Abstract
Objective
To evaluate feeding efficiency and weight gain in infants with cleft palate fed using 1 of the 2 specialty feeders.
Design
Retrospective cohort study.
Setting
Cleft palate clinic in a tertiary pediatric hospital.
Participants
Infants with cleft palate (with or without cleft lip) fed using the Medela SpecialNeeds® Feeder (n = 39) or the Dr. Brown's® Specialty Feeding System (n = 16) and who had documented feeding and growth data available from the time of initial assessment in the first month of life and at subsequent follow-up between 2 and 4 months.
Primary Outcome Measure
Feeding velocity (mL/min).
Secondary Outcomes Measures
Calorie velocity (kcal/min), weight gain, and complications associated with poor feeding.
Results
No statistically significant differences in feeding or calorie velocities were identified between infants with cleft palate fed with the Medela SpecialNeeds® feeder and those fed with the Dr. Brown's® feeder. Mean weight z-scores by month did not differ significantly between the 2 bottle groups at the time of initial assessment (P = .84) and follow-up (P = .20). Mean weight z-scores by month for the infants included in this study fell below the 50th percentile. The proportion of infants who developed otitis media, reflux requiring treatment, or who required hospital admission for nasogastric (NG) feeds did not differ significantly between the 2 groups.
Conclusions
Despite being adequately powered for the primary outcome, no significant differences were identified between infants fed with the Medela or the Dr. Brown's feeders in terms of feeding velocity, calorie velocity, weight gain, or complications.
Keywords
Introduction
Nutritional management of infants with cleft palate remains a challenge. 1 Lengthy feeding times, decreased volume intake, and nasal regurgitation are commonly encountered issues in infants with cleft palate that can result in reduced calorie intake, inadequate nutritional status, and poor growth.2–4 These in turn can lead to failure to thrive and compromise social and emotional health.2,5–8 Additionally, feeding difficulties during infancy have been associated with an increased risk of cognitive impairment and delayed achievement of major developmental milestones.9,10
Feeding difficulties in infants with cleft palate vary depending on factors such as cleft severity and the presence of a syndrome and result from an inability to generate negative intraoral pressure, which is a prerequisite for breastfeeding or feeding from a standard bottle.11–14 The production of negative intraoral pressure requires the creation of a firm seal between the lips and the nipple or teat, separation of the oral and nasal cavities via the action of the soft palate and associated muscles, and depression of the tongue and mandible.4,13 Compression of the nipple or teat against the palate with the tongue creates positive pressure, which forces fluid into the oral cavity. In most children with an isolated cleft lip, normal feeding is possible because the infant retains the ability to generate negative intraoral pressure.6,15,16
To ensure adequate nutrition and weight gain in infants with cleft palate, educational programs, obturators, and specialized bottles are commonly used to eliminate the need for negative intraoral pressure generation.1,4,13,14,17 While these specialty feeding systems improve oral intake in infants with cleft palate, they do not entirely normalize feeding compared with infants without a cleft palate. 18 A variety of specialized bottles are commercially available, including the Cleft Lip/Palate Nurser by Mead Johnson and the Pigeon feeder. The 2 specialized bottles supplied by our institution are the Medela SpecialNeeds® feeder (formerly the Haberman feeder) and the Dr. Brown's® Specialty Feeding System. The Medela feeder contains a one-way valve that isolates milk within the teat; once inside, the infant is able to extract milk through compression alone, which enables feeding in the absence of negative pressure generation.4,13,14 While this design allows the infant to control the rate of feeding, the squeezable nipple in the Medela feeder may also be compressed by the caregiver if the infant requires assistance or additional milk during feeding.4,13 Additional fluid delivery may also be achieved by changing the orientation of the teat in the infant's mouth, which alters the flow rate. The presence of a venting system in the neck of the bottle is meant to prevent vacuum formation within the nipple; by decreasing air intake during feeding, this design claims to reduce vomiting and colic. 19 Similar to the Medela feeder, a one-way valve in the Dr. Brown's feeder enables the infant to feed without suction by isolating milk within the teat. Unlike the Medela feeder, the Dr. Brown's feeder does not require compression of the nipple by the caregiver; instead, flow of the bottle can be modified by changing the nipple aperture. 20 Prior to 3 months of age, most infants require a level 1 nipple, transitioning to a level 2 nipple based on their intake. The presence of a vent in the Dr. Brown's feeder has been proposed to reduce vomiting and colic by decreasing air intake during feeding, but to date there has been little data to substantiate this claim. 4
While these 2 bottles are functionally similar, the 2 feeders differ in their cost. To date, the 2 feeders used at our institution have not been compared directly in terms of their feeding efficacy. The purpose of this observational study was to assess the feeding velocity and subsequent weight gain in infants with cleft palate fed using either the Medela or the Dr. Brown's feeders.
Methods
Study Design and Participants
This retrospective observational study examined feeding velocity (mL/min) and calorie velocity (kcal/min) in infants with an isolated cleft palate or cleft lip and palate from birth to 4 months of age fed using either the Medela or Dr. Brown's feeders.
Inclusion criteria included infants with an isolated cleft palate or cleft lip and palate who were assessed in the cleft palate program at a single tertiary pediatric center within the first 2 weeks of life and who subsequently underwent primary palatoplasty by the senior author between January 1, 2014 and December 31, 2019. Prior to January 1, 2014, all patients seen through the cleft palate program were offered the Medela feeder; after this date, both the Dr. Brown's or Medela feeders were offered. All families received counseling from the team nurse regarding the use of both the Medela and Dr. Brown's feeders. Families were then free to select one or the other. No other significant changes in postnatal cleft care were otherwise implemented over the study period.
Patients with an isolated cleft lip, a syndrome precluding them from oral feeding (eg, severe neuromuscular dysfunction with aspiration, tracheoesophageal fistula) or those patients who did not use either the Dr. Brown's or Medela feeders were excluded from this study. Infants with Robin sequence (airway obstruction, glossoptosis, and micrognathia) were included in this study. 21
Data Collection
Institutional research ethics board approval was obtained. Demographic and clinical data were extracted from the patients’ medical records. The first 3 digits of the patient's postal code were collected to determine proximity to the tertiary center or a peripheral center. Peripheral birth center was recorded as patients who received follow-up care, there would conceivably have fewer in-person supports from our cleft team nurse and dietician. A history of a sibling with cleft was also collected as a measure of past parental experience with specialty feeders. Gestational age and presence of Robin sequence (airway obstruction, glossoptosis, and micrognathia) or other syndrome were also collected as greater feeding difficulty and an increased likelihood of failure to thrive have been documented in premature infants and those with syndromic cleft palate compared with nonsyndromic cases.3,6,14 Otitis media, nasal regurgitation, and gastroesophageal reflux requiring medication therapy, conversion from one feeder to another, and need for nasogastric (NG) tube feeding at any point during their care were also recorded. While NG tube feeding was not an exclusion criteria, patients were only included if the NG tube feed was used to “top up” after an attempted oral feed; patients were exclusively NG tube fed at any point after the introduction of one of the feeders were excluded.
Feeding data extracted from patient charts included the feeding system used, volume of feed consumed per feed, type, and calorie density of feed used (formula type, expressed breast milk, and presence of fortification), feeding time, and feed frequency. Families were routinely instructed to record this information which was then collected in the patient chart every 2 weeks by telephone or during clinic visits by the team nurse. For patients using the Dr. Brown's feeder, the nipple level was determined based on feeding time; once feeding times consistently exceeded 20 min, the team nurse instructed families to change to the next level nipple. For the Medela feeder, parents were instructed to apply pressure to the upper chamber in time with the rhythm of the baby's sucking and swallowing. Families that converted from one feeder to the other were considered to have incomplete data for time points for their original feeder after conversion. The monthly average for these parameters were calculated and recorded up to 4 months of age. Feeding entries were included for analysis only when the above variables were documented in their entirety. To calculate calorie velocity, the calorie concentration of breast milk was estimated to be 0.67 kcal/mL based on previous reports.22–24 For formulae, the calorie concentrations reported by the suppliers were used.
Patient weights were obtained from growth charts and subsequently converted to z-scores using World Health Organization (WHO) standards according to infant sex and age (in days) to allow for the pooling of anthropometric data for both sexes as previously described.8,25–28 Premature birth was defined as birth occurring prior to 37 weeks’ gestation; in the event of prematurity, corrected weight was obtained using the Fenton growth chart. 29
Data Analysis
The primary outcome of average monthly feeding velocity (mL/min), as well as the secondary outcomes of average monthly calorie velocity (kcal/min) and average infant weight z-score by month, was compared between groups with a two-tailed student's independent t-test, with P < .05 considered significant. Differences in the proportion of infants in each feeder group who developed otitis media, required treatment for gastroesophageal reflux, required NG feeding, were premature, had a syndromic diagnosis, or were born at a peripheral center, were compared using Fisher's exact test. Statistical analyses were carried out using Prism (Version 7; GraphPad Software).
Based on the previous published estimates 18 and defining a clinically relevant increase in feeding velocity of 33% (3 vs 4 mL/min with a standard deviation of 1 mL/min), with a sample size of 16 participants and an α of 0.05, the calculated post hoc power for our primary outcome was 0.80.
Results
Sample Description
A total of 55 infants for whom adequate feeding data could be obtained using the Medela (n = 39) or Dr. Brown's feeders (n = 16) during the study period. Briefly, the composition of each bottle group was similar in terms of sex and cleft type (Table 1). Assessment of initial bottle use according to birth center revealed no statistically significant differences in terms of bottle uptake in the tertiary center (where the study site cleft palate program was located) versus smaller, peripheral centers (P > .99). Average birth weight (P = .23), as well as the proportion of infants born premature (P > .99) did not differ significantly between infants fed using either the Medela or Dr. Brown's feeders. Similarly, there were no differences in the number of syndromic patients in each group (P = .47). In the Medela feeder group, 8 patients had a Robin sequence with no identified mutation on microarray, 1 had Wolf–Hirschhorn syndrome, and 1 had a 15p14 microdeletion syndrome; in the Dr. Brown's feeder group, 2 patients had a Robin sequence with no identified mutation on the microarray.
Participant Characteristics.
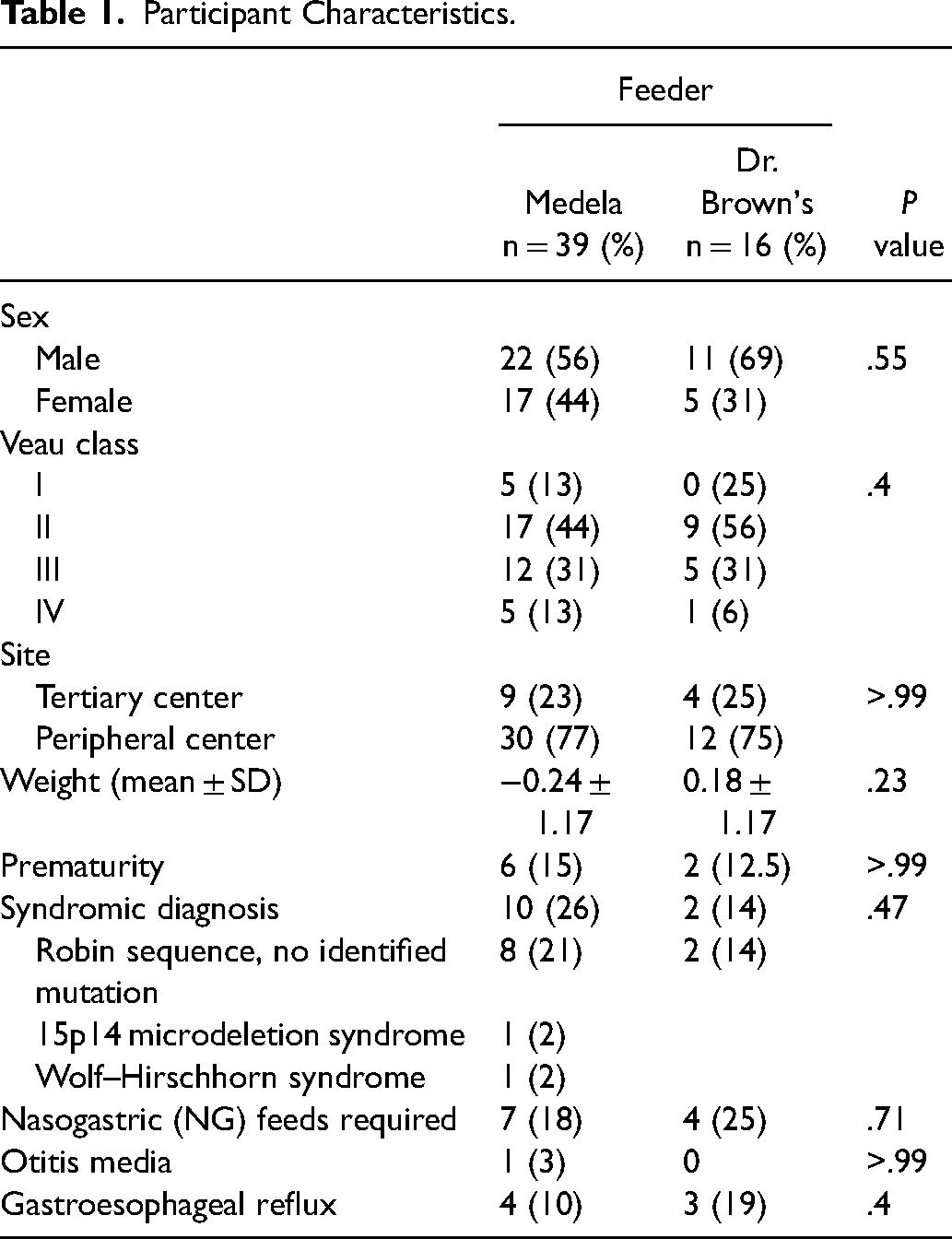
Comparison of the 2 specialty feeder groups revealed no significant reduction in commonly reported feeding sequelae observed in infants with cleft palate. For both bottle groups, no statistically significant differences were observed in terms of the proportion of infants who required NG feeds during their care due to poor weight gain (P = .71). Similarly, the proportion of infants who developed otitis media (P > .99) or nasal or gastroesophageal reflux necessitating therapy (P > .40) was not found to differ significantly between groups. After 1 month, 5 patients who began with the Medela feeder converted to the Dr. Brown's feeder; no Dr. Brown's feeder patients converted to the Medela feeder.
Feeding Efficiency
Feeding efficiency was assessed by examining feeding velocity (mL/min) and calorie velocity (kcal/min) for infants fed using either the Medela or Dr. Brown's feeders during the first 3 months of life. By 3 months of life, all patients in the Dr. Brown's feeder had transitioned to a level 2 nipple. Feeding data for both bottle groups according to the infant's age in months is summarized in Table 2 and represented graphically in Figure 1. Mean feeding and calorie velocities increased with age for both groups, though no statistically significant differences were observed between bottle groups at the time of initial assessment (P = .89 for both feeding and calorie velocities) or at subsequent follow-up (P = .34 for feeding velocity and P = .36 for calorie velocity). No differences were observed between patients with or without Robin sequence in either feeder group or when comparing all patients with Robin sequence to non-Robin sequence patients.
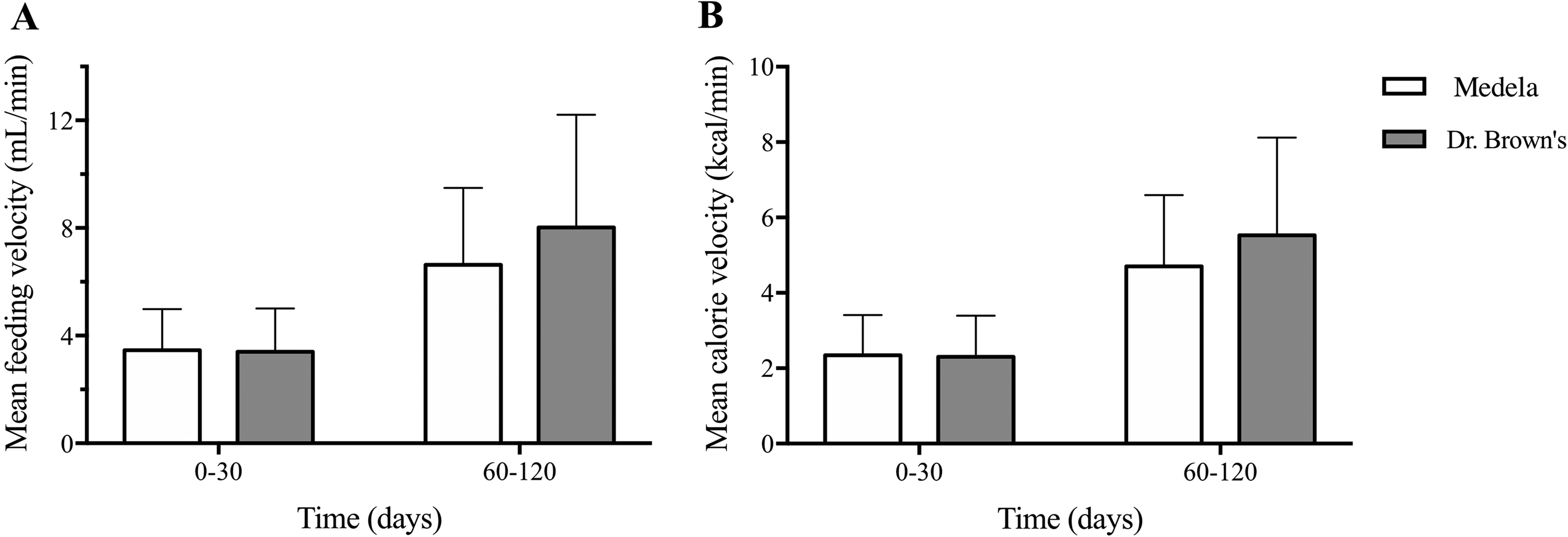
Mean feeding velocity (A) and calorie velocity (B) according to age range (in days) at the time of initial assessment (0-30 days) and follow-up (60-120 days) for infants with cleft palate or cleft lip and palate fed using either the Medela or Dr. Brown's feeders.
Comparison of Feeding Efficiency by Feeder Type.
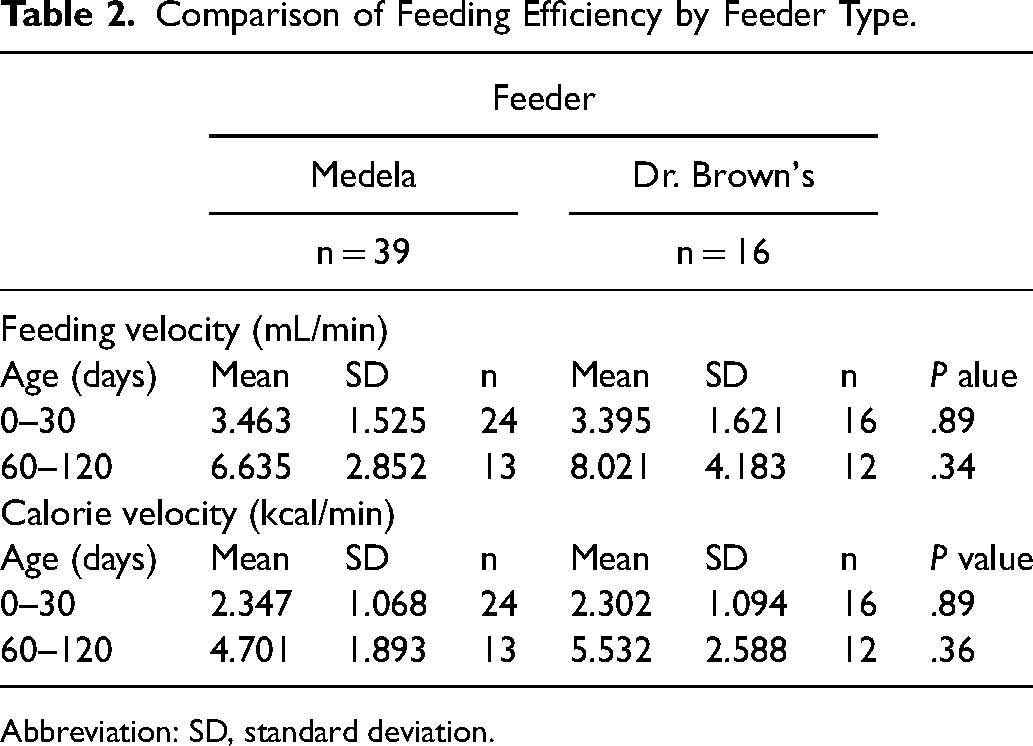
Abbreviation: SD, standard deviation.
Weight Gain
Mean weight z-scores for infants fed using the 2 specialty feeding systems are summarized in Table 3. Trends in weight change over the first 3 months of life are represented in Figure 2. The mean weight z-scores did not differ significantly between the 2 feeder groups at the time of initial assessment (P = .84) or when reassessed at follow-up (P = .20). At every time point assessed, mean weight z-scores for both bottle groups were below the 50th percentile according to WHO growth standards.
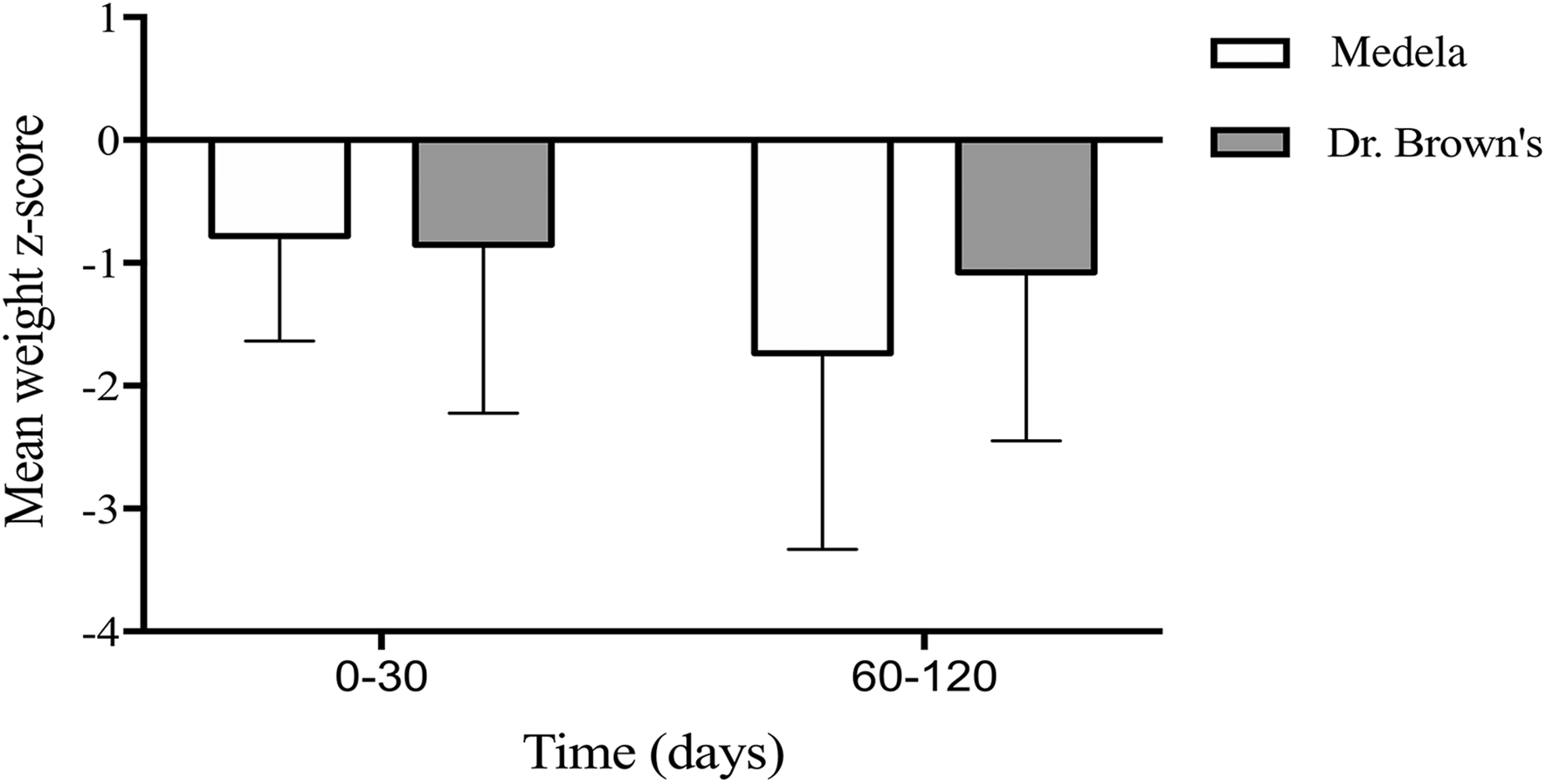
Mean weight z-scores grouped by age range (in days) at the time of initial assessment (0-30 days) and follow-up (60-120 days) infants with cleft palate or cleft lip and palate fed using either the Medela or Dr. Brown's feeders.
Comparison of Weight z-Scores by Feeder Type.
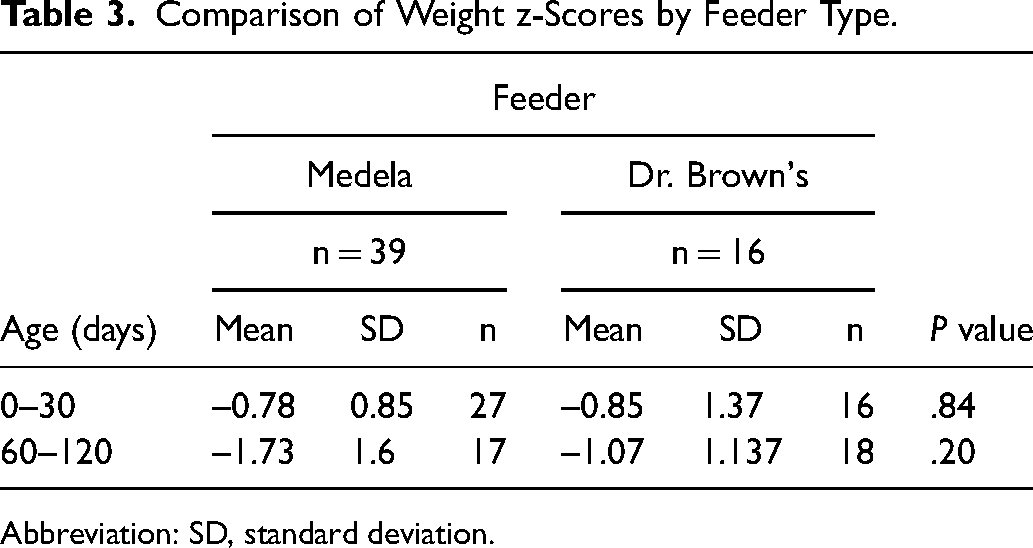
Abbreviation: SD, standard deviation.
Discussion
The primary aim of this study was to assess feeding velocity in infants with an isolated cleft palate or cleft lip and palate fed using either the Medela (formerly the Haberman bottle) or the Dr. Brown's feeders. When feeding data was grouped according to the infant's age in days, no statistically significant differences in feeding or calorie velocities were detected between the 2 bottle groups, despite being adequately powered for our primary outcome. Given the lack of data to suggest that feeding efficiency differed by feeder type, it is unsurprising that no statistically significant differences were observed in infant body weight between feeder groups. Comparison of the prevalence of select adverse feeding sequelae commonly seen in infants with cleft palate failed to identify any benefit of one feeder type over the other. Indeed, the proportion of infants who developed otitis media, required treatment for nasal or gastroesophageal reflux, or required NG feeds due to poor weight gain did not differ significantly between infants fed using either feeder.
Evidence quantifying feeding velocity with specific bottles designed for use in infants with cleft conditions is limited. Turner et al examined feed volume and duration in 8 infants with cleft palate (with or without cleft lip) who were fed with either a Haberman bottle alone, an obturator plate, or with both a Haberman bottle and an obturator plate. 30 Mean feeding velocities documented with the Haberman bottle in that study were 1.06 mL/min (36.5 mL/34.3 min) during the first week of life and 1.15 mL/min (37mL/32.3 min) when assessed during the second or third week after birth. A study by Reid et al 15 evaluated feeding efficiency in infants with cleft lip, cleft palate, or cleft lip and palate during the first month of life. This study found that 12 of the 15 infants assessed were unable to generate feeding velocities exceeding 1 mL/min. This observation must be interpreted with care as infants were required to feed from an unfamiliar, noncompressible bottle. In the current study, mean feeding velocity during the first month of life was found to be 3.46 mL/min and 3.40 mL/min for the Medela (formerly Haberman) and Dr. Brown's feeders, respectively. The fundamental differences in the properties of the bottles used in the study by Reid et al 15 versus the current study may explain the higher feeding velocities documented in the current study. The study by Turner et al 30 also assessed feeding velocity with the Haberman feeder at similar time points, though again mean feeding velocity was substantially lower than that observed in this study (1.15 mL/min vs 3.46 mL/min). This may be attributable to differences in feeder technique. In Turner et al study, families were instructed to compress the upper chamber with gentle, steady pressure, while in our study, parents were specifically instructed to squeeze the bottle to match the rhythm of their baby's sucking and swallowing. When fed using a familiar bottle, another study by Reid et al 3 found that feeding velocities of <2.2 mL/min in the first month of life and <3.3 mL/min during the third month of life were useful cutoffs for the identification of clinically relevant feeding difficulties. According to these cutoffs, few infants in the current study would be documented as poor feeders, even though they all fell below the 50th percentile for weight gain.
This study is not the first to suggest a lack of association between weight gain and specific type of specialty feeding system for infants with cleft conditions. A large body of literature exists to suggest cleft type and severity are the predominant determinants of feeding difficulty and impairments in weight gain.9,14,15,31 The disparities in weight gain according to cleft type have persisted in recent literature, irrespective of the type of feeder used. 32 While claims regarding the efficiency of various specialty feeding systems have been made to suggest the use of one system over another, there is a lack of evidence to support these. 14
In the current study, weight gain was not found to vary significantly between infants fed using the Medela or Dr. Brown's feeders; however, 5 infants initially fed using the Medela feeder eventually switched to the Dr. Brown's feeder. No infants fed using the Dr. Brown's feeder were switched to the Medela feeder. The reasons for feeding system conversion in this study were not explored, though other studies have reported caregivers’ frustration with the Haberman bottle, particularly its assembly and cost. 32 This and the lack of a formal cost analysis are notable limitations of the current study.
The current study is one of the first to directly compare 2 specialized cleft palate feeders. While a larger body of literature has documented the role of assisted feeding systems in infants with cleft palate, there is scant evidence exists to recommend the use of one feeder over another. Moreover, many studies of feeding interventions for infants with clefts frequently include infants with an isolated cleft lip, despite the well-documented finding that feeding and weight gain in these infants more closely resemble that of normal infants than their counterparts with cleft palate.3,5,6,10,15,33 Similarly, many feeding studies in infants with clefts have excluded infants with syndromes, despite the fact that a considerable percentage of infants with cleft palate have Robin sequence or an associated syndrome. 31 Indeed, in the current study, 21.8% of infants (12 of 55) with a cleft palate or cleft lip and palate had Robin sequence or an associated syndrome. By only selecting infants with clefts involving the palate and including infants with syndromic clefts, the current study offers insights into the feeding management of a more clinically challenging subset of patients. Unfortunately, the number of patients with Robin sequence in each of our feeder groups was too small to draw any meaningful conclusions on how this population differed from other children in terms of their feeding outcomes.
While we did have adequate power for our primary outcome, a major limitation of this study was the small sample size due to incomplete recording of infant feeding data. This was most apparent at follow-up assessments occurring after the first 30 days of life among infants who demonstrated successful feeding and acceptable weight gain early on. In these cases, omission of feeding duration, the type of feed consumed (expressed breast milk vs formula), or whether fortification was used was more common, making it impossible to determine feeding or calorie velocities. Thus, the current data may be skewed toward infants who struggled more with feeding and weight gain and ultimately required more intensive and detailed follow-up as a result. Owing to our low sample size, we were unable to formally explore the influence of factors such as sex, calorie density of feed (expressed breast milk vs formula), and syndrome diagnosis on feeding.
While patients were provided with instructions for the use of both specialty feeders and could choose between the 2, without formal randomization other factors may have confounded our results. Cost of specialty feeding systems has been identified as one such factor. 32 Given that the Medela feeder is more expensive than the Dr. Brown's feeder, families from lower socioeconomic status may have preferentially selected the latter feeding system. Unfortunately, our low patient numbers meant we were unable to stratify participants based on median income using parental postal code as previously described. 16
While impairments in weight gain in infants with cleft palate are generally corrected by 5 to 9 months of age, there is evidence to suggest that addressing feeding and growth issues during the first 2 to 3 months of life improves developmental and cognitive outcomes.9,33 The current study suggests that among our selected groups neither feeder type performed better nor the other in terms of feeding velocity. The limitations of the current study, including the inability to explore differences in feeding outcomes based on syndromic diagnosis, sex, prematurity, and birth center, or perform a formal cost analysis, could be addressed with a larger randomized controlled trial. Additionally, a greater rate of conversion from the Medela to the Dr. Brown's feeders observed in this study points to the need for a closer exploration of parental satisfaction and other family-reported outcomes.
Conclusion
The current study compared the Medela (formerly Haberman) and the Dr. Brown's feeders with regard to feeding velocity, calorie velocity, and weight gain in a nonrandomized population of infants with cleft palate. While no statistically significant differences in our primary outcome of rate or intake or secondary outcomes of calorie velocity or weight gain were observed between feeder types, this report points to the need for larger studies to explore feeding outcomes using different feeders among specific patient populations, including syndromic and premature infants as well as the need for robust cost-effectiveness methodology.
Footnotes
Author’s Contribution
CP: Contributed to study design, data acquisition, data extraction, analysis of results, drafting of the manuscript, final approval of the manuscript, and agrees to be held accountable for all aspects of the work. KN: Contributed to data acquisition, data extraction, analysis of results, drafting of the manuscript, final approval of the manuscript, and agrees to be held accountable for all aspects of the work. HG: Contributed to study design, drafting of the manuscript, final approval of the manuscript, and agrees to be held accountable for all aspects of the work. MB: Contributed to study design, data acquisition, analysis of results, drafting of the manuscript, final approval of the manuscript, and agrees to be held accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
