Abstract
Objective
To characterize the onset and prevalence of conductive hearing loss (CHL) in pediatric patients with cleft palate (CP) prior to palatoplasty with an enhanced audiologic protocol.
Design
Retrospective cohort study
Setting
Multidisciplinary cleft and craniofacial clinic at a tertiary care center.
Patients
Patients with CP who received audiologic workup pre-operatively. Patients with bilateral permanent hearing loss, expiration prior to palatoplasty, or no pre-operative data were excluded.
Interventions
Patients with CP born February 2019 to November 2019 who passed newborn hearing screening (NBHS) received audiologic testing at 9 months of age (standard protocol). Patients born December 2019 to September 2020 underwent testing prior to 9 months of age (enhanced protocol).
Main Outcome Measures:
Age of identification of CHL in patients after implementation of the enhanced audiologic protocol.
Results
The number of patients who passed their NBHS in the standard protocol (n = 14, 54%) and the enhanced protocol (n = 25, 66%) did not differ. Infants who passed their NBHS, but demonstrated hearing loss on subsequent audiologic testing did not differ between enhanced (n = 25, 66%) and standard cohort (n = 14, 54%). Of patients who passed NBHS in the enhanced protocol, 48% (n = 12) had CHL identified by 3 months, and 20% (n = 5) by 6 months of age. With the enhanced protocol, patients who did not undergo additional testing post NBHS significantly dropped from 44.9% (n = 22) to 4.2% (n = 2) (P < .0001).
Conclusion
Even with passed NBHS, CHL is still present for infants with CP pre-operatively. Earlier and more frequent testing for this population is recommended.
Introduction
The relationship between cleft palate (CP) and conductive hearing loss (CHL) has been well-established.1,2 This hearing loss is primarily attributed to chronic otitis media with effusion that can develop into recurrent otitis media in early childhood. The association between CP and chronic otitis media with effusion and recurrent otitis media is attributed to abnormal insertion of the tensor veli palatini and levator veli palatini laterally due to failure of the palate to fuse during fetal development, resulting in Eustachian tube dysfunction.3–6 While only 18% of infants with cleft palate fail their newborn hearing screening (NBHS), 7 69% of school-age children with CP demonstrate some degree of hearing loss. 6 Greater than 90% of patients with cleft lip and palate have been shown to have middle ear effusion at the time of lip repair around 3-4 months. 8 Myringotomy with tympanostomy tube insertion is often performed at the time of palate repair, around 9 to 12 months of age, 9 although some have advocated for earlier tube placement. 10 Palate repair aids in correcting the tensor veli palatini and levator veli palatini muscle structure to reduce the incidence and duration of middle ear effusions and therefore timely palate repair may lead to improved long-term middle ear function.10–13
Early detection and treatment of hearing loss is important for speech and language outcomes. Children with CP have been shown to have significant delays in vocabulary development compared to their peers by 24 months of age.14,15 CHL in this population has been shown to affect speech development, contributing to cleft speech characteristics, with recommended audiology monitoring from birth in order to provide timely and appropriate interventions. 16
The Joint Committee on Infant Hearing's (JCIH) 2019 Position Statement 17 notes the importance of early hearing loss detection to long-term quality of life and promotes a universal 1-2-3 screening. NBHS should be performed for all patients by 1 month of age. For those who refer on NBHS, diagnostic evaluation should be performed by 2 months of age, and appropriate interventions established by 3 months of age. Patients who passed the NBHS with a diagnosis of CP, are recommended for re-assessment at 9 months of age. 17 Therefore, prevalence of hearing loss in children who pass their NBHS prior to this timeframe is unknown. Although CP patients are at a high risk for CHL at an early age, and there is agreement that early detection is critical, there is currently no consensus protocol for timing of hearing monitoring during the first 9 months of life for these patients.
The purpose of this study was to evaluate an enhanced audiologic protocol designed for patients with CP in the first year of life. The rate of detection of CHL was compared before and after implementation of the enhanced protocol to determine if enhanced testing would be effective in detecting CHL prior to planned CP repair. Our hypothesis was that due to the amount of CHL associated with CP, hearing loss would be identified earlier than the current 9-month recommendation period, despite a passed NBHS result. Secondary endpoints included the detection of CHL in patients who passed NBHS, age of onset of CHL, and severity of CHL.
Methods
This retrospective cohort study was approved by the Vanderbilt University Institutional Review Board (#190840). No consent from patients was required. Patients with CP from February 2019 to June 2021 who were seen by the multidisciplinary Cleft and Craniofacial Team prior to CP repair were included. Patients with bilateral sensorineural hearing loss, microtia, or atresia, expiration prior to 9 months of age, or who failed to complete the audiologic workup were excluded.
Retrospective chart review was performed. Demographic data, severity of the CP (complete, incomplete, or submucous), presence of genetic abnormalities (eg, Stickler Syndrome), NBHS results, pre-operative hearing evaluations, and date of palate repair were collected. Traditional risk factors for hearing loss including prematurity and extended NICU stay secondary to various medical complexities were noted. Hearing data was collected on all patients until completion of palatoplasty or 9-12 months of life.
Patients with CP born from February 2019 to November 2019 were evaluated according to the standard protocol. It was recommended that these patients receive audiologic testing at 9 months if they passed their NBHS, as recommended by the JCIH. 17 Additional audiologic testing occurred only in the cases of failed NBHS, by physician order (typically ordered for pre-operative hearing data prior to tympanostomy tube placement), or parental concern. An enhanced audiologic protocol (Figure 1) was implemented in January of 2020. Patients born December 2019 to September 2020, regardless of the result of NBHS, received serial hearing evaluations beginning at 3 months of age, with monitoring until palate repair.
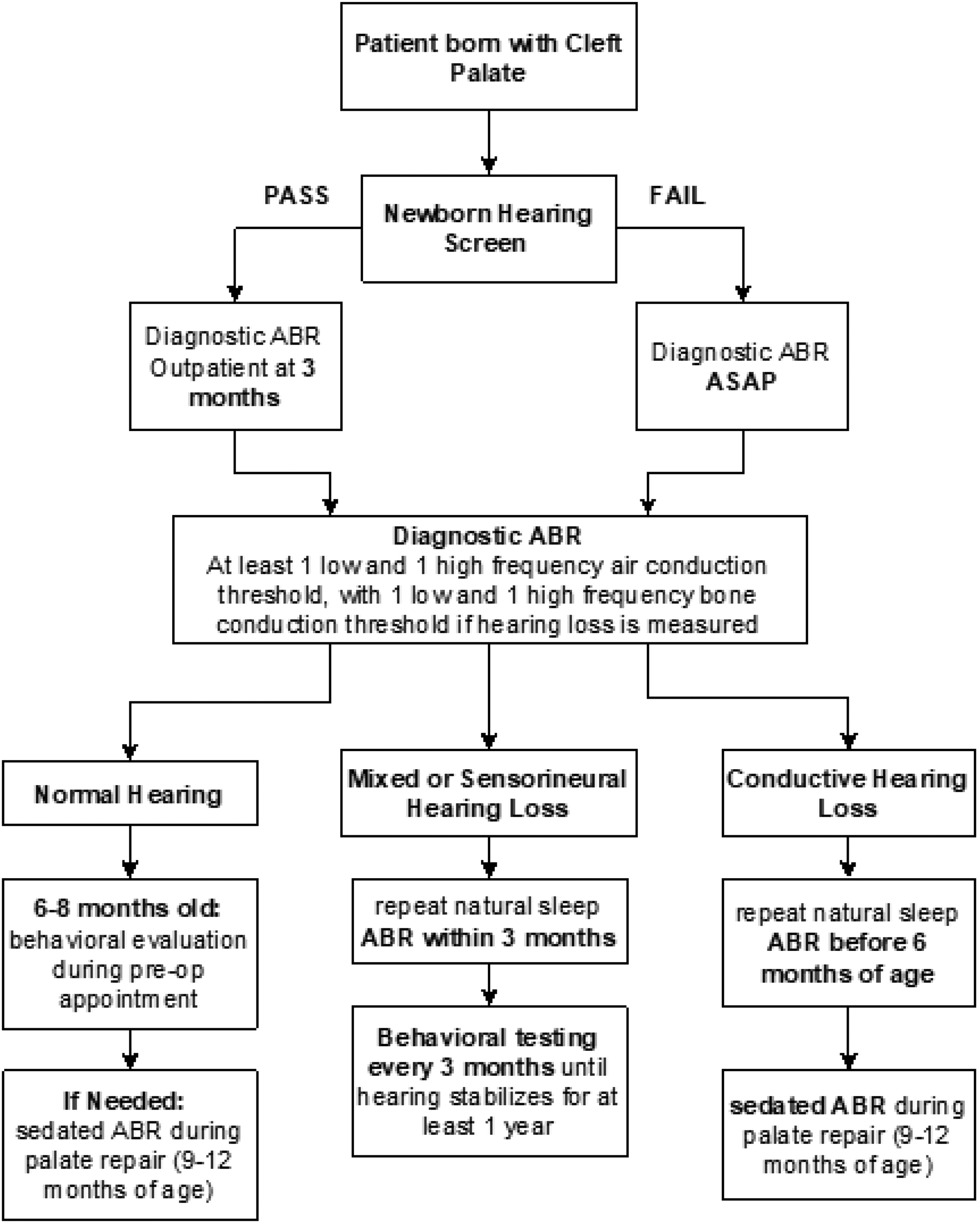
Enhanced audiologic protocol.
Auditory brainstem response (ABR) was used for all NBHS. Bilateral, frequency-specific (ABR) testing during natural sleep was performed in patients aged 0 to 6 months as part of the enhanced protocol or when otherwise indicated. This included click stimuli to determine auditory nerve integrity, minimum of one low frequency toneburst stimuli, minimum of one high frequency toneburst stimuli, and frequency specific bone conduction testing. Additional audiologic testing was determined by patient age and developmental status including Tympanometry, Distortion Product Otoacoustic Emission (DPOAE), and Visual Reinforcement Audiometry (VRA).
The visual reinforcement audiometry (VRA) protocol was used for behavioral testing when developmentally appropriate. Initial testing was performed in the sound field from 500 to 4000 Hz. Pending child compliance, ear-specific testing was attempted via insert earphone transducers or supra-aural headphone transducers from 500 to 8000 Hz. If hearing loss was measured at any frequency, unspecified bone conduction testing was performed to assess for CHL vs SNHL. Contralateral masking for bone conduction testing was not attempted due to cohort's developmental status.
DPOAEs (2000 to 6000 Hz) were also a considered testing modality, notably when subjects were noncompliant for natural sleep ABR or behavioral testing. When present, DPOAEs can indicate normal to near normal hearing thresholds at the level of the cochlea. 18 It should be noted however, that prior studies have demonstrated that the amplitude response of DPOAE is altered in the presence of middle ear dysfunction. 19 For the purposes of our study, patients who were not acquiescent for ABR or behavioral testing, but presented with abnormal DPOAEs, were categorized as “unclassified” hearing loss, as hearing severity cannot be determined by DPOAE responses alone.
Clinically, hearing loss is categorized according to severity as defined by the American Speech-Language-Hearing Association guidance: 26-40 dB defined as mild, 41-55 dB defined as moderate, 56-70 dB defined as moderately severe, 71-90 dB as severe, and greater than 91 dB defined as profound. For this study, a child was considered to have hearing loss that required intervention if there was mild or greater severity hearing loss present at two frequencies in one ear or three frequencies across both ears. Our hearing loss categories were constructed based upon most common severity ranges identified in the two cohorts: normal hearing, mild, mild-moderate, moderate, moderately-severe, and unclassified (abnormal DPOAEs). Severity of hearing loss was defined as the average severity level across all frequencies tested (500 Hz-4000 Hz).
Chi-squared tests were used to analyze the descriptive statistics of demographics, cleft characteristics, genetic abnormalities, risk factors for CHL, severity of hearing loss, differences in NBHS results between groups and the incidence of hearing loss detection between cohorts overall and within the subgroup of patients who passed their NBHS.
Results
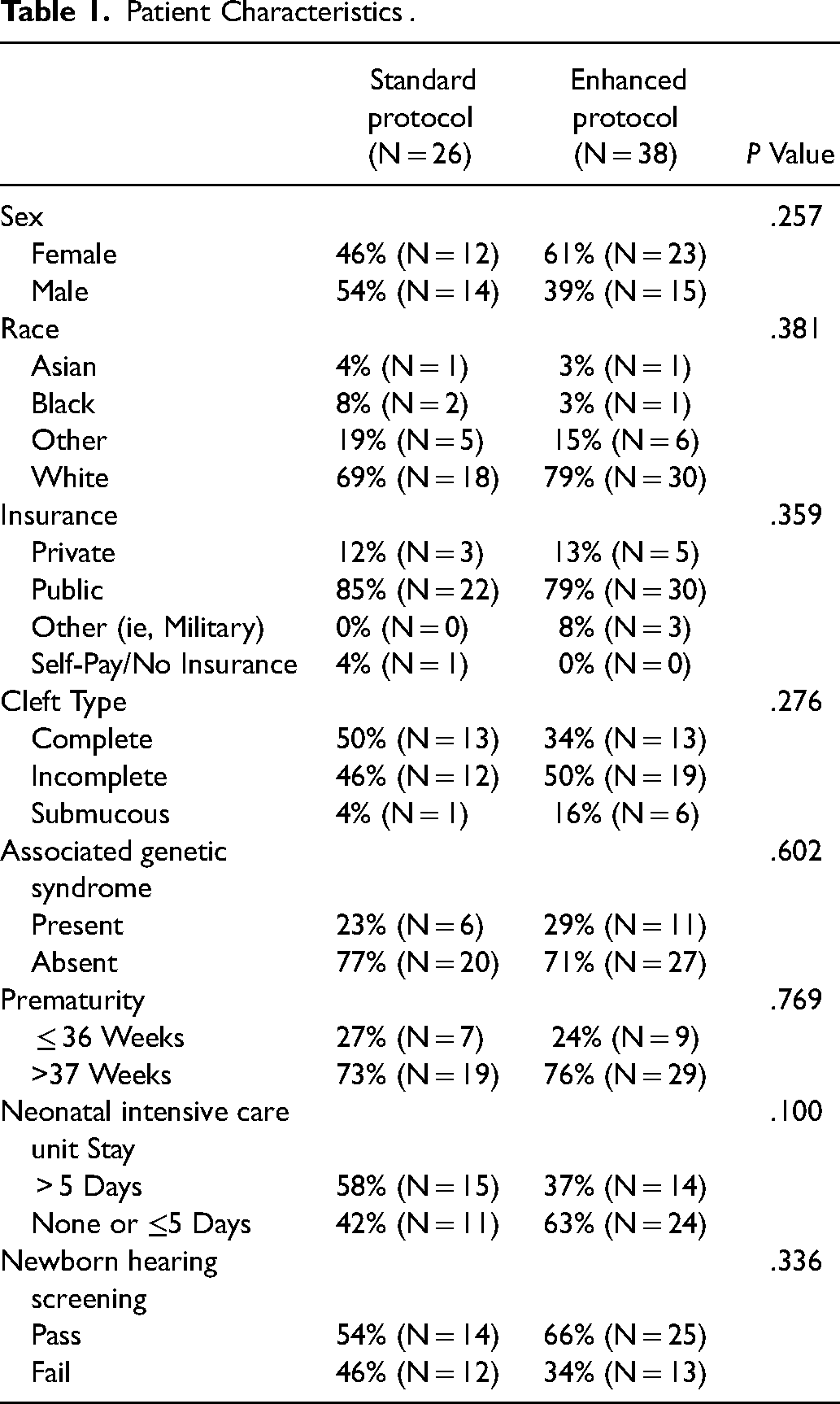
Twenty-six participants met inclusion criteria for the standard audiologic protocol and 38 met criteria for the enhanced audiologic protocol. Infants in these groups did not differ with regards to sex (P = .26), race (P = .38), insurance type (P = .36), cleft type (P = .28), presence of genetic syndrome (P = .60), or traditional risk factors for hearing loss including prematurity (P = .77) and extended NICU stay (greater than 5 days) (P = .1) (Table 1).
Patient Characteristics .
The number of patients who passed their NBHS did not differ between the standard protocol (n = 14/26, 54%) and the enhanced protocol (n = 25/38, 66%) (P = .34) (Table 1). The percentage of patients with CHL present prior to CP repair also did not differ in the enhanced protocol cohort (n = 29/38, 76%) compared to the standard protocol (n = 20/26, 77%) (P = .96). After initiation of the enhanced protocol, the number of patients who did not undergo additional pre-operative monitoring post passed-NBHS and by palatoplasty/9-12 months of age significantly dropped from 45% (n = 22/49) to 4% (n = 2/38) (X2[1, N = 49] = 21.46, P < .0001).
The severity of hearing loss detected did not differ between the two protocols (P = .66) (Table 2). There was no difference between the two protocols for infants who passed their NBHS, with 68% (n = 17/38) of patients in the enhanced protocol cohort having detected hearing loss on subsequent audiologic testing compared to 57% (n = 8/26) in the standard protocol cohort (P = .10) (Table 3). Twenty patients (60%) in the standard protocol that passed their NBHS did not have subsequent hearing testing prior to their CP repair, compared to 2 patients (6%) in the enhanced protocol. Of those patients who passed their NBHS in the enhanced protocol group, 48% (n = 12/38) had CHL identified by 3 months, with an additional 20% (n = 5/38) by 6 months of age.
Hearing Loss Severity.

*Represents absent DPOAE results when unable to obtain behavioral information.
Hearing Loss with Passed NBHS.

Conclusion
Patients with CP are known to be at high risk for early childhood hearing loss. However, to this date, no additional hearing assessment following a passed NBHS has been routinely performed prior to 9 months of age for this population. Prior studies have shown the necessity of early comprehensive audiologic assessment and intervention, but no standard protocol has been established. 20 In this study, an enhanced audiologic protocol was implemented for patients with CP to detect hearing loss prior to palate repair and/or 9-12 months of age. Prior to implementation of the enhanced protocol, 45% of patients did not undergo additional diagnostic audiologic testing pre-operatively following a passed NBHS. This declined to 4% after implementation of the enhanced protocol. This protocol resulted in a significant improvement in early detection of hearing loss. Overall, 76% of patients were identified as having hearing loss prior to 9 months of age after the enhanced protocol was implemented, and 77% in the standard protocol. This data indicates that CHL was prevalent in cohorts well before the 9-month age mark, and while there was no significant difference between the two groups, it can be speculated that values would have been higher in the standard protocol group if 45% of these subjects received more frequent audiologic monitoring.
The increased detection of hearing loss was primarily among patients who passed their NBHS, as patients who failed NBHS undergo additional audiologic workup as soon as possible. Among those who passed the NBHS, 68% had detected hearing loss with the enhanced audiologic protocol compared to 57% with the standard protocol. In addition, the onset of CHL in patients who pass NBHS was identified earlier than the recommended 9-month guideline, with 48% (n = 12) had CHL identified by 3 months, and 20% (n = 5) by 6 months of age.
While an improvement in early detection of hearing loss was achieved with the enhanced protocol, it was not known whether there would be a difference in the severity of hearing loss being detected by increasing the frequency of testing. Results indicated no significant difference amongst the two groups in hearing loss severity. Mild hearing loss was the most common severity of hearing loss detected with the standard protocol (39%, n = 10), compared to the enhanced protocol, with mild hearing loss and mild to moderate hearing loss being tied for the most common severities (29%, n = 11).
These results have important implications as patients who are not receiving routine hearing evaluation prior to CP repair or prior to 9 months of age are not receiving appropriate interventions for potential undetected hearing loss. Our study showed 68% of CP patients that passed their NBHS eventually developed CHL prior to 9 months of age with an enhanced protocol in place. It has been well-established that even mild hearing loss can directly impact a child's speech and language, as well as their psychoeducational and psychosocial development. 21 This study demonstrates a benefit to comprehensive audiologic testing in addition to a passed NBHS result within the first few months of life for patients with CP.
The goal of early detection of hearing loss is early intervention and improved speech and language outcomes. Most hearing loss in patients with CP ranges from mild to moderate CHL in nature, which is amenable to non-invasive intervention via amplification. Infants identified with CHL at this center were fit with softband bone conducting hearing aids until more permanent interventions (ie, tympanostomy tubes and palate repair) were performed and hearing was confirmed normal post-operatively. Future studies will measure longitudinal audiometric and linguistic outcomes following this method of early intervention.
There were several limitations to this study. As a retrospective single institution study with a small sample size, additional multi-institutional studies are necessary to confirm these results and to determine the true incidence of hearing loss during infancy in this population. As a tertiary referral center, the patients seen at this institution may be more complex with a higher rate of comorbid conditions than that seen at lower acuity centers. However, the rates of genetic syndromes seen in this CP population were similar to global estimates, suggesting that this population is likely representative of that seen outside of tertiary referral centers. Limited sleep state and missed cleft team appointments resulted in two patients unable to complete the enhanced protocol prior to palate repair.
The current recommendation for hearing assessment in patients with CP who passed NBHS is at 9 months of age according to the JCIH guidelines. 17 However, a significant proportion of patients who pass their NBHS will develop CHL within the first 3-6 months of life, and this hearing loss will not be identified by following the current guidelines. This study demonstrates a need for earlier routine audiologic evaluation in this population, regardless of NBHS results. An enhanced protocol with routine diagnostic ABR at 3 months of age for this at-risk population will improve early detection and intervention strategies, possibly improving long-term audiometric outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. This retrospective cohort study was approved by the Vanderbilt University institutional Review Board (#190840).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
