Abstract
Spontaneous reossification following a cranial defect is described by only a few case reports. A 6-month-old male with epidural hematoma underwent decompressive craniotomy, subsequently complicated by scalp abscess requiring removal of the bone flap. On serial outpatient follow-up, the patient demonstrated near-complete resolution of cranial defect over the course of 18 months, thus deferring the need for future cranioplasty. Prior articles have identified this occurrence in children and young adults; however, the present case is the first to report of this phenomenon in an infant less than 1 year of age. A brief review of the literature is provided with the proposed physiologic underpinning for the spontaneous reossification observed. While prior studies propose that recranialization is mediated by contact with the dura mater and pericranium, new investigations suggest that calvarial bone repair is also mediated by stem cells from the suture mesenchyme.
Case Description
A 6-month-old previously healthy male presented to the emergency department with traumatic brain injury following a fall from his mother's arms. On examination, the patient was noted to be somnolent with a Glasgow Coma Scale score of 8. Computed tomography (CT) revealed a depressed diastatic skull fracture of the right parieto-occipital bone and large epidural hematoma along the right cerebral convexity with 6 mm of midline shift (Figure 1A).
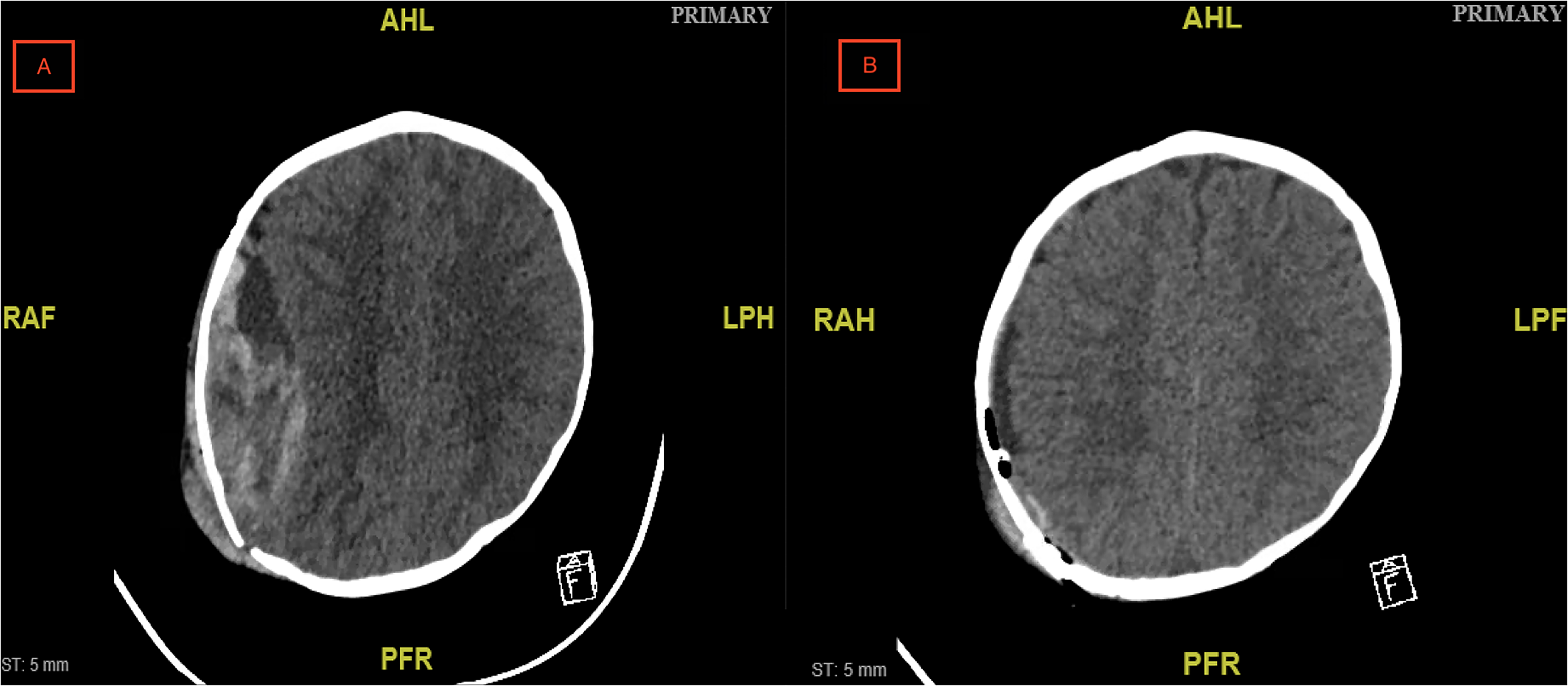
(A) Computed tomography (CT) image demonstrating right parietal epidural hematoma with 6 mm of midline shift and (B) CT image showing successful hematoma evacuation with mild residual mass effect.
The patient was taken to the operating room (OR) for emergent craniotomy and surgical evacuation of epidural hematoma. The epidural hematoma was evacuated with suction, and hemostasis of the middle meningeal artery was meticulously achieved. The bone flap was plated into an anatomic position, and a subgaleal drain was placed. Estimated blood loss was 300 mL. Postoperative CT demonstrated successful evacuation of right parietal epidural hematoma with mild residual mass effect (Figure 1B). The patient's hospital course was uncomplicated, and he was discharged 4 days postoperatively.
Four days after discharge, the infant presented back to the emergency department with edema and erythema of the right craniotomy site and a fever of 101.2° F. Magnetic resonance imaging (MRI) demonstrated a large right scalp abscess overlying the prior craniotomy without intracranial extension or involvement of the dura (Figure 2). He was taken to the OR for wound exploration. Purulent material was expressed immediately upon incision. The bone flap was removed and washout and debridement of the wound were performed. Postoperative CT documents a large residual calvarial defect (Figure 3). The patient was discharged 14 days after completion of antibiotic therapy with a referral for outpatient follow-up in the craniofacial clinic. The speciation of the intraoperative cultures showed methicillin-sensitive Staphylococcus aureus and the patient was subsequently treated with nafcillin at 200 mg/kg/day for 14 days.
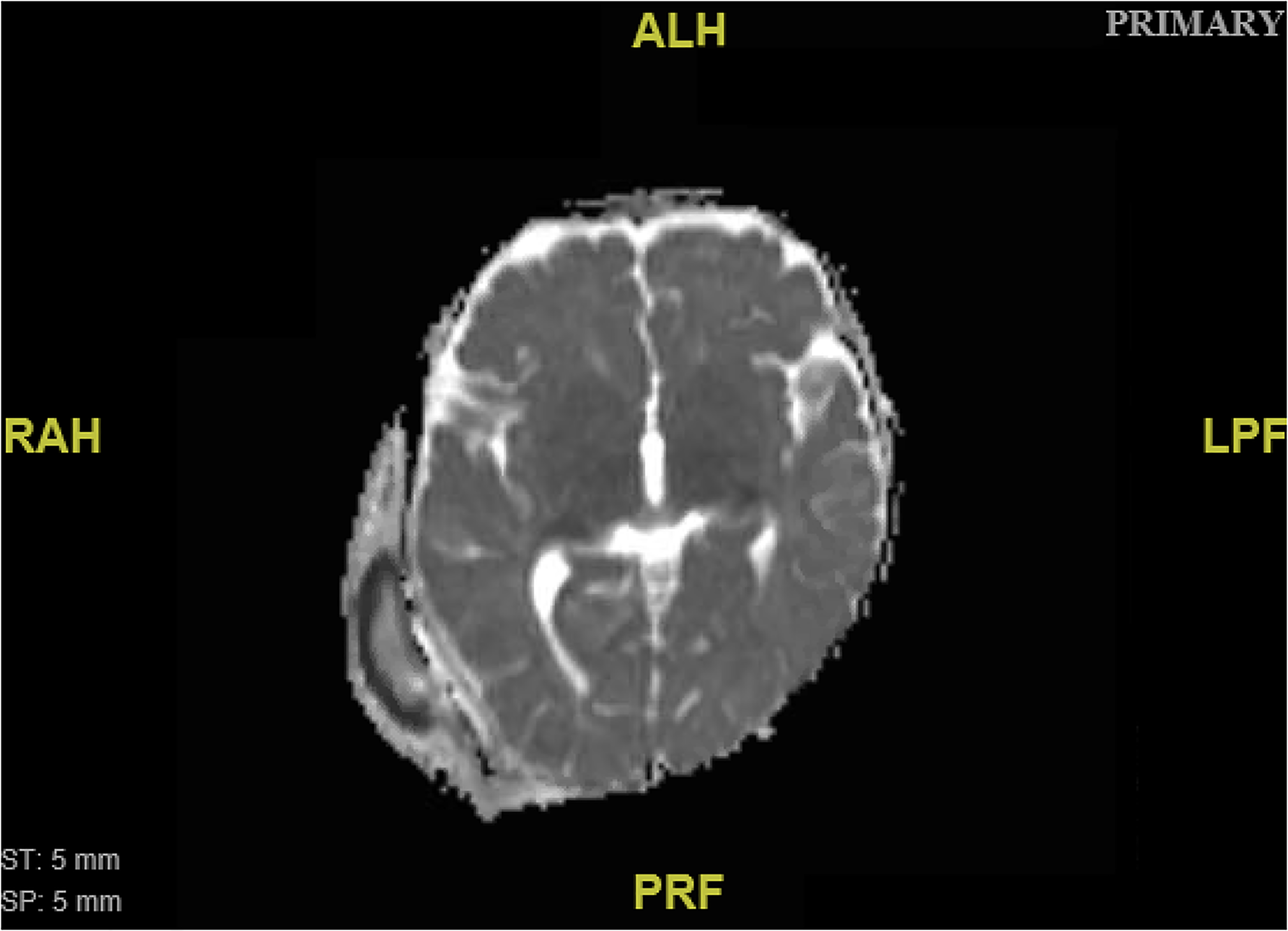
Diffusion-weighted magnetic resonance imaging (MRI) demonstrating a large right scalp abscess overlying the prior craniotomy without intracranial extension or involvement of the dura.
(A) Computed tomography (CT) image demonstrating large right cranial defect following right craniectomy for epidural wound washout and (B) three-dimensional reconstruction of postoperative CT.
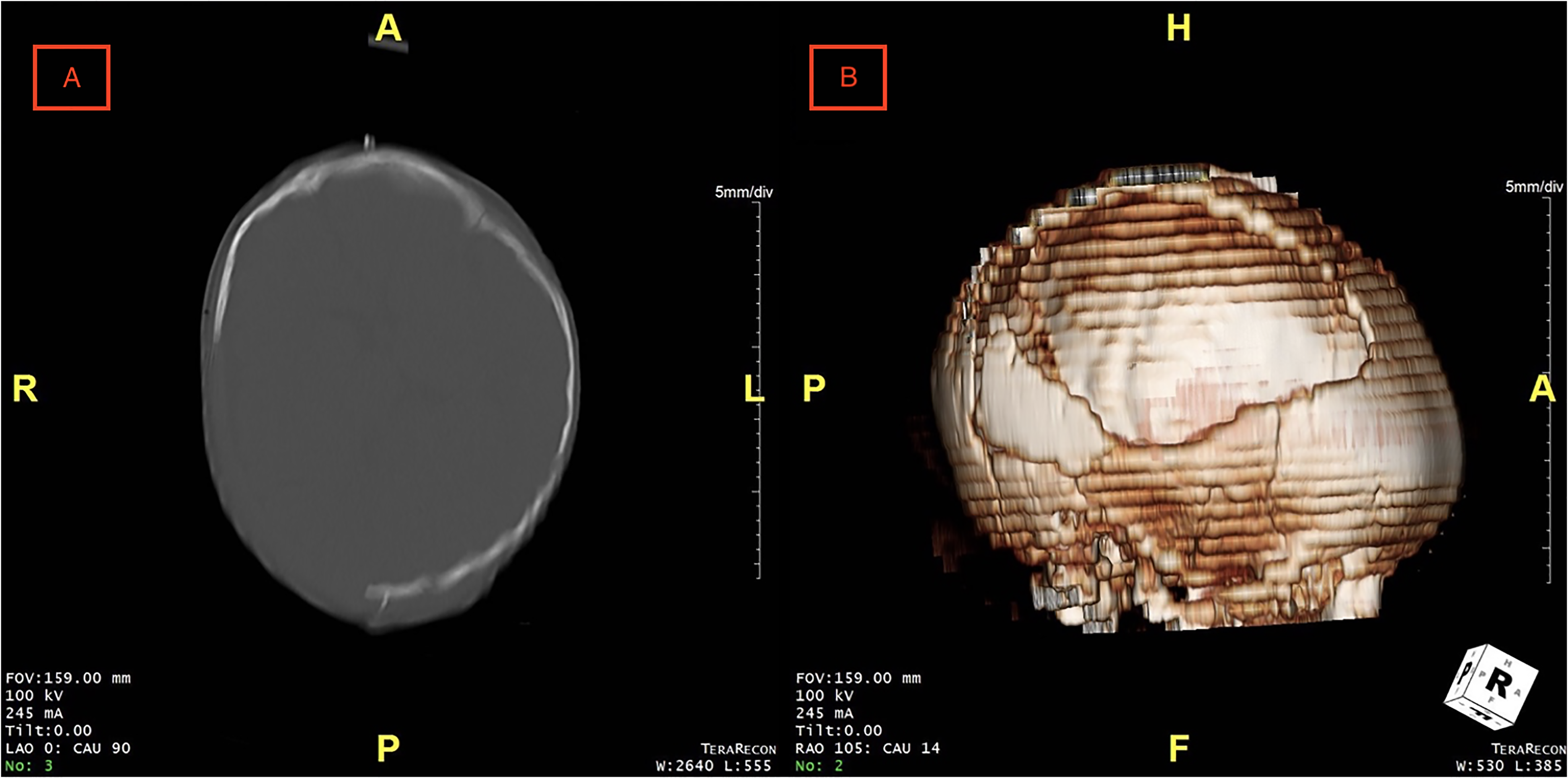
The 7-month-old patient was evaluated for cranial reconstruction 10 days after discharge. A custom implant was not thought to be a viable option due to the patient's young age and likelihood of rapid skull growth. Instead, plans for a cranioplasty with autologous bone grafting were favored at 1 year of age after skull maturation. At subsequent evaluation 4 months later, it was noted on x-ray that there was significant interval regrowth of the right anterior parietal calvarium (Figure 4). Approximately 80% of the bone had regrown with an 8 × 2 × 3.2 cm persistent irregular gap along the inferior border. Given the remarkable reossification, surgery was deferred and the patient was scheduled for continued follow-up with conservative management. On interval three-dimensional CT imaging 1 year later, the 2-year-old male showed near-complete reossification of the cranial defect (Figure 5). To date, the patient demonstrates no focal neurologic deficits and has met most childhood developmental milestones, other than the demonstration of some mild speech delay.
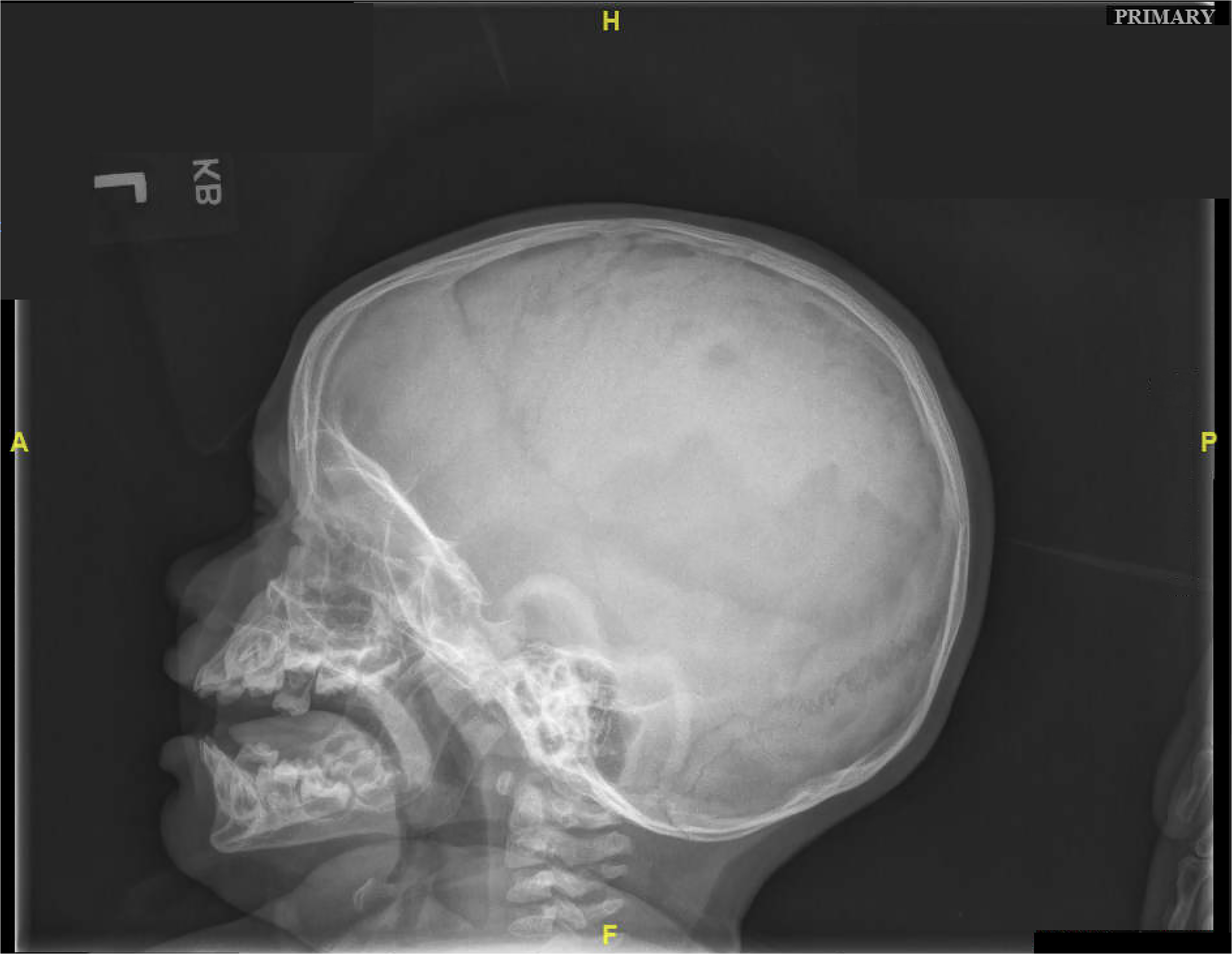
Postoperative x-ray at 4-month evaluation demonstrating significant interval regrowth of the right anterior parietal calvarium, with an 8 × 2 × 3.2 cm persistent irregular gap along the inferior border.
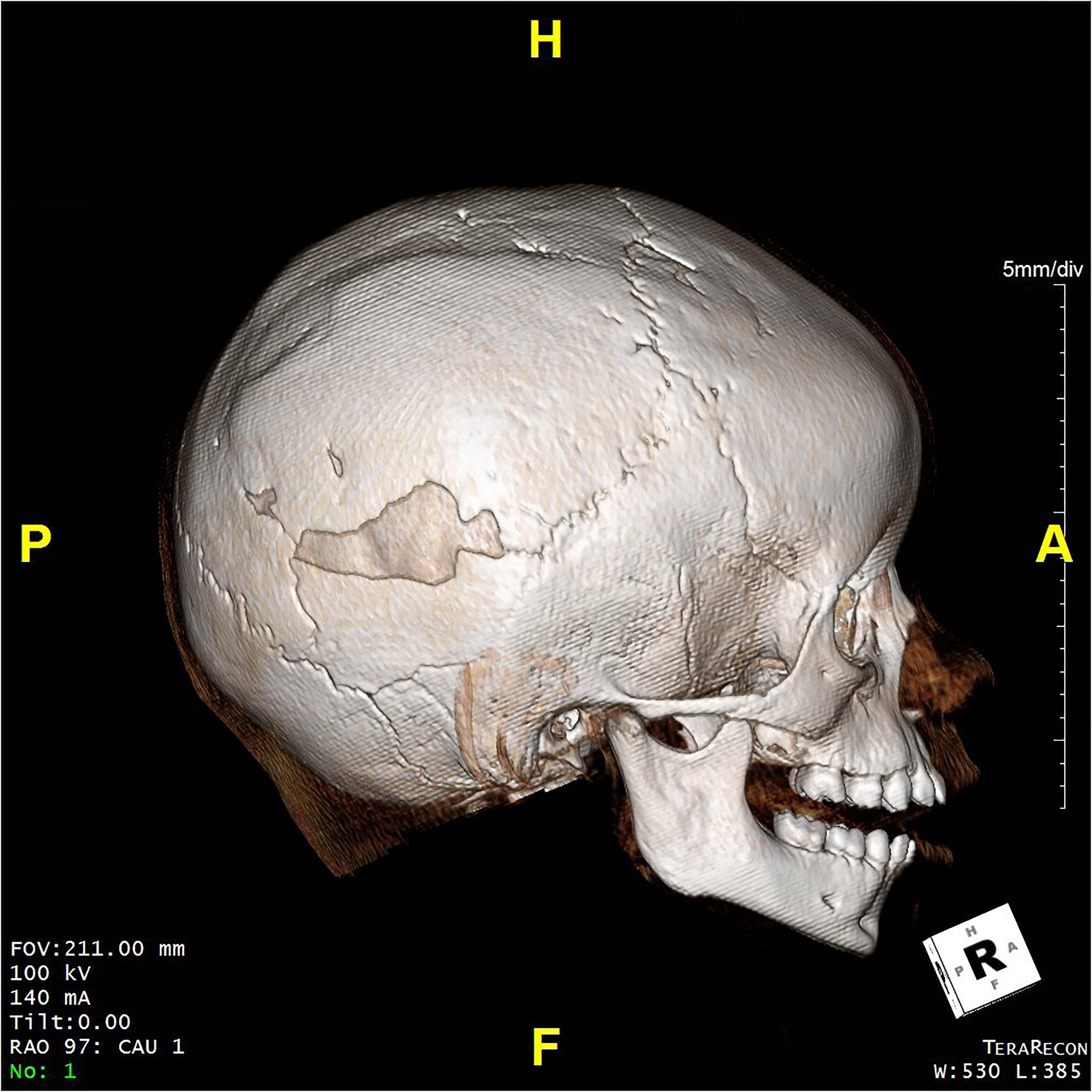
Three-dimensional reconstruction of computed tomography (CT) demonstrating near-complete reossification of skull defect 18 months after craniectomy.
Discussion
We present a case in which a 6-month-old infant demonstrated remarkable spontaneous bone regrowth following a large craniectomy, avoiding the need for reconstructive cranioplasty. We identified 9 total articles documenting this phenomenon across all age groups, 6 of which occurred in patients aged 17 and below, and the youngest so far reported in the present case.1–6 The remaining instances occurred in patients aged 18, 20, and 29, with various mechanisms of injury and patient-specific comorbidities.7–9 Generally, pediatric patients, particularly those under 1 year of age, possess strong osteogenic potential for calvarial regrowth. This is most notably remarked by craniosynostosis literature, in which spontaneous closure of surgical defects is frequently encountered.10,11 For this reason, cranioplasty in infants with large cranial defects should be judiciously considered given the possibility of spontaneous bone regeneration as in our patient, where we could also demonstrate the spontaneous reconstruction of the skull defect happening within a reasonable or acceptable time period.
There are several proposed mechanisms for spontaneous cranial reossification. For decades, the literature has suggested that calvarial bone repair is primarily mediated by progenitor cells arising from the pericranium and dura mater.12–14 In numerous studies, cells emerging from either the dura or pericranium have been shown to independently facilitate the repair of cranial defects within animal models, with dural-based reossification demonstrating greater relative efficacy.12–15 Dural contact with cranial defects enhances osteogenesis through the entirety of the developing bone graft, whereas regeneration from contact with pericranial tissue remains limited to the pericranial surface of the repair. 15 Importantly, osteogenic capacity of the dura mater appears to be inversely related to patient age, with infants demonstrating more rapid and complete bone regeneration than adults. 12
More recent studies have suggested that calvarial repair could be mediated in large part by stem cells emerging from the sutural mesenchyme. 16 The presence of skeletal stem cells in long bones has been widely studied; however, until recently, the presence of stem cells within craniofacial bones has remained uncertain. Unlike long bones, flat bones contain little marrow space to accommodate progenitor lines. As such, attention was previously directed toward the milieu of the defect, including the pericranial tissue and underlying dural tissue, to investigate calvarial regeneration. Nonetheless, 3 stem cell populations facilitating cranial reossification, termed Gli1 + , Axin2-expressing, and PRX1 + , have been discovered in the suture mesenchyme over the last decade.17–19 In an adult mouse model, Gli1 + cells were heavily recruited to the site of cranial defect following injury, and ablation of these cells led to compromised repair, craniosynostosis, and skull growth arrest. 17 Likewise, Axin2-expressing cells identified within the suture mesenchyme in mice were also shown to facilitate suture closure and promote calvarial healing by direct engraftment. 18 Most recently, PRX1 + cells were found to be essential for cranial bone regeneration in the setting of global ablation, regional suturectomy, and orthotopic suture transplantation.16,19,20 Despite these advances, additional studies are necessary to further elucidate the relationships among these cell populations, as well as the specific mechanistic underpinnings that permit calvarial bone regeneration.
While the process of spontaneous reossification remains poorly understood, this phenomenon is likely multifactorial, with dural-based and calvarial suture-based stem cells playing a significant role. Much like their dural counterparts, calvarial stem cells have been found to decrease with age. 19 Wilk et al. demonstrated in an animal model that there was a 75% reduction of PRX1 + stem cells between 8 and 32-week-old animals. 19 Gli1 + and Axin2 + populations have been shown to grow consistently from birth to 1 year of age, however, unlike their PRX1 + counterparts, reduction of these cell lines during infancy has not been well characterized. Further investigation is needed to assess the growth rate later in life.17,18 Other research demonstrates impaired geriatric mesenchymal stem cell proliferation in vitro, further bolstering the claim that these cells are more active in pediatric populations. 21 Therefore, these findings suggest that calvarial stem cells, like other stem cell populations, are more robust in pediatric patients, and may present as a reserve for flat bone regrowth following cranial injury. As such, we suspect that calvarial stem cells likely contribute to the osteogenic capabilities of flat bones in pediatric patients, functioning in tandem with osteogenic precursors from the periosteum and dura mater to facilitate wound healing following skeletal injury.
In our case, an additional factor potentially contributing to bone regeneration is the presence of surgical site abscess. Paradoxically, infectious states have been shown to facilitate bone regeneration, as bone healing depends on the presence of pro-inflammatory mediators. 22 It is proposed that inflammation is necessary early in reossification to initiate the repair cascade. Transient inflammation can therefore be beneficial for the regenerative process. If the infection persists, however, leading to a chronic inflammatory response, the regenerative process will be impaired. Mediators of bone reformation during inflammatory states include cytokines such as IL-1, IL-6, and TNF-α. Prostaglandin E2 (PGE2) has also been shown to inhibit osteoclast function, and administration of cyclooxygenase inhibitors (eg, non-steroidal anti-inflammatory drugs) which impair PGE2 synthesis have been found to impair fracture healing.23–26
This report adds to a growing body of literature documenting spontaneous recranialization in a pediatric patient. There exist, however, significant gaps in knowledge regarding the underlying mechanism and reliability of such a process. As such, implications for altering operative management and decision-making currently remain poorly understood. Nonetheless, it stands to reason that if appropriately selected patients demonstrate spontaneous reossification within a clinically reasonable timeframe, they may be able to forgo operative intervention for expectant management.
Whereas the risk of potential direct traumatic brain injury during prolonged periods without calvarial support is not trivial, we found that reossification occurred while preparing for future alloplastic reconstruction. As such, it may be reasonable to balance a reossification trial alongside anticipated alloplastic or autologous reconstruction. Furthermore, our patient demonstrated significant reossification at 4-month follow-up, indicating that a modest trial of several weeks in this specific population may be sufficient to note the initial filling of calvarial defect. Such an approach allows for the demonstration of spontaneous osteogenic potential, while still offering the current standard of care within an acceptable timeframe. We advocate that reossification trials may be most appropriate for patients 18 months of age or younger. While previous reports have documented recranialization in adolescents and even young adults, the exceedingly low likelihood of such an event in the face of significant risk discourages the prospect of a trial in older patients.
Future basic science work is critical to better elucidate the mechanistic underpinnings of spontaneous cranial reconstruction, and particularly how these mechanisms relate to age. A stronger understanding of this aspect may serve to guide patient selection for trials of spontaneous reossification. In addition, prospective clinical trials are necessary to determine the natural history of spontaneous reossification across age groups and over time and assess the reliability of such a phenomenon in patients with acquired bony defects of the skull.
This case is among the first to document the enhanced osteogenic capacity of pediatric patients following calvarial injury. Within 4 months of insult, our patient regenerated 80% of the defected area without any operative intervention. We hypothesize that reossification in our patient was mediated in part by mesenchymal stem cells from calvarial sutures. Further studies are necessary to assess the reliability of spontaneous reossification in an infant population. Should basic science and clinical evidence better describe this phenomenon, a brief trial of spontaneous reossification may be considered in appropriately selected patients alongside anticipated cranioplasty.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
