Abstract
Stroke is a disease with a heavy social and familial care burden that can cause permanent brain damage, long-term disability, and/or death. This study aimed to determine the effect of technology-based health promotion training on the daily life activities, quality of life, and self-care of stroke patients. The study design was a Randomized Controlled Trial. The study sample included persons diagnosed with stroke diagnosed with stroke and were receiving inpatient treatment in the neurology clinic of a university hospital. The sample size was calculated as a total of 70 patients, 35 interventions and 35 controls. The intervention group patients received telephone-based education and follow-up grounded in Orem’s Self-Care Theory over a 12-week period subsequent to their discharge. The educational content was divided into three distinct categories: self-care needs with regard to health deviations, developmental self-care needs, and universal self-care practices. Data were collected using the Montreal Cognitive Assessment Scale, the Katz Index of Independence in Activities of Daily Living, Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale. The Independent Sample T-Test was used for intergroup comparisons, and the Dependent Sample T-Test was used for intragroup pre-test and post-test comparisons. Independent variables affecting the post-test scores, such as age and gender, were analyzed using the multiple linear regression model. The scale sub-dimension variables were compared using the multivariate analysis of variance test according to the groups. When compared with the control group patients after the training, it was determined that there was a statistically significant difference in the intervention group patients’ mean scores for the Stroke-Specific Quality of Life Scale (t = 11.136, p = .001) and the Exercise of Self-Care Agency Scale (t = 14.358, p = .000). Training interventions led to enhanced awareness and knowledge about stroke among the intervention group patients. They also fostered the development of healthier lifestyle behaviors and bolstered both self-care abilities and quality of life.
Introduction
Stroke is a disease that can cause permanent brain damage, long-term disability, and/or death (Centers for Disease Control and Prevention, 2022). It is the second most common cause of death according to the global health estimates of the World Health Organization (WHO) (Feigin et al., 2022). It is believed that 15 million people across the world have a stroke each year, of whom 5 million die and 5 million are permanently disabled (WHO, 2023). In many developed countries, the incidence of stroke has decreased as a result of better control of high blood pressure and reduced smoking (WHO, 2023). On the other hand, stroke has increased dramatically in low- and middle-income countries (Yan et al., 2016). In the 2017 data of the Turkish Statistical Institute, stroke ranked second after coronary heart disease for total deaths in Türkiye, and the frequency of cerebrovascular disease was 1.8% in men and 2.2% in women (Republic of Turkey, Ministry of Health General Directorate of Public Health, 2019).
Background
The maintenance of a healthy lifestyle and the control of risk factors is crucial in both the primary and secondary prevention of stroke. However, stroke patients may need better self-management and self-care skills, as well as caregiver and family support, in order to achieve optimal health outcomes (Yan et al., 2016). In the transition from hospital to home, it is necessary to support the independence and physical and emotional well-being of stroke patients (Lin et al., 2022). Post-discharge follow-up of patients can improve health outcomes (Lin et al., 2022; Yan et al., 2016). Ongoing education about the disease is a major determinant of gaining self-efficacy in stroke care (Lin et al., 2022).
Technology can be used in the management of care in stroke nursing (Camicia et al., 2021). Digital health technologies using tablets and smartphones are examples of this (Yan et al., 2016). The self-care skills of stroke patients can also be increased by providing education and counseling by telephone in a planned manner after discharge (Dennis et al., 2013). Stroke is a disease with high healthcare costs. By providing stroke patients with care, training and counseling services both in the clinic and after discharge (Chen et al., 2018), the burden of the disease on the healthcare system can be reduced (Dennis et al., 2013).
To the best of the authors’ knowledge, this is the first study in the north of Türkiye to determine the effect of post-discharge mobile application-based health promotion and support training on the quality of life and self-care abilities of stroke patients. This study thus provides evidence for the use of technological devices such as smartphones in the prevention and control of stroke. It also contributes to the expansion of international knowledge on this subject. At the end of the study, the effect of mobile application-based health promotion training on patients’ quality of life and self-care will be determined, and appropriate recommendations will be made about the strategies to be developed in this regard.
Conceptual Framework
The conceptual framework of this study is based on Orem’s Self-Care Theory. Orem defines self-care as “the actions performed by oneself in order to maintain one’s life, health and well-being” (Alligood, 2014; Masters, 2015). According to Orem, an individual must have the ability to meet their self-care needs (Alligood, 2014; Masters, 2015). Orem states that a nurse “can help the individual by acting or doing on behalf of the individual, guiding, supporting, creating an environment that enables development and education.” Therefore, nurses play an important role in increasing the individual’s self-care skills and ability (Alligood, 2014; Masters, 2015).
Improving the self-care skills of individuals with stroke is very important in terms of effective management of the disease and improving the quality of life. The WHO defines Quality of Life as “an individual’s perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns” (WHO, 2023). At the same time, through education and counseling, the ways in which stroke patients perform their daily life activities can be improved. Activities of daily living (ADL) can be defined as the basic skills required for independent self-care, such as eating, bathing and mobility (Katz, 1983). In this context, education and counseling provided to stroke patients and structured according to Orem’s Self-Care Theory may improve the self-care ability and the disease-related quality of life of stroke patients (Figure 1). Stroke-related complications may thus be prevented and re-hospitalizations avoided. In addition, this may contribute to the management of home care (Alijanpour et al., 2020; Aslani et al., 2016; Camicia et al., 2021).
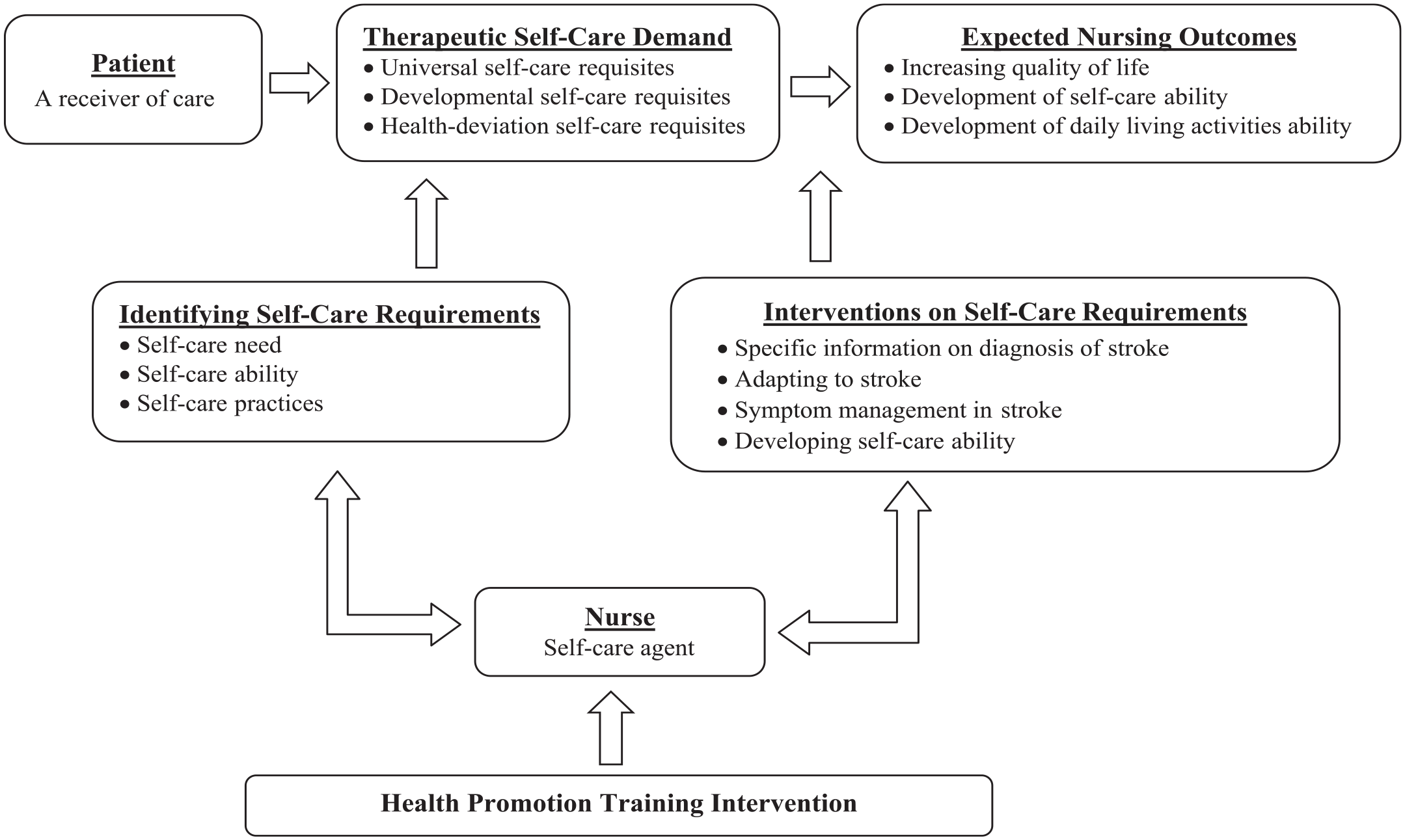
Theoretical framework of the health promotion training program.
Research Hypothesis
Hypothesis: After 12 weeks of mobile phone application-based educational support, there will be a difference between the scores of the intervention and control group patients for quality of life, self-care ability, and ADL.
Methods
Study Design and Sampling Method
The study was conducted as a longitudinal, randomized controlled study using the pre-test–post-test trial model. The Clinical Trial Registry number is NCT05484843. The consolidated standards of reporting trials were used to report the current study. It was carried out with the participation of patients who were diagnosed with stroke and were receiving inpatient treatment in the neurology clinic of a university hospital. The G*Power 3.1 program was used to determine the sample size in the study. The sample size was calculated as a total of 70 patients, 35 interventions and 35 controls, with a 95% confidence interval, 5% margin of error, 1.66 effect size, and 99.9% representation power.
Within the framework of this study, the intervention group patients received telephone-based education and follow-up grounded in Orem’s Self-Care theory. Notably, the clinic overseeing the study did not administer discharge training to the patients or furnish them with a training manual concerning the management of stroke-related ailments. On discharge, the patients received guidance solely on their follow-up appointments with the doctor, instructions regarding medication usage, and an all-encompassing epicrisis report detailing the procedures carried out during their hospital stay.
Inclusion and Exclusion Criteria
Those who had been diagnosed with hemorrhagic or ischemic stroke for the first time, were over the age of 18, literate, able to communicate, scored 21 or higher on the Montreal Cognitive Assessment (MoCA) Scale, could be reached by phone after discharge, and were willing to participate were included in the study. Patients who had a significant disease accompanying stroke (cancer, heart failure, etc.), were in the terminal period, had a mental illness or impaired consciousness, had a secondary stroke, had aphasia or dysphasia, had physical deficits that could greatly impact physical function, and did not complete the 12-week training period were excluded from the study.
Randomization
Simple randomization was used in the present study. Using a free computer program (www.randomizer.org), a randomized list of patients to be assigned to either the intervention and control groups was created. The patients were assigned to the intervention and control groups by an independent researcher not involved in the study. The research was conducted as a single-blind study due to the fact that the patients remained unaware of both the study hypotheses and their respective group assignments. Patients who met the inclusion criteria were invited to participate in the study before being discharged from the clinic. The patients in the both groups were asked not to disclose any information about the study to any other patients. A total of 158 patients was reached during the data collection process. Of these, 55 patients were not included in the study because they did not obtain the required score from the MoCA Scale, and 5 patients who met the research criteria did not want to participate in the study and were also excluded. A total of 60 patients were excluded (Figure 2).
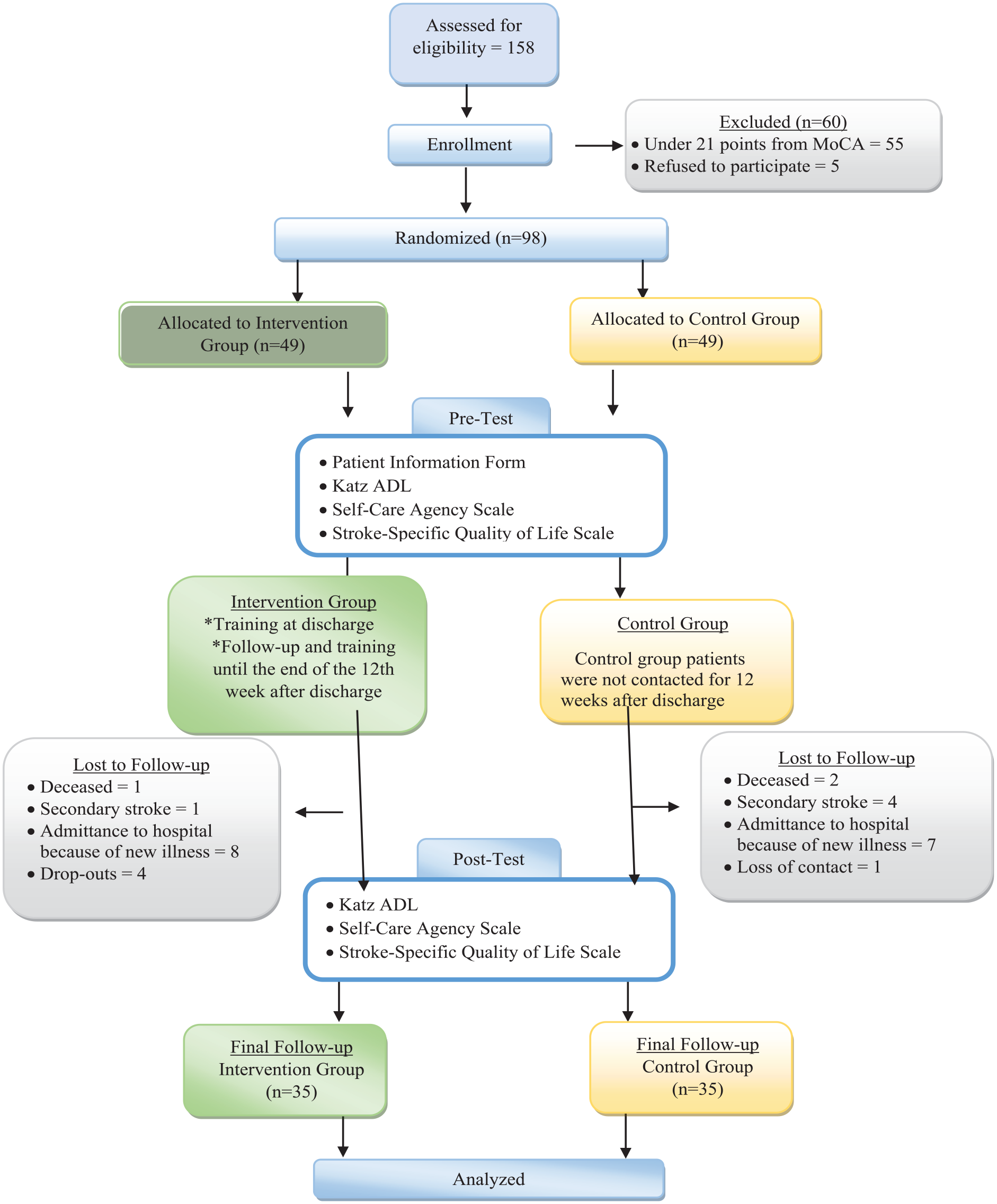
Study flowchart.
Intervention Procedures and Fidelity
Intervention Group
Following the randomization, eligible patients who met the inclusion criteria and achieved the requisite score on the MoCA Scale were enrolled into the intervention group. Those patients who expressed their willingness to participate provided written informed consent. Considering potential follow-up challenges post-discharge, the initially determined sample size of 35 was augmented by 40% to a total of 49.
The preliminary stage of the study involved crafting a training manual rooted in Orem’s Self-Care Theory, intended for delivery to the intervention group patients. This training manual was developed in adherence to evidence-based practice guidelines and manuals (Kleindorfer et al., 2021). To refine the manual, a panel of three stroke research experts critically reviewed and contributed suggestions, culminating in the final version. The panel also offered insights into the process of patient education.
The educational intervention’s foundational theoretical framework and components were aligned with Orem’s Self-Care Theory, drawing from clinical guidelines pertinent to stroke management. The research intervention protocol integrated components such as discharge education, a stroke education booklet, telephone follow-up, SMS reminders, the ability to contact the researchers if necessary, ADL, and aspects related to quality of life.
A dedicated room within the ward was arranged to facilitate the application of the measurement tools and the discharge education. Prior to discharge, the intervention group patients were met face to face in the clinic. During this session, baseline data were collected through the application of measurement tools, including the Patient Information Form, the Katz Index of Independence in Activities of Daily Living (Katz ADL), the Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale. Concurrently, the patients’ proficiency in performing ADL and self-care were evaluated. This assessment encompassed crucial factors influencing their ADL, self-care routines, quality of life, identified needs, and potential barriers. The preferred timing and day for receiving post-discharge education and counseling services were determined through discussion. The patients were also informed that they could contact researchers for queries outside the scheduled times, and the researchers’ contact details were shared for this purpose.
During the training sessions, one researcher (CB) assumed the role of the primary trainer, whereas the other researcher (ZK) participated in the sessions as an observer, abstaining from direct intervention. The observer researcher (ZK) used a comprehensive checklist to meticulously assess the training proceedings. This checklist was designed to gauge whether the training content, aligned with the stroke management framework outlined in the educational booklet prepared for the intervention group patients, was being appropriately conveyed at the intended level (Supplemental Table S1).
In accordance with the educational booklet (Figure 3), the intervention group patients received telephone-based educational and counseling sessions weekly until the culmination of the 12-week period. As part of the procedure, patients were sent SMS reminders the day before each weekly educational session, detailing the scheduled day and time of the forthcoming session.

Photos of patient’ education.
The educational content delivered to patients was divided into three distinct categories: self-care needs with regard to health deviations, developmental self-care needs, and universal self-care practices (Supplemental Table S1). The schedule was follows:
Week 1: Coverage of stroke-related concepts, encompassing definition of stroke, signs, symptoms, and the impact of stroke on bodily functions.
Week 2: In-depth exploration of stroke risk factors, including both modifiable and non-modifiable factors.
Week 3: Focus on expectations of recovery post-stroke, methods to enhance quality of life, hypertension management, and drug therapies.
Week 4: Emphasis on the significance of attending rehabilitation appointments, engaging in therapeutic exercises, and the role of physical activity.
Week 5: Discussion of the necessity of quitting smoking, addressing obesity, and implementing weight loss strategies.
Week 6: Stress management techniques, coping with stroke-related fears, and recognizing warning signs of secondary stroke.
Week 7: Prioritizing emergency intervention for stroke, managing chronic conditions beyond stroke, and emphasizing regular health checkups.
Week 8: Identifying individuals at high risk of stroke, managing high-risk behaviors, and ensuring adequate air and fluid intake.
Week 9: Dietary alterations to reduce fat and salt consumption, maintaining regular excretory functions, and adapting to daily life post-stroke.
Week 10: Fall prevention measures, infection prevention, oral hygiene, and maintaining body cleanliness.
Week 11: Strategies to cope with fatigue, the value of adequate rest and sleep, measures to prevent pressure sores, and sustaining overall well-being.
Week 12: Addressing post-stroke mental challenges, fostering a positive mindset, balancing being alone with social interaction, and promoting engagement in social activities and hobbies.
Considerations were given to the educational levels, cultural characteristics, and individual differences of the patients throughout the training and subsequent monitoring phases. Broadly speaking, it was ascertained that the patients’ needs regarding educational material matched the material contained in the instructional handbook. The same educational content was administered to all patients during the educational program, and no circumstances were encountered necessitating the revision of the education and training at any time while it was being implemented. The training was provided weekly for 12 weeks, with each session lasting around 30 minutes. Meticulous attention was devoted to providing the training and monitoring the patients during periods when their general well-being was good and they were most receptive to learning. In situations where the patient was not prepared to engage with the program, a joint decision was made to identify an alternative day and time within the same week for the training session. The procedures applied to the intervention group patients included post-discharge weekly phone calls, telephone-based training, a booklet, and visual materials (figures, images) (Supplemental Table S2).
In the training and discussion with the intervention group patients, a variety of techniques were used, including question–answer, demonstration, discovery, problem-solving, inference, linking with prior knowledge, comparison, and cooperation. At the end of the 12-week period, the examiner researcher administered post-assessment tests (including the Katz ADL, the Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale) during the evaluations at the outpatients’ clinic, thereby bringing the study to an end. Throughout the period of patient education and follow-up, no unforeseen or unexpected circumstances occurred.
During the research process one patient died during the follow-up period, one patient had a secondary stroke, eight patients were rehospitalized due to another disease, and four patients decided to withdraw from the study. Within the 12-week follow-up period, 35 of the patients in the intervention group finished all of the components of the training program. Their health status was then examined. The analysis did not include the 14 patients who were in the intervention group at the beginning of the study, but who it was not possible to follow up for 12 weeks after discharge and who thus did not complete all the components of the training.
Control Group
In accordance with the inclusion criteria and the randomization, individuals within the control group who attained the requisite score on the MoCA scale were incorporated into the study. Written informed consent was obtained from those patients who willingly agreed to participate. To guard against potential risks, the originally determined sample size of 35 for the control group was augmented by 40% (n = 49), taking into account the anticipated attrition of patients post-discharge.
During the initial face-to-face interaction with the control group patients before their discharge, baseline data were collected through the application of the measurement instruments (Patient Information Form, Katz ADL, the Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale). Following discharge, there was no active communication with the control group patients for 12 weeks, and no interventions were carried out during this period. Post-assessment tests (the Katz ADL, the Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale) were administered when the control group patients’ attended the outpatients’ clinic at the conclusion of the 12th week, bringing the study to an end. The final follow-up was completed with 35 patients. During the research process two patients died during the follow-up period, four patients had a secondary stroke, seven patients were re-hospitalized due to being diagnosed with another disease, and one patient could not be reached by mobile phone after 12 weeks. To mitigate any ethical issues arising from the difference in how the intervention and control group cohorts had been treated, the researcher responsible for the study conducted training sessions for the control group patients after the study had ended. During these sessions, any questions that the patients in this group had were addressed and resolved.
Outcome Measures
The data were collected by the researchers using the Patient Information Form, the MoCA Scale, the Katz ADL, the Stroke-Specific Quality of Life Scale, and the Exercise of Self-Care Agency Scale. The Patient Information Form and scales were pilot-tested on a group of five patients. Patients in the pilot study were not included in the sample.
Patient Information Form
The Patient Information Form, prepared by the researcher, included questions about the socio-demographic and clinical characteristics of the patients.
MoCA Scale
This scale was developed by Nasreddine et al. (2005) and adapted into Turkish by Selekler et al. (2010). It was developed as a rapid screening test for mild cognitive impairment. The scale assesses different cognitive functions, including attention and concentration, executive functions, memory, language, visual construction skills, abstract thinking, calculation, and orientation. Applying the scale takes approximately 10 minutes. The lowest score that can be obtained is 0, and the highest score is 30. In its initial formulation by Nasreddine et al. (2005), the MoCA had a threshold of 26 as a cut-off score. Selekler et al. (2010), in their study of the Turkish version of this scale, established the cut-off score as 21. Consequently, MoCA scores of 21 and higher are categorized as indicative of cognitive adequacy in this regard. Divergent thresholds emerge in variations of the MoCA: the Korean version adheres to a cut-off point of 22/23, whereas in other Far Eastern nations (such as China, Japan, Thailand) and Western counterparts (Canada, England, France, Germany, Denmark), the benchmark is set at 26/30. The deviation from the original version in the Turkish MoCA’s cut-off score is attributable to potential cultural and educational influences, as noted by Selekler et al. (2010). They determined the Cronbach’s α reliability coefficient of the MoCA Scale as .83. In this present study, the Cronbach’s α reliability coefficient of the scale was .79.
Katz Index of Independence in Activities of Daily Living
The Katz ADL was developed by Katz et al. (1963) and adapted into Turkish by Pehlivanoğlu et al. (2018). It evaluates the dependence/independence of the individual in performing ADL. The scale consists of six subheadings: bathing, dressing, going to the toilet, carrying, continence, and feeding. Scores of 0 to 6 points are evaluated as “dependent,” of 7 to 12 points as “semi-dependent,” and of 13 to 18 points as “independent.” Pehlivanoğlu et al. (2018) determined the Cronbach’s α reliability coefficient of Katz ADL as .83. In this study, the Cronbach’s α reliability coefficient of the scale was determined as .85.
Stroke-Specific Quality of Life Scale
This scale is a 5-point Likert-type scale which was developed by Williams et al. (1999) and adapted into Turkish by Yönt and Khorshid (2012). The scale was developed to assess the quality of life of stroke patients. The Turkish version consists of eight sub-dimensions and 48 items, including activities, energy, temperament, social and family role, vision, language, thinking, and personality traits. A high total score on the scale indicates a high quality of life, and a low score indicates a low quality of life. Yönt and Khorshid (2012) determined the Cronbach’s α reliability coefficient of the Stroke-Specific Quality of Life Scale to be .97), while in this study the Cronbach’s α reliability coefficient of the scale was .92.
Exercise of Self-Care Agency Scale
The Exercise of Self-Care Agency Scale is a 5-point Likert-type scale developed by Kearney and Fleischer (1979) and adapted into Turkish by Nahcivan (2004). The scale is used to measure an individual’s self-care ability or self-care ability and is focused on individuals’ self-assessment of their involvement in self-care. It consists of 35 items. The maximum score that can be obtained from the scale is 140. A high score obtained from the scale indicates a high level of self-care or self-care ability. The scale has no cut-off value. Nahcivan determined the Cronbach’s α reliability coefficient of the scale to be .89. In this study, the Cronbach’s α reliability coefficient of the scale was found to be .85.
Data Analysis
The study data were analyzed using the IBM SPSS Statistics 28 package program. Frequencies (number, percentage) were given for categorical variables and descriptive statistics (mean, standard deviation) were given for numerical variables. The normality of the numerical variables was analyzed by the skewness and kurtosis coefficients and the coefficients were within the range of ±1.5 and showed normal distribution. Therefore, parametric statistical methods were employed in the study. The chi-square test and Independent Sample T-Test were used to compare the descriptive characteristics of the intervention and control groups. The Independent Sample T-Test was used for intergroup comparisons, and the Dependent Sample T-Test was used for intragroup pre-test and post-test comparisons. Independent variables affecting the post-test scores, such as age and gender, were analyzed using the multiple linear regression model. The scale sub-dimension variables were compared using the multivariate analysis of variance (MANOVA) test according to the groups. The variance-covariance assumption was satisfied since the data were regularly distributed. Cohen’s d effect size criterion was employed to ascertain the magnitude of the effect. Subsequent to the research, an assessment of whether the sample size adequately accommodated the measurements was conducted using Bartlett’s test and the KMO sample measurement value adequacy test. The statistical significance level in the analyses was 0.05.
Ethical Considerations
The study was implemented after written permission was obtained from the Clinical Research Ethics Committee of Ondokuz Mayıs University (Nr: B.30.2.ODM.0.20.08/1856). To collect the data, written informed consent was obtained from the university hospital (Nr: 15374210-302.08.01-E.376).
Results
The study was completed with a total of 70 patients. The mean age was 61.59 ± 9.95 years and the mean monthly income was 4707.14 ± 1285.29 Turkish Lira. Most of the patients were married (n = 64, 91.4%), high-school graduates (n = 21, 30.0%), had social security (n = 69, 98.6%), were unemployed (n = 43, 61.4%), were retired (n = 29, 41.4%), lived in a city (n = 36, 51.4%), had children (n = 66, 94.3%), and lived with their children and spouse (n = 34, 48.6%). Most of the participants had ischemic stroke (n = 47, 67.1%) and had a chronic disease other than stroke (n = 65, 92.9%). The baseline characteristics of the patients in the intervention and control groups were compared and no statistically significant difference was found between the groups (p > .05, Supplemental Table S3).
The statistical analysis revealed that the mean total scores for the post-test evaluations on the Stroke-Specific Quality of Life Scale exhibited notable increase (pre-test = 2.61 ± 0.39; post-test = 3.94 ± 0.31; p = .000). A similar pattern was observed in the scores of the Exercise of Self-Care Agency Scale (pre-test = 61.80 ± 14.53; post-test = 104.06 ± 9.85; p = .000) and the Katz ADL (pre-test = 14.00 ± 2.70; post-test = 15.46 ± 2.12; p = .000). Notably, all these post-test mean total scores exhibited statistically significant increases in comparison to their respective pre-test counterparts (Supplemental Table S4).
In the intervention group, the mean pre-test score for the Stroke-Specific Quality of Life Scale was 2.61 ± 0.39 (Supplemental Table S5) and the mean post-test score was 3.94 ± 0.31 (Supplemental Table S7); the mean pre-test score for the Exercise of Self-Care Agency Scale was 61. 80 ± 14.53 (Supplemental Table S5) and the mean post-test score was 104.06 ± 9.85 (Supplemental Table S7); the Katz ADL mean pre-test score was 14.00 ± 2.70 (Supplemental Table S5) and the mean post-test score was 15.46 ± 2.12 (Supplemental Table S7).
In the control group, the mean pre-test score for the Stroke-Specific Quality of Life Scale was 2.78 ± 0.47 (Supplemental Table S5) and the mean post-test score was 2.92 ± 0.44 (Supplemental Table S7); the mean pre-test score for the Exercise of Self-Care Agency Scale was 65. 34 ± 13.59 (Supplemental Table S5) and the mean post-test score was 64.91 ± 12.77 (Supplemental Table S7); the Katz ADL mean pre-test score was 14.74 ± 2.45 (Supplemental Table S5) and the mean post-test score was 16.40 ± 1.48 (Supplemental Table S7).
In Supplemental Table S6, since the sub-dimension variables of the pre-test Stroke-Specific Quality of Life Scale were related to each other, the data were normally distributed and the variance-covariance assumption was met, the intervention and control group sub-dimension variables were compared using the MANOVA test. It was determined that all sub-dimension variables of the pre-test Stroke-Specific Quality of Life Scale showed statistically significant differences between the groups. Before the training, the total and sub-dimension scores from the Stroke-Specific Quality of Life Scale of the control group were higher compared to the intervention group (p < .05) (Supplemental Table S6).
According to these results, there was a statistically significant increase in the post-training, Stroke-Specific Quality of Life Scale (t = 11.136, p = .001) and Exercise of Self-Care Agency Scale (t = 14.358, p = .000) mean scores of the intervention group patients compared to the control group patients (Supplemental Table S7). It was seen that all sub-dimension variables of the post-test Stroke-Specific Quality of Life Scale except the Vision sub-dimension showed statistically significant differences between the groups. In Supplemental Table S8, since the sub-dimension variables of the post-test Stroke-Specific Quality of Life Scale were related to each other, the data were normally distributed and the variance–covariance assumption was met, the intervention and control group sub-dimension variables were compared using the MANOVA test. It was determined that all sub-dimension variables of the post-test Stroke-Specific Quality of Life Scale showed statistically significant differences between the groups. After the training, the total and sub-dimension scores from the Stroke-Specific Quality of Life Scale of the intervention group were higher compared to the control group (p < .05) (Supplemental Table S8).
Although the post-training Katz ADL scores of the intervention group patients were lower than the scores of the control group patients (Figure 4), a statistically significant increase was found when the pre- and post-training Katz ADL scores of the intervention group patients were compared (t = −5.278, p = .000) (Supplemental Table S9).
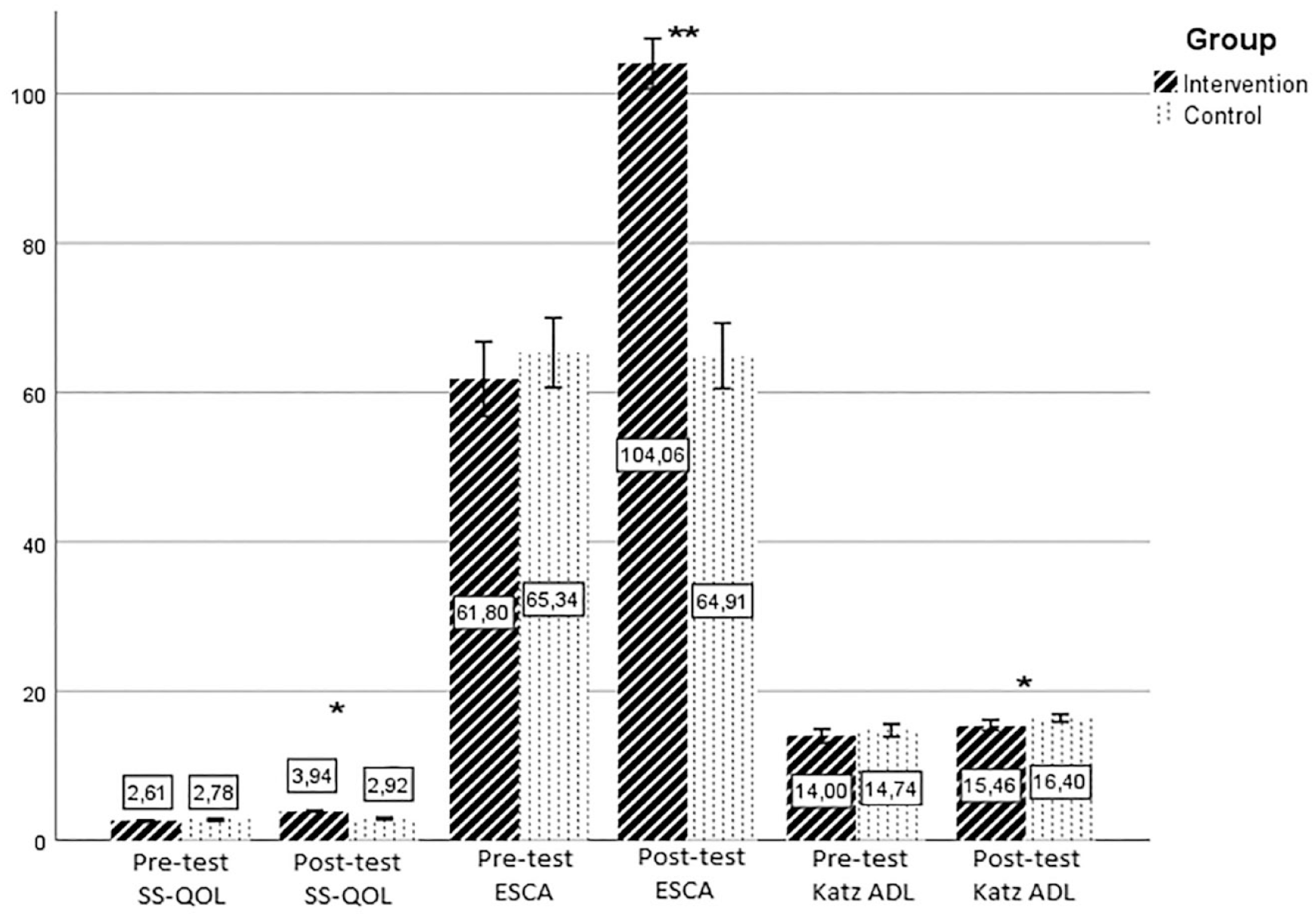
Pre- and post-training, SS-QOL, ESCA and Katz ADL Scores of the intervention and control group patients.
The multiple linear regression models established to examine the effect of age and gender on the post-test Katz ADL (F = 0.100; p = .905), Stroke-Specific Quality of Life Scale (F = 0.212; p = .810), Exercise of Self-Care Agency Scale (F = 0.117; p = .890) scores in the intervention group patients were not statistically significant (Supplemental Table S10).
The impact of the patient education and follow-up approach on the baseline data was assessed using Cohen’s d effect size criterion. In accordance with established conventions, a Cohen’s d value below 0.2 signifies a weak effect size, while a value of 0.5 indicates a moderate effect size, and a value exceeding 0.8 can be construed as a strong effect size, as delineated by Kılıç (2014). In this study, the calculated Cohen’s d effect size for the Katz ADL was 0.51, indicating a moderate difference between the intervention group and control group, suggesting that there was a meaningful practical impact. The calculated Cohen’s d effect size for the Exercise of Self-Care Agency Scale was 3.43, indicating a substantial difference between the intervention group and control group, suggesting a highly significant and robust impact. The calculated Cohen’s d effect size for the Stroke-Specific Quality of Life Scale was 2.68, indicating a substantial difference between the intervention group and control group, suggesting both a statistically significant and practically significant impact (Supplemental Table S11).
At the conclusion of the study, Bartlett’s test and the KMO sample measurement adequacy test were employed to evaluate the adequacy of the sample measurements. Notably, the KMO test stipulates a lower limit of 0.50, below which the dataset is unsuitable for factorization (Field, 2009). On the basis of this criterion, it was anticipated that Bartlett’s test would yield a significant result, while the KMO test would exhibit a value exceeding 0.50.
For the Stroke-Specific Quality of Life Scale, the computed KMO value of 0.755 affirmed the sufficiency of the sample size for data analysis. Similarly, although the Bartlett's test yielded a significant chi-square value, χ2(1128) = 5881.361; p < .001, the data’s reliability can be considered quite high, given the Cronbach’s α value of .971.
Likewise, the Exercise of Self-Care Agency Scale displayed a KMO value of 0.761, indicating adequate sample size for analysis. Examining the Bartlett’s test results, a notable chi-square value, χ2(595) = 225.756; p < .001, emerged. In light of this, the data’s reliability is high, as evident from the Cronbach’s α value of .952 (Supplemental Table S12).
Similarly, the KMO value for the Katz ADL was determined as 0.695, indicating sufficient sample size for data analysis. Furthermore, the significant chi-square value from Bartlett’s test, χ2(15) = 136.747; p < .001, underscores the data’s reliability, which is confirmed by the Cronbach’s α value of .775 (Supplemental Table S12).
Discussion
Due to the protracted and challenging nature of recovery from stroke, the transition from hospital to home can impose significant stress on both patients and their relatives. The post-discharge support services provided may sometimes be inadequate (Bretz et al., 2014; Cheng et al., 2011; Lin et al., 2020; Lo et al., 2016; Sit et al., 2016). Consequently, the recommendations outlined in secondary stroke prevention guidelines often underscore alterations in lifestyle risk factors, along with the necessity of active education and behavioral counseling. These measures may entail making informed changes to one’s behavior, incorporating self-care, and interventions designed to teach self-management, all facilitated through education and subsequent monitoring (Kleindorfer et al., 2021; Lennon et al., 2018).
Within the framework of this study, a telephone-based education and follow-up intervention rooted in Orem’s Self-Care Theory was administered to the intervention group patients. This approach led to improved knowledge and awareness concerning stroke disease management among the patients. While several studies have focused on patient education and follow-up programs based on other theories, such as the Chronic Disease Care Model, Bandura’s Self-Efficacy Theory, Health Empowerment Theory, and various stroke-related guidelines, the use of Orem’s Self-Care Theory here remains relatively distinct (Cheng et al., 2011; Kalav et al., 2022; Lin et al., 2022; Lo et al., 2018; Wan et al., 2016).
It is evident that theory-guided stroke programs have proven beneficial in bolstering patients’ ability to take care of themselves, and increasing their ability to monitor and manage their own health status and level of functioning (Chen et al., 2018; Cheng et al., 2011; Lo et al., 2013, 2018; Wolf et al., 2016). For those with limited access to healthcare services in particular, theory-based stroke programs can shorten the recovery time (Dennis et al., 2013). Such programs can empower stroke patients to adopt healthier lifestyles, enhance their ADL, and augment their overall quality of life (Alijanpour et al., 2020; Aslani et al., 2016).
Although Türkiye lacks a specific program aimed at preventing secondary strokes, the integration of theory-based health promotion and supportive programs, including telephone education and follow-up, within the healthcare system is deemed crucial and impactful.
Various studies have underscored the positive impact of post-discharge follow-up and educational programs on stroke patients and demonstrated their contribution to improved clinical outcomes. Such interventions have been associated with improvements in patients’ knowledge regarding stroke, self-management behaviors, self-efficacy, self-care abilities, ADL, quality of life, functional capacities, and adherence medication. Furthermore, training and education make it easier to detect the signs and symptoms of recurrent stroke at an early stage, consequently reducing hospitalizations (Chau et al., 2019; Chen et al., 2018; Huang et al., 2021; Kalav et al., 2022; Lin et al., 2020, 2022; Magwood et al., 2020; Sarfo et al., 2018; Si et al., 2021; Sit et al., 2016; Wan et al., 2016; Xu et al., 2021).
By maintaining consistent telephone communication with patients, real-time updates on their health status can be swiftly obtained. This approach also makes it easier to transmit essential reminders and information about their condition (Chen et al., 2018; Eames et al., 2013; Irewall et al., 2015; Kalav et al., 2022; Lin et al., 2020; Lo et al., 2018; Wan et al., 2016; Wong et al., 2015). In the present study, the intervention group patients received approximately 30 minutes of stroke management education and counseling once a week, extending until the 12th week post-discharge. This intervention led to heightened self-care abilities and improved the stroke-specific quality of life scores among the patients by the 12th week.
The increase in post-test scores for stroke quality of life, self-care, and ADL in the intervention group could be because the patients who exited the study during the training and follow-up phases were those who were more ill or had poorer functional status. Similar to our findings, Wan et al. (2016) reported a dropout rate of around 20%, with 11 out of 91 subjects lost to follow-up, underscoring the potential impact of selection bias on research outcomes. Consequently, it is imperative to conduct future investigations on this subject with larger sample sizes, whether based on Orem’s Self-Care Theory or alternative theoretical frameworks, to yield more robust conclusions.
The duration of telephone-based follow-up and counseling varies in different studies. Follow-up periods spanning 3, 6, and 12 months are often recommended to track changes over time following patient training (Lo et al., 2018; Wan et al., 2016). However, any form of telephone-based intervention holds substantial potential for effectiveness in nurse-guided care after stroke (Sarfo et al., 2018). Similarly, a telephone-based lifestyle coaching system was found to complement standard treatment and help reduce cerebro-cardiovascular risk factors (Spassova et al., 2016). Providing continuous nursing care through mobile phone applications has also shown promise in enhancing self-efficacy, quality of life, and motor functions among stroke patients in community settings (Xu et al., 2021).
In the study conducted by Wan et al. (2016), three telephone follow-up interviews were conducted with patients in the 1st week, 1 month, and 3 months after discharge. Data were collected both before discharge and at the 3rd and 6th months post-hospitalization. The intervention group patients exhibited higher medication compliance compared to the control group. Similarly, Kalav et al. (2022) found that a 3-month telephone education and follow-up program increased knowledge about stroke, fostered improved healthy lifestyle behaviors, self-management, and quality of life, and ultimately led to increased satisfaction levels among patients. In another study by Wong et al. (2015), a 4-week transition period from hospital to home, including telephone follow-up, resulted in elevated Barthel Index scores and increased satisfaction among intervention group patients compared to the control group. Additionally, this intervention was linked to a decrease in the rate of hospital readmissions and emergency department usage.
In a study conducted by Chen et al. (2018), patients received discharge training followed by a 4-week period of telephone follow-up. This approach yielded notable improvements in both self-efficacy and ADL among the intervention group patients, observed 3 months post-discharge, in contrast to the control group patients. Similarly, a study carried out by Lo et al. (2018) introduced a self-efficacy-based stroke self-management program encompassing a home visit, a group meeting, and three subsequent telephone follow-ups. This comprehensive intervention demonstrated its efficacy by enhancing patients’ self-efficacy, outcome expectancy, and satisfaction with their execution of self-management behaviors, marking significant progress within an 8-week timeframe.
Irewall et al. (2015) discovered that nurse-led telephone-based secondary preventive follow-up yielded superior results compared to the usual care in terms of enhancing medical adherence, reducing blood pressure, and lowering LDL-C levels 12 months post-discharge. A study by Eames et al. in 2013 reported that education, comprising an information booklet and telephone-based verbal support before and for 3 months after discharge, resulted in elevated self-efficacy among patients in accessing stroke-related information. Lin et al. (2020) conducted a study demonstrating that a 12-week program involving weekly telephone support after discharge and face-to-face coaching sessions every 2 weeks brought about improvements in patients’ health and self-efficacy by both the 12th and 24th weeks.
The implementation of “health empowerment” education and counseling has emerged as a pivotal factor in shaping stroke patients’ ability to care for themselves (Lin et al., 2020, 2022). The process of health empowerment helps patients to take an active role in managing their own health, increasing their sense of control over their lives, ensuring continuity of care, and improving their self-confidence (Sit et al., 2016). Health professionals’ roles in delivering appropriate care and ensuring its continuity is crucial for supporting self-management in stroke patients (Kuo et al., 2021; Si et al., 2021). Optimizing stroke patients’ capacity to manage their own condition is of paramount importance (Lo et al., 2016).
Nurses have significant responsibilities, encompassing education, rehabilitation, and fostering patient independence. Self-care training for stroke survivors is a transformative process, enhancing patients’ ability to function and transitioning them from a state of dependency on others to self-reliance. When patients are better able to care for themselves, their resilience in facing stressors associated with their condition may also be heightened (Kalav et al., 2022).
Limitations
The current study has several limitations that need to be acknowledged. First, due to the lack of blinding of the researchers to the allocation of patients into intervention and control groups, the study could not adhere to the standard of a double-blind randomized controlled trial. Instead, the research was executed as a single-blind randomized controlled study, wherein patients were kept unaware of their assigned groups and with the study’s hypothesis. Nevertheless, the lack of blinding among the researchers introduces a potential threat to the study’s internal validity. Second, the study involved literate patients who were experiencing their first stroke. Excluding illiterate patients, those with secondary strokes, and individuals with significant physical impairments affecting their functionality, raises uncertainties regarding the applicability of telephone-based health promotion training to patients in these specific categories. As such, the study has to remain silent on the effectiveness of the intervention for these subgroups. A third limitation pertains to the absence of a stroke severity assessment scale within the study's methodology, which means that there is a gap in the characterization of the severity of the participants’ strokes. The study’s fourth limitation stems from the sole involvement of a single researcher in delivering the training program. This precludes standardization, as it remains uncertain whether the intervention could be consistently applied by multiple researchers. The fifth limitation lies in the statistically significant increasing post-test stroke quality of life, self-care, and ADL scores within the intervention group, surpassing the pre-test mean score. This trend may have been because the patients who discontinued participation during the training and follow-up phase were predominantly those facing severe illness or impaired functionality. Another limitation arises from only assessing the training program's impact at 12 weeks post-discharge. The absence of long-term evaluation means that it was not possible to achieve a comprehensive understanding of the program's extended effectiveness. Lastly, the study’s scope was confined to the neurological service of a single hospital, thus limiting the generalizability of findings to a broader context.
Conclusion
In this study, interventions encompassing follow-up, education, and counseling were administered weekly for 12 weeks post-discharge, guided by Orem’s Self-Care Theory. These interventions led to enhanced awareness and knowledge about stroke among the intervention group patients. They also fostered the development of healthier lifestyle behaviors and bolstered both self-care abilities and quality of life. In future research in this area, it is advised that interventions be grounded in established theories. Additionally, conducting double or triple-blinded studies with a multidisciplinary team will enhance the internal validity of randomized controlled trials. Given the likelihood of patient attrition during follow-up, the use of larger sample sizes should be considered. Employing a standardized scale to measure stroke severity would enhance the comprehensiveness of any future study. Monitoring the effects of interventions for durations exceeding 3 months would provide insights into their long-term impact. Including patients with multiple occurrences of stroke would mean that the research would have a wider and more diverse application. Finally, capturing the perspectives of patients who have received education and counseling in qualitative interviews would enrich our understanding of the efficacy of such interventions and is recommended.
Relevance to Clinical Practice
The regular application of health promotion education to stroke patients can increase their knowledge about the disease and lead to healthy lifestyle changes. Stroke patients’ self-care ability and quality of life can be improved through training. In addition, complications and re-hospitalizations can be prevented by providing training and counseling services to stroke patients after discharge.
Supplemental Material
sj-docx-1-cnr-10.1177_10547738231211980 – Supplemental material for Technology-Based Health Promotion Training Among Stroke Patients: A Randomized Controlled Trial
Supplemental material, sj-docx-1-cnr-10.1177_10547738231211980 for Technology-Based Health Promotion Training Among Stroke Patients: A Randomized Controlled Trial by Cansev BAL and Zeliha KOÇ in Clinical Nursing Research
Footnotes
Acknowledgements
We thank the patients who participated in the study.
Authors’ Note
The manuscript, including related data, figures, and tables, has not been published previously and that the manuscript is not under consideration elsewhere.
Author Contributions
CB and ZK have involved in the conception and design of the study, or acquisition of data, or analysis and interpretation of data, drafting the article or revising it critically for important intellectual content, and final approval of the version to be submitted.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study began after written permission was obtained from the Clinical Research Ethics Committee of Ondokuz Mayıs University (B.30.2.ODM.0.20.08/1856). To collect the data, written informed consent was obtained from the university hospital and the patients included in the study. Persons visible in photos provided consent for these photos to be used in the publication of this article.
Data Availability Statement
The data supporting this study’s findings are available upon reasonable request from the corresponding author, ZK.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
