Abstract
Purpose
A palatal fistula is an adverse outcome of cleft palate repair. It is unknown if a palatal fistula will influence velopharyngeal closure, even after repair of the fistula. This study determines the effect of a soft palate fistula on the risk of developing velopharyngeal insufficiency.
Methods
A retrospective chart review was conducted on patients who underwent primary cleft palate repair between 2000 and 2015, with complete records at 4 years of age. Fistulae involving the secondary palate following primary palatoplasty were classified as the soft or hard palate. A forced-entry multivariate logistic regression model was built to detect predictors of velopharyngeal dysfunction.
Results
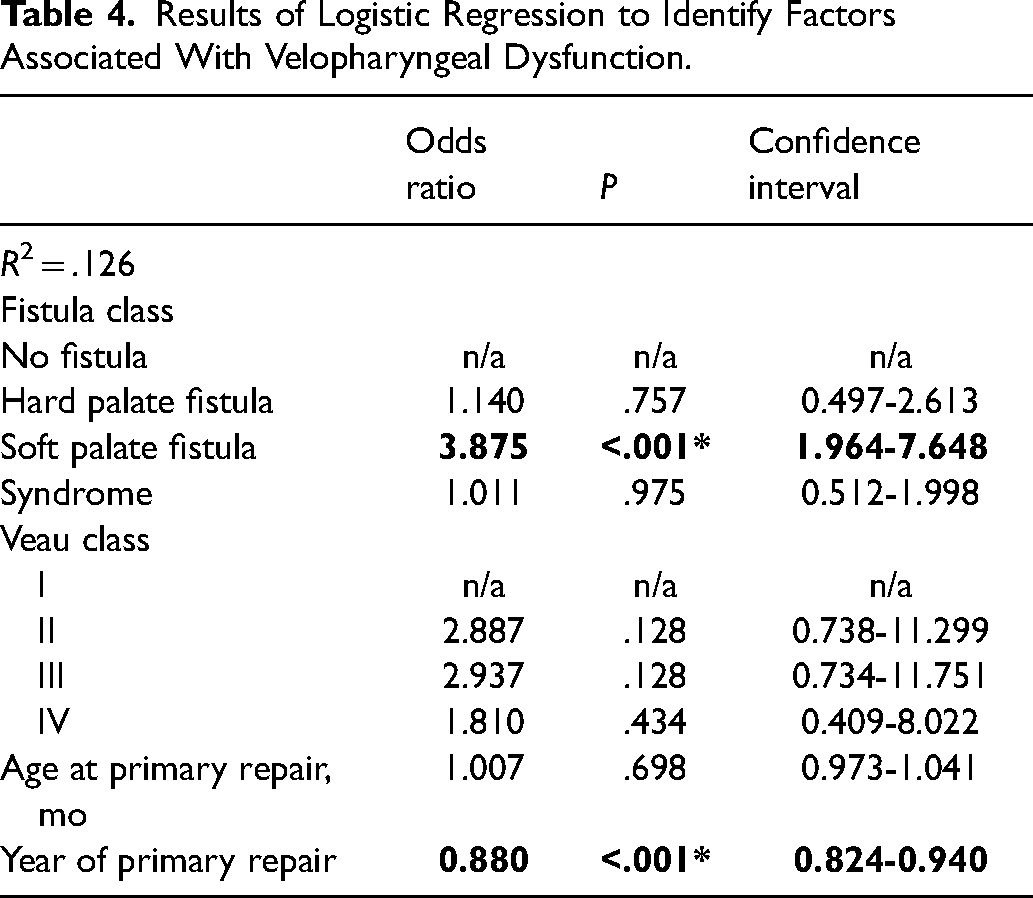
Records of 329 patients were analyzed with a mean follow-up of 8.7 years. A palatal fistula was identified in 89/329 patients (27%) and 29/329 patients (9%) underwent an independent fistula repair. Of the patients with fistula, 44% were located in the hard palate only and 56% had soft palate involvement. Compared to patients without a fistula, rates of velopharyngeal dysfunction were significantly higher in patients with a fistula involving the soft palate (OR 3.875, CI: 1.964-7.648, P < .001) but not in patients with a hard palate fistula (OR 1.140, CI: 0.497-2.613, P = .757). Veau class, age at primary repair, and syndromic status were not significant predictors of VPI (0.128≤P ≤ .975).
Conclusions
A palatal fistula involving the soft palate is a significant predictor for development of velopharyngeal dysfunction after primary palatoplasty. Surgical intervention, at the time of fistula repair, to add vascularized tissue may be indicated to prophylactically decrease the risk of velopharyngeal dysfunction.
Introduction
The primary goal of cleft palate repair is to create an intact palate capable of normal feeding and speech production. The achievement of velopharyngeal competence is the primary measure of success. Velopharyngeal dysfunction is when the velopharyngeal valve does not close completely during the production of oral sounds, which can be caused by abnormal anatomy or neurophysiology. 1 Velopharyngeal dysfunction after cleft palate repair is most often due to a structural defect where the velum is short relative to the posterior pharyngeal wall. 2 The velopharyngeal mechanism acts in unison to control airflow through the nose and mouth. Any disruption of this mechanism can result in abnormal, poorly intelligible speech manifesting as hypernasality, nasal emission, and/or decreased intraoral pressure for consonant production.3,4 Patients with velopharyngeal dysfunction frequently develop persistent maladaptive articulations to compensate for speech difficulties. 5 Rates of this outcome vary widely, with reported rates ranging between 5% and 30%.6–9 Factors contributing to this variation in speech outcomes are largely unknown. Treatment of velopharyngeal dysfunction often depends on closure pattern, gap size, surgeon and institutional preferences. 10 Multiple secondary palatoplasty techniques exist, including pharyngeal flap, double opposing Z-palatoplasty, sphincter pharyngoplasty, and buccinator myomucosal flaps.11–14 Velopharyngeal dysfunction is usually diagnosed years after primary palatoplasty, once patients are old enough to undergo speech imaging studies.
Compared to velopharyngeal dysfunction, a palatal fistula is an early adverse outcome after primary palatoplasty. Fistulae can result in regurgitation of food or fluid and nasal air escape with speech.15–18 Fistula rates vary from 0% to 78% and are thought to be a result of tension and compromised vascularity at the wound site.6,15,19,20 The incidence has been correlated with initial cleft type, with the highest rates generally occurring in patients with Veau 3 and 4 clefts.15,19,21–24 Other reports have also associated fistula incidence with an associated syndrome.20,25,26
Indication for surgical intervention for a palatal fistula is generally conducted when it is considered to be symptomatic. 27 Symptomatic oronasal fistulas can result in speech distortion due to nasal air escape, and/or nasal regurgitation of liquids and food. 15 The persistence of symptomatic palatal fistula in school-age children is associated with a lower self-reported quality of life and perceived speech outcomes due to lower self-reported oral health and speech scores. 28 Fistula repair techniques vary primarily based on fistula location, availability of local tissue, fistula size, patient history, and surgeon experience. 29 Although success rates of surgical repair have improved, fistula reoccurrence rates remain high.23,25,30,31
In this study, we aim to quantify the effect of a fistula involving the soft palate on the subsequent development of velopharyngeal dysfunction. We hypothesize that decreased mobility of the soft palate due to scarring caused by a soft palate fistula interferes with the velopharyngeal mechanism. Our secondary objective is to identify additional predictors of velopharyngeal dysfunction in patients after primary cleft repair.
Methods
A retrospective chart review was conducted on patients who underwent primary cleft palate repair at St. Louis Children's Hospital at Washington University in St. Louis between 2000 and 2015. Only patients with complete records at 4 years of age were included. Collected variables included sex, date and age at primary repair, surgeon of primary repair, syndromic status, cleft type, presence and type of palatal fistula using Pittsburgh Fistula Classification System, presence of velopharyngeal dysfunction, date and age at correction of velopharyngeal dysfunction, and age at last follow-up. 32 All fistulae noted on exam or intraoperatively were included, regardless of size or associated symptoms, until final surgical correction of velopharyngeal dysfunction. Fistula repair was classified as isolated or combined with other procedures such as alveolar bone graft or secondary palatoplasty for velopharyngeal dysfunction.
Speech Assessment
A perceptual speech evaluation was conducted to assess resonance and articulation. Perceptual speech evaluations were video recorded and consisted of a standard set of 17 CVC words that included oral and nasal consonants in spoken English. If the child presented with a limited inventory of correctly produced oral consonants, 2- to 3-word phrases and simple sentences, with the nose unplugged and plugged were used to identify distortion related to velopharyngeal dysfunction and to verify that the consonant motor patterns were sufficiently stabilized to be used in imaging assessments. If the child's consonant inventory included a larger number of appropriately articulated oral stops and fricatives, the perceptual recording included complex phonemic sequences and sentence imitation tasks with specific consonant loads, nose unplugged and plugged, to identify distortion related to velopharyngeal dysfunction and to verify that the consonant motor patterns were sufficiently stabilized to be used in the speech imaging assessments. Audible nasal emission, nasal turbulence, or nasal/facial grimacing were rated as present or absent.
For velopharyngeal dysfunction to be identified, patients had to have at least one allophonic pair of consonants that were placed appropriately, without active extraoral production or co-production, consistently, in phrase imitation. If nasal air leakage or decreased intraoral pressure was evident on the appropriate articulations, patients without fistula were referred for speech imaging studies. If the patient had a fistula, it was next determined if the fistula was in the hard palate, or in the soft palate. Soft palate fistulas are difficult to plug during perceptual speech exams, as it is difficult to get the plugs to maintain adherence, or to place the plug without causing significant gagging. Those patients with fistula involving the soft palate and nasal air leakage with speech were referred for speech imaging studies. Those with a hard palate fistula only underwent repeat perceptual speech evaluation with the fistula plugged if possible. The fistula was plugged with cotton and surgical lubricant contoured to the hard palate. If the nasal air leakage disappeared with the fistula plugged, speech imaging studies for velopharyngeal dysfunction were deemed unnecessary, as the source of the air leakage was clearly the fistula. If nasal air leakage persisted with the hard palate fistula plugged (suggesting that the velopharynx was dysfunctional), the patient was then referred for speech imaging studies.
If velopharyngeal dysfunction was a concern during perceptual speech exam, the diagnosis was definitively made using speech imaging studies: video nasopharyngoscopy, and speech video fluoroscopy. These studies were performed using only consonants for which the child used the correct place of articulation (no active extraoral productions or co-productions) during the studies. Most patients underwent both procedures, consistent with protocols at our center. In some cases, patients may have undergone only speech video fluoroscopy, if cooperation was problematic for completion of an exam.
Diagnosis of velopharyngeal dysfunction was made when a velarpharyngeal gap was seen on speech imaging (Figure 1). In a multidisciplinary fashion, the decision on type of secondary procedure in patients with velopharyngeal involved members from Speech-Language Pathology, Otolaryngology and Plastic Surgery. Isolated fistula repairs were done in patients with symptomatic fistulae with no velopharyngeal dysfunction. Palatal fistulae involving the secondary palate were recorded and classified according to the Pittsburgh Fistula Classification System as types II, III, and IV. 32 Pittsburgh II and III were defined as soft palate fistulae, and isolated Pittsburgh IV were defined as hard palate fistulae.
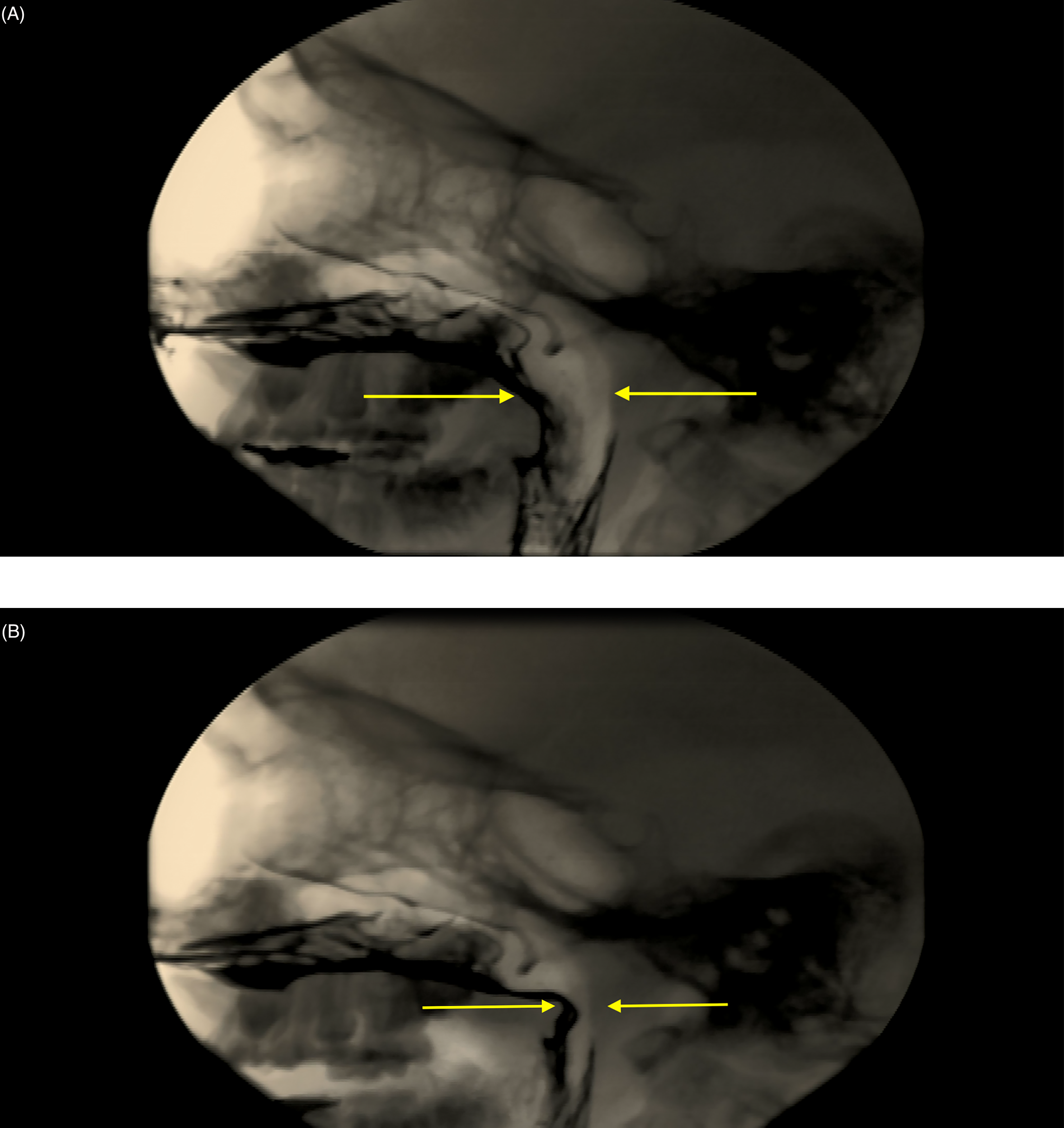
Speech videofluoroscopy, lateral view: (A) preoperatively, at rest and (B) preoperatively, during active speech showing the velopharyngeal gap.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics Version 27 (IBM Corp., Armonk, NY). Bivariate analysis of continuous variables was performed using Student's or Welch's t-tests as appropriate. A forced-entry multivariate logistic regression model, including age at primary repair, year of primary repair, syndromic status, Veau class, and fistula class, was built to predict VPI occurrence after primary repair. Significance was set a priori at P = .05.
Results
Patient Characteristics
We identified 466 patients who underwent primary cleft palate repair between 2000 and 2015. We excluded 117 patients due to insufficient follow-up; an additional 20 patients were also excluded due to missing records, submucous cleft palate, or repairs performed elsewhere. A total of 329 patients were included. The repairs were done by a total of eight surgeons, with a range of 2 to 146 repairs per surgeon. Primary repair techniques varied over the course of these 15 years.
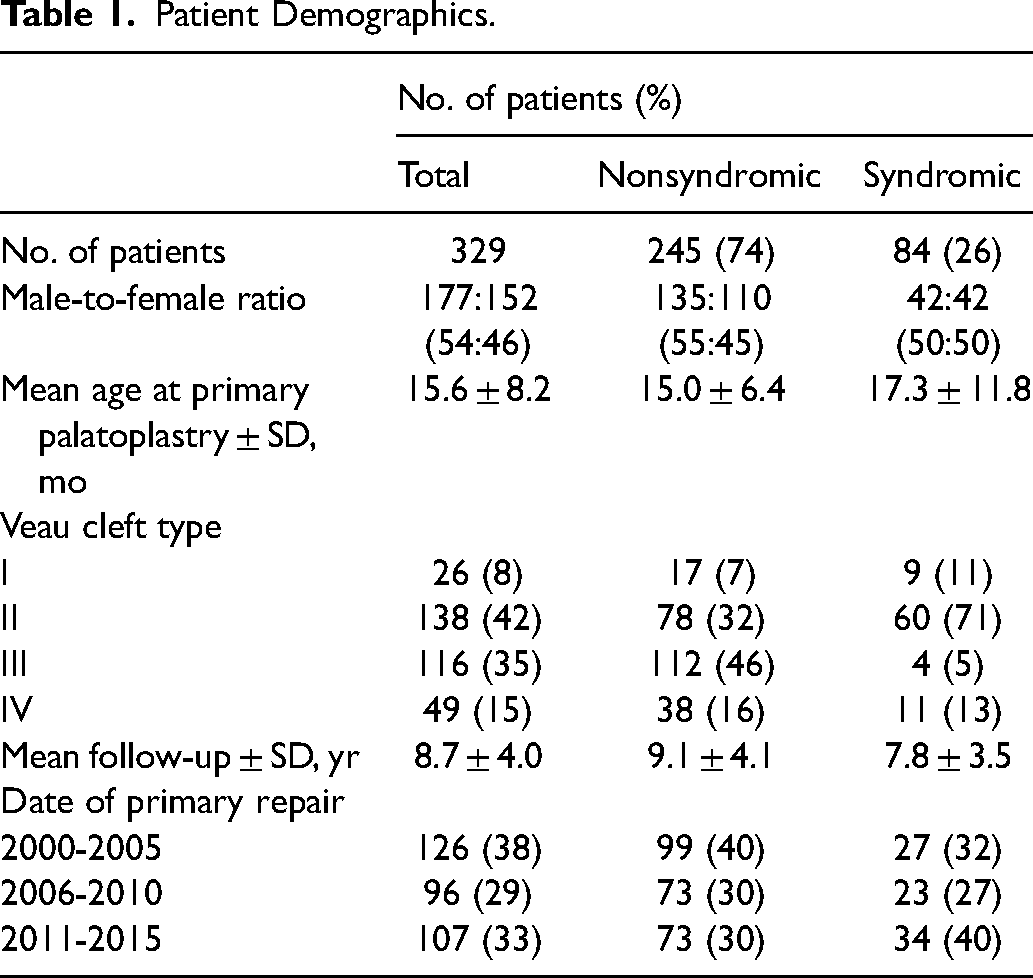
Of the 329 patients, 245 (74%) were nonsyndromic and 84 (26%) were syndromic. The mean age at primary repair was 15.6 months (SD 8.2), and there was a mean interval of 8.7 years (SD 4.0) between primary repair and the most recent follow-up. Additional patient demographics are listed in Table 1.
Patient Demographics.
Palatal Fistulae
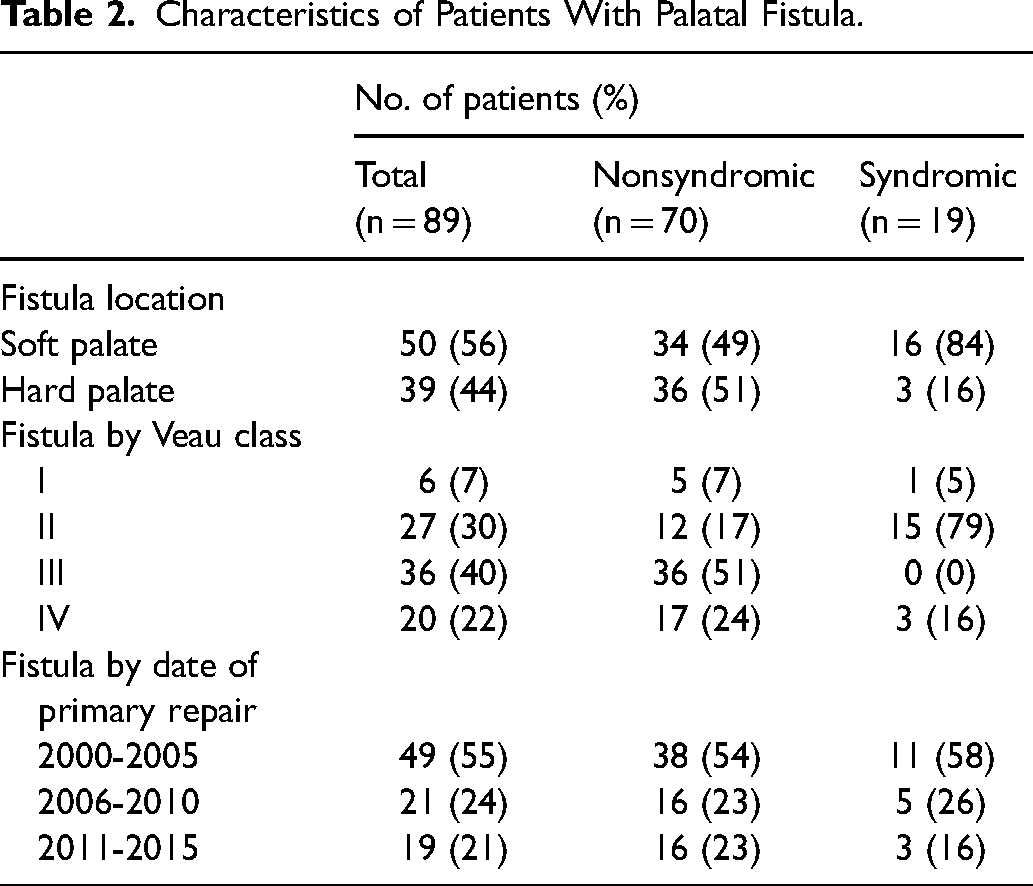
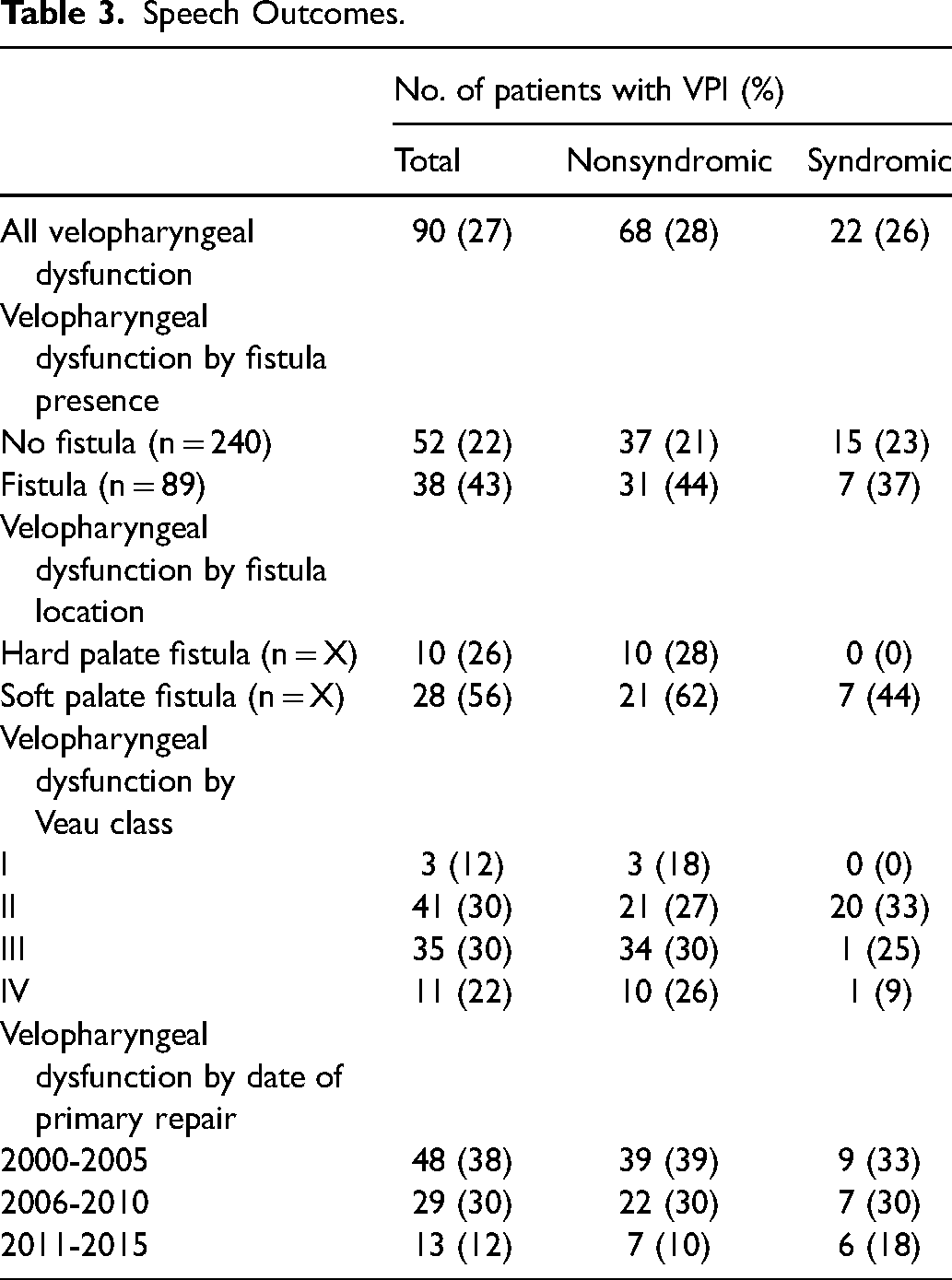
Postoperative palatal fistulae were identified in 89/329 patients (27%); 29/329 patients (9%) underwent an independent fistula repair, 49 (15%) patients underwent a fistula repair in combination with another procedure, and 11 (3%) underwent no fistula repair (Figure 2). Of the 89 patients with fistula, 39 (44%) were hard palate only and 50 (56%) involved the soft palate (29 Pittsburgh type II and 21 Pittsburgh type III) (Table 2).
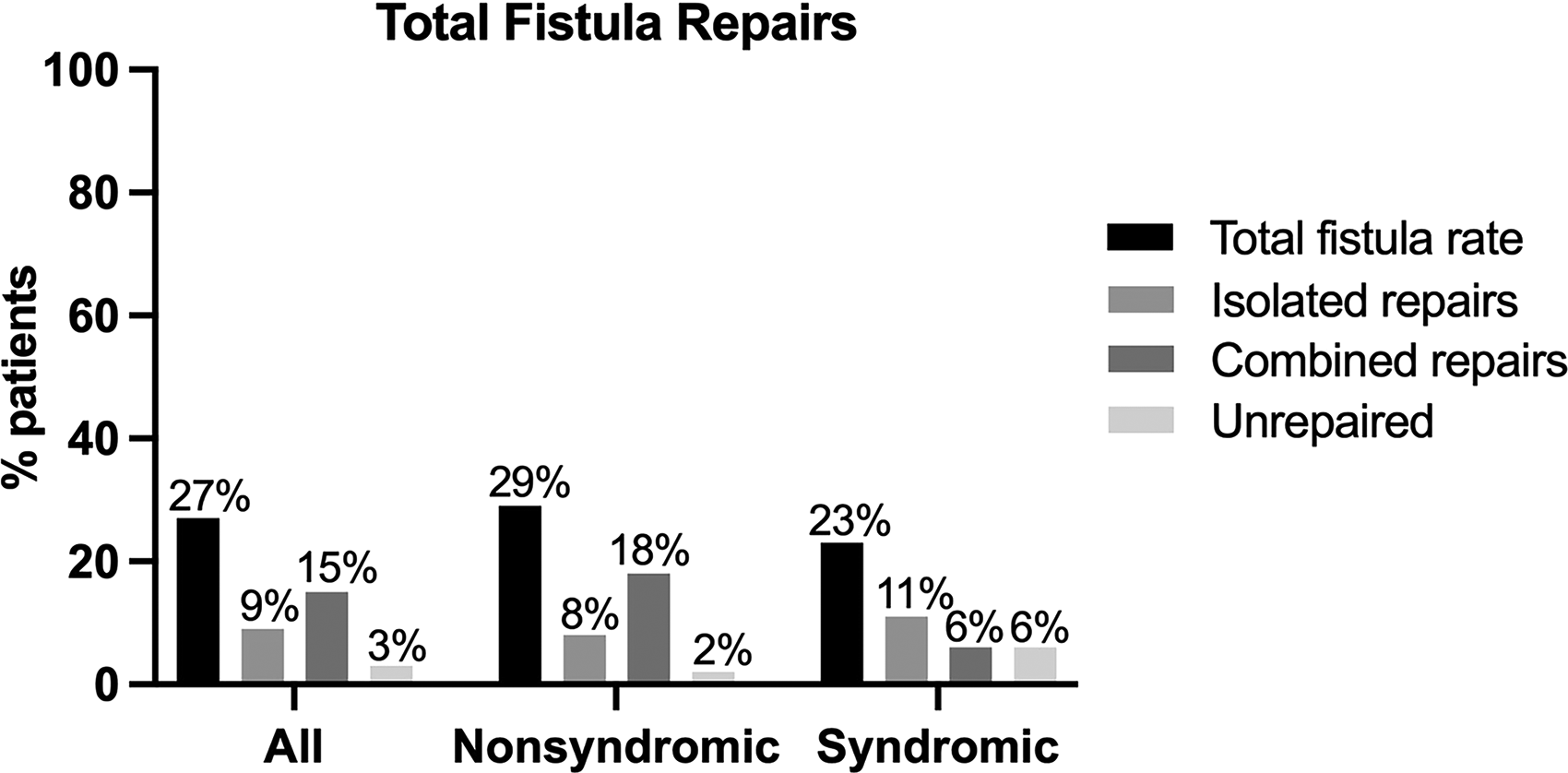
Total fistula rate compared to the rate of fistulae requiring surgical repair. Percentages were calculated for the 329 total patients. Combined fistula repairs were performed during alveolar bone grafting or secondary palatoplasty for velopharyngeal dysfunction.
Characteristics of Patients With Palatal Fistula.
Speech Outcome
Ninety patients (27%) developed velopharyngeal dysfunction. Velopharyngeal dysfunction was reported in 28 (56%) of 50 patients with soft palate fistula compared to 52 (22%) of 240 patients without palatal fistula and 10 (26%) of 39 patients with hard palate fistula (Table 3). Five of 18 patients (28%) with an isolated soft palate fistula repair developed velopharyngeal dysfunction.
Speech Outcomes.
Multivariate regression analysis showed significantly higher rates of velopharyngeal dysfunction in patients with soft palate fistula compared to those with no fistula (OR 3.875, CI: 1.964-7.648, P < .001) (Figure 3). The presence of a hard palate fistula did not increase rates of velopharyngeal dysfunction above that for patients without palatal fistula (OR 1.140, CI: 0.497-2.613, P = .757). Rates of velopharyngeal dysfunction decreased significantly over the course of the study (OR 0.880 per year, CI: 0.824-0.940, P < .001). Veau class, age at primary repair, and syndromic status were not significant predictors of velopharyngeal dysfunction (.128≤P≥.975). Additional results from the regression analysis are shown in Table 4.
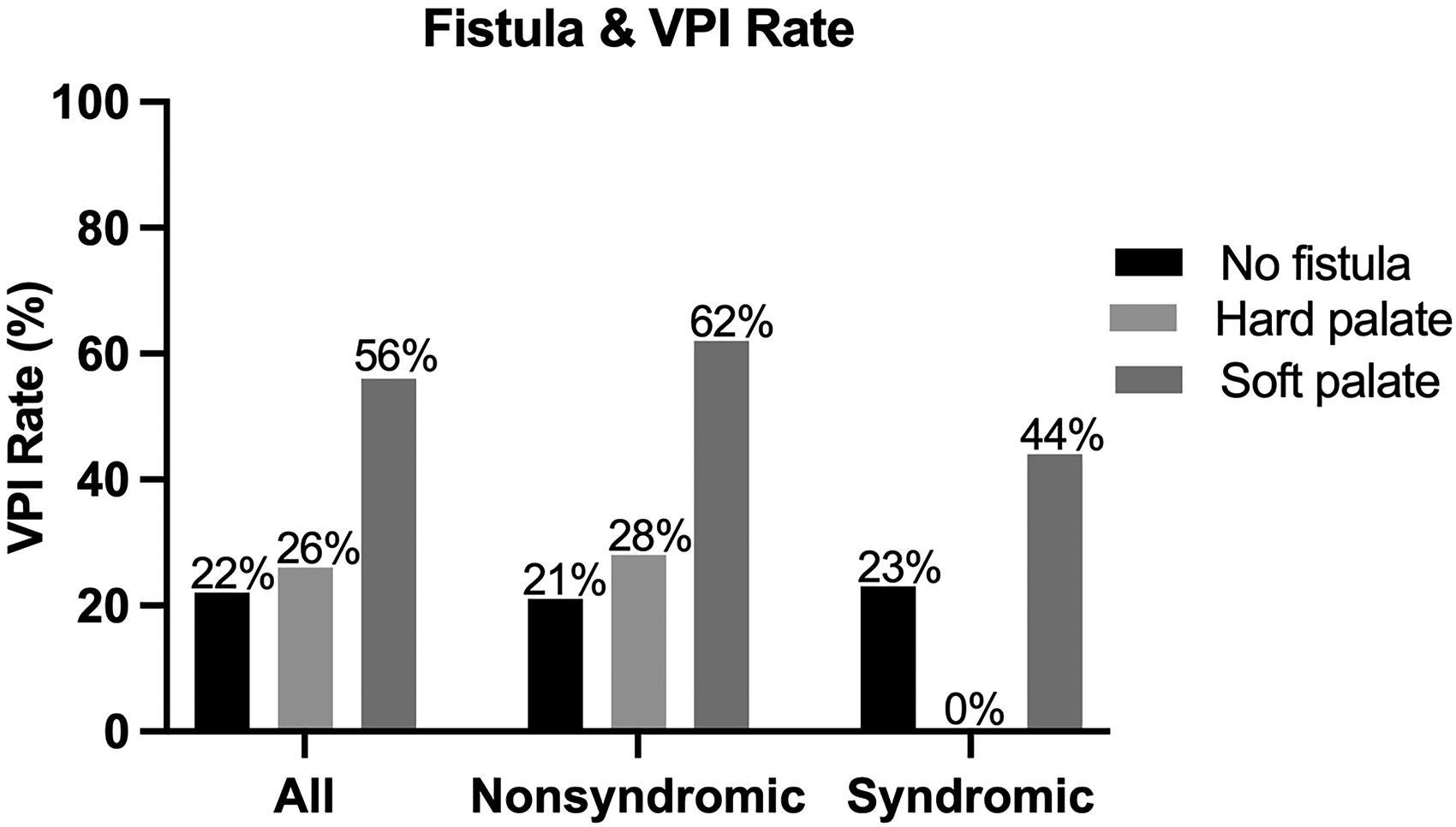
Palatal fistula location compared with velopharyngeal dysfunction. Soft palate fistula was associated with increased incidence of VPI (OR 3.875, CI: 1.964-7.648, P < .001); hard palate fistula was not (OR 1.140, CI: 0.497-2.613, P = .757).
Results of Logistic Regression to Identify Factors Associated With Velopharyngeal Dysfunction.
Discussion
The prevention and management of velopharyngeal dysfunction remains a major challenge in cleft care. In this study, we sought to determine if the development of a fistula involving the soft palate increased the risk of developing velopharyngeal dysfunction. Our findings showed that patients with a soft palatal fistula had significantly increased rates of velopharyngeal dysfunction compared to patients with a hard palatal fistula and patients without fistulae. We propose that this finding results from decreased mobility of the soft palate caused by scarring from the fistula, which in addition, can impact the orientation of the velar muscles.
Our total fistula rate included small and asymptomatic fistulae, and was high at 27% compared to reported rates.6,20,33–35 This could be due to the lack of a standardized definition of oronasal fistula throughout the field, where studies often only include clinically significant or symptomatic fistulae.34,35 Independently repaired fistulae serve as the best measure of clinically significant fistulae for our study. We differentiated independently repaired fistulae from less significant fistulae that were incidentally repaired during another procedure. Our rate of independently repaired fistulae was 9%, and is more consistent with the literature and supports this idea.6–9
Our overall velopharyngeal dysfunction rate decreased throughout the course of the study from 38% to 12% and is also reflected in other studies, as is our overall decrease in fistula rate from 39% to 18%.6–9,36 Given that our study included 8 surgeons, the decrease in velopharyngeal dysfunction rate is most likely due to broader evolution in surgical technique and more aggressive intravelar veloplasty. 10 We did not see differences in velopharyngeal dysfunction rates based on Veau class, although several single-surgeon studies have shown those results.6,7,9
Surgical intervention, or less commonly a prosthetic, can be used for secondary management for hypernasality in velopharyngeal dysfunction in patients with cleft palate. Surgical management of velopharyngeal dysfunction can be placed into 2 categories, physiologic with various palatal re-repair techniques or by narrowing the velopharyngeal sphincter (pharyngeal flap or sphincter pharyngoplasty). The use of buccal flaps is growing in popularity during both primary and secondary palatoplasty. Palatal lengthening with buccal myomucosal flaps has been shown to be particularly effective at reducing rates of fistula and velopharyngeal dysfunction even in very wide clefts during primary palatoplasty, and the use of double opposing buccal flaps during secondary palatoplasty has been shown to effectively treat velopharyngeal dysfunction.37–39 We now offer buccal flaps either alone or in combination with double opposing Z-palatoplasty for velopharyngeal dysfunction management to increase palatal length and improve mobility of the soft palate.
Our study examined the outcomes of 329 cleft palate repairs performed over a 15-year period. We found that the presence of a soft palatal fistula was significantly associated with an increased rate of developing velopharyngeal dysfunction. We hypothesize that scarring, secondary to a soft palate fistula, limits the mobility of the soft palate, increasing the risk for velopharyngeal dysfunction. Additionally, scarring may contract the soft palate anteriorly, thereby shortening palatal length, in addition to limiting mobility. The authors believe it is important to add vascularized tissue, using buccinator myomucosal flap(s) at the time of soft palate fistula repair, to enable improved velopharyngeal function.
Limitations
Although our study had a large sample size relative to earlier investigations into factors related to velopharyngeal dysfunction following cleft repair, our main regression model was not able to explain the majority of the variation in outcomes seen in our patient population. (The overall model r2 was .126.) Therefore, this study is limited in the extent to which it can predict velopharyngeal dysfunction. Future studies may identify factors besides fistula location that are also predictive of the development of velopharyngeal dysfunction. Additionally, although our minimum age of 4 years at last follow-up is equal to or older than the cutoff of similar studies, there is always the possibility of missing later velopharyngeal dysfunction diagnoses.7,12,40,41 The use of multiple surgeons and primary repair techniques also adds an element of variability not seen in single surgeon studies. Due to these limitations and the retrospective nature of the study, our findings will need to be confirmed by other studies before they can be definitively generalized to all patients and primary repair types. Lastly, although incomplete velopharyngeal closing was confirmed via speech imaging evaluation for all patients, it is impossible to definitively determine the cause of speech dysfunction in patients with concomitant velopharyngeal dysfunction and soft palate fistula because soft palate fistulae cannot be occluded.
Furthermore, this study is correlational and does not address the mechanism underlying any relationship between the presence of a soft palate fistula and velopharyngeal dysfunction. Our proposed management strategy assumes that the relationship is caused by scarring and decreased tissue mobility based on anecdotal evidence, but that hypothesis remains untested. Follow-up studies are required to assess outcomes of surgical intervention to add vascularized tissue at the time of soft palatal fistula repair.
Conclusion
This study demonstrated that the presence of a fistula involving the soft palate after primary palatoplasty increases the incidence of velopharyngeal dysfunction. We propose that this is a result of decreased mobility of the palate caused by scarring from a soft palate fistula, with a potential impact on the orientation of the velar muscles. Surgical intervention to add vascularized tissue at the time of fistula repair may be indicated to prophylactically decrease the risk of velopharyngeal dysfunction.
Footnotes
Acknowledgments
This work was sponsored in part by the NIH T35 NHLBI Training Grant Fellowship (grant number 5T35 HL007815) and the Office of Medical Student Research of Washington University in St. Louis School of Medicine.
Financial Disclosure Statement
The authors declare no conflicts of interest regarding commercial associations nor have any financial disclosure related to this paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Heart, Lung, and Blood Institute (grant no. 5 T35 HL007815).
