Abstract
Objective
To investigate the subjective risk for obstructive sleep apnea (OSA) in adolescents and young adults with isolated Robin sequence (IRS). Additionally, to investigate the association of OSA risk with respiratory signs/symptoms, and retrognathia.
Design
Prospective, observational, and cross-sectional study.
Setting
Tertiary reference hospital for the rehabilitation of craniofacial anomalies.
Participants
Adolescents and adults (n = 30) with IRS were clinically evaluated and screened through the Berlin Questionnaire (BQ) and Respiratory Symptoms Questionnaire. The maxillomandibular relationship was assessed on lateral cephalograms of those that reached skeletal maturity (n = 13). Polysomnography (PSG) was performed in a subgroup of 4 individuals.
Results
The mean age of the sample was 18.2 (±3.4) years, 17 (56.7%) were adolescents (14-19 years), and 16 were (53.3%) female, all presented a repaired cleft palate.
Clinical Parameters
Systemic arterial pressure (118.0 ± 4.1/76.3 ± 4.9 mmHg), body mass index (BMI) (20.9 ± 2.8 kg/m2), neck (33.2 ± 2.3 cm), and waist circumferences (72.0 ± 5.8 cm) were within normal ranges. A skeletal class I pattern was observed in 61.5% of the participants while a class II was seen in 15.4% of them. A high risk for OSA was detected in 16.7%, and it was associated with nasal obstruction, snoring and drowsiness, and a skeletal class II pattern (P ≤ .05). One patient presented with mild OSA (apnea–hypopnea index [AHI] = 10.1 events/hour) at the PSG exam.
Conclusions
A high risk for OSA can be observed with a moderate frequency among adolescents and young adults with IRS, especially among those who are concurrently suffering from nasal obstruction, snoring and retrognathia.
Introduction
Robin sequence is a developmental anomaly affecting a body system, especially the face, oral cavity, or teeth (ICD-11 – LA56). 1 A triad with micrognathia, glossoptosis, and upper airway obstruction characterizes it. It is characterized by micrognathia, glossoptosis, and upper airway obstruction. 2 Robin sequence is presented as an isolated form (IRS) or can occur as a part of more than 50 different known syndromes (in approximately 30% of cases), most frequently Stickler, Velocardiofacial, and Treacher Collins.3,4
Due to morphological bone alterations and retrognathia, neuromotor dysfunction of muscular structures, a more posteriorly and superiorly displaced tongue, and a marked reduction of the posterior airway space, respiratory problems are highly prevalent across the lifespan of the population with IRS.5–7 Consequently, they are also considered more prone to develop obstructive sleep apnea (OSA) than the general population.8,9 Still, previous studies reported that OSA would be associated with respiratory symptoms, especially chronic nasal obstruction,10–12 which is a chronical condition frequently detected in patients with IRS. 13
Heterogeneous severity of respiratory symptoms and OSA would be expected among these patients due to the substantial variability of IRS’ clinical spectrum. According to debatable previous evidence, they would also be age related and dictated by the catch-up growth potential of each patient's mandible.6,13 With advancing age, other predisposing factors for OSA begin to become more pronounced, including weight gain, increase in neck and waist circumferences, and also alcohol and tobacco consumption.8,9,15,16 However, the effects of these variables on the risk for OSA and its severity are incompletely understood in adults with IRS due to the lack of evidence.
Early diagnosis of OSA and predisposing factors is a matter of public health, well-being, and quality of life, as intermittent hypoxemia promotes disorders of homeostasis and high morbidity. 17 Therefore, this study aimed at the conjoint evaluation of the risk for OSA and occurrence of excessive daytime sleepiness in adolescents and young adults with IRS using validated questionnaires. Additionally, associations of a high risk for OSA with the occurrence of respiratory symptoms, and the maxillomandibular discrepancy pattern, were investigated. Hypothetically, this specific population with IRS should be at high risk for OSA and consequently present excessive daytime sleepiness; this sleep-disordered breathing should affect those with respiratory symptoms or with a retrognathic mandible.
Material and Methods
Ethical Compliance and Methodological Considerations
The [details omitted for double-anonymized peer review] Institutional Review Board approved the present study (process number [details omitted for double-anonymized peer review]). All procedures are fully compliant with the Declaration of Helsinki and its later amendments or comparable ethical standards. All the participants signed an informed consent form authorizing clinical data collection and images used for scientific purposes.
Study Design, Settings, and Sample Characteristics
A prospective and observational study was conducted at a tertiary reference hospital for the rehabilitation of craniofacial anomalies. A convenience sampling strategy was applied, and 137 patients, in routine consultation with a confirmed diagnosis of IRS were invited to participate. They included adolescents (10-19 years old) and young adults (20-29 years old), male or female, of any racial or ethnic background, and willing to participate. The exclusion criteria were syndromic Robin Sequence, history of tongue lip adhesion, previous adenoidectomy and or tonsillectomy, individuals submitted to orthognathic surgery, current use of antibiotics or anti-inflammatories for management of upper airway pathologies, and previously diagnosed physical or mental limitations that would restrain the participants’ autonomy.
Calculation of the sample size was performed considering the frequency of OSA recently detected in a pediatric population with IRS studied in the same settings where the present study was conducted. 13 From a total population of young adults with an IRS of 137 individuals, a sample size of 30 participants was estimated to predict an expected frequency of 38.78% of OSA, with a margin of error of 15% and test power of 90%.
Of the whole population (137), 96 did not respond to the invitation requesting their participation in this study, and two (2) refused to take part. Nine patients willing to participate did not meet the inclusion criteria of age.
Clinical Assessment
Sociodemographic data including sex, age, educational level, and habits (alcohol and tobacco consumption) were obtained through a structured questionnaire. One trained researcher performed the physical examination and verified: body mass index (BMI = kg/m2) and neck and waist circumferences (cm).
The criteria for adult overweight and obesity classification of the Centers for Disease Control and Prevention (CDC) were considered, which resulted in the following stratification of BMI: <18.5 = underweight, 18.5 to <25 = normal, 25 to <30 = overweight, and 30 or higher = obesity. 18 A neck circumference ≥ 36cm (♀) and ≥ 40cm (♂) and a waist circumference ≥ 80 cm (♀) and ≥ 94 cm (♂) were considered risk factors for OSA.15,16 The American College of Cardiology (ACC)/American Heart Association (AHA) blood pressure guidelines (2018) were used so values <130/80 mm Hg were considered normal. 19
Risk for OSA and Clinical Respiratory Symptoms
To predict OSA in the sample, the self-administered, adapted, and validated Portuguese version of the Berlin Questionnaire (BQ) was used.20,21 This instrument contains 10 questions stratified in 3 domains assessing: (1) snoring, (2) daytime sleepiness, (3) hypertension, and BMI history. Participants can be classified into high risk or low risk based on their responses. Persistent symptoms (> 3-4 times/week) in at least 2 questions of 2 out of the 3 categories indicate a high risk for OSA. 20
A Respiratory Symptoms Questionnaire adapted from Caouette-Laberge et al. 22 was used to evaluate respiratory distress over the day- and nighttimes. Self-perceived symptoms were assessed as dichotomic variables (No/Yes).
Cephalometric Analysis
Those who reached skeletal maturity (≥ 16 years of age for females and ≥ 17 years of age for males) 23 and had a high-quality lateral cephalogram obtained within 3 months of the survey were considered eligible for cephalometric analysis. One experienced and calibrated researcher performed all measurements twice, with a month interval between the first and second assessments, ensuring intra-observer reliability. An intra-class correlation coefficient above 0.80 was considered satisfactory and obtained for all the variables assessed.
The following variables were measured using Dolphin Imaging version 11.95 (Dolphin, Image & Management Solutions): SNA (angle formed by the intersection of sella-nasion and nasion-A lines, normative value = 82 ± 2°), SNB (angle formed by the intersection of sella-nasion and nasion-B lines, normative value = 80 ± 2°), and ANB (angle formed by the intersection of nasion-A and nasion-B lines, normative value = 2 ± 2°). An SNA measure above the normative value indicated a protruded maxilla and a retruded jawbone when reduced. An augmented SNB indicated mandibular protrusion, and its reduction, a retruded mandible. Classes II and III were observed when the ANB value measured was increased and reduced, respectively.23,24
Polysomnography
Four participants of the present study, who had a medical prescription, were submitted to overnight type 1 polysomnography (PSG) exam (EMBLA N7000, Natus Medical Inc.). A minimum of 6 h was registered, and the participants were monitored in audio and video. The following biological characteristics were assessed: cerebral activity (electroencephalogram), eye movement (electrooculogram), muscular activity of inferior and superior limbs (electromyography), respiratory parameters (airflow, nasal pressure, snoring, oxyhemoglobin saturation), and body positioning. The criteria of the AASM Manual for the Scoring of Sleep and Associated Events were adopted. 25 OSA was considered as a cessation of airflow in the presence of breathing effort for 10 or more seconds. Based on the apnea–hypopnea index (AHI), the severity of OSA was classified as mild (AHI ≥ 5), moderate (AHI ≥15 and < 30), or severe (AHI ≥ 30). The number and duration of apneas and hypopneas, the awakening index, and the AHI/hour of rapid eye movement (REM) sleep were registered. 25
Data Analysis
Sample characteristics were described as mean ± standard deviation (SD) as the continuous variables were normally distributed (Kolmogorov–Smirnov analysis). Categorical data were presented as frequencies and percentages. The unpaired two-tailed t-test and Fisher's exact test were, respectively, used to detect significant differences between means, and associations between categorical data, using high risk for OSA as the dependent variable. In sequence, associations obtained through Fisher's exact test were reassessed using a multiple regression model via ordinary least squares or through point-biserial correlation coefficient (Pearson's correlation coefficient), considering a high risk for OSA as the dependent variable. Confidence intervals (CIs) and their lower and upper limits for all analyses were set at the 95% level, and P values ≤ .05 were considered statistically significant. SPSS v.26 and Graph Pad Prism v.9.1.2 were used for data analysis.
Results
Thirty (n = 30) individuals with IRS accepted to participate in the study and responded to the survey. The mean age of the sample was 18.2 (±3.4) years, 17 (56.7%) were adolescents (aged 14-19 years), and 16 (53.3%) were female, 16 (53.3%) reported secondary level education, and 17 (56.7%) were students. All the participants had a repaired cleft palate, either complete (19) or incomplete (11). Mean values of BMI (20.9 ± 2.8 kg/m2) were within normal ranges, and 3 participants were marginally overweight (25.5 ± 0.3 kg/m2). Smoking was not reported, and 1 participant regularly consumed alcoholic beverages.
Variables stratification by age and sex (Table 1) showed that the mean ages of adolescents and adults were significantly different. In addition, the systolic pressure was higher in females than males (P ≤ .05) but remained within the reference ranges. Contrarily, male subjects had larger neck and waist circumferences than women (P ≤ .05), although the mean values of these anthropometric measures were within normal reference values.
Demographics.
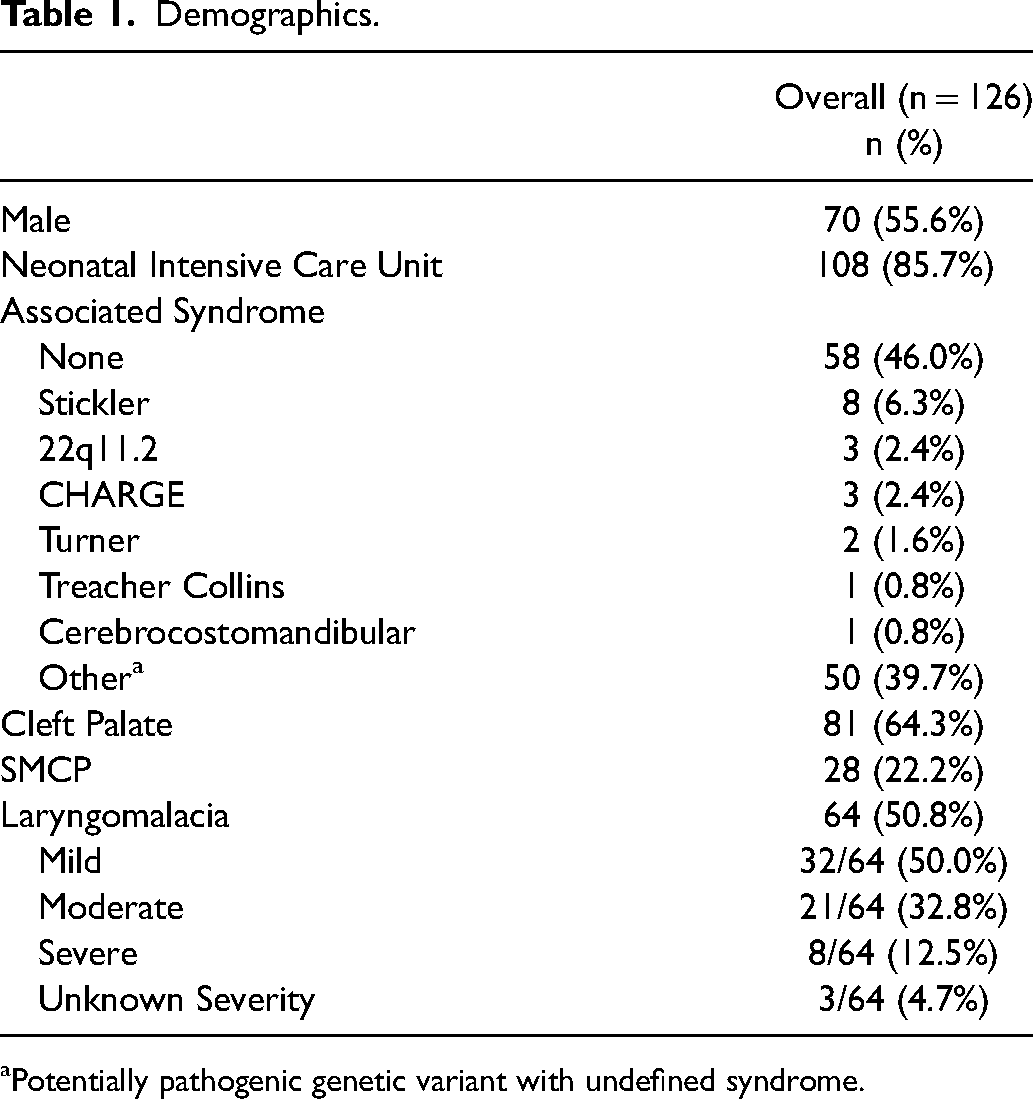
Potentially pathogenic genetic variant with undefined syndrome.
A high risk for OSA (BQ) was detected in 16.7% (5) of the sample. There were no statistically significant associations of having a high risk for OSA with demographical data—sex and age range (Table 2).
Demographics and Comorbid Airway Disorders in Patients With PRS With and Without Laryngomalacia.
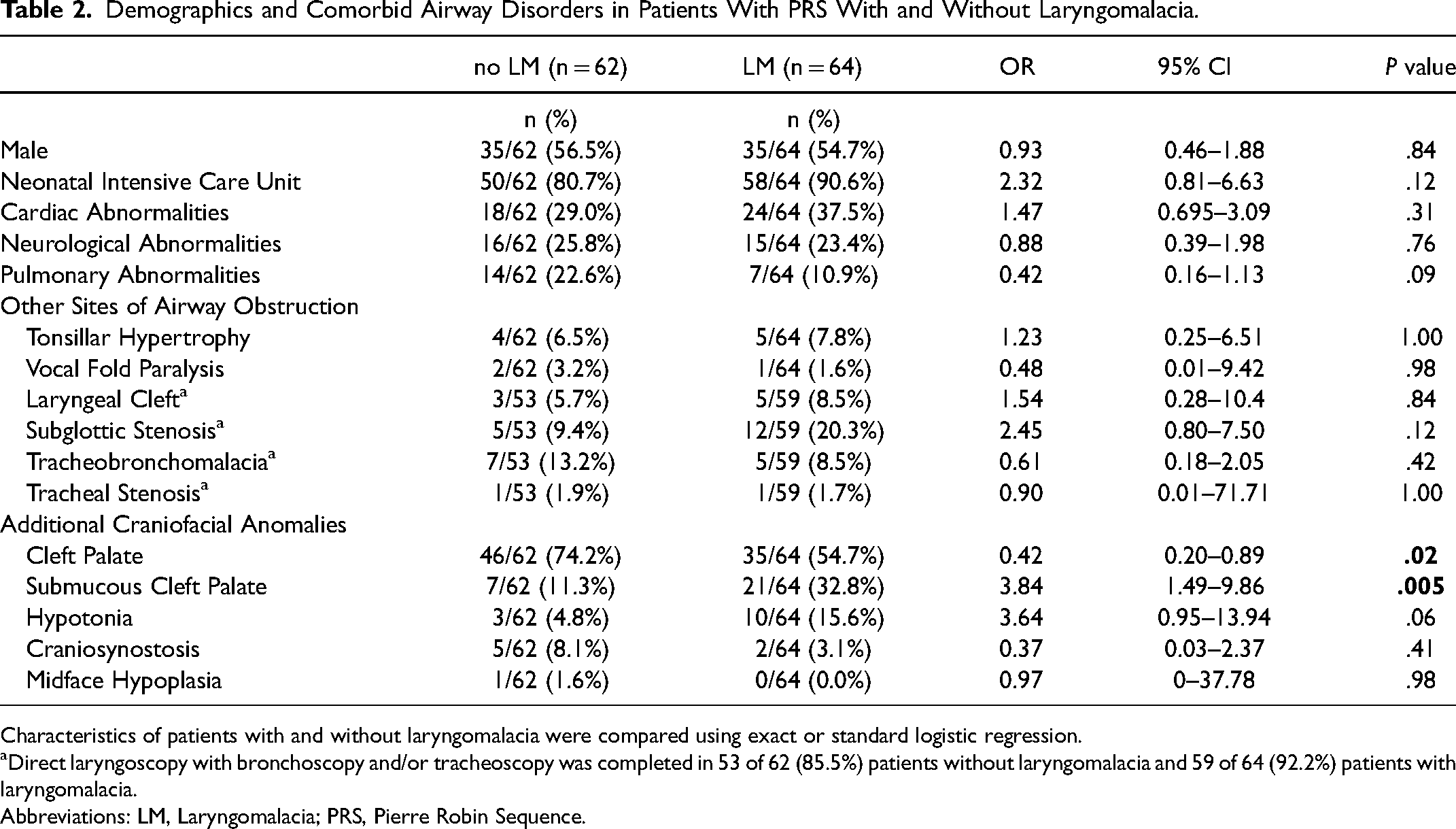
Characteristics of patients with and without laryngomalacia were compared using exact or standard logistic regression.
Direct laryngoscopy with bronchoscopy and/or tracheoscopy was completed in 53 of 62 (85.5%) patients without laryngomalacia and 59 of 64 (92.2%) patients with laryngomalacia.
Abbreviations: LM, Laryngomalacia; PRS, Pierre Robin Sequence.
The participants reported frequent nasal obstruction (33.3%) and secretion (30.0%), altered olfaction (10.0%), mouth breathing (26.7%), and regular use of topical nasal decongestants (20.0%). They also reported difficulty sleeping in dorsal decubitus position (10.0%), restless sleep (33.3%), snoring (36.7%), airway obstruction while sleeping (13.3%), drowsiness (26.7%), and constant tiredness (10.0%).
Table 3 summarizes the results of the Respiratory Symptoms Questionnaire that exhibited statistically significant associations with high risk for OSA, while no differences concerning sex or age range were detected. When reassessed through multiple regression model via ordinary least squares, only snoring remained significantly associated with a high risk for OSA in the BQ, at a statistically significant level (P ≤ .05).
Presenting Symptoms in Patients With PRS With and Without Laryngomalacia.
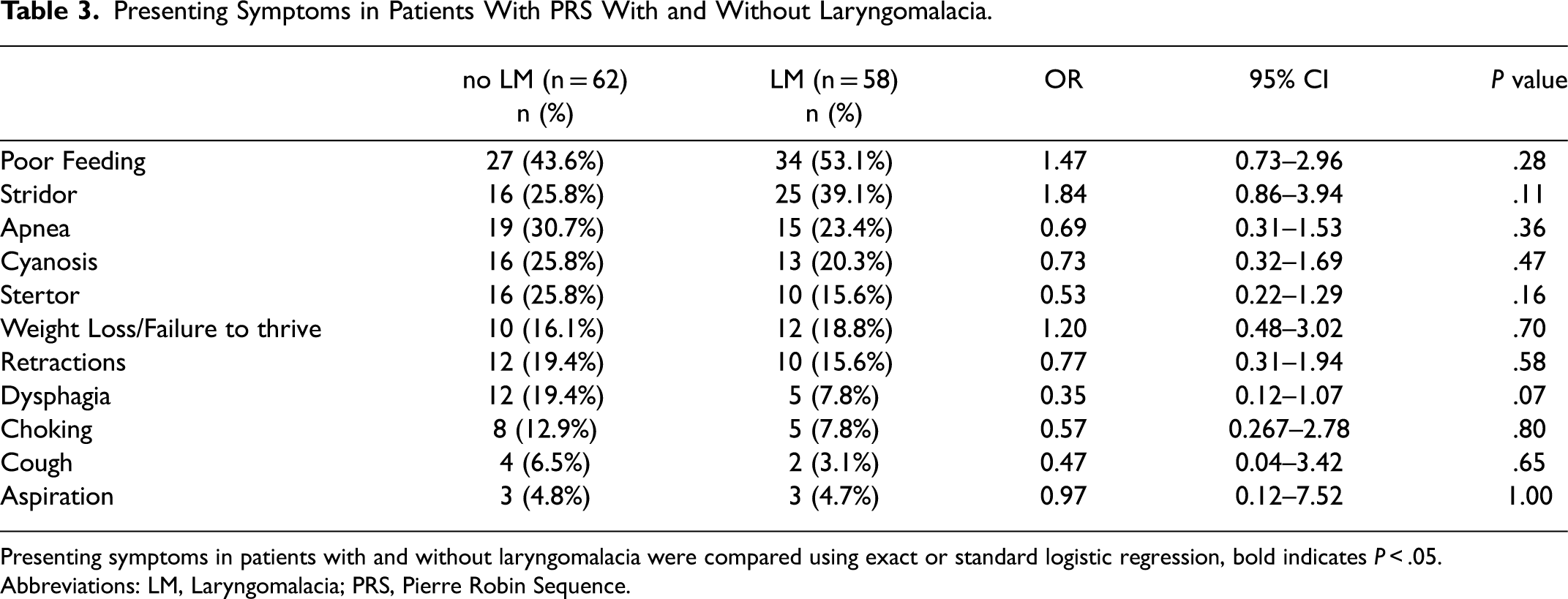
Presenting symptoms in patients with and without laryngomalacia were compared using exact or standard logistic regression, bold indicates P < .05.
Abbreviations: LM, Laryngomalacia; PRS, Pierre Robin Sequence.
Of the total sample, 56.7% (17) had available lateral cephalograms, but only 43.3% (13) of the exams belonged to participants who have already competed maxillofacial growth, 23 which were therefore evaluated. Considering the mean values of SNA, SNB, and ANB, of these 13 individuals, 61.5% (8) were class I, 15.4% class II (2), and 23.1% class III (3) (Table 4). Having a retruded mandible (angle's class II) was associated with a high risk for OSA in the BQ (Figure 1, P ≤ .05). Also, a high risk for OSA showed a strong and positive correlation with ANB angle (r = 0.85; CI = 0.55 to 0.95; P = .0001).
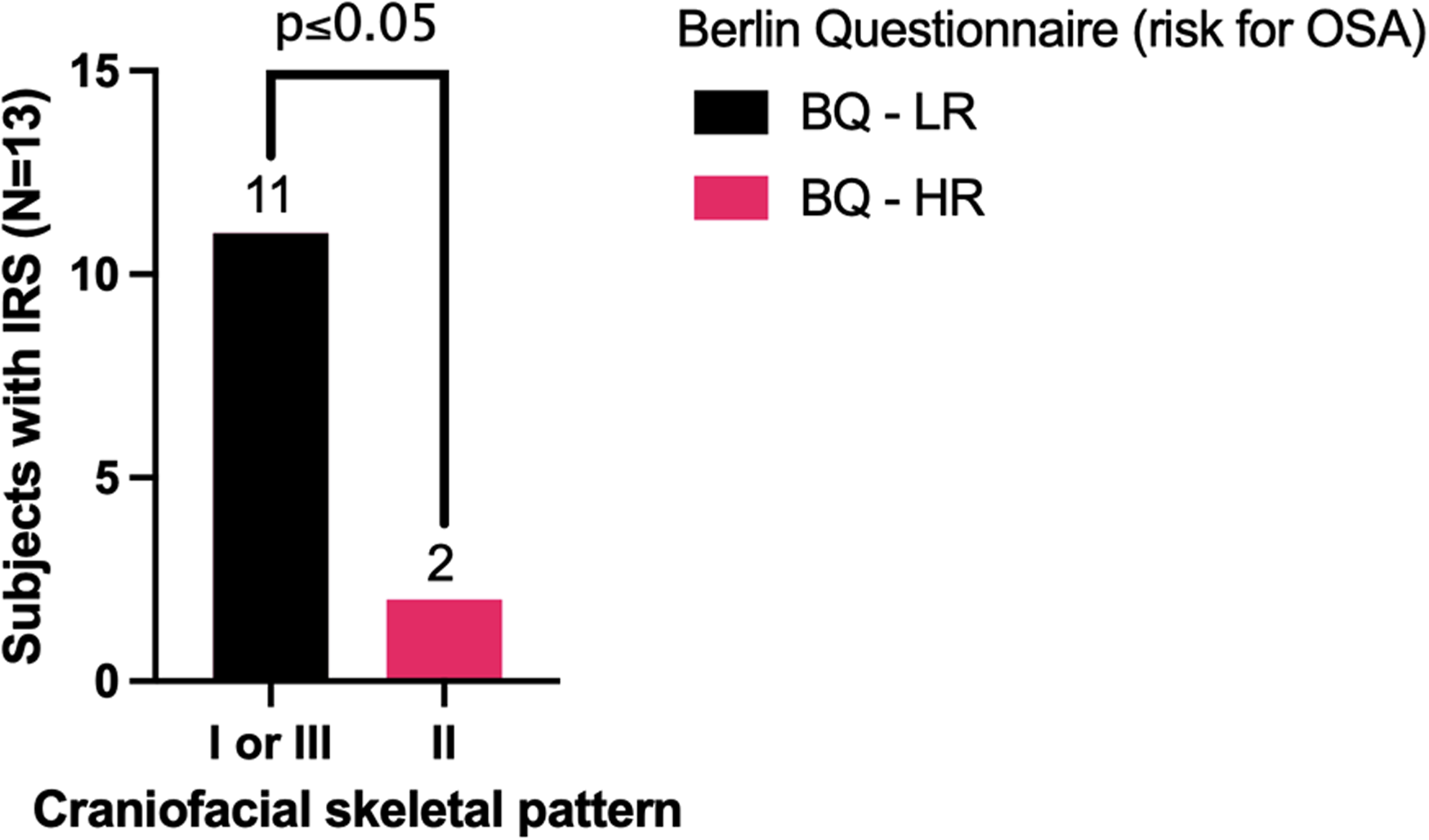
Having a high risk for obstructive sleep apnea (OSA) in the Berlin Questionnaire (BQ) was associated with having a retruded mandible (angle's class II) in the cephalometric analysis.
Surgical Interventions in Patients With PRS With and Without Laryngomalacia.
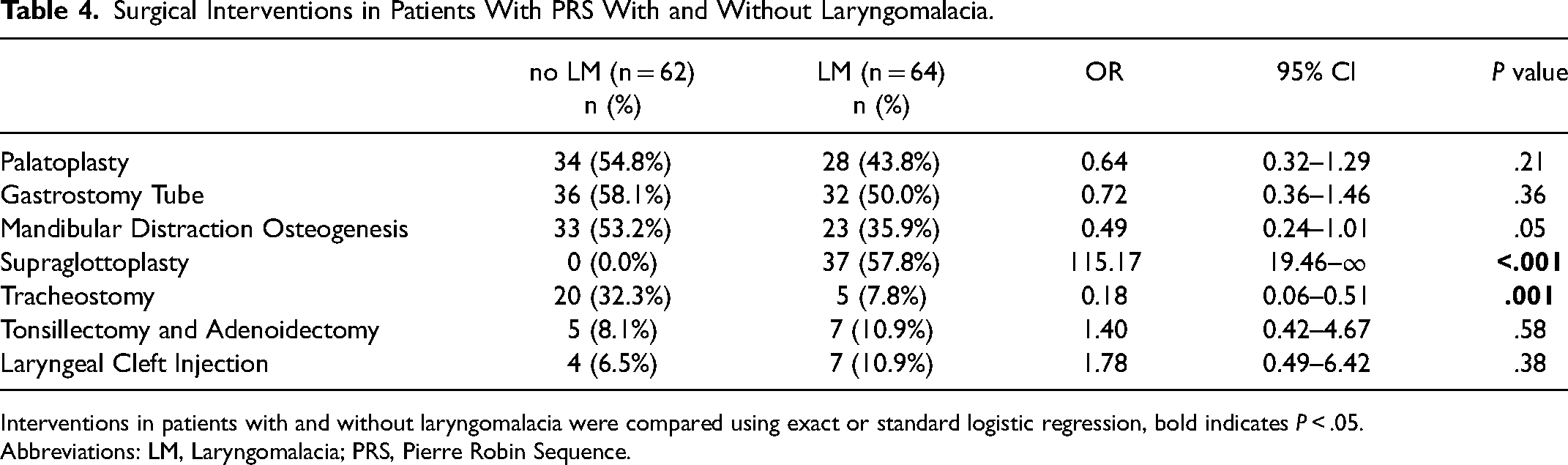
Interventions in patients with and without laryngomalacia were compared using exact or standard logistic regression, bold indicates P < .05.
Abbreviations: LM, Laryngomalacia; PRS, Pierre Robin Sequence.
As shown in Table 5, the mean value of the AHI was 3.9 (±4.2) events/hour and 1 participant presented mild OSA (AHI = 10.1 events/hour).
Pre-Intervention Polysomnography (PSG) Results in Patients With PRS With and Without Laryngomalacia.
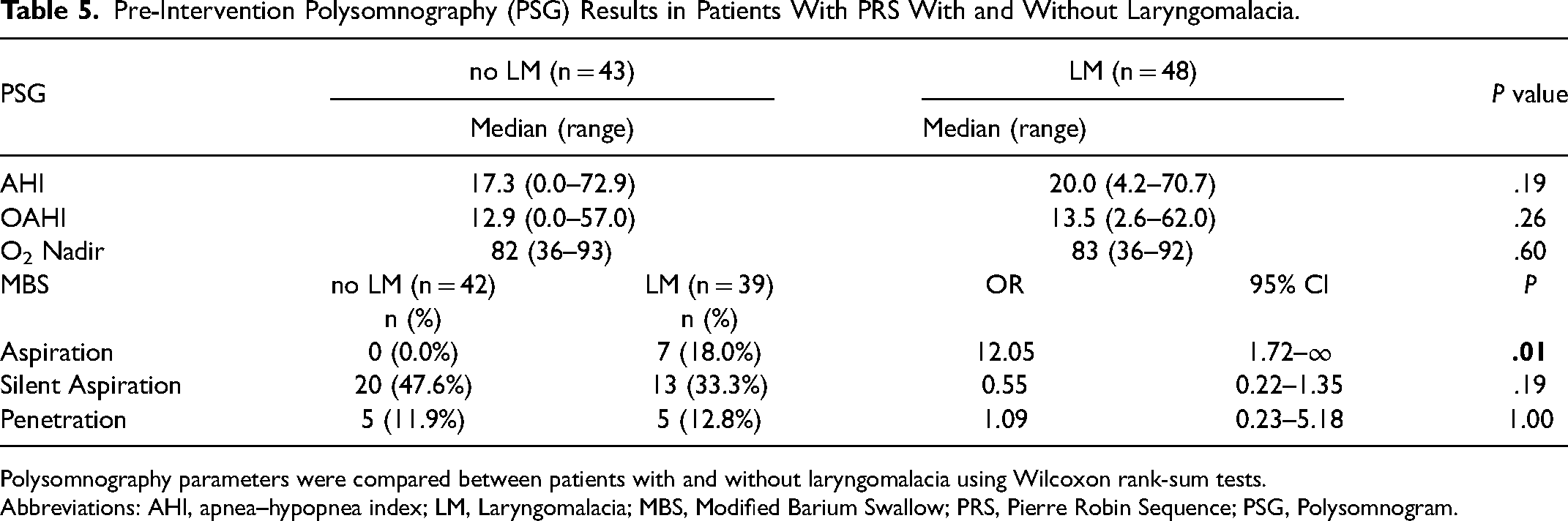
Polysomnography parameters were compared between patients with and without laryngomalacia using Wilcoxon rank-sum tests.
Abbreviations: AHI, apnea–hypopnea index; LM, Laryngomalacia; MBS, Modified Barium Swallow; PRS, Pierre Robin Sequence; PSG, Polysomnogram.
Discussion
It is well known that IRS is a significant cause of neonatal sleep-disordered breathing. 7 OSA occurred in 85% of infants submitted to 16-channel PSG, and 55% of them suffered from the severe form of the disease (obstructive AHI = 39.5 events/hour). 14 Currently, longitudinal studies in patients with IRS assessing OSA progression with age are lacking. However, it is estimated that neurodevelopmental changes in respiratory control, upper airway changes, and craniofacial growth potential should contribute to a resolution of OSA across the lifespan.5,26–28
Two aspects are seen as the studies evaluated school-aged children and young adults with IRS. First, there is a trend of OSA prevalence decrease in the first years of life. To cite an instance, a high risk for OSA was detected through the Sleep Disturbance Scale for Children in 38.78% of patients with IRS aged 6 to 12 years. 13 Second, the severity of this disorder becomes milder. As seen in the study of Spier et al., 8 7 of 8 individuals with IRS aged 16.3 (±4.6) years, evaluated through 13-channel PSG, presented a higher occurrence of sleep disturbances and apneas than controls (P ≤ .05), though they did not suffer from frank OSA. 8 As reported, only mild alterations were observed in the group with IRS than in controls (apnea + hypopnea/ hours of sleep = 1.8 ± 1.0 vs 0.54 ± 0.77; minimum oxygen saturation = 94.1 ± 1.5 vs 96.0 ± 0.8). 8 Both studies8,13 indicate a clinically significant reduction of the sleep-disordered breathing frequency and symptoms than reported among infants. 14
However, these observed trends would be spurious since different methods have been used to assess OSA prevalence across the lifespan of patients with IRS at different age ranges. Additionally, sample sizes are relatively small due to the infrequency of the anomaly, and most studies are observational. Moreover, the appearance of sleep-disordered breathing or the aggravation of OSA severity, are expected as individuals age. 9 Probably among those with IRS, despite confirmatory evidence, this trend would be expected; either due to habits modifications, unfavorable anthropometric measures, or physiological changes the current of aging. 9
In the present study, 16.7% of the sample with IRS (14-28 years old), screened for sleep-disordered breathing through the BQ, presented a high risk for OSA. The frequency found in the surveyed individuals with IRS was not compared to a control group, which is a limitation of this study. However, in subjects without craniofacial anomalies, aged 21.87 ± 1.85, and assessed through the same instrument, a high risk for OSA was detected in 11.20% of the sample. 29 In this case, the OSA frequency reported in our sample suggests that young adults with IRS might have a discrete increase (< 5%) in the subjective risk for OSA than individuals without craniofacial anomalies at a similar age range, 29 when the same assessment instrument is employed. 8
Among individuals without craniofacial anomalies, aged 20 to 29 years, living in the same geographical area as those in the present sample, mild OSA was detected through the gold-standard exam in 12.4% of males and 1.4% of female subjects. 9 In the present study, no type 1 PSG was requested solely for research purposes due to the high cost and limited access in our settings. However, 4 subjects were submitted to type 1 PSG exams for medical purposes during the period of this study, as shown in Table 5. Mean value of the AHI was 3.9 (±4.2) events/hour. Unexpectedly, despite average values of BMI, neck and waist circumferences, and low subjective OSA risk, 1 participant, presented mild OSA (AHI = 10.1 events/hour). It is underscored that considering the low number of evaluated exams, this finding should be interpreted with caution, as it might represent a selection bias, despite the subjects were erratically selected as they returned for their follow-up appointments.
Hence, when exclusively considering the objective assessment, the frequency of OSA in adolescents and adults with IRS raised from 16.67% to 25.00% (1 of 4) in this study. Therefore, almost double the frequency proposed by Tufik et al. 9 for the general population. Consequently, it would be suggested that questionnaires should be used for screening purposes only since false-negative results limits its performance. While evaluation through in-lab sleep studies or at-home sleep apnea testing seems mandatory for OSA definitive diagnosis. 25 In underdeveloped regions, though, access to these technologies can be limited. Consequently, OSA screening through a widely used and validated questionnaire is a cost-effective manner for resource allocation and in clinical and research scenarios.
Evidence is scarce concerning airway obstruction and sleep-disordered breathing in patients with IRS that reached skeletal maturity. Therefore, the strength of this study is that screening for OSA was performed in 13 individuals that have completed their maxillomandibular development process. The complete resolution of the maxillomandibular discrepancy in the present sample was predominant. According to the mandibular “catch-up” concept, there is a potential for individuals with IRS to develop a faster mandibular growth rate than that of non-IRS subjects, which would lead to a resolution of the maxillomandibular discrepancy and airway obstruction among adults. 6
In agreement with this theory, it was observed that having a class II craniofacial skeletal pattern, was associated with a high risk for OSA (P ≤ .05), which did not occur among those with a class I or III. From a clinical point of view, retrognathia favors a more posterior displaced tongue, and consequently, leads to retroglossal oropharynx constriction, impairing the airflow passage, in states of alertness and sleep.5,7,8,30 As the presented evidence suggest retrognathia seems to be related with a high risk for OSA in individuals with IRS that reached skeletal maturity, despite the majority of them seemed to achieve a resolution of the maxillomandibular discrepancy and of airway obstruction.6,26,27
In the present study, there was an association of high risk for OSA with nasal obstruction, nasal secretion, reduced sense of smell, frequent use of nasal decongestants, and mouth breathing (P ≤ .05). However, these associations were not confirmed through multiple regression model via ordinary least squares using risk for OSA as the dependent variable.
In the literature, it is a well-documented relationship between nasal obstruction and OSA, either in individuals without10,11 or with craniofacial anomalies, 12 including children with IRS. 13 Reportedly, nasal obstruction increases airway resistance, interfering with airflow dynamics and predisposing to pharyngeal collapse, aggravating OSA symptoms.10,11
A high prevalence of persistent allergic rhinitis, sinusitis, hypertrophic turbinates, and nasal septum deformities would be causal factors of respiratory symptoms in our sample. Effective management of these affections should result in long-standing improvement of nasal breathing with an additional benefit in controlling OSA severity in this population.31,32,33–35
The prevalence of snoring observed in this study was equivalent to the frequency detected in individuals over 50 years of age without craniofacial anomalies. 25 Additionally, the sample showed a high risk for OSA in those who snored (P ≤ .05). These results are alarming since this cardinal sign of obstructed airways may reduce alveolar ventilation and increase systemic and pulmonary pressure, precociously leading to OSA-related morbidities. 8
The present sample's BMI was within normal ranges, smoking and alcohol were not regular habits, and the high risk for OSA was not sex related.9,36 Therefore, no association of these variables with snoring or OSA was found. Other morphological craniofacial characteristics that might be altered in adults with IRS and would predispose them to OSA and related symptoms were not investigated. Although they should be of interest for future studies in order to elucidate OSA physiopathogenesis in adolescents and adults with IRS.8,26,28,30,37
The physiopathogenesis of OSA is associated with excessive production of reactive oxygen species and, subsequently, increased oxidative stress; blood pressure elevation; disturbance of hormonal axes via increased sympathetic activation; and endothelial dysfunction with vascular inflammation. 17 These alterations lead to severe systemic problems, including cardiovascular and metabolic diseases. Therefore, OSA screening and early diagnosis should be a matter of concern in clinical settings, including in the field of craniofacial anomalies. These patients should be screened through clinical and subjective predictors of OSA, and referred for in-lab sleep study, aiming for early definitive diagnosis and proper management. Systematic follow-up of the multidisciplinary team is recommended for this population, across their lifespan.
Conclusions
A high risk for OSA was observed with a moderate frequency among adolescents and young adults with IRS, especially among those who were concurrently suffering from drowsiness, nasal obstruction, or snoring, and those with retrognathia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (grant number Finance Code 001).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, [details omitted for double-anonymized peer review], upon reasonable request.
Ethical Approval
All procedures performed in human participants were in accordance with the ethical standards of the institutional and national research committees, and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
