Abstract
Binder’s syndrome is a rare congenital deformity characterized by midface hypoplasia, particularly around the nasomaxillary area. Genetic etiology or developmental failure caused by prenatal exposure to teratological agents has been considered. In this article, we present 3 related rhesus monkeys born with orofacial deformities similar to those found in infants with the Binder phenotype. For the first time, a primate biomodel for this condition is presented. The clinical description and association with management and environmental factors are discussed. These findings reinforce the knowledge about the relationship between possible vitamin K metabolism interference and Binder’s syndrome.
Keywords
Introduction
Binder’s syndrome (BS) is an uncommon developmental anomaly of the middle third of the face characterized by nasomaxillary hypoplasia. It is a very rare facial syndrome that is considered part of the skeletal dysplasia spectrum of heterogeneous etiology. 1 The current understanding is that BS is not a disease but rather a phenotype with several causes. 2 BS is characterized by a shortened nose with a depressed nasal bridge, flat facies, and a convex upper lip. Malocclusion, underdevelopment of frontal sinus (FS), and anomalies of the cervical spine may be present. 1 Most of the individuals with the Binder phenotype have skeletal dysplasia, but the range of manifestations is wide, from mild to severe. Other associated alterations have been observed sporadically: some cases show orofacial clefting,1,3–5 dental anomalies,5,6 and mental retardation.1,6,7 A typical semilunar shape nostril was also described.3,7,8 The most important diagnostic criterion is aplasia of the anterior nasal spine, which can be observed only in the lateral skull radiograph. Patients affected by BS present a flattening of the anterior nasal spine area compared with normal patients, a thin anterior vestibular plate of alveolar bone, and usually proclined upper incisors. In normal patients, the anterior nasal spine should have a sharp aspect, and vestibular alveolar bone covering the upper incisors root should appear a bit thicker. A sporadic association with cleft palate has been recognized, but there is no reference to the prevalence of clefts among those affected. 9 While in some cases only the facial features are present, other cases display the full clinical picture. 10 Other studies on the etiology of BS have underlined the possible role of drugs involved in vitamin K deficiency during pregnancy (warfarin and phenytoin). Phenotypic characteristics were observed in the Sebitoli chimpanzee community of Kibale National Park (Uganda) such as a flattened nose, reduced nostrils, and concave midface resembling those presented in human BS. It has been proposed that such a defect could have been caused by prenatal exposure to agents that cause vitamin K deficiency, such as lithium, ethanol, warfarin, or phenytoin. 11 Other 3 chimpanzees with facial dysplasia were described in Kibale National Park, Uganda. It was considered that the possibility of genetic etiology or developmental failure was caused by prenatal exposure to teratological agents. Agent Orange, a highly teratogenic substance, was used in Sebitoli in the 1970s as an arboricide in fig trees, which is one of the main foods for chimpanzees. These chimpanzees were also exposed to chemicals used to treat the tea plantations and corn gardens around Sebitoli. 12 In this work, we present for the first time, a clinical description of orofacial anomalies in 3 related rhesus macaques held in captivity, discuss them in the environmental and management context of the breeding colony, and correlate the findings with human BS.
Case Report
The aim of this report is to describe orofacial anomalies in 3 rhesus monkeys (Macaca mulatta) of the same familial group living in captivity at an animal breeding center in Brazil. Those animals that were born between 2016 and 2017 in the same breeding group showed a lip retraction characteristic similar to a healed cleft lip with a pair of superior labial frenulum and diastema associated. They were 1 male and 2 females and the deformities were very similar. The rhesus macaques in this breeding colony live in single male groups of 25 to 30 animals in cages of 48 m2. Receive filtered, treated water ad libitum through automatic stainless steel water drinking and fed a commercial primate chow in the morning, supplemented with fruits and vegetables in the afternoon, which first were immersed in a 2% sodium hypochlorite solution for parasitological and bacteriological control. The enclosures are swept clean daily and washed with pressurized water 3 times each week; no chemical is used. Periodic control of vectors is carried out in the colony where the product RODILON SOFT BAIT Bayer® (Difetialone) has been used. Each enclosure has perches, drums, trunks of trees, swings, and environmental enrichment items (offering fruit sorbet, popcorn, tire swing, and pool on hot days). It has a covered part, which serves as a shelter or refuge containing a stainless steel feeder. The enclosures are located in a natural lighting environment with no controlled ambient temperature. Clinical and behavioral history of these animals raised of individual records containing data from birth as housing conditions, feeding, exposure to drugs, diseases, trauma, and surgeries. The breeding colony was maintained in compliance with Brazilian law, registered in Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA) under protocol number 77933 and approved by the Ethics Commission on Animal Experimentation. The 3 animals that presented the orofacial defects belong to 1 of the 18 family groups kept in the animal breeding center. During the annual clinical management of the colonies the animals were sedated with ketamine (10 mg/kg) and midazolam (0.1 mg/kg), clinically examined, photographed, and radiographed. A pedigree was drawn to visualize the degree of kinship. From each animal was collected 2 mL of blood to proceed to blood count, biochemistry tests, and cytogenetic analysis. The orofacial deformities found in the clinical evaluation of the 3 animals showed great similarity. An unaffected animal of the same generation identified as AN23 was used as normal control. AN27 is a male, presenting upper lip with 2 retraction points similar to a scar shape, without cleft palate but with a flat nose. The dentition presented diastema between the upper central incisors and double upper labial frenulum. AN28 is a female, presenting the same retraction with scar aspect of the upper lip, without cleft palate, double upper labial frenulum, flat nose, and diastema between the upper central incisors. AO6 is a female, also presenting the retraction aspect in the upper lip without cleft palate. The dentition presented diastema between the upper central incisors and fusion of the upper right lateral incisor with the canine besides the presence of a double upper labial frenulum and flat nose. The oral alterations observed in common to the 3 animals included diastema between the upper incisors, absence of teeth, and insertion in the anomalous position of molars and premolars, in addition to the duplication of the upper labial frenulum and shortening of the lower lip. By radiographic images, in all 3 macaques the nasal bridge remained underdeveloped, resulting in recessed frontal and nasal bones when compared to the normal animal (Figure 1). These affected macaques lived in a family group of 25 animals. They are all offspring of the same male with different mothers. According to family pedigree (Figure 2), they are related and belong to close generations, born in 2016 (AN27 and AN28) and 2017 (AO6). By cytogenetic evaluation, no chromosomal anomalies were observed. Blood count and biochemistry tests did not provide any noteworthy data.
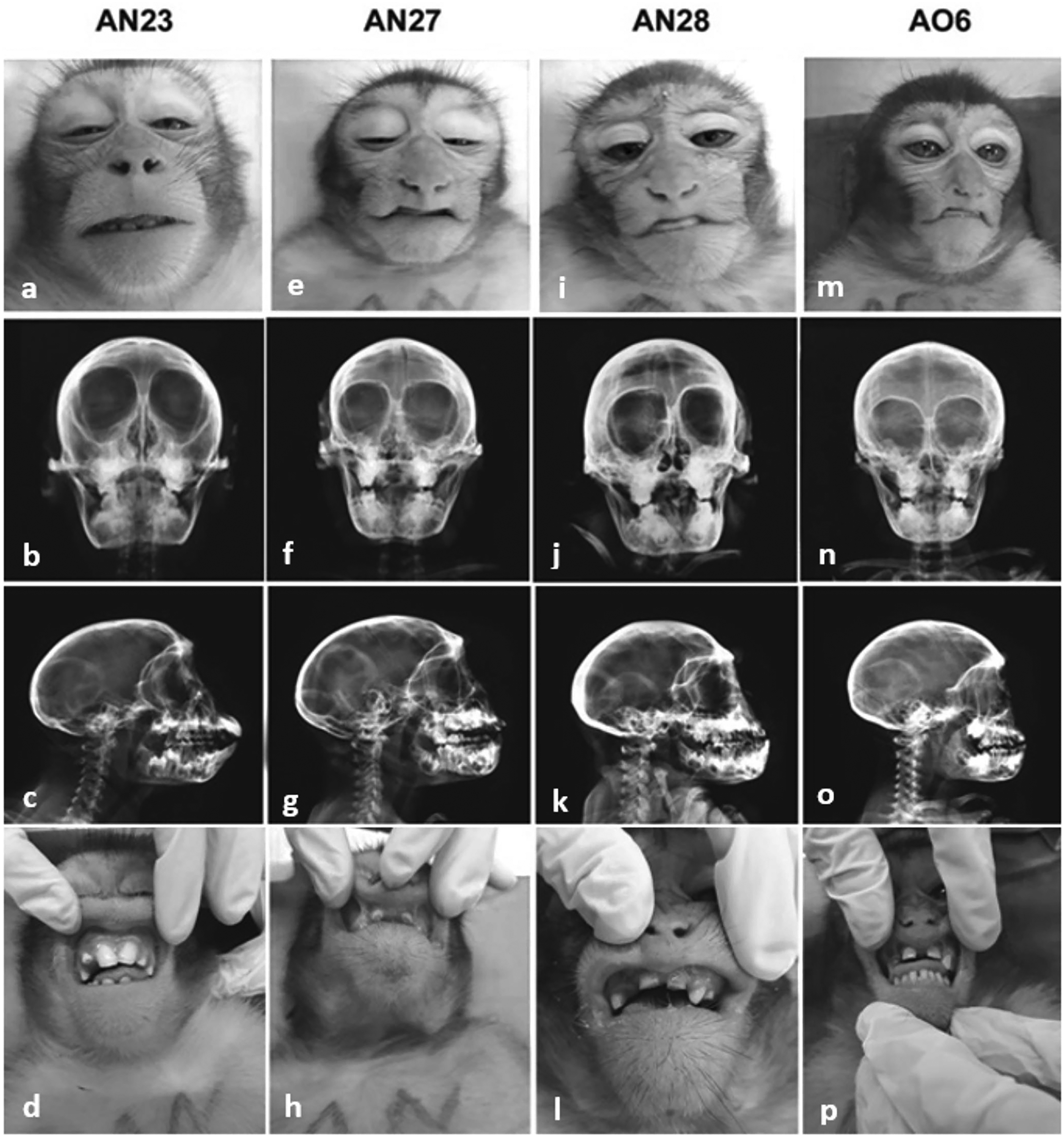
Normal rhesus monkey AN23: (a) frontal face, (b) frontal radiographic image, (c) profile radiographic image, and (d) dentition and upper labial frenum. Affected rhesus monkey AN27: (e) frontal face showing retraction aspect in upper lip, (f) frontal radiographic image showing an underdeveloped nasal bridge, (g) profile radiographic image showing hypoplasia of the anterior nasal spine, and (h) altered dentition and doubling of the upper labial frenum. Affected rhesus monkey AN28: (i) frontal face showing retraction aspect in upper lip, (j) frontal radiographic image showing an underdeveloped nasal bridge, (k) profile radiographic image showing hypoplasia of the anterior nasal spine, and (l) altered dentition and doubling of the upper labial frenum. Affected rhesus monkey AO6: (m) frontal face showing retraction aspect in upper lip, (n) frontal radiographic image showing underdeveloped nasal bridge, (o) profile radiographic image showing hypoplasia of the anterior nasal spine, and (p) altered dentition and doubling of the upper labial frenum. None of the cases showed cervical abnormalities.
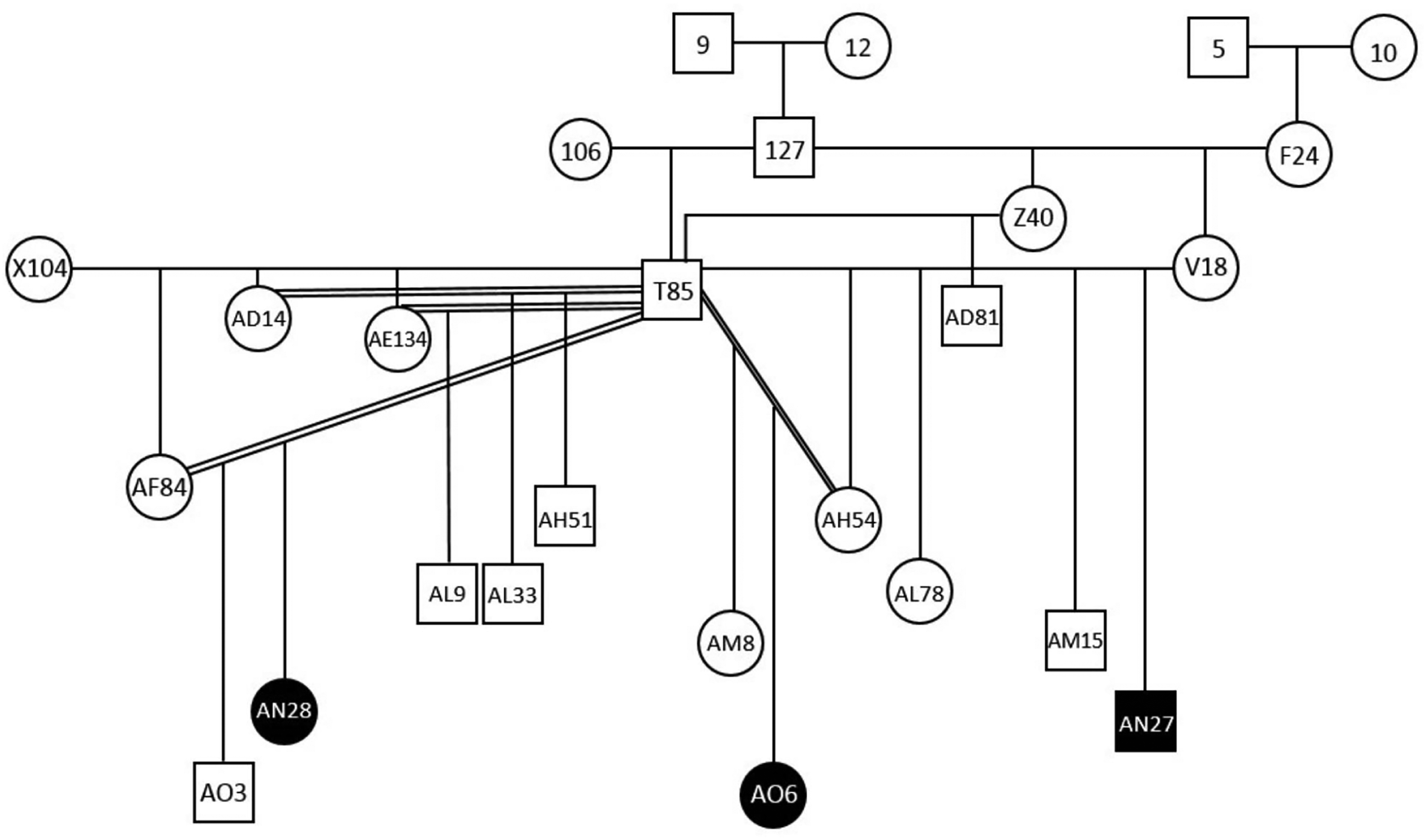
Pedigree of the rhesus macaque family group showing the kinship of affected animals.
Discussion
Human Binder phenotype is characterized by midface hypoplasia with the absence of the nasal spine leading to characteristic facial features such as flat profile and depressed nasal bridge with obtuse or flat frontonasal angle, short nose, short columella, acute nasolabial angle, perialar flattening, and tendency to a class III malocclusion.6,13 The 3 affected rhesus monkeys showed the same typical facial morphology, with a retracted maxilla, a flat vertical nose, and a convex upper lip with healed cleft lip. As described by Binder 14 and confirmed by others, 15 in the 3 monkeys, the anterior nasal spine was also hypoplastic and in a more posteroinferior position than normal which is considered a pathognomonic sign of BS. 16 The FSs are absent or hypoplastic in 40% to 50% of human patients with the Binder phenotype, 1 however, it is completely lacking in macaques (Macaca sp.), which possess only the maxillary sinus. 17 Oral alterations were observed, including diastema between the upper incisors, absence of teeth, and insertion in an anomalous position of molars and premolars, in addition to the duplication of the upper labial frenulum and shortening of the lower lip. Abnormal upper lip midline frenulum attachment is one of the most frequently described causes of diastema. 18 As can be seen in Figure 1, the normal rhesus monkey (AN23) does not have a labial frenum, however, the 3 affected animals that have a reared cleft lip also have a marked upper central lip retraction that could be related to the presented diastema. These findings suggest a correlation with human BS even because they have a great phylogenetic similarity. Data on the etiology of BS is scarce and unclear. Many theories have been presented regarding etiopathogenesis. 6 Most cases are sporadic, although hereditary factors may play a role. A genetic component could be partly responsible for this condition. A hereditary connection was found in 16% of 50 patients with BS, 19 and inheritance was interpreted as an autosomal recessive trait with incomplete penetrance. Also, a positive family history of 36% in 97 patients with BS was also reported. 20 Autosomal dominant transmission of Binder dysplasia reported in some cases is doubted by other authors. 13 Retrospective studies showed that some patients had facial similarities with mild forms of chondrodysplasia punctata (CDP), 21 and concluded that Binder syndrome was a heterogeneous condition caused by various genetic and extrinsic factors with CDP underlying most of the cases, rather than a distinct clinical entity. The close kinship of the 3 animals presented here allows the interpretation of genetic etiology. A causal relationship between the development of BS and trisomy of the pericentromeric region of human chromosome 5, suggested the role of chromosomal abnormalities 22 although no changes were observed in the monkeys’ chromosomes. Binder phenotype has also been associated with warfarin exposure, sialic acid storage disease, and deficiency in vitamin K or functional interference with its metabolism. 13 A patient with a Binder phenotype who had prenatal exposure to warfarin was reported. 23 Pregnancy histories of 3 cases of BS with prenatal exposure to warfarin, phenytoin, and alcohol were described. They suggested for the first time that BS could be caused by prenatal exposure to agents that cause vitamin K deficiency. 24 The impact of vitamin K deficiency (induced by the administration of warfarin and other therapeutic drugs such as phenytoin) in rat models was related to uncontrolled calcification in the normally uncalcified nasal septal cartilage preceding the facial malformation and decreasing the cartilage longitudinal, resulting in maxillonasal hypoplasia. 25 Matrix γ-carboxyglutamic acid protein (MGP), a γ-carboxyglutamic acid-rich, vitamin K-dependent, and apatite-binding protein, is a regulator of hypertrophic cartilage mineralization during development. 26 Developing septal cartilage is normally rich in the MGP. The periodic control of vectors carried out in the colony with the use of the product RODILON SOFT BAIT Bayer® (Difetialone), could be related to the deformities, since it has an antivitamin K activity. It could have been carried by rodents to the monkeys’ feeder and ingested by pregnant females for some period. Craniofacial abnormalities have consistently been produced in rhesus monkey (M mulatta) by administration of the synthetic corticosteroid triamcinolone acetonide at critical times during early development. 27 Both genetic and environmental involvement of the vitamin K-dependent metabolic pathway may be a major contributing cause to the Binder phenotype. 28 As molecular genetic techniques are rapidly evolving and improving, there is an increasing interest in biomodels of rare diseases. Identifying the etiology of a disease is the most important step in correctly classifying that disease. Morphological observations of nonhuman primates can provide valuable models and clues to the understanding of human disorders, in addition to being important to identify and prevent etiological factors.
Conclusions
Although there is a degree of consanguinity among the affected rhesus, the fact that they have different mothers and are restricted to a temporal range, in this case, the correlation with the Binder phenotype seems to be more subject to the action of interference with vitamin K metabolism promoted by the possible anticoagulant ingestion during pregnancy.
Footnotes
Authors’ Note
The breeding colony was maintained in compliance with Brazilian law, registered in IBAMA under protocol number 77933, and approved by the Ethics Commission on Animal Experimentation of the Oswaldo Cruz Foundation under protocol number LW5/16.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
