Abstract
Palatal fistulae are a recognised complication in individuals who have undergone surgical repair of a cleft palate, however, congenital or idiopathic palatal fistulae are rare. This report discusses the presentation and treatment of a 16-year-old female with a submucous cleft palate, who presented with a recent onset change in speech and evidence of a new palatal fistula. There was no history of recent infection or known trauma, and the patient had not undergone any previous palatal surgery. This report discusses the clinical presentation, recommended management and relevant literature for this rare phenomenon.
Introduction
Although cleft lip and palate are among the most common congenital malformations and palatal fistulae are a recognised complication following the surgical repair of a cleft palate, congenital and acquired palatal fistula are rare. 1 Acquired palatal fistulae can be related to local trauma, or more rarely, primary or secondary malignancies, conditions such as Wegener's granulomatosis or radiation arteritis, deep mycoses in the immunocompromised, cocaine abuse or syphilis.1,2
The congenital palatal fistula mainly occurs in patients with a submucous cleft palate (SMCP) and is the most commonly identified in the first few months of life, with symptoms such as nasal regurgitation, or in the early years of life with hypernasal speech.3,4 However, the spontaneous development of a palatal fistula in older children and adults does occur, and the risk of this should be mentioned to patients and their families.5,6
We discuss the presentation of an idiopathic palatal fistula in a 16-year-old female, identified due to a recent onset change in speech.
Case Report
A 16-year-old female with a conservatively managed SMCP was reviewed by the cleft speech and language therapists (SLT) following a concern raised by the mother of new speech unintelligibility. Her medical background included macrocephalus, mild learning difficulties, sleep apnoea, gastro-oesophageal reflux and mild tracheolaryngomalacia. Her only regular medication was Desmopression. She was a non-smoker and resided with her mother.
A reduction in speech clarity was noted by the parent shortly following the patient's fitting of orthodontic brackets, with no recollection of recent trauma. The speech and language assessment determined that the patient had mild and consistent hypernasal resonance with some inconsistent audible nasal emission. She had no active cleft speech characteristics but had developed new weak consonants in connected speech. Previous speech assessments noted mild turbulence only, with clinical notes describing the palate as intact with the features of a SMCP. Therefore, the fistula was considered to have occurred spontaneous or as a result of an innocuous everyday activity, such as eating.
Referral for lateral videofluoroscopy and review by the cleft surgical team were arranged. The clinical assessment demonstrated a fistula in the midline of the patient's soft palate. The videofluoroscopy showed good palatal length with a mild anterior lift but effective closure (Figure 1).
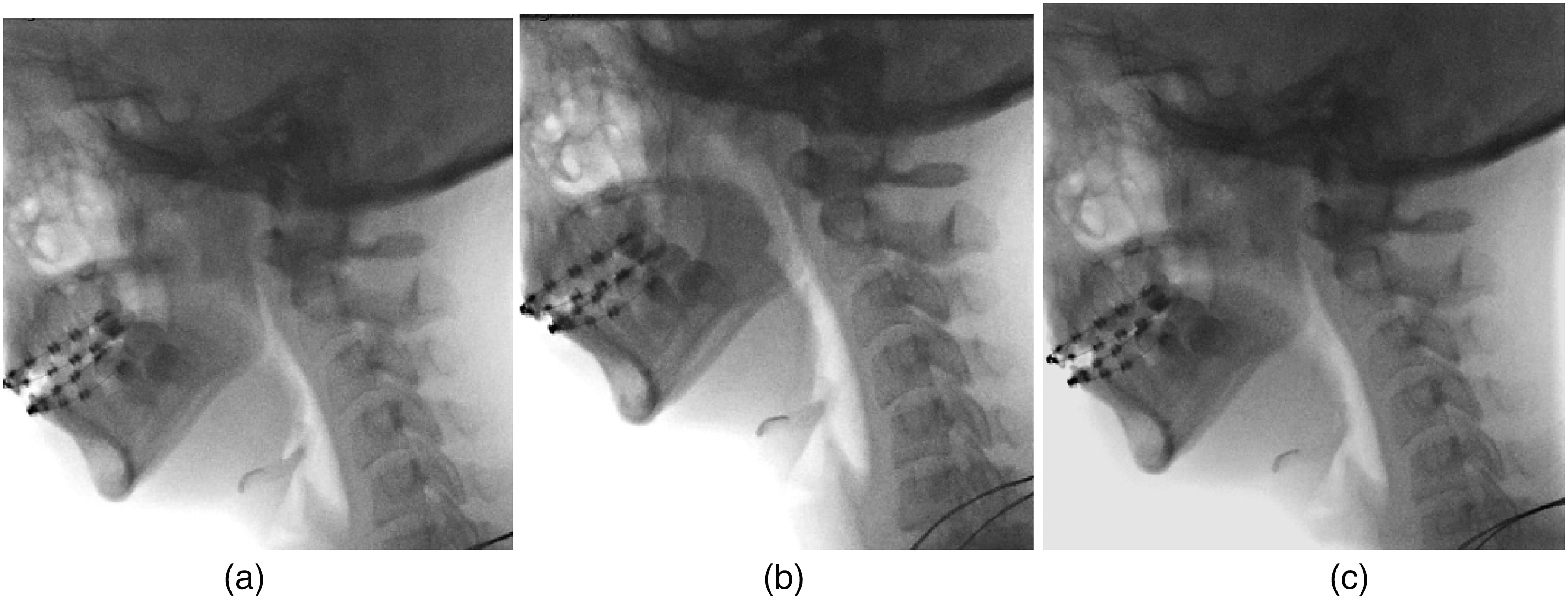
(a–c) Lateral videofluoroscopy demonstrating effective closure.
The diagnosis of velopharyngeal dysfunction (VPD) relating to nasal air escape through this new palatal fistula was made. The patient was listed for repair of SMCP and closure of the fistula under a general anaesthetic.
Peri-operatively, a central blue line, bifid uvula and a moderate hard palate notch were identified. The central soft palate fistula measured 8 mm × 5 mm in size (Figure 2).
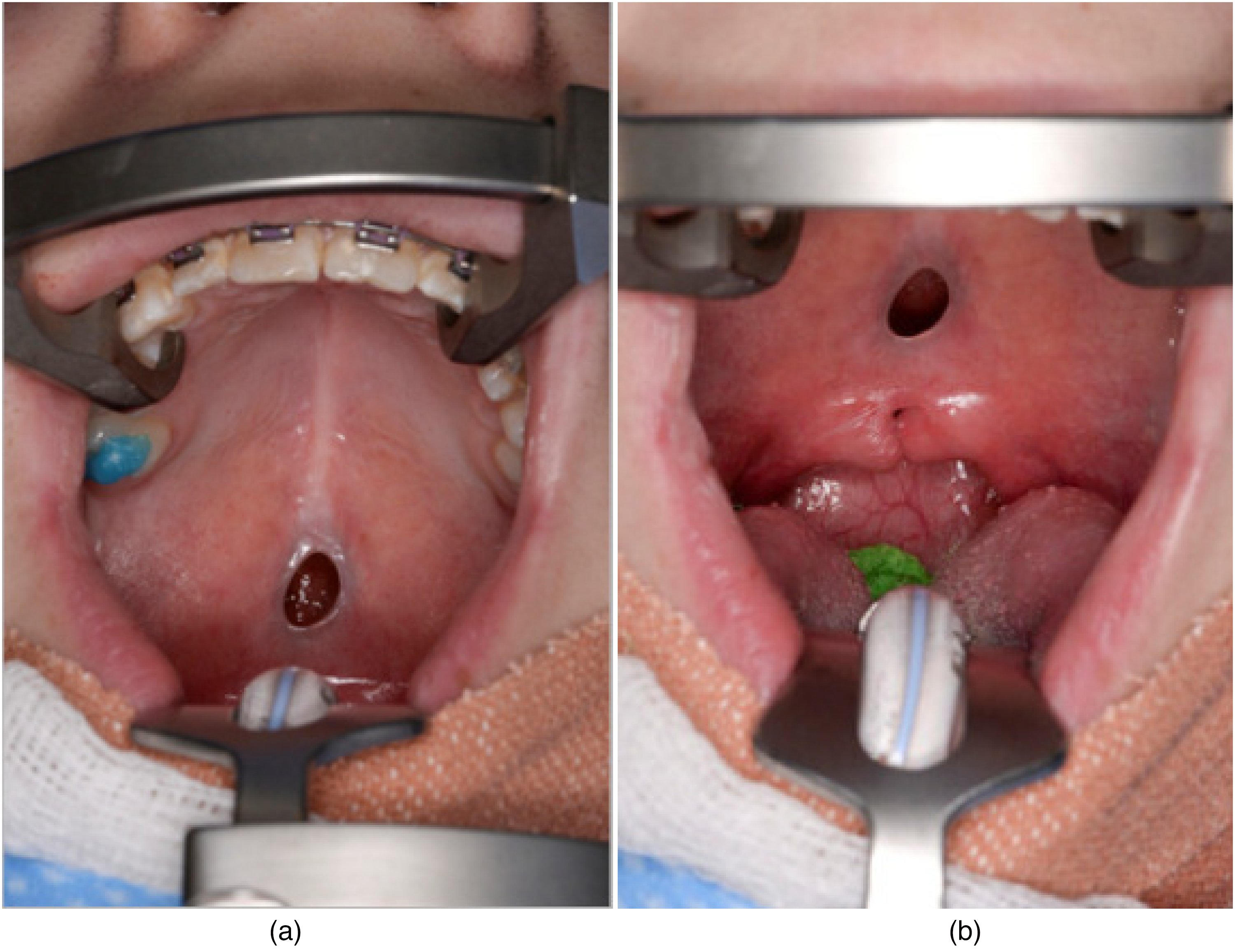
(a and b) Photograph of palatal fistula.
A midline incision including fistula margins was performed following infiltration with lignocaine local anaesthetic with adrenaline. The nasal layer was repaired with 4-0 vicryl. The muscles were found to be running anteriorly, as expected. An intravelar veloplasty with dissection of the levator veli palatini muscle and retropositioning of the muscle bundles of the soft palate was performed. The oral layer was repaired with 4-0 Vicryl and Chirocaine 0.25% was infiltrated for post-operative analgesia.
The patient had an uneventful recovery. Post-operative antibiotics were administered as per the unit protocol – two post-operative intravenous infusions of co-amoxiclav followed by five days of co-amoxiclav oral suspension. Regular analgesia was advised and chlorhexidine mouth-rinse for use three times daily for seven days was prescribed. A soft diet for six weeks was recommended. The patient was discharged the day after surgery, with a specialist cleft nurse review four months after surgery where it was found that the patient had made a full recovery, her palate had healed well and both patient and her mother recognised improvement in her speech. Formal speech assessment with cleft SLT demonstrated normal resonance with no evidence of VPD.
Discussion
Congenital and idiopathic palatal fistulae are a rare entity. In 1931, Veau and Borel 7 first reported the phenomenon of congenital palatal fistula, although there were two previous documentations of congenital palatal fistulae in 1870 and 1908, by Trelat and Metcalf, respectively.8,9
Since then, little over 40 cases of congenital or idiopathic cases of palatal fistulae have been reported in the literature worldwide (see Table 1). The majority of the reported cases presented with palatal fistula at birth or in the early years of life.3,4,10,11 These palatal fistulae appear as oval-shaped, full-thickness fenestrations, most often in the palatal midline and at the hard–soft palatal junction and do not extend fully anteriorly or posteriorly. 4
Literature Review of Congenital or Idiopathic Palatal Fistulae via PubMed Database.
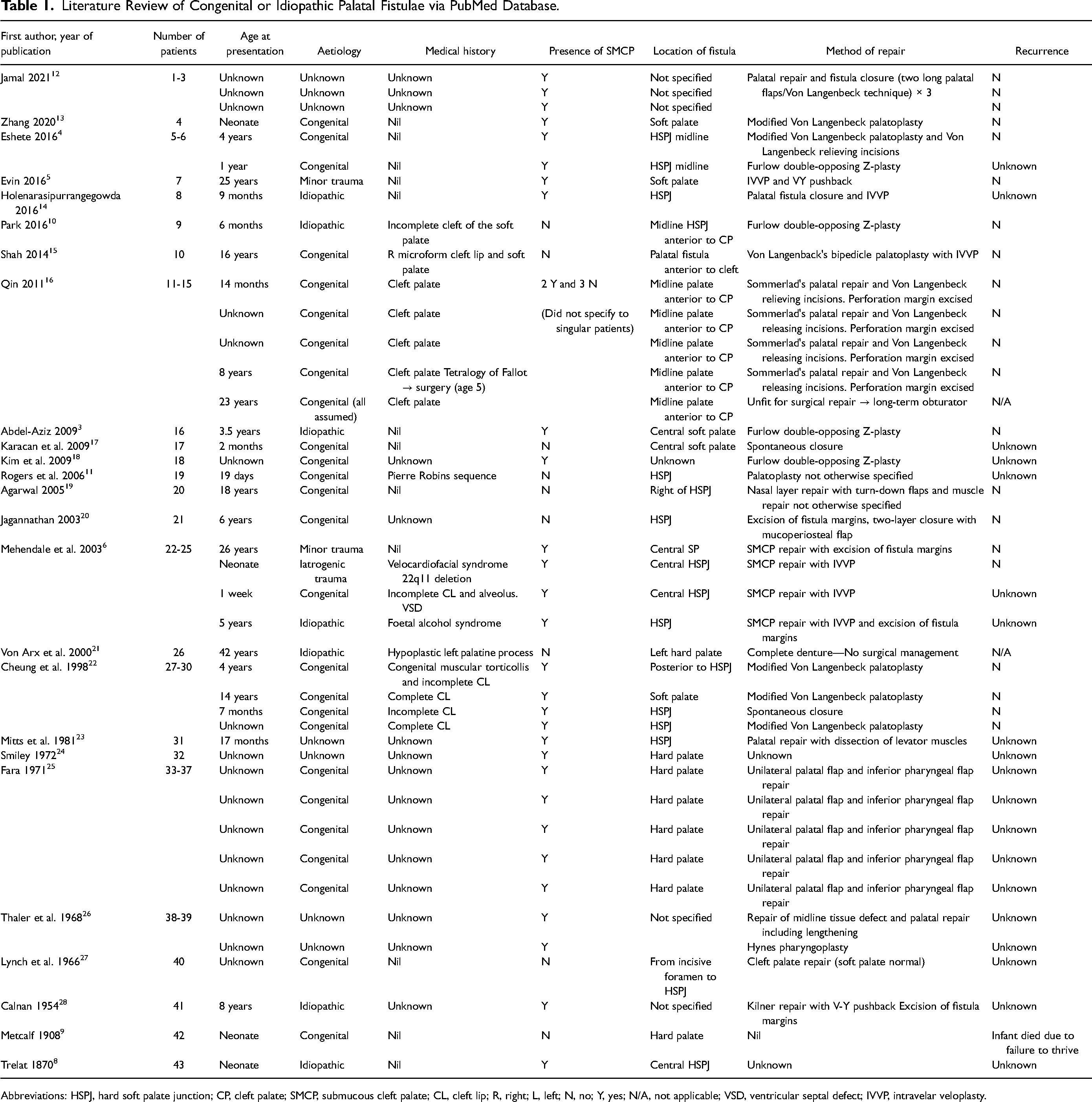
Abbreviations: HSPJ, hard soft palate junction; CP, cleft palate; SMCP, submucous cleft palate; CL, cleft lip; R, right; L, left; N, no; Y, yes; N/A, not applicable; VSD, ventricular septal defect; IVVP, intravelar veloplasty.
These fistulae are mainly identified in patients with a SMCP. The classic presentation of SMCP is described by Calnan's triad with a palpable bony notch of the posterior hard palate, a midline translucent zone (zona pellucida) of the soft palate and a bifid uvula. 28 The abnormal muscular anatomy impairs normal function and can result in difficulties with swallowing and VPD affecting speech. Not all patients with SMCP require surgical intervention, only those with affected speech and/or nasal regurgitation.4,29
Therefore, it is the practice of our unit that any child in whom a diagnosis of SMCP is suspected is registered with the cleft service. If the child is under 18 months of age, they are reviewed by the cleft surgeon. If an obvious SMCP is diagnosed, with nasal regurgitation, then surgery will be offered at this point as operating early can be associated with better velopharyngeal function. 30 If not, then a babble assessment at 18 months of age is undertaken by the SLT. Where there is evidence of velopharyngeal insufficiency, or cleft type speech characteristics, then treatment is offered tailored to the individual child. In cases where treatment is not indicated, speech is further assessed at age 3. If there are still no concerns regarding speech, then the child is discharged with advice on re-referral should concerns be raised in the future. Older children referred to the service are assessed first by the SLT to determine if there are speech difficulties and for the role of further investigations such as lateral videofluoroscopy. As a significant proportion of individuals with an unrepaired SMCP will have VPD, SLT should and do play a key role in the identification and subsequent investigation of these children. 30 Therefore, a period of follow-up for the asymptomatic individual is recommended to monitor for deterioration in speech, hearing or nasal regurgitation. Individuals who undergo repair continue with the same follow-up and aftercare as those with an overt cleft of the palate.
The theories for the aetiology of congenital palatal fistula are conflicted. Veau 7 believed the fistula resulted from prenatal rupture of a SMCP. This was contradicted by Lynch et al., 27 who reported SMCP was missing in some patients with such fistula. Fara 25 reported that the fistula was defined as a postnatal appearance of the wide SMCP and collapse of the palatal tissue in the area of maximal tension during the fetal period. Accidental or artificial intraoral trauma was also frequently proposed.5,12 It is generally agreed that the congenital or idiopathic palatal fistula usually occur in those with a SMCP.3,6,17,22 There is the odd exception, for example, a case report by Karacan et al.. 17 described a congenital palatal fistula in a 2-month-old boy with no predisposing factors that was not associated with any cleft and closed spontaneously 18 months after presentation.
A literature search of congenital or idiopathic palatal fistulae was conducted via the PubMed database, using Boolean search terms “congenital” AND “palatal fistula”; “submucous cleft palate” AND “palatal fistula”, “trauma” AND “palatal fistula.” In addition to this, review of relevant references in all reports analysed was undertaken.
Inclusion criteria:
Fistula/perforation/aperture of palate Congenital/idiopathic or related to minor trauma Description of fistula + /− repair detailed in article Article available in English language History of surgical intervention of palate Acquired causes, for example infective or neoplastic aetiology No description of fistula or its repair in article Article not available in English language
Exclusion criteria:
From the PubMed literature search, 19 articles were identified, with 4 articles meeting the inclusion criteria. In the Google Scholar literature search, the initial results totaled 354, and 6 articles met the inclusion criteria, following removal of duplicates. An additional 15 articles were identified from review of all references of existing reports.
The results of the literature review are available in Table 1.
The completeness of findings was restricted by clinical details included in article reports, as multiple more historic articles listed palatal perforation among other findings or included a captioned clinical photo of a palatal fistula without further information.24,28 This did affect detail of information complicated in the table above. Within the total of 25 articles that met the inclusion criteria, a total of 43 cases of congenital or idiopathic palatal fistula were reported.
It is apparent from available literature, that such congenital or idiopathic fistulae are frequently identified in those patients with an existing SMCP (31/43 identified cases).3–6,8,12,13,16,18,22–26,28 This demonstrates the importance of informing the families of patients with SMCP that there is a risk of early or late palatal perforation, that can impact speech intelligibility and lead to nasal regurgitation. This finding also suggests that the presence of a SMCP should be explored in all patients presenting with a palatal fistula, as it is more common in this population cohort.5,12
From the available literature, it has been concluded that repair should not focus on fistula closure alone but also on providing adequate palatal length and appropriate muscle positioning to provide effective velopharyngeal function, as in any cleft palate repair. 3 Speech evaluation by a cleft SLT should be performed to assess velopharyngeal function pre-operatively and post-operatively, especially when there is overlap of a palatal fistula with the spectrum of a submucous cleft. 4 This case also identifies the benefit of including an oral examination in speech assessments by SLT, even for long-term patients.
In this case, the new onset of hypernasality and previous clinical assessment makes it unlikely a fistula was previously present. Given the patient and family's lack of awareness of the fistula, with no declaration of significant events in the history, it is unlikely this was acquired from an infection or significant trauma. However, it is likely it was related to a minor trauma in the thin central mucosa of the secondary palate. Other considered possibilities include increased palatal tension from placement of orthodontic fixed appliance, or spontaneous rupture of the friable mucosa.
Conclusion
From this case and with review of available literature, we have concluded it is important to warn SMCP patients and their families of the increased risk of developing an idiopathic palatal fistula and inform the families of signs, symptoms and potential risk factors of this rare phenomenon. Patients presenting with a new fistula of unknown origin should be assessed for the presence of a SMCP and repair of the palatal fistula should be completed alongside functional repair of the palate.3–6,9–16,18,22,25
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
