Abstract
Background
This study characterizes the potential loss of velar length in patients with a wide cleft and rescue of this loss of domain by local flap reconstruction, providing anatomic evidence in support of primary lengthening of the soft palate during palatoplasty.
Methods
A retrospective review was conducted of all patients with a cleft palate at least 10mm in width, who underwent primary palatoplasty with a buccal flap prior to 18 months of age over a 2-year period. All patients underwent primary palatoplasty with horizontal transection of the nasal mucosa, which was performed after nasal mucosa repair, but prior to muscular reconstruction. The resulting palatal lengthening was measured and the mucosal defect was reconstructed with a buccal flap.
Results
Of the 22 patients included, 3 (13.6%) had a history of Pierre Robin sequence, and 5 (22.7%) had an associated syndrome. No patients had a Veau I cleft, 7 (31.8%) had a Veau II, 12 (54.5%) had a Veau III, and 3 had (13.6%) a Veau IV cleft. All patients had a right buccal flap during primary palatoplasty. The mean cleft width at the posterior nasal spine was 10.6 ± 2.82mm, and mean lengthening of the velum after horizontal transection of the nasal mucosa closure was 10.5 ± 2.23mm. There were 2 (9.1%) fistulas, 1 (4.5%) wound dehiscence, 1 (4.5%) 30-day readmission, and no bleeding complications.
Conclusions
Patients with a wide cleft palate have a potential loss of 1cm velar length. The buccal flap can rescue the loss of domain in palatal length, and potentially improve palatal excursion.
Keywords
Introduction
Principles of primary palatoplasty entail closure of the oronasal fistula and anatomic reconstruction of the levator sling. Despite the application of these principles, there is a known incidence of velopharyngeal insufficiency (VPI), which occurs in approximately 6% to 8% of patients with a cleft palate.1–4 Yet, the rates of reported VPI vary widely in the literature, and up to approximately 30% of patients having undergone cleft palate repair may require secondary surgery.5–7 In particular, wide cleft palates (10 mm or greater) have a known increased incidence of VPI, potentially challenging contemporary principles of primary palatoplasty techniques.4, 8–10
With regards to the closure of the wide cleft palate, the senior author has made 2 observations. First, complete mucosal reconstruction of the soft palate is achievable using a radical intravelar veloplasty (IVV); however, the soft nasal mucosa closure tends to be tight and can lack the capacity for normal excursion. Second, in patients who later present with VPI after undergoing primary palatoplasty, the problem tends to be a deficiency in palatal length rather than poor muscle position. These observations support the concept popularized by Dr Robert Mann of adding mucosal tissue to the nasal mucosa at the time of primary cleft palate repair.
In 1997, Dr Mann described the addition of bilateral buccal flaps to the Furlow double opposing Z-plasty. 11 One of the buccal flaps would be used to close the nasal lining of the soft palate in situations where complete nasal mucosa closure is not possible. The second buccal flap would be used to close the hard palate reconstruction in a manner that would limit palatal dissection, theoretically preserving facial growth. Dr Mann advocated that the addition of buccal flaps to the soft palate repair facilitated easier interdigitation of Z-plasty muscle and mucosal flaps across clefts of nearly all widths.11, 12
The concept that speech results in patients with a wide cleft would benefit from addition of mucosa to the soft palate nasal lining during primary palatoplasty is based on the assumption that there is a paucity of soft palate nasal mucosa. However, this potential loss of domain has yet to be quantified. This anatomic study characterizes this potential loss of domain, and specifically this potential loss of velar length of the nasal soft palate mucosa in patients with a wide cleft palate.
Methods
A retrospective review of all patients who underwent a primary cleft palate repair with a buccal flap prior to 18 months of age by the senior author over a 2-year period from 2019 to 2021 was performed. Inclusion criteria were defined as patients with cleft palate at least 10 mm in width at the area of the posterior nasal spine. All patients who met inclusion criteria underwent primary soft palate repair using the radical IVV technique13, 14 and with horizontal transection of the nasal mucosa during palatoplasty. This horizontal transection was performed after nasal mucosa repair, but prior to muscular reconstruction and occurred just distal to the posterior border of the hard palate (Figure 1A). Specifically, at the junction of the hard and soft palate with a 2mm cuff with nasal mucosa to sew too that was attached to the hard palate. The horizontal width of the cleft palate at the posterior nasal spine and the resulting mucosal defect created by the transection of the nasal mucosa at the soft palate/hard palate junction was measured with a caliper (Figure 1B). The mucosal defect was then reconstructed with a buccal flap (Figure 1). Patient demographics, intraoperative palatal measurements, and postoperative outcomes were analyzed.
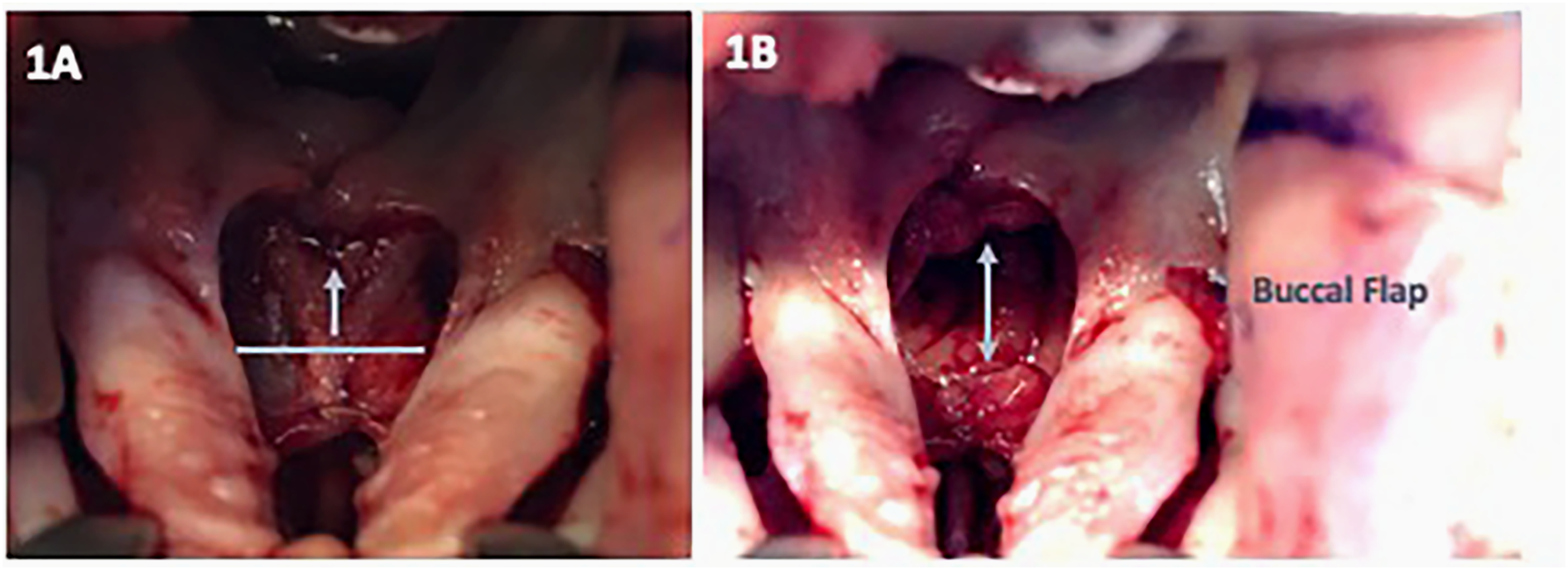
Patients underwent primary cleft palate repair with horizontal transection of the nasal mucosa during palatoplasty. Notably, this transection was performed after nasal mucosa repair, but prior to muscular reconstruction (A). The resulting mucosal defect was measured with a caliper and reconstructed with a buccal flap (B).
The surgical technique for buccal flap dissection was previously described by Mann.11, 12 Following identification of Stenson's duct, the right buccal flap incisions are marked in blue; the width of the buccal flap is approximately 1 to 1.5 cm, and the length of the buccal flap is predicated upon the distance from the oral commissure to the retromolar area, which is approximately 4 to 5 cm. The right flap is sharply elevated after local anesthesia with 1% lidocaine with 1:100,000 epinephrine is administered. Specifically, the buccal flap is mobilized using a 15-blade scalpel for the initial mucosal incision, and subsequently, tenotomy scissors are used with meticulous attention to ensure a uniform thickness to the flap and to maintain the submucosal vascularity of the flap as it is elevated distally to proximally. Then, the flap is transposed behind the greater palatine vessel into the defect created by the horizontal transection of the nasal mucosa during palatoplasty. The buccal flap is transposed and inset into the defect of the palate using interrupted 3-0 chromic sutures. The mucosa of the buccal flap is inset facing the nasal side of the cleft repair (Figure 2). The donor site for the buccal flap is closed in the V-Y fashion using interrupted 3-0 chromic sutures.
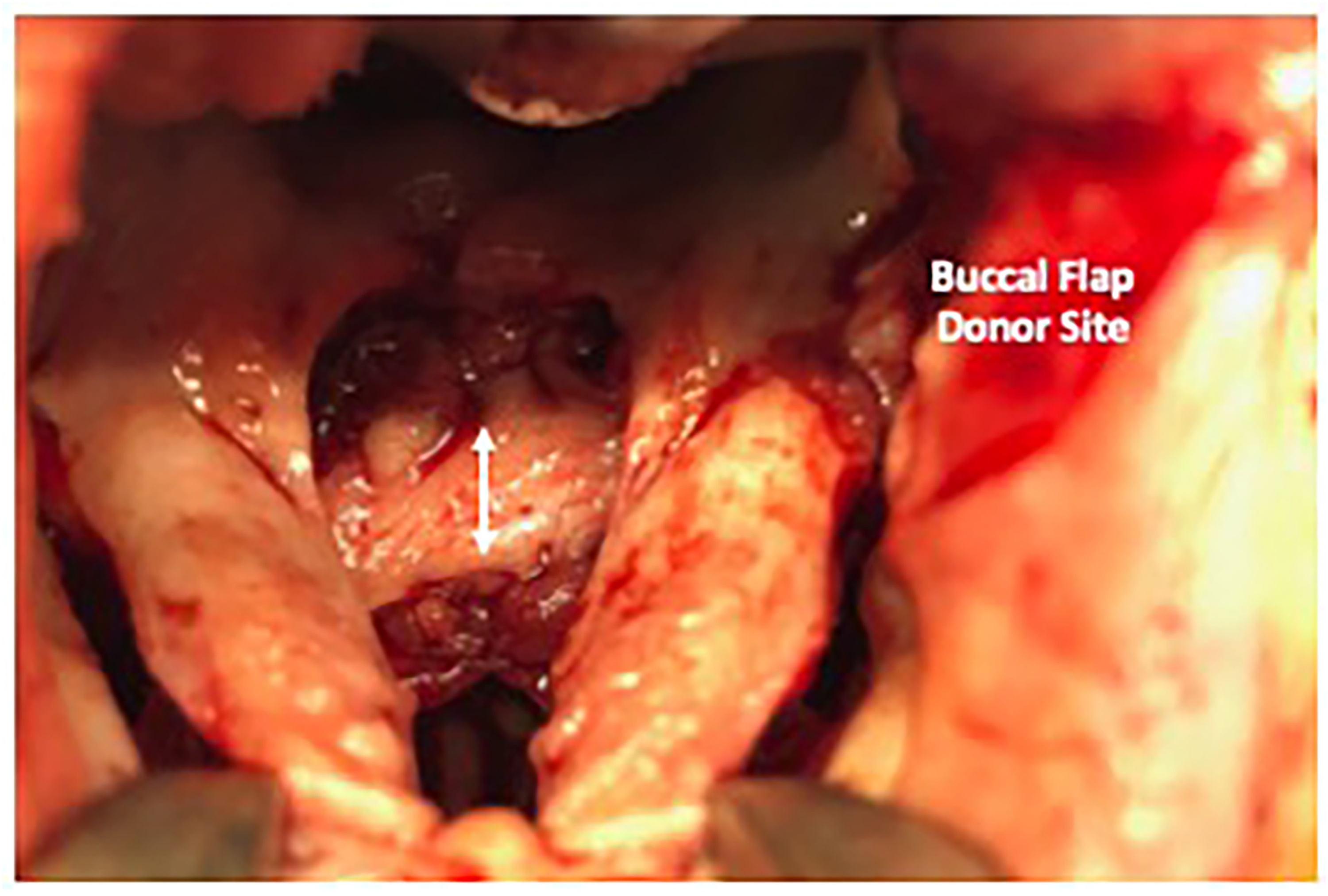
Specifically, the buccal flap is mobilized using a 15-blade scalpel for the initial mucosal incision, and subsequently, tenotomy scissors are used with meticulous attention to ensure a uniform thickness to the flap and to maintain the submucosal vascularity of the flap as it is elevated distally to proximally. The buccal flap is transposed behind the greater palatine vessel into the defect created by the horizontal transection of the nasal mucosa during palatoplasty. The buccal flap donor site is then closed primarily with no tension.
Results
Twenty-two patients met the inclusion criteria for this anatomic study. Fifty-nine percent of patients were female and 41% male. The mean age at surgery was 10.68 ± 1.04 months, mean gestational age at birth was 38.14 ± 1.75 weeks, and mean weight at surgery was 8.75 ± 1.22 kg. Two (9.1%) of the patients had a family history of cleft lip and/or palate. Three (13.6%) had a history of Pierre Robin sequence, and 5 (22.7%) had an associated cleft syndrome. Thirteen patients (59.1%) had a history of nasoalveolar molding, and 15 (68.2%) had a cleft lip and palate. No patients had a Veau I cleft, 7 (31.8%) had a Veau II, 12 (54.5%) had a Veau III, and 3 (13.6%) had a Veau IV cleft (Table 1, Figure 3).
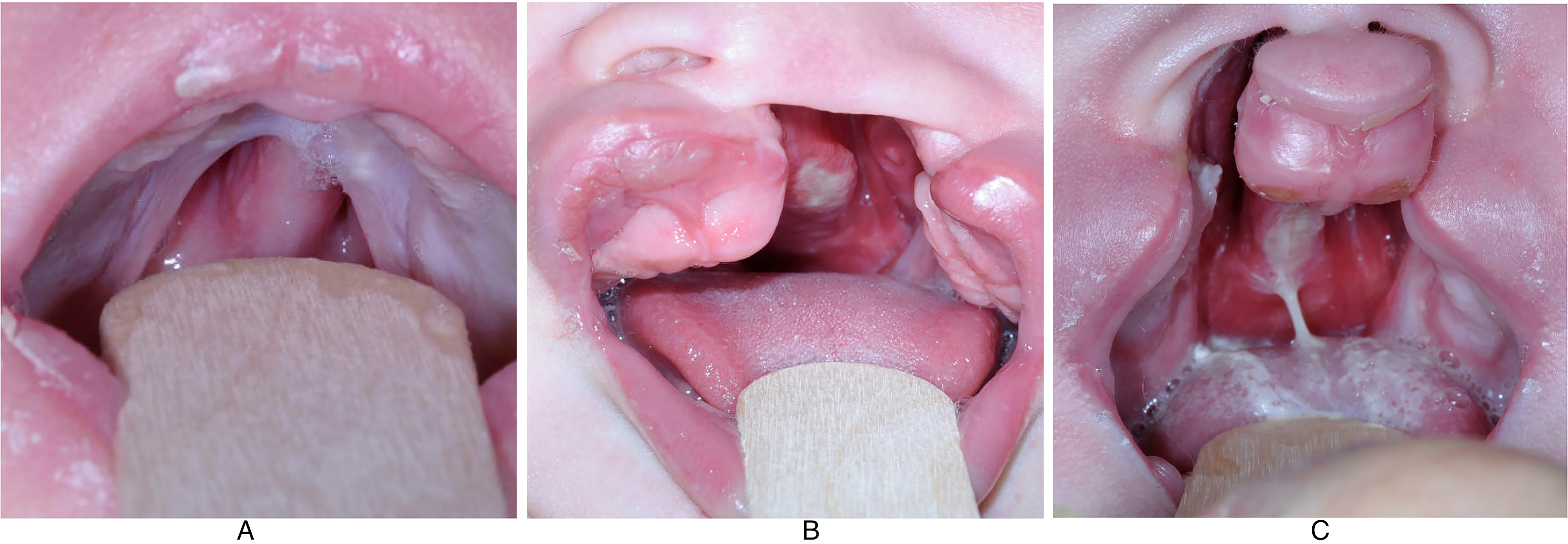
Representative clinical photograph of one of the patients with a Veau II cleft (A), Veau III cleft (B), and Veau IV cleft (C), respectively.
Cleft Palate Repair With Buccal Flap: Patient Demographics.
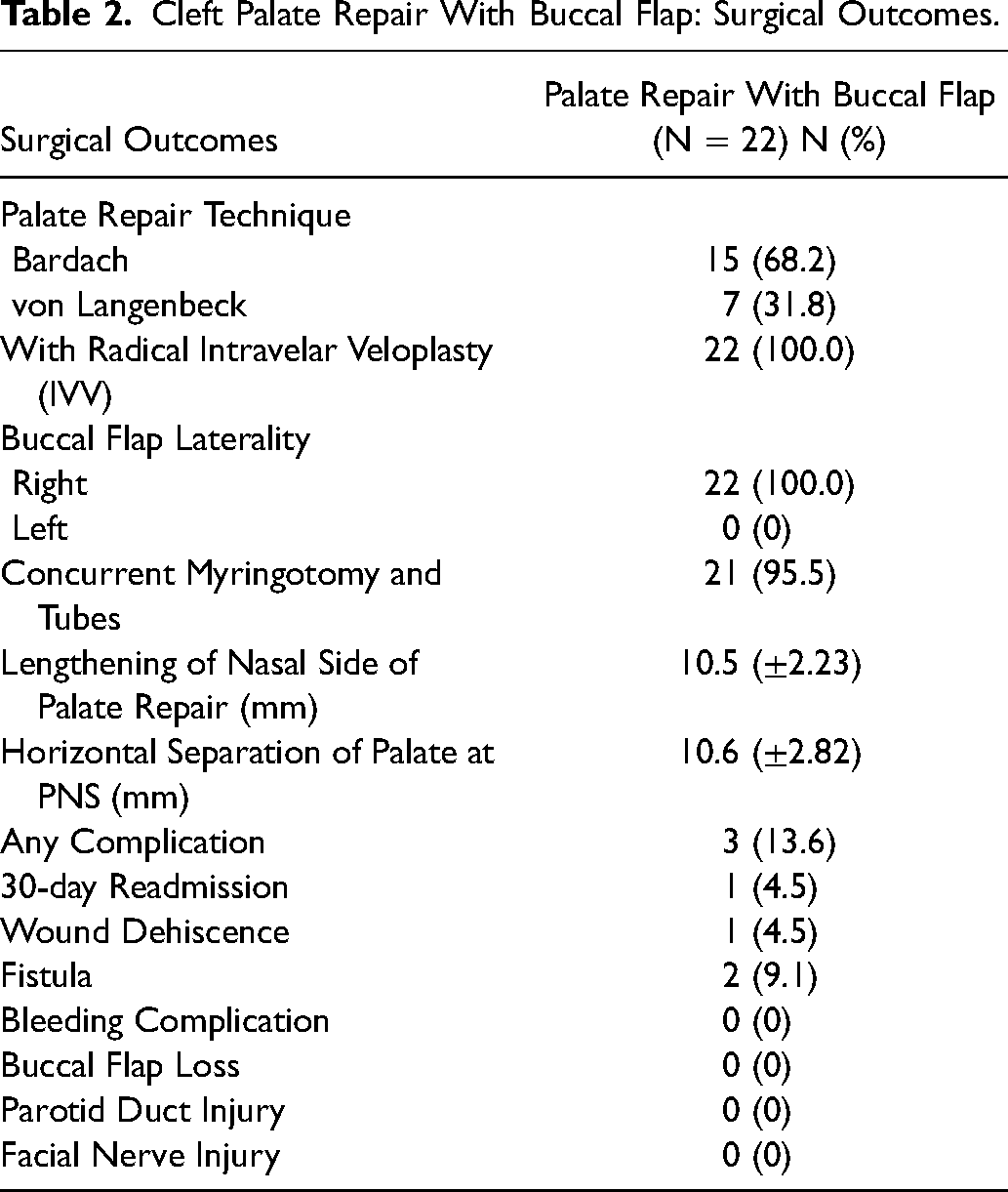
Regarding the hard palate repair techniques employed, 15 (68.2%) had a Bardach 2-flap palatoplasty, and 7 (31.8%) had a con Langenbeck. The von Langenbeck technique was used for Veau II clefts, and the Bardach technique was used for Veau III and IV clefts. All study patients underwent radical IVV repair of the soft palate. All 22 (100%) patients had a right buccal flap during primary palatoplasty. Twenty-one patients (95.5%) of patients had concurrent myringotomy and tubes. The mean cleft width or horizontal separation of the palate at the posterior nasal spine was 10.6 ± 2.82 mm. After reconstruction of the nasal lining of the soft palate but prior to reconstruction of the levator sling, horizontal transection of the nasal mucosa at the junction between the soft and hard palate resulting in an average of 10.5 ± 2.23 mm lengthening of the soft palate (Table 2, Table 3). In terms of operative time, the unilateral buccal flap adds a mean of 44 min to the average surgical time for primary palatoplasty cases in patients with a wide cleft palate, which we compared to a control cohort of 22 patients without buccal flaps (Table 4).
Cleft Palate Repair With Buccal Flap: Surgical Outcomes.
Cleft Palate Repair With Buccal Flap: Outcomes by Veau Classification.

Data are represented as mean (±std. dev.).
Cleft Palate Repair With Buccal Flap: Operative Surgical Time.
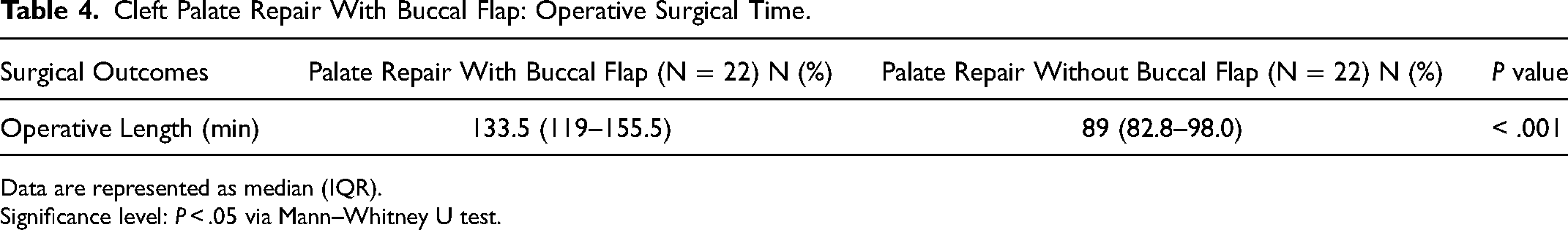
Data are represented as median (IQR).
Significance level: P < .05 via Mann–Whitney U test.
The complication rate was 13.6%: 2 (9.1%) fistulas and 1 (4.5%) superficial wound dehiscence, which was successfully treated with observation. There were no bleeding complications and no episodes of airway obstruction. One patient (4.5%) had a 30-day readmission for an unrelated condition (RSV bronchiolitis) (Table 2). There were no buccal flap losses, no parotid duct injuries, and no facial nerve injuries.
Discussion
This is an anatomic study on the potential loss of soft palate domain in patients with wide clefts. More specifically, this study calls attention to a vertical deficiency of velar nasal mucosa. The deficiency of soft palate nasal mucosa is a limitation that has been previously reported.11, 12, 15, 16 The challenges of complete nasal mucosa reconstruction in patients undergoing Furlow double opposing Z-plasty has prompted the use of Alloderm to close the mucosal defect remaining after nasal flap transposition.17, 18 Mann subsequently reported the use of bilateral buccal flaps in conjunction with Furlow double opposing Z-plasty.11, 12 The purpose of 1 buccal flap was to fill the remaining mucosal defect created after transposition of the nasal flaps during soft palate repair. The second buccal flap was used to repair the hard palate defect. The nasal mucosa (velar) buccal flap was posited to increase palatal length, therefore optimizing velopharyngeal closure and improving speech outcomes.11, 12 Although these studies highlight the deficiency of soft palate nasal mucosa more commonly seen in patients with wider palatal clefts, no study has quantified the potential loss of domain or, more specifically, the potential loss of velar length in patients with a cleft palate.
In this anatomic study, the mean cleft width or horizontal separation of the palate at the posterior nasal spine was 10.6 ± 2.82 mm. After straight-line repair of the nasal mucosa but prior to reconstruction of the levator sling, horizontal transection of the nasal mucosa at the junction between the hard and soft palate resulted in a mean velar lengthening of 10.5 ± 2.23 mm. These results suggest that patients with a wide cleft palate have a potential loss of vertical velar length of approximately 1 cm. Considering that patients with a wide palatal cleft are predisposed to developing VPI,4, 8–10, 19, 20 these data provide supportive evidence that acute palatal lengthening during primary palatoplasty should be considered for this patient population. We believe that a buccal flap technique is well suited for this type of repair, although other types of repair techniques can be considered, such as the Furlow double-opposing Z-plasty. The overall complication rate for using a buccal flap during primary palatoplasty in our study was 13.6%, including 2 (9.1%) fistulas, 1 (4.5%) wound dehiscence, no bleeding complications, no flap loss, no parotid duct injuries, and no facial nerve injuries. Regarding the fistula rate, the first fistula occurred at 1 week postoperatively with separation of the buccal flap at the junction of the hard and soft palate, and this was closed by secondary intention with no need for revisional surgery. The second fistula occurred at 1 week postoperatively at the junction of the hard and soft palate; this fistula healed by secondary intention with no further intervention or surgery required. While our cohort's fistula rate of 9.1% is higher than most series, this reflects the introduction of a new unilateral buccal flap technique and is a limitation of this study in a small cohort of patients. Neither patient required a revisional surgery secondary to these small fistulas that closed by secondary intention, and we anticipate that the fistula rate will be lower as the technique is utilized in a larger number of patients, which we are now prospectively reviewing.
If one observes the vectors of force used during oral and nasal mucosal closure during cleft palate repair, these findings become predicable. Oral mucosa repair at the level of the hard and soft palate follows medially directed vectors. Provided that there is an adequate release of the periosteum around the greater palatine vessel, the oral mucosa flap should translate medially and without tension. The vectors used in nasal mucosa repair are variable depending on the location within and width of the cleft palate. At the level of the hard palate, nasal mucosa repair (when used) follows a medial vector. This is easily appreciated during the transposition of a vomer flap. At the area of the soft palate, the vector of closure is affected by the width of the cleft. In small to medium-sized clefts, the direction of closure follows a medial trajectory. In wider clefts, increased separation at the posterior nasal spine creates a paucity of lining options for nasal mucosal repair at the junction between the hard and soft palate. This is because the lateral palatal shelves have limited ability to transpose nasal lining and the posterior border of the nasal septum (another source of nasal lining) has been reached. This paucity of nasal lining options at the junction of the hard and soft palate will produce a mucosal gap between a fixed unit anteriorly (hard palate) and a mobile unit posteriorly (soft palate). Hence, the only feasible way of closing this central mucosal gap at the junction of the hard and soft palate is by closing the nasal mucosa of the soft palate using a medial and anteriorly directed vector, or by replacing this missing mucosal tissue with a graft (Alloderm) or a flap (buccal mucosa). Therefore, wide cleft palates are predisposed to anterior displacement of the nasal mucosa during closure, resulting in foreshortening of the velum on the nasal side, compromising soft palate excursion and (ultimately) palatal function. It should be noted that even if this nasal mucosa gap at the junction of the hard and soft palate is not closed, the subsequent healing by secondary intention and scar contracture will eventually shorten the soft palate over time.
The theoretical benefit of augmenting velar length at the time of primary palatoplasty to speech results was a major impetus behind the popularization of buccal flap reconstruction. In a retrospective review of 505 patients treated with double opposing Z-plasty plus or minus buccal flap approach, Mann et al. reported a 6.6% secondary surgery rate for VPI, a 6.1% fistula rate, and a 2.7% large fistula rate, which was defined as greater than 2 mm in size. 12 For context, VPI occurs in approximately 6% to 8% of patients with a cleft palate, and while the rates of VPI vary widely in the literature, up to approximately 30% of patients having undergone cleft palate repair may require secondary surgery.1–7 Additionally, recent reports on fistula following primary palatoplasty cite rates of 2.4% to 35%.21–42
Buccal flaps are well-vascularized reconstructive adjuncts that are a compelling match for palatal mucosa in the closure of wide cleft palates. While the addition of myomucosal buccal flaps to palatal repair has previously been described, the characterization of the potential loss of domain in palatal length rescued by buccal flap reconstruction has not been previously reported.43–49 We believe this study provides quantifiable, anatomic support for the implementation, in select patients, of this technique to lengthen the soft palate at the time of primary palatoplasty. A recent study on the use of buccal flaps during primary palatoplasty by Jagannathan et al. reported that for Veau I clefts and narrower clefts, there was less palatal lengthening exhibited, 46 which further underscores the supporting evidence for performing a unilateral buccal flap only in the setting of a wide cleft, only for Veau II or greater clefts, as we found.
In this study, we utilized a unilateral buccal flap in conjunction with a Bardach or von Langenbeck hard palate repair with radical IVV for soft palate repair. Regarding the utility of a unilateral buccal flap in this described technique, a critical point to emphasize is that primary closure of the soft palate nasal mucosa is achievable in all cases. The purpose of the buccal flap technique is to rescue the loss of nasal mucosa domain, restore soft palate excursion and, ultimately, improve velar function. In wide cleft palates, there is a tendency for the nasal side closure to be very tight, akin to a drum, and the recruited buccal flap facilitates palatal movement that restores a more supple nature to the soft palate similar to a parachute. Of note, the average restoration of nasal mucosal palatal length in this reported cohort is 1 cm, a significant distance in the pharynx. Therefore, the pivotal benefit of the buccal flap to primary soft palate reconstruction is to restore a potential loss of domain, of approximately 1 cm of vertical length in patients with a wide cleft.
The aforementioned association of wide palatal clefts with an increased incidence of VPI has been well described.4, 8–10, 19, 20 This study is the first to quantify the potential loss of vertical velar length associated with this more challenging group of patients with a wide cleft palate. We believe that buccal flap restoration of the soft palate nasal mucosa has the potential to achieve lower VPI rates by mediating improved palatal excursion in this at-risk patient population. Long-term follow-up will determine if the palatal function is, indeed improved for this more challenging subset of patients. We note that our indications for the use of the buccal flap are more selective than those described by Mann. We believe that most patients with a cleft palate repaired at the cleft center (over 90%) do not need VPI surgery, and this is reflected in the literature. Therefore, it is only a subset of patients, such as those with a wide cleft, who would benefit from the additional morbidity of buccal flap reconstruction at the time of primary palatoplasty. Furthermore, we are implementing the buccal flap technique to patients undergoing radical IVV rather than a Furlow repair. Although no speech outcomes data is yet available on the use of buccal flap augmentation of radical IVV repair, we believe the benefit should be at least equivalent if not greater. Implementation of the buccal flap technique to the Furlow double opposing Z-plasty will only augment nasal mucosa lining on one side. In the technique described therein, the buccal flap extends to the opposite side of the junction between the hard palate and soft palate, further rescuing the loss of nasal mucosa domain, theoretically providing greater benefit to the palatal excursion.
The limitations of this study include its retrospective nature and the relatively small patient cohort size, as it is a newly adopted technique and a growing experience at our institution. Furthermore, a limitation is that the measurements were all performed by 1 senior author surgeon intraoperatively, and thus there is no ability to measure inter-rater reliability within this study. Importantly, this study is specifically an anatomic study with supportive clinical evidence, and is not a functional or primary clinical study. Long-term speech outcomes will be required to determine the impact of this type of buccal flap reconstruction at primary palatoplasty, including rates of VPI and speech outcomes.
Conclusion
Patients with a wide cleft palate have a potential loss of vertical velar length of approximately 1 cm. Considering that patients with a wide palatal cleft are predisposed to developing VPI, these data provide supportive evidence that acute palatal lengthening during palatoplasty should be considered for this patient population. Buccal flap transposition to the soft palate nasal mucosa can rescue the loss of domain in palatal length, improving palatal excursion and potentially benefiting long-term speech outcomes in patients with a wide cleft palate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
