Abstract
Background:
Common surgical techniques aim to turn the entire vomerine mucosa with vomer flaps either to the oral side or to the nasal side. The latter approach is widely performed due to the similarity in color to the nasal mucosa. However, we lack a histologic description of the curved vomerine mucosa in cleft lip and palate malformations.
Methods:
We histologically examined an excess of curved vomerine mucosa in 8 patients using hematoxylin–eosin, periodic acid–Schiff, Elastin van Gieson, and Alcian blue stains. Tissue samples were obtained during surgery at 8 months of age.
Results:
Our histological analysis of the mucoperiosteum overlying the curved vomer revealed characteristics consistent with those of an oral mucosa or a squamous metaplasia of the nasal mucosa, as exhibited by a stratified squamous epithelium containing numerous seromucous glands. Some areas showed a palisaded arrangement of the basal cells compatible with metaplasia of respiratory epithelium, but no goblet cells or respiratory cilia were identified. Abundant fibrosis and rich vascularity were present.
Conclusion:
The vomer mucosa showed no specific signs of nasal mucosa. These findings should be considered in presurgical cleft orthopedics and palatal surgery for further refinement. Shifting the vomer mucosa according to a fixed physiologic belief should not overrule other important aspects of cleft repair such as primary healing and establishing optimal form and function of palatal roof and nasal floor.
Keywords
Introduction
One in 500 to 1000 newborns (Genisca et al., 2009; Mastroiacovo et al., 2011; Doray et al., 2012; Wang et al., 2017) is affected by cleft deformities of the lip, jaw, or palate. These orofacial clefts constitute the most common congenital disorders in humans that require surgical correction after birth. Cleft deformities are believed to be caused by a combination of genetic factors and yet-to-be identified environmental factors (Mossey et al., 2009). The most common manifestation of cleft deformities involves a complete unilateral cleft lip, jaw, and palate. Complete unilateral clefts typically comprise 30% to 40% of all cleft deformities, occurring in 1 in 1000 to 2000 births (Genisca et al., 2009; Mastroiacovo et al., 2011; Doray et al., 2012; Wang et al., 2017). A unique characteristic of the malformation is the curved vomer that can be already present at week 11 in fetal development and then persists (Atherton, 1967; Latham, 1969). Figure 1 shows the curved vomer where the nasal and palatal mucosa merge.
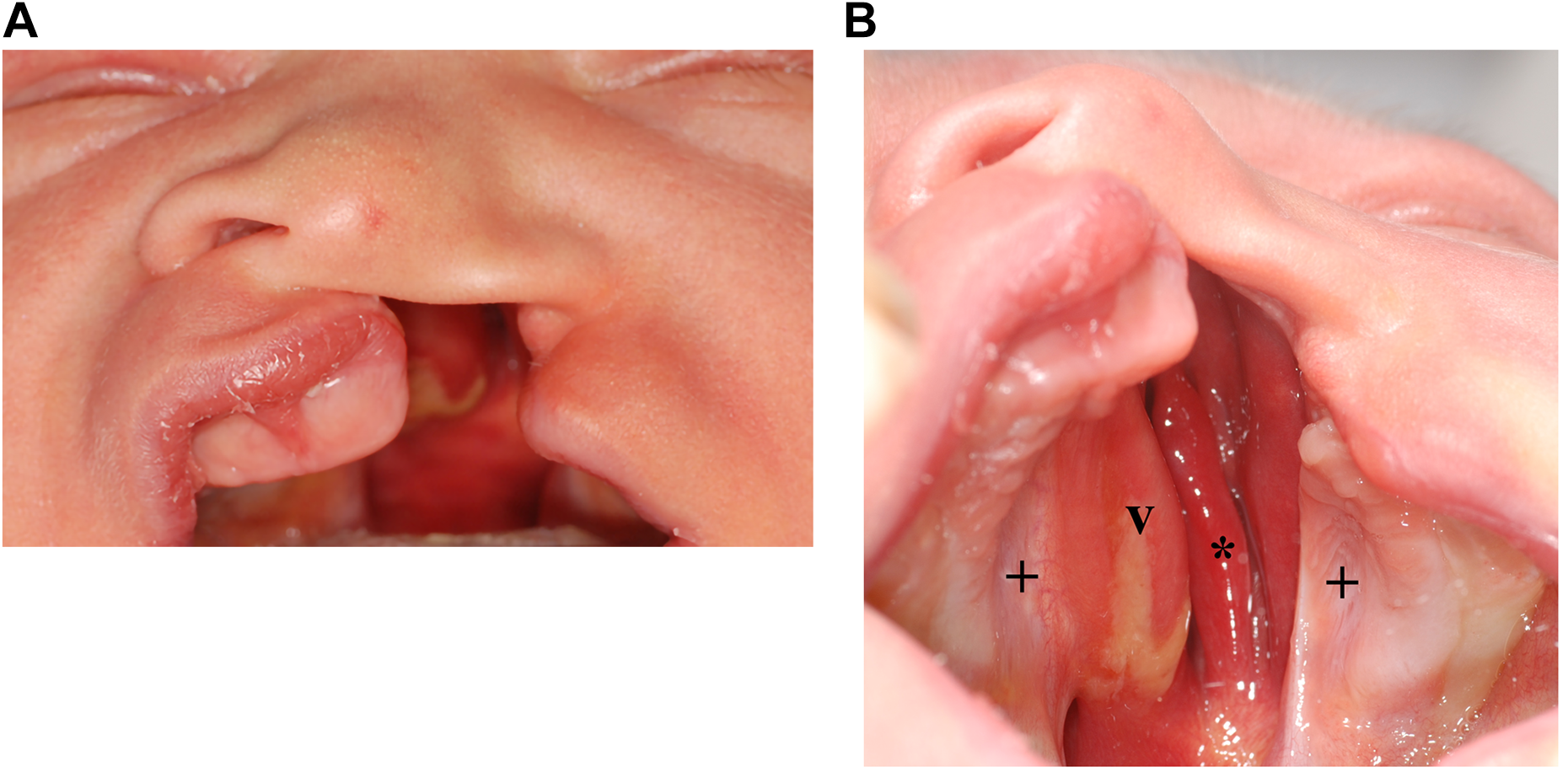
Newborn with complete unilateral cleft lip, jaw, and palate. (A) Front view. (B) View on the palate with deficiency of the bony nasal floor on the cleft side, thus connecting the oral and nasal cavities. This situation results in the nasal concha (*) being visible through the cleft. The hard palate is covered by light pink mucosa in the area of the separated palatal plates (+) and by dark red mucosa in the middle part over the curved vomer bone (v).
Understanding the spatial and histological characteristics of the cleft region is crucial when surgically repairing the cleft (Nalabothu et al., 2020). The evolution of cleft surgical techniques has been decisively based on the work of Veau (1931). Veau reported on the morphology of the cleft in detail and also the important characteristics in the vomer region for consideration when performing surgical palatal cleft closure (Veau, 1931). The vomer bone is connected to the palatal plates and premaxilla by the vomeromaxillary and vomeropremaxillary suture with fibrous tissue (Burdi, 1971). The cleft palate can be divided into 3 main parts: the palatal plates, the true cleft, and the curved vomer (Figure 2A and B).
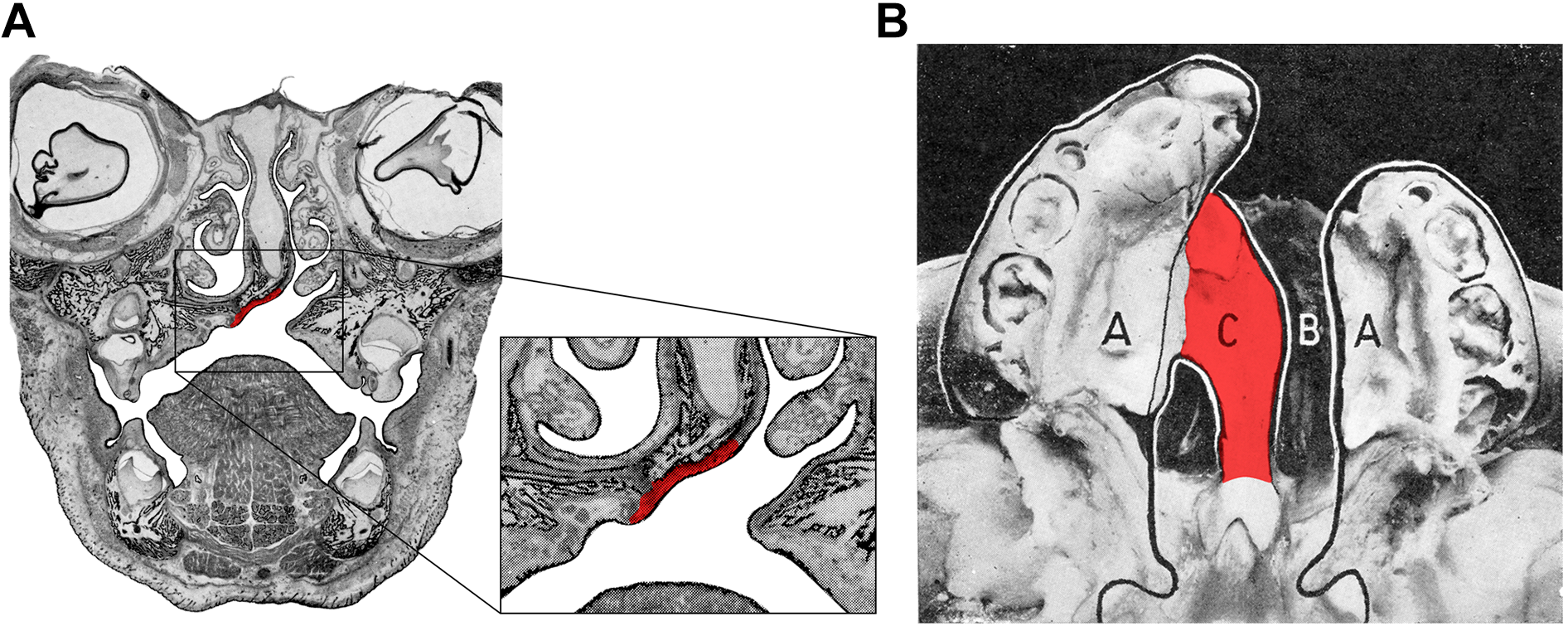
Illustration of a left-sided complete cleft lip and palate. A, Frontal section of a 4-month-old fetus with the mucoperiosteum overlying the curved vomer in red. B, View on the bony palate with the bony part of the curved vomer indicated in red: A = “les lames palatines”: “the palatal plates”; B = “la fente vraie”: “the true cleft”; C = “portion du vomer incurvé”: “curved vomer”. The figures in (A) and (B) are derived from figures 79 and 78, respectively, on pages 41 and 42 of reference (Veau, 1931). Division palatine. anatomie. chirurgie. phonétique. V. veau avec la collaboration de Mlle S. Borel, Dijon and Paris: impr. darantière masson et cie éditeurs; 1931. Elsevier did not object to the use of these figures. The creative commons license does not apply to these pictures.
The overlying vomerine mucoperiosteum has long been routinely used as a flap in palatoplasty (Pichler, 1926; Dunn, 1952; Widmaier, 1966), and surgical variants based on its use have been classified (Kumar, 1985; Agrawal & Panda, 2006; Agrawal, 2009). Cleft palate surgical techniques attempt to turn the entire vomerine mucosa either purely to the oral side (Campbell, 1926; Widmaier, 1966; Kobus, 1984; Bütow, 1987) or to the nasal side (Lannelongue, 1872; Veau, 1931; Dunn, 1952; Kobus, 1984; Kumar, 1985; Bardach, 1995; Abyholm, 1996), or its use is advised against (Delaire & Precious, 1985). Veau hypothesized that the vomerine mucosa is embryologically derivated from the nasal mucosa and therefore must be turned into the nose during surgery, and this hypothesis has influenced generations of cleft surgeons despite the lack of any clear supporting evidence (Veau, 1931; Agrawal & Panda, 2006; Ogata et al., 2017). While the vomer region is referred to as “the center of its field of action” for the surgeon (Veau, 1931), we lack a histological description of the vomerine mucosa in cleft lip and palate malformations.
The aim of this study was to histologically characterize the tissue of the vomerine mucosa in cleft lip and palate and to challenge Veau’s hypothesis that “…normal structures are present on either side of the cleft, only modified by the fact of the cleft…” (Talmant et al., 2007).
Material and Methods
Data Collection
This retrospective observational study investigated 8 patients with unilateral complete cleft lip and palate who underwent cleft repair in one surgical procedure comprising full primary healing (clinicaltrials.gov: NCT04108416). All of the parents and their guardians signed an informed consent form for the surgical procedures and for releasing their medical information and photographs for use in scientific investigations. Patients were treated according to the standard cleft treatment at the institute of the author (A.A.M.). The study was performed in accordance with the Declaration of Helsinki after approval from the ethics commission.
Treatment of Cleft Lip and Palate
Presurgical orthopedics using a passive palatal plate were started during the first days after birth and were continued until cleft surgery was performed. The surgical technique consisted of simultaneously repairing the cleft nose, lip, and palate in one single surgical intervention at the age of 8 months (Benitez et al., 2021).
Histology of the Vomer
In 8 patients, an excess of vomerine mucoperiosteum (Figure 3) had to be trimmed off to allow for straight contact between tissue borders. This small pieces of excess tissue were subsequently examined histologically using hematoxylin–eosin, Elastin van Gieson, periodic acid–Schiff, and Alcian blue stains.
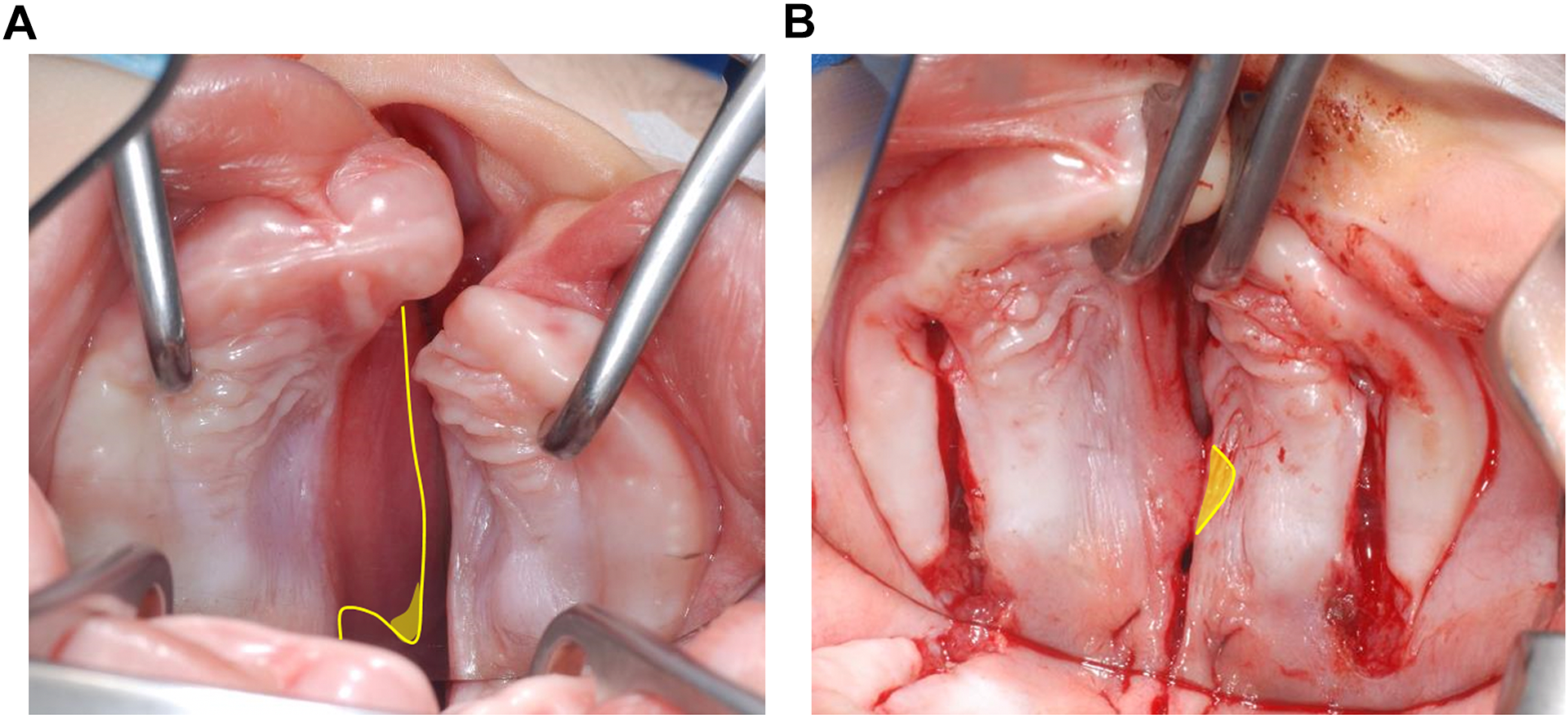
Intraoperative view of the cleft palate. A, Oblique vomer with incision outline (yellow line) and region where a mucosa biopsy was performed (yellow area). B, Incisions made with visible tissue overlap and region of biopsy (yellow area).
Statistical Analysis
Descriptive statistical analysis was applied using Stata (version 15.1; StataCorp) to calculate median and interquartile range (IQR) values of the age at surgery.
Results
Out of 9 patients initially assessed for eligibility, histological analysis of 8 patients could be included in the study (consent not being provided by 1). The median age at surgery was 7 months (IQR = 7-8 months), and 1 patient was female. The histological analysis revealed that the curved vomerine mucoperiosteum comprised a stratified squamous epithelium with numerous seromucous glands. Some areas showed a palisaded arrangement of the basal cells compatible with metaplasia of the respiratory epithelium. In none of the 8 samples, goblet cells or respiratory cilia were identified, and abundant fibrosis and rich vascularity were present (Figure 4).
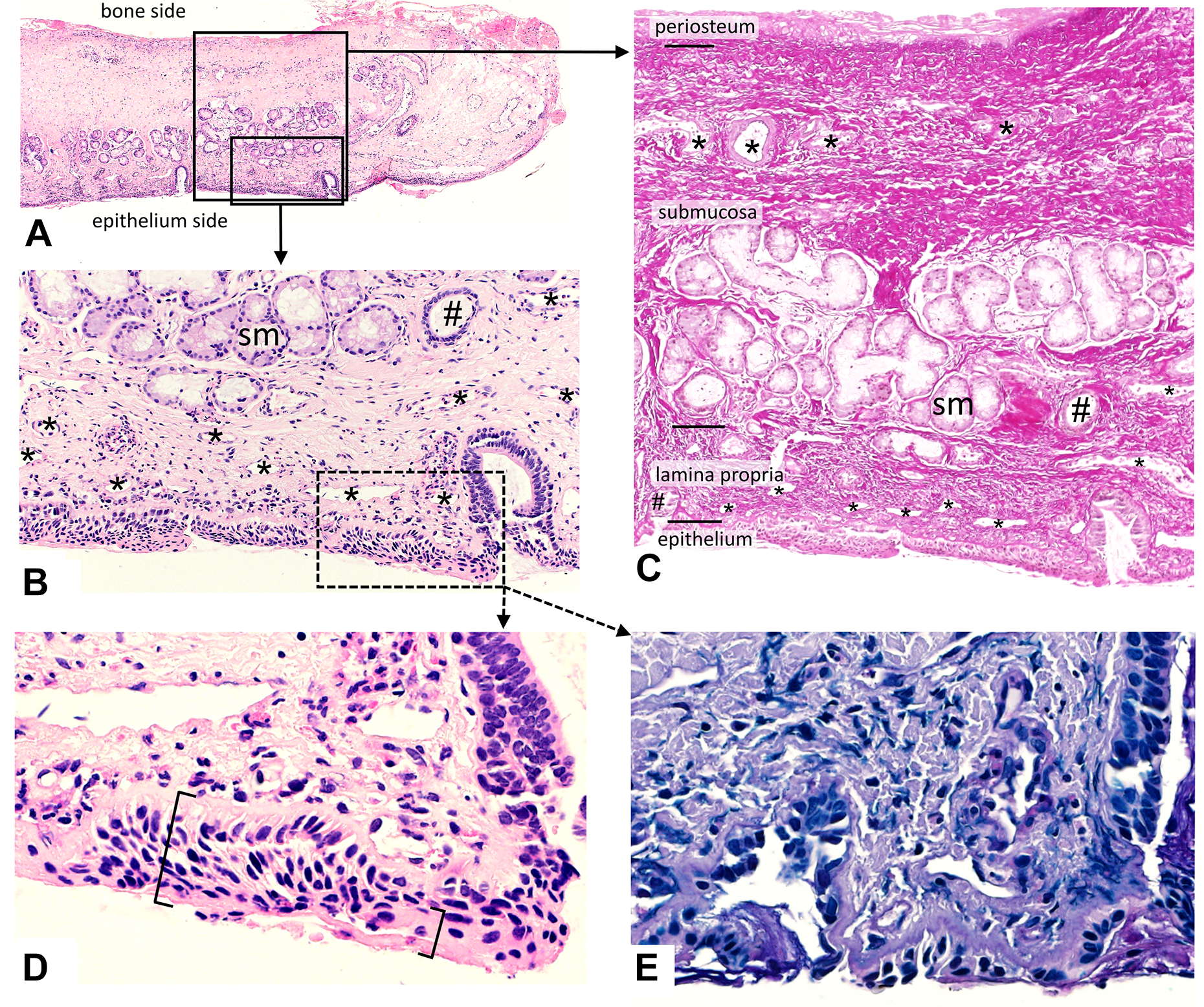
Histology of the curved vomerine mucosa. A, Overview of the curved vomerine mucoperiosteum (compare with the donor site in Figure 3A and B). B, The superficial half of the submucosa contains seromucous glands (sm) and cross sections of the secretory ducts (#). The lamina propria contains multiple small vessels (*). C, The deep half of the submucosa contains a dense network of collagen fibers and some vessels (*). The collagen fiber network reaches from the periosteum to the basal membrane of the epithelium. D, The epithelium appears as a stratified squamous epithelium ([). In some areas, the epithelium shows parakeratosis (]), and the corneal layer therefore contains pycnotic cell nuclei. No ciliated cells were detected. E, No goblet cells with intracellular mucus were detected. A, B, D, Hematoxylin–eosin stain. C, Elastin van Gieson stain. E, Alcian blue and periodic acid–Schiff stain.
Discussion
Cleft lip and palate in nonsyndromic infants is considered a failure of tissues fusion rather than a consequence of mesodermal deficiency (Veau, 1931; Mulliken et al., 2003; Talmant et al., 2007). Veau hypothesized that “…normal structures are present on either side of the cleft, only modified by the fact of the cleft…” (Talmant et al., 2007), which implies that the goal of any surgical approach for cleft repair should be to construct the normal anatomy by relocating the present structures. Various approaches of utilizing incisions in the vomer region have been outlined for determining the optimal positioning from a clinical point of view (Veau, 1931; Dunn, 1952). Some authors (including Veau) have argued on a post hoc basis that the exclusive use of the vomerine mucosa for nasal reconstruction is justifiable from an embryological or physiological point of view, in the absence of histological evidence (Veau, 1931; Agrawal & Panda, 2006; Ogata et al., 2017).
Histology of the Vomer
Our histological analysis of the mucoperiosteum overlying the curved vomer revealed no specific signs of the nasal mucosa, with neither ciliated cells nor goblet cells being present. Therefore, the histological findings of the mucoperiosteum are compatible with an oral mucosa or with squamous metaplasia of the nasal mucosa. During the 7 to 8 months of presurgical plate therapy, the vomerine mucosa was not exposed to any physical irritation since the palatal plate did not contact the underlying mucosa and the tongue was kept away from the vomerine mucosa (Figure 5).
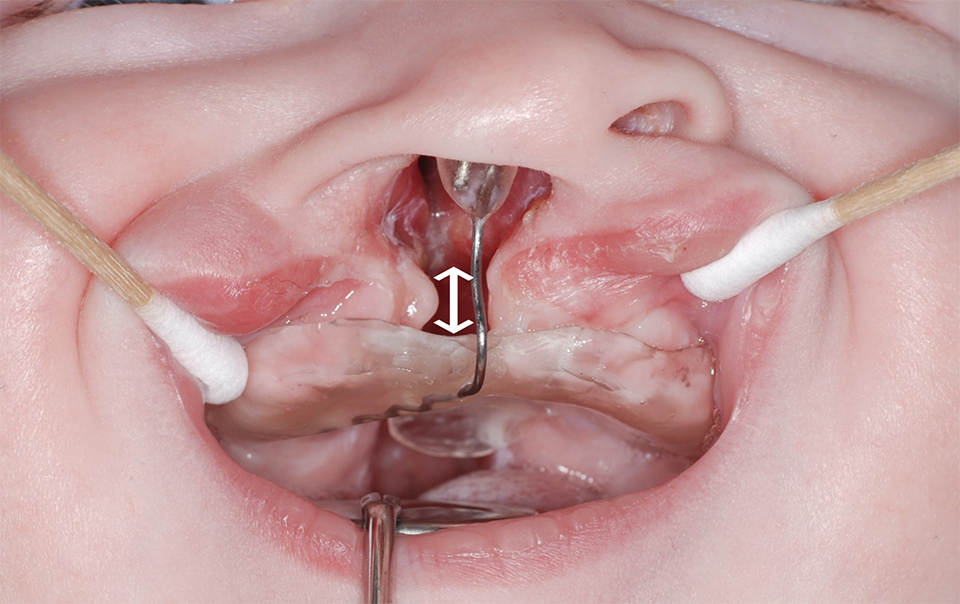
A passive palatal plate with free space (white ↕) between the vomerine mucosa and palatal plate. The plate kept the tongue out of the cleft and thus away from the vomerine mucosa in a newborn with a right-sided complete cleft lip and palate.
Figure 6 shows that during the further postoperative course and normal pediatric development, the vomer mucosa still showed macroscopic differences from the palatal mucosa, as has also been shown by other authors (Ogata et al., 2017). This further highlights that metaplasia of the vomerine mucosa is not reversible. The permanent presence of metaplasia must therefore be assumed, which is analogous to the intestinal metaplasia of the esophagus in gastroesophageal reflux disease (King, 2007).
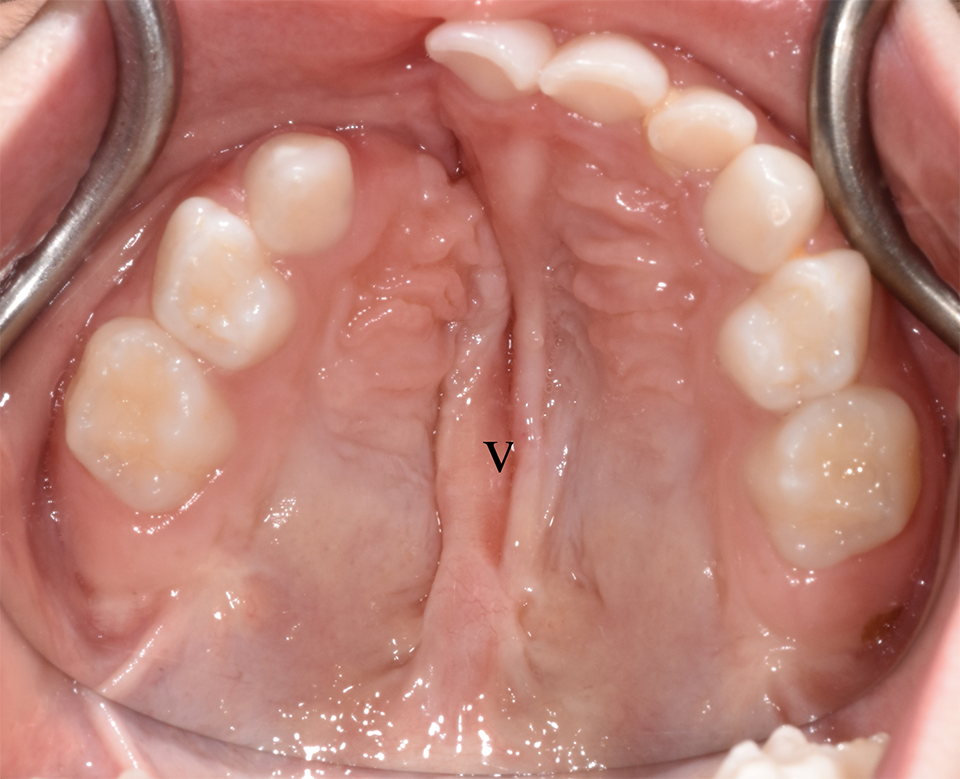
Visible difference between the vomer mucosa (v) and the palatal mucosa in a patient with a right-sided complete cleft lip and palate at 3 years of age. The surgical repair was performed at 8 months of age. The primary lateral incisor (tooth 52) is absent. Hard palate repair was performed in 2 layers using bilateral bipedicle flaps, soft palate repair was performed using medial pterygoid detachment and intravelar muscle repair, and lip repair comprising primary rhinoplasty.
Clinical Relevance for Cleft Lip and Palate Repair
The vomer mucosa showed no specific signs of nasal mucosa. From this histological point of view, surgical techniques should be reconsidered. We must challenge the pure use of the entire vomer mucosa as a cranial pedicled flap for nasal floor reconstruction (eg, 2-flap palatoplasty) (Bardach, 1995). Veau (1931) has already subdivided the cleft palate into vomer and the true cleft (Figure 2B). The former spanning across the noncleft nasal floor and bottom of the osseous septum, the latter spanning across the absent nasal floor that opens into the nasal cavity on the cleft side. This distinct division of the palatal cleft area has not been given the full attention in surgical and presurgical protocols so far. In staged surgical concepts, typically the first step—whether lip or soft palate repair (Gundlach et al., 2013)—has the mere intention to simplify or make the subsequent hard palate cleft repair less invasive. Whereas cleft orthopedics has shaped the definition and measurements of cleft palate width and cleft area mostly ignoring the vomer region (Berkowitz et al., 2005). The vomer region is more difficult to identify on cleft impressions, even more so after the first step of surgical repair that leads to growth alteration. In contrast, in unilateral clefts without surgical treatment with undisturbed growth after birth until adulthood, the vomer area and true cleft area can be clearly distinguished, as can be seen repeatedly in photographs of unoperated adults (Shetye, 2004). The vomer region has been generally considered as part of the nose without question. This has shaped outcome research to quantify the change in cleft palate morphology (Prasad et al., 2000; Braumann et al., 2002; Neuschulz et al., 2013; Bruggink et al., 2020) as well as led to clinical recommendations for the timing of surgery (Berkowitz et al., 2005). Although this central region of the vomer has not been studied, it has had and continues to have an impact on treatment approaches. Therefore, the concept of the nasal opening “the true cleft” has not been widely used in surgical concepts or in measuring the effect of orthodontic treatment (Nalabothu et al., 2020). Current research on cleft palate morphology and thus surgical concepts still imply that the vomer belongs to the cleft palate. Considering the true cleft described by Veau and the finding that the vomer mucosa is not a nasal mucosa, the exact definition of cleft palate needs to be challenged. What we define as cleft palate and how we translate it into orthodontic research and surgery should be reopened for discussion based on our findings. The clinical relevance of our results will therefore depend on whether we as cleft surgeons and orthodontists have the flexibility to rethink “the center of our field of action” (Veau, 1931). To anatomically reconstruct a symmetrical nasal floor, only a small part of the vomer mucosa is necessary. Figure 3A shows an incision outline on the vomer that contributes minimally to the reconstruction of the nasal layer and leaves substantial tissue for oral closure. We propose allocation of the vomer tissue to reconstruct the oral and nasal layer likewise. If only as little as necessary is used for the symmetrical reconstruction of the nasal floor, most of the vomer mucosa can be used to reconstruct the oral layer. This would support the hypothesis that the structures are present in the case of a cleft and they are only modified by the cleft (Talmant et al., 2007). Technically, a small 5/8 needle with a cone point (eg, FR-10 Art.6O15132 T; Serag Wiessner) is advisable to sew the nasal layer tightly in spite of limited vomer tissue turnover. Without previous lip surgery (Figure 3), the nasal layer can be sutured from the oral side, and especially in the case of narrow clefts, it is technically easier to suture the nasal layer anteriorly from the vestibular (extraoral) side. From a biological point of view, additional tissue to close the oral layer could enable minimal lateral incisions (Karsten et al., 2003) or eliminate the need for lateral relief incisions even in unilateral cleft lip and palate repair (Brusati & Mannucci, 1994; Brusati, 2016). Reduced secondary healing lead to less scarring, having a negative impact on growth (Kim et al., 2002; Pigott et al., 2002). The preservation of vascularity could also facilitate primary wound healing to achieve lower fistula rates (Losken et al., 2011). The amount of tissue required to close the true cleft depends on treatments performed prior to hard palate cleft repair. With passive presurgical orthopedics or lip taping, the true cleft can be reduced in a clinically relevant amount even without prior lip surgery (Abd El-Ghafour et al., 2020; Nalabothu et al., 2020). By combining presurgical reduction of the hard palate cleft and limiting reconstruction to the true cleft by proportional distribution of vomer tissue, cleft palate repair could be facilitated. In this regard, prior lip surgery aiming to reduce the hard palate cleft could be questioned. When the curved vomer mucosa is surgically rotated into the oral cavity, it retains its difference in color from the palatal mucosa. However, it remains uncertain as to whether the vomer mucosa would ever transform into a typical nasal mucosa when it is surgically turned into the nasal cavity. It also remains unclear whether the vomerine mucosa is embryologically formed as a typical nasal mucosa and undergoes reparative adaptive changes or whether it is a mucosal transition zone between the oral and nasal mucosa. This is supported by the common, yet unspecific characteristics of both types of mucosa found in the vomer mucosa. Even if the macroscopic aspect of the vomer mucosa differs from the palatal mucosa (Veau, 1931; Agrawal & Panda, 2006; Ogata et al., 2017), our histological results indicate that the vomerine tissue is adequate for use in oral reconstruction from a histoanatomical point of view. These findings should be considered when further refining the optimal anatomical reconstruction method to apply to cleft palates.
Limitations
The number of examined cases is small, but they showed a uniform histological aspect. However, in the area of this malformation, the collection and examination of tissue is limited and can only be done from an ethical point of view as in our case with excess tissue. Since von Langenbeck described the palatal cleft closure in 1861 (Pigott et al., 2002), we are now able to describe to cleft surgeons, on the basis of a small number of cases, the histology of “the center of its field of action” (Veau, 1931) in cleft, lip, and palate malformations, which must be emphasized as a strength. Further analysis of other centers with larger numbers of cases is necessary to confirm the consistency of our findings. Following findings on the histology of the vomer mucosa, different techniques should be investigated for their influence on tissue perfusion and wound healing. We used a presurgical orthopedic plate without any contact with the examined vomer. To exclude the possibility of metaplasia due to presurgical treatment, an external control without any pretreatment is planned. Further evidence of tissue differentiation could be provided by molecular differentiation and testing for specific antigens.
Conclusion
The areas of mucosa analyzed in this study were suggestive of the occurrence of massive regenerative changes. Nonetheless, the histological characteristics of a respiratory epithelium were not present in any of the samples analyzed. The mucoperiosteum overlying the curved vomer did not exhibit any of the specific signs of nasal mucosa, such as ciliated cells and goblet cells. These results substantiate that the curved vomerine mucosa may be used to reconstruct both the nasal floor and palatal roof in cleft lip and palate, since the mucoperiosteum of the curved vomer forms a tissue that is similar (but not identical) to those at the nasal floor and palatal roof. Thus, the distinct use of the vomerine tissue in a cleft protocol should be made by other justifications than the assumption that it is nasal mucosa.
Footnotes
Authors’ Note
E.B. and A.A.M. have contributed equally to this work. A.A.M. and B.K.B. were involved in the conception and design of the study and in the drafting of the manuscript. E.B. was involved in the analysis and interpretation of the histological findings. A.B., P.N., J.A.v.J. and E.B. were involved in revising the manuscript critically for important intellectual content. All of the authors have read and approved the final version of the manuscript. This paper was presented at the 24th Congress of the European Association for Cranio-Maxillo-Facial Surgery (EACMFS), 2018, Munich, Germany. The data sets generated and analyzed for this study are available on request from the correspondence author. The study was reviewed and approved by the Ethics Commission of Northwest and Central Switzerland (EKNZ) (project-ID: EKNZ 2020-00739). All parents and their guardians signed an informed consent form for the surgical procedures and for releasing their medical information and photographs for use in scientific investigations.
Acknowledgments
The authors thank Marco Weber Academy of Art and Design, University of Applied Sciences and Arts Northwestern Switzerland for producing the visualizations in Figure 2A and ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Benito K. Benitez acknowledges that he received financial support for his PhD program through a grant from the Department of Surgery at the University Hospital of Basel. The funding source was not involved in the study design, the collection, analysis, or interpretation of data, the preparation of the manuscript, or the decision to submit the manuscript for publication. This research did not receive any other specific grants from funding bodies in the public, commercial, or nonprofit sectors.
