Abstract
Background:
The majority of research to understand the risk factors of nonsyndromic orofacial clefts (NSOFCs) has been conducted in high-income populations. Although patients with NSOFCs in low- and middle-income countries (LMICs) are at the highest risk of not receiving care, global health infrastructure allows innovative partnerships to explore the etiologic mechanisms of cleft and targets for prevention unique to these populations.
Methods:
The International Family Study (IFS) is an ongoing case–control study with supplemental parental trio data designed to examine genetic, environmental, lifestyle, and sociodemographic risk factors for NSOFCs in 8 LMICs (through August 2020). Interview and biological samples are collected for each family. The interview includes demographics, family history of cleft, diet and water sources, maternal pregnancy history, and other lifestyle and environmental factors.
Results:
Seven of 8 countries are currently summarized (2012-2017) for a total of 2955 case and 2774 control families with 11 946 unique biological samples from Vietnam, Philippines, Honduras, Madagascar, Morocco, Democratic Republic of the Congo, and Nicaragua. The phenotype distribution was 1641 (55.5%) cases with cleft lip and palate, 782 (26.5%) with cleft lip (CL), and 432 (14.6%) with cleft palate (CP).
Discussion:
The International Family Study is the largest case set of NSOFCs with an associated biobank in LMICs currently assembled. The biobank, family, and case–control study now include samples from 8 LMICs where local health care infrastructure cannot address the surgical burden of cleft or investigate causal mechanisms. The International Family Study can be a source of information and may collaborate with local public health institutions regarding education and interventions to potentially prevent NSOFCs.
Introduction
Cleft lip with or without cleft palate is one of the most common birth defects worldwide with a global incidence of 1 in 700 live births (Mossey et al., 2009). Patients with cleft face significant health risks from birth due to feeding difficulties leading to malnutrition and associated health risks (Masarei et al., 2007; Tungotyo et al., 2017; Munabi et al., 2017). Throughout their lifetime, when adequate care is available, a child born with a cleft lip and palate (CLP) will require multiple orofacial surgeries, specialized dental and orthodontic care, and speech therapy at a minimum. Some individuals will experience lifelong difficulties from the condition and may experience lasting psychosocial effects (Hunt et al., 2007). Current estimates are that over 5 billion people globally lack access to safe and affordable surgical care for any surgically treatable condition (Meara et al., 2015). Given the complexity of cleft surgery and the necessity of ancillary services with the large number of impacted children and adults awaiting care, it is unlikely that a global solution dependent solely on surgical intervention will ease the burden of cleft disease.
Nonsyndromic orofacial cleft lip and/or cleft palate (NSOFCs) is defined as those cases with no other major malformations or syndromes, and these represent approximately 70% of all orofacial clefts (Rittler et al., 2004; Calzolari et al., 2007; Leslie and Marazita, 2013). Multiple risk factors influence risks for NSOFCs are largely uncharacterized. The incidence of cleft varies widely by race/ethnicity. The highest rates of cleft are found in Asian populations with an incidence as high as 1 in 500 births (Cooper et al., 2006). The lowest rates are in African populations (1 in 2000 births) with populations of European ancestry falling between 1 in 1000 births (Gundlach and Maus, 2006; Mossey et al., 2009; Kadir et al., 2017). The most consistently identified risk factors for nonsyndromic cleft are family history, race/ethnicity, mother’s nutritional insufficiency of folic acid (Shaw et al., 1995; Tolarova and Harris, 1995; Czeizel et al., 1999; Wehby and Murray, 2010; Li et al., 2012), maternal smoking, and environmental tobacco smoke (Wyszynski et al., 1997; Sivertsen et al., 2008; Genisca et al., 2009; Hackshaw et al., 2011; Sabbagh et al., 2015), periconceptional alcohol use (Romitti et al., 2007; Bell et al., 2014), low maternal education (Yang et al., 2008; Acuña-Gonzalez et al., 2011), advanced maternal age (Mai et al., 2014; Berg et al., 2015), and diabetes (either pregestational or gestational) (Lebby et al., 2010; Figueiredo et al., 2015).
The International Family Study (IFS) was designed to address the lack of comprehensive data available on the genetic, environmental, lifestyle, and sociodemographic factors influencing the risk of NSOFCs in low- and middle-income countries (LMICs). Through an ongoing partnership with Operation Smile (OS), a well-established international cleft organization providing free cleft care (https://www.operationsmile.org), we conducted a “population-sampled” case–control study (see discussion for limitations related to design—hereinafter referred to as case–control) to explore risk factors for nonsyndromic cleft in 8 multiethnic LMICs—collecting both questionnaire data and biological samples. Although the etiologic mechanisms controlling risk of NSOFCs may be similar globally, the specific factors contributing to these mechanisms, the prevalence of exposures, and the numbers of cases attributable to specific mechanisms may vary by region of the world. Our effort to collect and document the experience of a large, regionally diverse set of children attending OS missions may inform risk mechanisms through novel factors or serve to replicate or elucidate findings from populations of European ancestry. The study will add to our understanding of determinants of NSOFCs in multiethnic populations (Asian, African, Latino) and will evaluate environmental, genetic, and lifestyle factors included in the current body of cleft research. Using the growing volume of patients served by OS, this study will be statistically powered to test these risk factors by cleft phenotype, country, and region.
Methods
Study Design
The International Family Study (IFS) is an ongoing case–control study with supplemental parental trio data led by investigators at the University of Southern California (USC), a large research university, and Children's Hospital Los Angeles (CHLA), the affiliated pediatric hospital in collaboration, with Operation Smile (OS). It is designed to examine genetic, environmental, lifestyle, and sociodemographic risk factors for NSOFCs in 8 LMICs (as of August 2020). Many of these cases are treated only through not-for-profit organizations and are therefore difficult to locate, document, and characterize through other data sources. Because of the lack of centralized information on cleft cases in LMICs, less information is currently available in the medical literature on environmental and genetic risk factors for clefts compared to Europe, the United States, and China. The study is currently active in 8 countries: Vietnam, the Philippines, Honduras, Nicaragua, Guatemala, Madagascar, Morocco, and the Democratic Republic of the Congo (DRC). The data snapshot presented summarizes collections through 2017 and does not include Guatemala. All work was approved by the institutional review board (IRB) at USC including country-specific authorizations. Funding for the study has come from a variety of donors and foundations through CHLA and OS. Key definitions for the study are provided in Table 1.
Key Definitions.a

a The following definitions are utilized in the study to standardize methods and patient selection.
Specific Aims
The primary aims of IFS were designed to address the leading suspected risk factors for environmental, lifestyle, and genetic causes of NSOFCs: (1) to evaluate the relationship between personal and environmental exposure to paternal and maternal smoke sources, both prior to and during pregnancy, and NSOFCs including maternal and paternal use of tobacco products, exposure to tobacco smoke in the home environment, and use of an open flame for cooking in the home; (2) to evaluate the relationship between modifiable maternal prenatal lifestyle factors (diet, water source, vitamin, drug or medication, and alcohol use) with risk to NSOFCs; (3) to evaluate the relationship between current and past paternal occupation and exposure to agents known to be mutagenic and NSOFCs; and (4) to explore both rare and common genetic variants and their relationship to risk of NSOFCs using genome-wide association and tests for gene–environment interaction methodology in multiethnic populations. The study framework is shown in further detail in Figure 1.
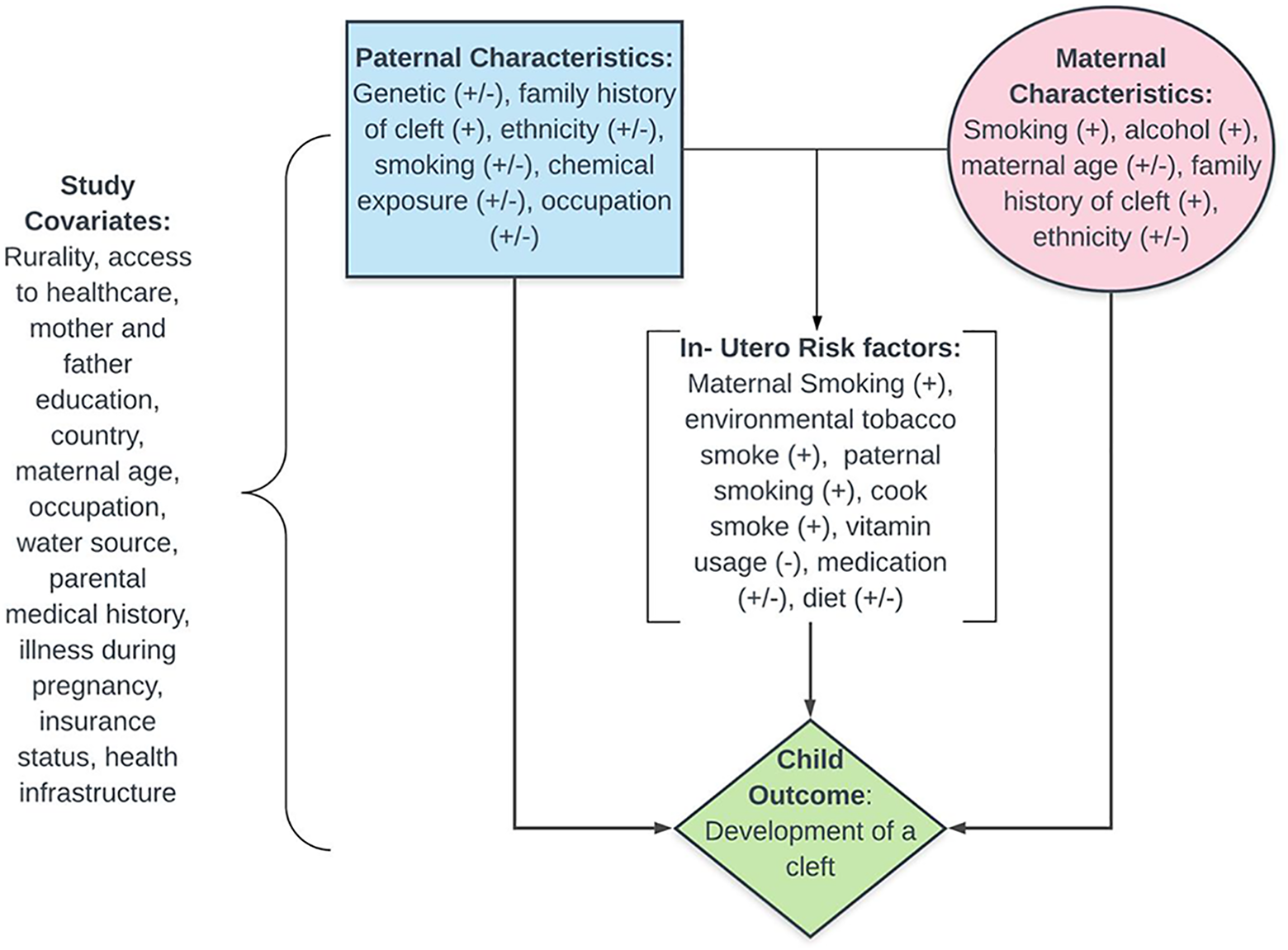
International Family Study (IFS) environmental and genetic study framework.
Operation Smile Partnership
The International Family Study was designed and is conducted by collaborators at USC, CHLA, and OS. An organizational chart for the IFS and OS program is shown in Figure 2. The epidemiology, biostatistics, laboratory, and other preventive medicine resources and personnel are based at the USC's Keck School of Medicine. Clinical study personnel and additional genetic laboratory resources are based at CHLA, which is an affiliated pediatric partner hospital of USC. These individuals make up the scientific portion of the core IFS study team, which is responsible for study design, data storage, genetic storage, IRB requirements, data analysis, and all other post-collection study needs. Multiple OS staff members dedicate time to work on the study, participate with the scientific team on field coordination, and manage related logistics. The OS teams (both international and local) are responsible for communicating and working with the local teams and partners.
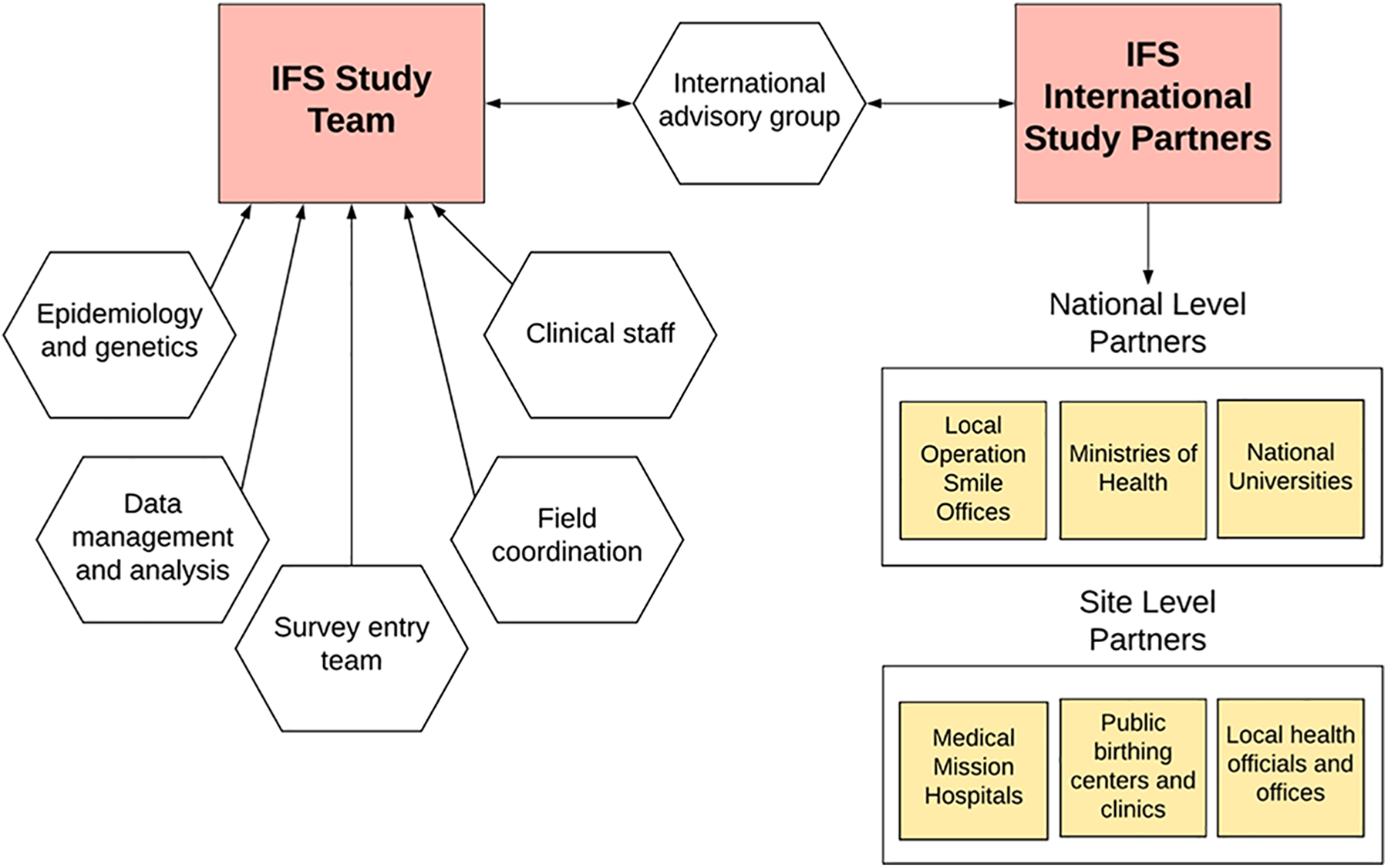
International Family Study (IFS) global structural organization chart.
In many OS mission countries, there is a local foundation functioning semiautonomously from the global organization. In these cases, there is often a large, dynamic in-country team with long-standing relationships and infrastructure that can support research. This is true for 7 of the 8 study countries (all but the DRC). Study countries are chosen based on both scientific and practical considerations by all stakeholders. The study team has chosen to work in what has been deemed a feasible number of the 27 currently active OS program countries, determined by staff, resources including available time, local infrastructure, presence of strong leadership, a participating volunteer base of research team members, and a representation of a regionally diverse group of LMICs. Allocating study resources to a smaller group of LMICs also strategically allows sample sizes to accumulate faster to enable country-level analysis. The project has both a full-time project coordinator and manager to ensure the teams work effectively and communicate weekly to meet research and quality control standards.
Case Definition
The International Family Study defines cases as any patients with nonsyndromic cleft lip and/or cleft palate (ICD10 35-37) presenting to an OS mission from ages 6 months to 4 years (ICD-10 Classifications of Mental and Behavioural Disorder: Clinical Descriptions and Disgnostic Guidelines, 1992). Nonsyndromic clefts are broken into 3 unique phenotypes: isolated cleft lip (iCL), isolated cleft palate (iCP), and CLP. Nonsyndromic orofacial clefts have a wide range of severity within phenotypes, ranging from a slight notch in the lip to the lip and palate being completely open. Cases were deemed nonsyndromic (as defined in Table 1) by credentialed OS clinical volunteers across a wide variety of specialties formally trained to work with patients with cleft.
Further eligibility criteria include that the biological mother or biological father of the child seeking medical care for cleft must be present to conduct the interview and provide informed consent for participation at the time of the OS mission. The consenting parent must be 18 years of age or older. A child is considered ineligible for IFS participation if the mother is pregnant, a biological sibling was born after the child-seeking treatment, the child has a clinically recognizable syndrome, the child is one of a multiple birth, or the child has a medical condition other than cleft. Criteria were selected to maximize the ability of the biological parent to recall the exposure window relevant to the most recent pregnancy with the child proband. If biological samples and interview data are only available for the biological father and the child (no maternal samples), the case-father dyads are used for genetic analyses and studies looking at paternal exposures, but they are excluded in studies based on maternal responses. Beginning in 2017, saliva samples were collected for OS mission children up to 7 years of age for inclusion in the genetic portion of the study only.
Case Ascertainment
Cases for IFS are recruited at selected OS surgical mission sites. The specific hospital used for each OS mission, and by default this study, is chosen based on its ability to meet the organization’s “Global Standards of Care.” Cases are considered representative of the region due to extensive OS community outreach. Recruitment methods differ slightly by country based on knowledge of geographical features and community structure established by local teams. The catchment area for study recruitment differs by country, and sometimes by region or province, based on the availability of specialized, affordable cleft care. Eligibility criteria were identical for case recruitment in all countries.
Control Definition
Control children are identified as any child born during an approximate 1- to 2-week window encompassing the mission date identified as a healthy newborn by local medical practitioners. Control child eligibility requires the child’s biological parent(s) to agree to participate in the study, complete the study interview (same questionnaire as case families), be 18 years of age or older, and provide informed consent for participation in the study. Consistent with case exclusion criteria, a child is considered ineligible for IFS participation if the mother is pregnant, the child has a syndrome or major birth defect (including cleft), or the child is one of a multiple birth.
Control Ascertainment
Multiple regional neighborhood, clinic, and hospital-based birth centers are identified prior to each mission by in-country OS partners to represent the catchment area of the OS mission and improve case–control comparability. All selected birth centers are public to better match demographics of the mission patients. Leadership is approached and debriefed on the study aims and procedures, and permissions are secured to recruit control families. Mothers are approached at the center by our study team members. Each site is visited daily or every other day during the mission to screen and recruit control families of healthy eligible newborns.
Study Area
The study area includes 8 different OS countries located across Asia, Africa, and Central America. The countries (number of unique sites, year of initial collection) in order that they were included are Vietnam (9 sites in 8 cities, 2012), Philippines (25 sites in 13 cities, 2012), DRC (7 sites in 1 city, 2012), Morocco (8 sites in 8 cities, 2014), Honduras (5 sites in 5 cities, 2013), Madagascar (5 sites in 4 cities, 2016), Nicaragua (2 sites in 1 city, 2016), and Guatemala (2 sites in 2 cities, 2018). These countries were chosen for the following reasons: (1) geographic diversity; (2) strong relationships with local stakeholders (both OS and medical volunteers) who support the research; (3) strong university, hospital, or government partners to obtain all necessary approvals; and (4) inclusion of OS countries underrepresented in current NSOFC literature. Additional detail including the collection of cities, hospital and university partners, and organizations utilized during these collections can be found in Supplemental Table 1.
Pre-Mission Control Site Recruitment
Beginning 1 year to 2 months prior to the mission, contact is made with the potential control sites (regional neighborhood, clinic, and hospital-based birth centers in the OS mission region) to assess the feasibility and appropriateness of each site for control recruitment, as well as to gauge interest in participating. Leadership from the selected sites are then invited to participate and debriefed on the study aims and procedures. Authorization from officials representing the local OS organization, center leadership, and/or Ministry of Health and related health agencies within the OS host country is obtained prior to recruitment.
Operation Smile Mission Recruitment Techniques
Operation Smile advertises its patient outreach extensively throughout local communities and often recruits hundreds of patients for its medical missions. This is done through radio broadcasting, television ads, flyers, billboards, and utilization of the local health system, among many other methods. Innovative techniques are being used by the organization to ensure that even remote regions are completely saturated with information and patients have the highest probability of accessing care. In Honduras, meter readers from the utility company, who visit every home in the country, were trained to recognize cleft conditions and provide contact information for OS follow-up when patients were identified. The reach of the recruitment initiatives depends on the country’s geography, the number of missions scheduled per year, and mission locations. In countries where medical missions happen throughout the year in multiple geographic sites, such as Vietnam, patients will be contacted by the organization about the next mission in their region. In Madagascar, where missions are less frequent, patients will often travel long distances to the mission because it is unknown if one will happen in their region in a timely window. Case recruitment initiatives often occur 1 year to at least 6 months prior to the mission to ensure regional saturation and prepare logistically. There are many countries where a patient tracking system has been established so patients are contacted about upcoming missions when they can most conveniently receive timely care.
Interviewer Recruitment and Training
International Family Study interviewers are selected from a previously identified volunteer pool wishing to support an OS mission. Specifically, they are chosen if they (a) can be present for the majority of the mission to volunteer, (b) have work experience with the study, (c) are proficient in English as well as their local language, and/or (d) have a medical or research background (such as medical or nursing students). Once they have been selected, they participate in a training conducted by the core study team member present on the mission. Interviewers are trained on how to assess each family for their eligibility, complete the consenting process, and explain the study to families, and how to record eligibility information for each family visiting the research station. The interviewers are then given copies of the questionnaire to familiarize themselves with the interview flow and the questions. The core study team member reviews questions with interviewers and procedures to ensure that each interviewer is able to conduct interviews appropriately with study families. Interviewers are trained in English and/or their native language.
Interviewers are also trained to collect saliva from the parents and child. Practice saliva kits are used to demonstrate how to properly collect saliva and the different strategies to ensure quality samples without cross-contamination for parents (spit kit) or children (swab). Interviewers are then trained on how to handle the collected saliva samples to ensure the samples and their corresponding questionnaires are properly linked. The core study team member is always present for both the saliva collection and questionnaire portions of the interview process to address any questions or issues that arise.
Interview Flow
The child and family proceed through clinical screening and recruitment as shown in Figure 3, which is the typical medical and comprehensive care screening for all OS surgical candidates in a mission setting. Clinical screening consists of creating a medical record, surgical evaluation, examination by anesthesiologists and pediatricians, vital signs taken by nurses, consultation from a speech language pathologist and child psychologist, and patient navigators who help guide the patients through the screening process. The research station follows clinical recruitment but is located prior to the phlebotomy station, which is only required for a subset of patients.
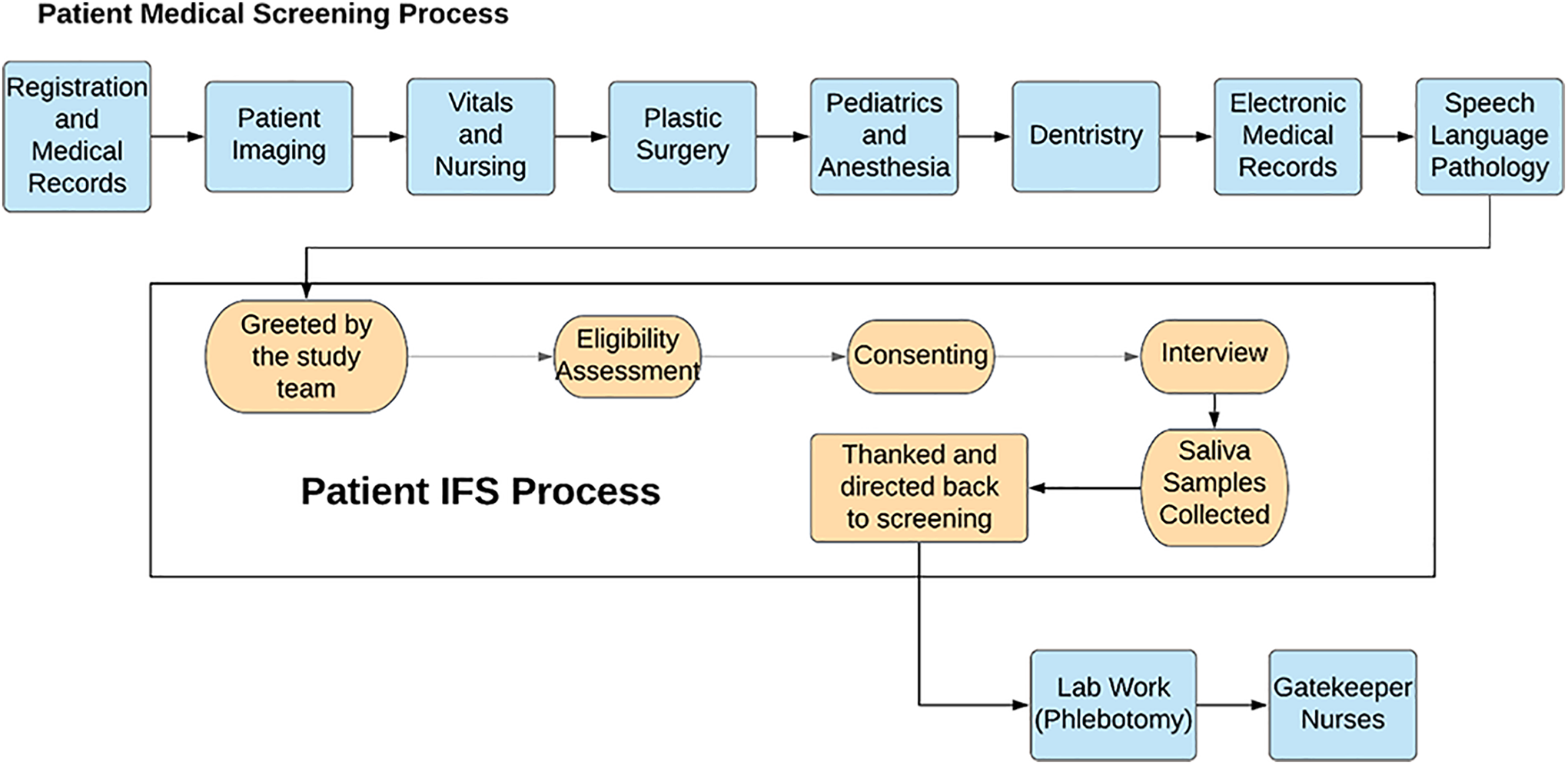
Operation Smile (OS) mission flow.
Case eligibility criteria are assessed by the core study team member. Eligible families are then assigned to an interviewer to complete the questionnaire. It is highly emphasized that their participation in the research study has no impact on the selection process for surgery with OS. Once families have agreed to participate in the study and are formally consented, they are brought into a private room or area whenever possible with an interviewer who can administer the questionnaire in the family’s native language. If both the mother and father of the patient are present, the interviewer tries to spend time with each parent individually. After the questionnaire is completed, the interviewer collects saliva samples from the mother, father, and proband. The barcodes from the saliva samples are added to the master data sheet, and parental questionnaires to ensure samples and questionnaire data can be linked without the use of identifiers.
Questionnaire Protocols and Procedures
Mothers are asked to complete a detailed interview, which takes approximately 40 minutes. Variables collected through the interview relevant to the etiology of cleft are summarized in Figure 1. All questionnaires, informed consents, and study materials have been translated and back-translated before being used in country. Extensive data are collected from the maternal questionnaire on family history of cleft (in the immediate and extended family on both sides), parental exposures prior to and during pregnancy (smoking, alcohol, diet, medical issues, medication, water usage, cooking behavior), child and parent demographic characteristics (age the child was born, pregnancy history, education, employment), and information about the father (smoking, alcohol, age the child was born, employment history and exposures, education). Questions are kept simple to decrease any potential mistranslation or confusion among both data collectors and study participants. Any individuals interested in obtaining a copy of the questionnaire or informed consent may contact the authors for a copy.
If the father is present, which is less common, he is asked to complete a short, 15- to 20-minute interview, which includes limited information on his medical history as well as environmental and behavioral characteristics. These characteristics include family history of cleft, smoking behaviors, alcohol intake, occupation, area of residence, education, and chemical exposures. The father is interviewed separately from the mother to increase the reliability of the response. The questions on the father questionnaire are written to a level of detail reasonable for the fathers to adequately respond.
Saliva Sample Protocols and Procedures
All biological samples are collected in the form of saliva using Mawi (Mawi DNA Technologies, ISWAB-DNA-250) and DNA Genotek (DNA Genotek, Inc, OGR-250, OGR-525, and OGR-575) collection kits. Adults are shown how to use the saliva tubes for collection and children are swabbed by a study member with gloves to ensure no contamination. Adults and older children (approximately 3+) most often have no issues providing samples. Saliva collection is often difficult in small children and babies due to contamination (if they were recently eating or drinking) as well as the realities of the setting; most cases have spent an entire day being screened for potential surgical care and are often agitated. Families are informed that if they are uncomfortable with providing a saliva sample, they are welcome to decline participation in this portion of the study and participate only in the questionnaire portion. The consent document ensures participants “To prevent this [loss of privacy], we will not put your name or other personal information on the surveys and saliva samples. Instead, they will be given a code number to protect your identity.”
Quality Control
There are multiple quality control measures taken throughout the interview, transportation, storage, and data entry processes of the study. A core study team member monitors all data collections.
Data Collection
Master data sheets are used to track study ID numbers, saliva kit numbers, cleft diagnosis information, and study eligibility criteria for each individual approaching the research station. This allows the study team to cross-check completed questionnaires with the master data sheets at the end of each day and again at the close of each mission to ensure all questionnaires are present and no duplicates have been collected. The study team also calculates participation rates and tracks the number of families who approach the research station but do not meet eligibility criteria.
Questionnaire Entry and Data Aggregation
All questionnaire data are entered by study staff trained in data management and computer programming. Data entry personnel are assigned to the study as interns for at least one year, ensuring familiarity with the questionnaire, acceptable responses, and other details that could be missed by untrained data entry personnel. Data are entered into Qualtrics, which has electronic quality control, including duplicate checks, built-in throughout the entry platform.
Genetic Data Storage and Processing
Saliva samples are shipped directly back to the university laboratory with all necessary approvals via courier from the respective mission sites to avoid issues with research personnel transporting biological samples out of the mission country or into the United States. Upon arrival, the saliva samples are inventoried, checked for the proper match against questionnaire data, and stored in the laboratory labeled with mission site and collection date. The location of each sample is kept in the study master file and database. All samples are stored in a temperature-controlled room at the pediatric hospital’s laboratory prior to extraction to increase the life of the stored saliva. Once DNA is extracted, the manifests are updated, and samples are stored in freezers monitored by the university’s Molecular Genetics laboratory.
Statistical Analysis
For this article, descriptive statistics were calculated to summarize cleft phenotype, country, year, maternal education, and paternal education separately for case or control. Chi-square tests for categorical data were used to assess differences between cases and controls using a conventional P = .05 for significance.
The most appropriate analysis for these aims will be logistic regression to analyze case–control data and estimate odds ratios with their 95% CI. Our proposed analyses to address the specific aims will include adjusted models based on knowledge of the literature, known confounders and interactions with respect to the main exposure of interest. Genetic analyses will be either case- and population-controls or case-trio data depending on the most appropriate comparison for the null hypothesis of independence between a genetic marker and case status. This will allow genome-wide association study (GWAS) analyses to look for common genetic variants in samples as well as exome or whole genome sequencing to test for association between aggregated rare variants and de novo mutations. The established environmental database, in combination with our genetic findings, will enable data from the IFS to be used to study a wide array of gene by environment interactions through traditional interaction analysis as well as 2- and 3-step approaches to preserve statistical power.
Results
From here forward, we reference only the 7 countries with data available, which excludes Guatemala. Table 2 briefly describes the complete IFS dataset through 2017. Between 2012 and 2017, there were 2955 (51.6%) case families and 2774 (48.4%) control families with a total of 11 946 individual saliva samples collected. The participation overall for cases was 90% and 85% for controls. The country-specific participations rates are (country [case %; control %]: Vietnam [86%, 93%], Philippines [92%, 82%], Congo [97%, 100%], Madagascar [93%, 90%], Morocco [90%, 72%], Nicaragua [77%, 85%], and Honduras [94%, 65%]). The majority of the data were collected in 2016 (25.6%), followed by 2017 (23.7%), 2015 (21.5%), 2014 (15.1%), 2012 (7.3%), and 2013 (6.8%). The highest yield countries were Vietnam (36.6%), the Philippines (21.4%), and Honduras (18.7%). Congo represents 8.7% of the IFS dataset, Madagascar 6.2%, Morocco 3.6%, and Nicaragua 4.7%. Most cases had the CLP phenotype (55.5%), followed by iCL (26.5%) and iCP (14.6%). We have collected 1020 (34.5%) case-trios compared to 467 (16.8%) control trios. Education was generally higher in the control group with 82.2% of control mothers compared to 65.3% of case mothers having a secondary education or higher (P < .01). This was also true among fathers with 78.7% of control fathers having a secondary education or more compared to 62.6% of case fathers (P < .01). A further breakdown of this information by country is provided in Supplementary Table 2.
Descriptive Characteristics of the Study by Case/Control Status Through December 2017.a
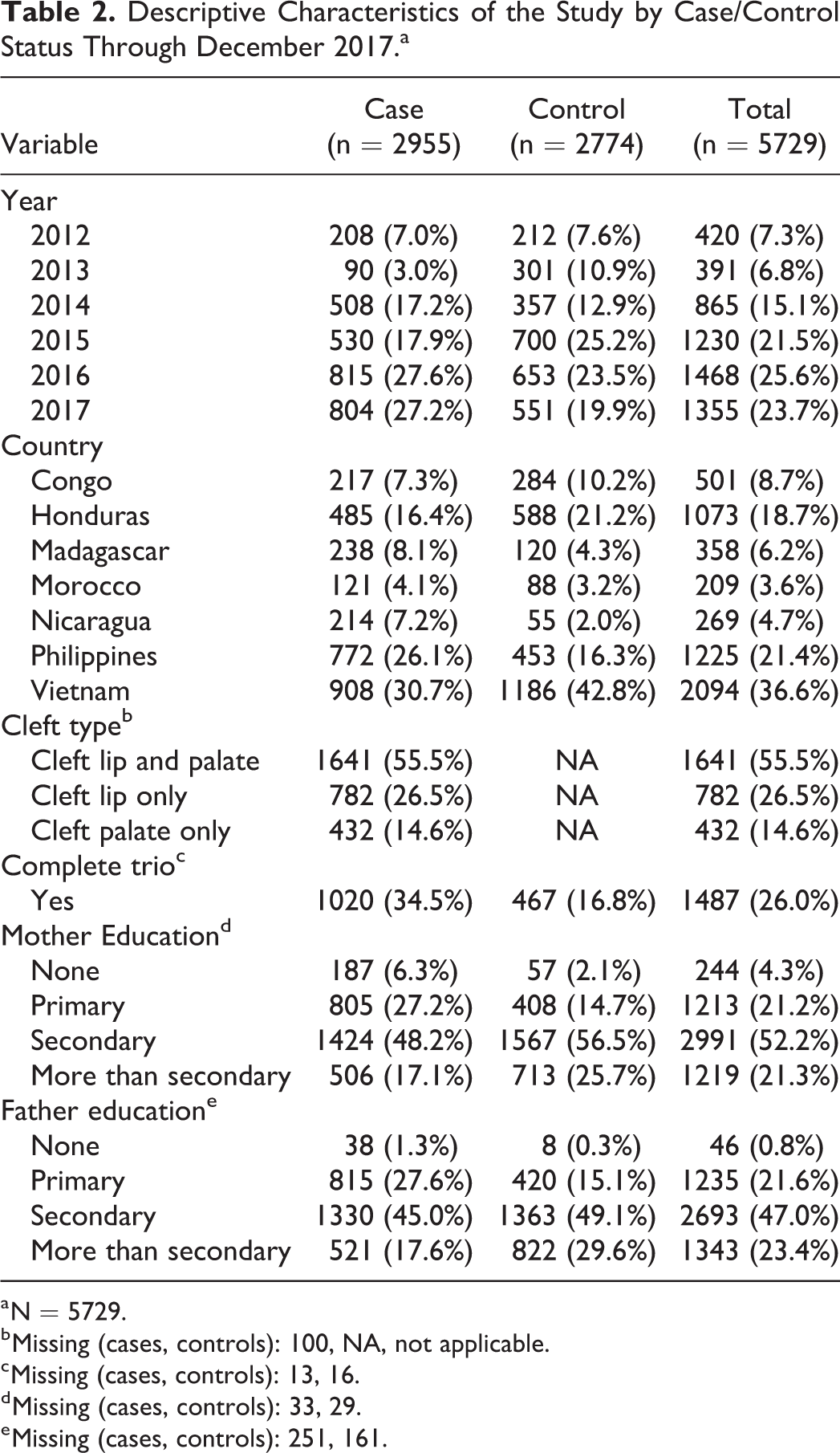
a N = 5729.
b Missing (cases, controls): 100, NA, not applicable.
c Missing (cases, controls): 13, 16.
d Missing (cases, controls): 33, 29.
e Missing (cases, controls): 251, 161.
Discussion
The International Family Study is the largest case–control study with a supplementary biobank, regionally sampled controls, and child–parent trios for exploration of etiologic questions around NSOFCs in a diverse group of LMICs with extensive population-wide recruitment. It was designed to have the unique ability to study genetic and environmental risk factors among racial/ethnic groups that have been underrepresented in the current literature, using the established network of a large, volunteer-based international nonprofit whose goal is to serve untreated children. Our study will be able to both explore populations not represented in the existing literature and strengthen and validate findings from other researchers who are working in similar populations (Zucchero et al., 2004; Beaty et al., 2010; Liu et al., 2016; Mbuyi-Musanzayi et al., 2018; Butali et al., 2018). The strengths of the study are the ability to reach LMICs that are underrepresented, an unconventional partnership between USC, CHLA, and OS, the large and diverse group of cases seen by OS, and the ability to study risk factors of NSOFCs that are highly prevalent in low-resource settings.
The key innovations include: Utilizing academic resources in collaboration with the networks and operational capacity of a large, international nonprofit organization allowing engagement of in-country stakeholders to ensure the success of the project. Providing a pathway for interviewers to join the core study team to improve local engagement, collect controls beyond the mission time frame if needed, and decrease study costs due to less travel being required from the United States. Creating a multiethnic database that can be used by other research teams to validate their findings and make comparisons by country, region, or cleft phenotype.
The data from this study can be used to identify unique environmental risk factors for cleft prevalent in low-resource areas and investigate genetic risk factors in multiple racial/ethnic groups underrepresented in cleft research. A recent paper by Sirugo et al. (2019) found that 78% of all GWAS studies are of European ancestry with only 2% African and 1% Hispanic or Latin American representation. Without studying populations like those included in IFS, not only will we be unable to fully understand cleft genetics but health inequalities will be further exacerbated as the era of personalized medicine continues to grow. This holds true for environmental exposures as well, which may not have been studied due to negligible prevalence in high-resource countries. For example, while maternal smoking may be an important contributor to cleft risk in westernized populations, in low-resource countries where few women smoke, the primary source of exposure to smoke-related agents and therefore attributable risk of NSOFCs, may be from biomass cooking (Mbuyi-Musanzayi et al., 2018; Auslander et al., 2020).
A potential limitation of this study is uncertainty about the representativeness of controls with respect to the base population for all environmental and lifestyle factors of interest. Controls are recruited from local birthing centers of similar socioeconomic status, restricted to similar age range, and sampled at the same time frame as case recruitment to improve comparability. Data were collected to control for other sources of selection bias in the analysis including household income, parental education, parental employment, and child’s place of birth (clinic, hospital, or home birth). Case-parent trios also extend our ability to explore genetic risk factors using a family-based design and help to better account for this possibility.
We used extensive, regional recruitment efforts to identify all eligible cases of cleft within the geographically targeted region. We recognize that the designation of population based is a difficult standard to meet and is likely to be achieved only for diseases or conditions identified in registries such as cancer or nested in population-based cohorts or clinical trials. We use the descriptor of “population-sampled” case–control study to distinguish our design from exclusively clinic-based or samples of convenience. Although we do not have adequate data to validate the representativeness of the final cases, the recruitment efforts of each mission were extensive through coordination with local government agencies, hospitals, and clinics to reach children and infants with unaddressed NSOFCs. Additionally, OS saturated the region in the months leading up to each mission by distributing recruitment materials through radio announcements, roadside banners, fliers, and direct conversations with local stakeholders. The organization’s 38-year history, well-recognized brand, and strong in-country relationships have made them a large part of the cleft landscape in all IFS countries ensuring a high likelihood of interaction with untreated patients with NSOFCs.
The potential of generalizing to the region or countries with a similar gross domestic product will continue to be explored further and has high potential to increase the impact of the IFS findings. There is also the possibility that subtle signs and symptoms of malformation syndromes may be missed, and these patients could be included in our study population. Through the screening process, patients were reviewed by multiple medical providers with expertise in cleft, increasing the likelihood for a syndrome to be identified.
In summary, IFS aims to fill a sizable gap in our current understanding of cleft risk factors because we cannot assume that the risk profile for NSOFCs is comparable between LMICs and high-resource regions. It is imperative to take risk factors specific to LMIC settings into account, as those individuals are at the highest risk of being unable to access care and live with the negative health consequences of this common craniofacial malformation. This information can inform public health interventions and education to potentially prevent disease in populations where care is sparse, and where children are most likely to feel the detrimental, lifelong medical and social effects of cleft. Modifiable, patient-centric solutions, such as providing a clean-burning cookstove, will be critical for efforts to decrease the burden of this malformation globally and improve lives around the world.
Supplemental Material
Supplemental Material, sj-pdf-1-cpc-10.1177_10556656211018956 - The International Family Study of Nonsyndromic Orofacial Clefts: Design and Methods
Supplemental Material, sj-pdf-1-cpc-10.1177_10556656211018956 for The International Family Study of Nonsyndromic Orofacial Clefts: Design and Methods by Allyn Auslander, Roberta McKean-Cowdin, Devin Feigelson, Frederick Brindopke, Melissa DiBona, Kathy Magee, Lili Arakaki, Rijuta Kapoor, Stephanie Ly, David V. Conti, Sylvia Rakotoarison, Fouzia Mahmoudi, Albertina McGregor, Melissa Giron, Angela Rose Hernandez, Thi-Hai-Duc Nguyen, Anselme Mwepu, Pedro A. Sanchez-Lara and William Magee in The Cleft Palate-Craniofacial Journal
Footnotes
Acknowledgments
The authors are indebted to the mothers, fathers, and their children who participated in this study as well as the local medical centers and staff who made this work possible. The authors also want to thank The Marguerite Foundation, The Rallis Foundation, The Sorenson Legacy Foundation, and the many CHLA donors for their support of this research. Our global research team is made up of countless volunteers who have worked with us over the years. Specifically, we’d like to thank our partners and volunteers from:
Operation Smile International
Vietnam: Thuy Bui, Hung Nguyen, Nguyen T. Thanh, Vu Nguyen
DRC: Nunda Misabe’o Pacifique, Tamlin Abrahams, Angie Sifa
Madagascar: Miadana Rakotondranaivo, Andriambelo Valisoa
Nicaragua: Julio Chamorro, Mayerlis Cuesta, Domingos Diogo
Philippines: Maria Irene Tangco, Leo Angelo Doble, Edith Y. Villanueva, Rhea Mae Magbanua, Vivina Chiu, Cathy Catubac, Jay-R Lado, Andrea Bernardino
Morocco: Othmane El Amouni
Honduras: Giannina Guell, Leticia Lopez, Seydi Sanchez, Paola Bustamante, Madelyn Ponce, Lastenia Zepeda, Maylin Herrera, Jeanie Barjum
Guatemala: Karen Acajabon, Alejandra Ortega
University of Southern California
Annie Pham, Yvette Estrada, Louella Gonzalez, Nikki Aflatooni, Chantel Aftab, Ashkan Nasr, Mahmoud Hamza
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded from a wide variety of philanthropic donors who played no role in the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
