrices in fistula repair. This is different with the survey study carried out by Rothermel et al. reporting 64% of participant occasionally using ADMs and 36% never using ADMs. 13 This noticeable difference could partly be explained by differences in sample size (eighteen versus 141), but also because the Rothermel et al. study was performed under several surgeons working in the US (where ADMs are freely available) and in Canada, were resources are more limited (and ADMs are used more judiciously) while the current study has representatives from all over the world. Simpson et al. performed a similar survey amongst members of the American Cleft Palate Craniofacial Association (ACPA), reporting 22% of which used ADMs on all patients, 45% introduced ADMs only if excessive tension was noted during surgery and 33% of participants which responded to never used ADMs. 23 Since participants in the current study originate from 74 countries, versus participants from the ACPA all working in the United States of America a national tendency to introduce ADMs could account for this difference. According to earlier research, up to 11–12% of recurrence rates were found when ADMs were introduced between nasal and oral mucosa.31,32 Higher fistula rates (17%) were found when mucosal defects were covered with ADM. 33 In addition to the cost of ADM, another reason to avoid ADM is that it is a non-vascularized graft that could also potentially act as a foreign body nidus for infection in an environment with a high bacterial load. 34
Several limitations of the current study should be addressed. Responses of our participants were based purely on digital pictures. In a normal outpatient consultation clinic, doctors are able to both look and palpate the palatal fistula, which might change surgical treatment approaches and therefore could skew results of this study. Moreover, surgeons may also change what they actually do on the day of surgery when they can better examine the patient under general anesthetic. Another major limitation in this study is recency bias (ie, a surgeon is after the webinar more likely to report a surgical method with which they have limited experience if they just heard an expert surgeon present their positive experiences with that technique). Additionally, the country of origin of all our respondents was not specifically asked in the survey for privacy reasons. However, we did know the country of origin for all participants of the webinar (242 participants from 74 unique countries) resulting in a large representative group of cleft specialists globally. Moreover, it is important to note that high volume cleft surgeons (question four in our survey [How many primary cleft palate surgeries do you perform per year? “None” versus “<10” versus “11–25” versus “26–50” versus “>50/year”) may not correlate with high volume cleft fistula surgeons. A substantial strength of this study is the large size of surgeons included in this cross-sectional survey. Moreover, the group of specialists is expected to be a proper representation of all specialists globally due to a high response rate, large sample size, multiple countries involved and different levels of expertise amongst participants.
Conclusion
In this study, an inventory is presented with frequently mentioned treatment options for surgical repair of palatal fistulae at different anatomical locations. Most craniofacial cleft specialists would close hard palate fistulae with oral mucoperiosteal flaps (both oral and nasal mucosa), while Furlow palatoplasty was mostly mentioned in soft palate fistulae. The majority of participants did not use ADMs or third layers in fistula repair.
Supplemental Material
sj-docx-1-cpc-10.1177_10556656241286864 - Supplemental material for Different Surgical Approaches to the Treatment of Cleft Palate Fistulae as Perceived by Cleft Surgeons
Supplemental material, sj-docx-1-cpc-10.1177_10556656241286864 for Different Surgical Approaches to the Treatment of Cleft Palate Fistulae as Perceived by Cleft Surgeons by Johannes A. Smit, Ruben P. Houkes, Nadia Lachkar, J. Peter W. Don Griot, Chantal M.A.M. van der Horst, Raymond W. Tse, Ghulam Qadir Fayyaz, Saleigh Adams and Corstiaan C. Breugem in The Cleft Palate Craniofacial Journal
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
