Abstract
This study sought to evaluate the impact of the subcutaneous tunneling technique on peripherally inserted central catheter (PICC) placement. We randomized 694 patients who needed PICC placement to either the tunneled PICCs (experimental group) or the non-tunneled PICCs (control group) from August to December 2021. The cumulative frequency of complications was assessed as the primary outcome. Secondary outcomes comprised of the amount of bleeding, catheter insertion time, self-reported pain score, and one-puncture success rate. After 6 months of follow-up, the tunneled PICCs group showed a significant decrease in the frequency of total complications, especially in infection (3.0% vs. 7.1%, p = .021) and catheter-related thrombosis (3.3% vs. 8.3%, p = .008), although approximately 0.5 ml bleeding and 3.5 min time were increased. This randomized multicenter study supports the efficacy of subcutaneous tunneling technology in reducing PICC-related complications, enhancing patient comfort, and encouraging using subcutaneous tunneling technology for PICC placement.
Background
Cancer has become the second leading cause of death worldwide, and its incidence is still increasing yearly (Soerjomataram & Bray, 2021). Chemotherapy is a standard treatment for cancer patients that can prolong the survival of metastatic malignancies. Medium-term access devices, peripherally inserted central catheters (PICCs), are preferred to infuse these vesicant and irritant agents owing to their advantages, such as safety, convenience, versatility, and cost-effectiveness (Park et al., 2021; Wang, Zhong, Huang, & Zhou, 2020).
Traditional PICCs are non-tunneled central venous access devices via a peripheral upper arm vein. Despite the many documented advantages of the conventional approach for preserving vessels in many settings and healthcare systems, some initial problems cannot be ignored. Catheter-related infection is a healthcare-associated severe problem that can result in septic embolism, infective endocarditis, osteomyelitis, and even death (Chan et al., 2017). Another complication with infection contributing to 30% of PICCs failing prior to completion of treatment is catheter-related thrombosis (CRT), which is associated with blood stasis, endothelial vessel damage, and blood hypercoagulation (McLendon et al., 2023; Nañez-Terreros et al., 2019). One potential strategy to prevent complications is to avoid using PICCs in patients at high risk for complications. For example, a 52-hospital quasi-experimental study (Chopra et al., 2022) showed improved PICCs appropriateness by reducing short-term PICCs use (<5 days), decreasing the use of multi-lumen devices, and avoiding PICCs placement in patients with chronic kidney disease. However, properly estimating or prioritizing these risks can be challenging when making choices on PICC placement.
Combined with the zone insertion methodTM (ZIMTM) (Dawson, 2011), a novel technology modified from non-tunneled PICCs by creating a subcutaneous tunnel, there might be similar advantages to centrally placed tunneled catheters. First, the primary mechanism of infection formation is the migration of microorganisms along the catheter (Rupp & Karnatak, 2018; Ullman et al., 2021), and the subcutaneous tunnel increases the retrograde difficulty. Second, the catheter-to-vein ratio (CVR) was improved, which has been recognized as an important determining factor in CRT (Sharp et al., 2021).
Previous studies have demonstrated that tunneled PICCs have technical advantages (Elli et al., 2017; Maria et al., 2019; Subramanian et al., 2013). However, the non-randomized methodology and small sample size did not provide sufficient evidence of tunneled PICCs’ effectiveness. In 2019, the first bi-center retrospective study (Kim, Shim, Lee, et al., 2019) showed that subcutaneous tunneling techniques could effectively reduce the occurrence of central line-associated bloodstream infection with nonsignificant catheter malposition, while the latest two trials showed the opposite (Dai et al., 2020; Xiao et al., 2021). To objectively evaluate the impact of the subcutaneous tunneling technique in PICC placement, a parallel, multicenter, and randomized controlled trial was designed.
Study Purpose
This study aims to evaluate the impact of the subcutaneous tunneling technique on PICC placement, thereby reducing catheter-related complications and promoting the use of subcutaneous tunneling for PICCs.
Methods
Study Design
A parallel, open-label, multicenter randomized controlled trial was performed on Chinese patients with cancer according to the CONSORT 2010 Statement (Schulz et al., 2010). All eligible participants were 1:1 ratio randomly assigned to a tunneled or control group. The prospective registry website for this study was https://www.chictr.org.cn/showproj.aspx?proj=124016
Study Settings
Three hospitals in northwestern China participated in this multicenter randomized controlled trial. The leading center is a 5,100-bed urban tertiary teaching hospital, consistently ranked among the top 20 hospitals in China. The other two centers are regional referral and educational hospitals with state-of-the-art diagnostic and treatment technologies.
Participants
The study population included all cancer patients who underwent PICC placement between August and December 2021, regardless of whether they were inpatients or outpatients. Patients aged ≥18 years and scheduled to undergo regular catheter maintenance at our hospitals were enrolled. The exclusion criteria were patients with any contraindications for PICC placement. Patients who missed two or more data points would be eliminated.
Sample Size
The sample size was determined using PASS 15 (NCSS, LLC; Kaysville, UT, USA). Effect sizes were calculated based on a previous study (traditional PICCs: 32.2%, tunneled PICCs: 18.4%) (Dai et al., 2020). Considering the attrition rate of approximately 20% in previous studies, 580 participants were approached to achieve 0.9 power with a two-tailed alpha of 0.025, and 290 patients were required in each group. Finally, the collection ratio between the three centers was determined to be 1.1:1:0.9 based on the hierarchical requirements and the ability of each center to complete cases.
Randomization and Blinding
The randomization sequence generated was performed by a statistician with SAS 9.3. Site-stratified block randomization with randomly varying block sizes was performed, and consecutive envelopes were assigned to each site. To protect the opaque envelope, each hospital has an independent researcher. When the subjects completed the baseline data assessment, the independent researcher randomly assigned the participants to the control or tunneled group according to numbered envelopes. Because of the different wounds and surgical procedures, interventionists and participants could not be blinded; however, data collectors and analysts were blinded to random assignments throughout the study.
Catheter Placement and Maintenance
The specialized team performed all PICC insertions following evidence-based institutional protocols with maximal barrier and antisepsis precautions.
The control group had PICC placement with the traditional method under a Doppler ultrasound device, including the function of ECG-EDUG (an abbreviation of ECG and Doppler Ultrasound Guidance devices). Before applying a tourniquet, a preprocedural ultrasound evaluates the vessel and surrounding anatomical structures. We implanted only 4Fr, open-ended, Silicone with a Proximal Valve PICC (Branden Tech, Inc., Dezhou, China). The CVR at the venipuncture site was also calculated before catheter placement.
The experimental group similarly inserts a trimmed distal catheter before tunneling. After the catheter entered the vessel, we would create a 3- to 7-cm retrograde tunnel using a 12- to 15-cm metallic tunneler from the venipuncture site to the exit site. In line with ZIMTM theory, the venipuncture sites need to be in the yellow area, and the exit sites are in green. A small amount of octyl cyanoacrylate skin adhesive was used to close the wounds, and sterile transparent waterproof dressings were used to fix the catheters. PICCs were trimmed to expose 5 to 7 cm outside the skin for connector installation, and the tips were checked using posteroanterior chest X-rays.
Catheter maintenance was performed following a stringent nursing protocol formulated by our hospital based on infusion therapy guidelines (Gorski et al., 2021). The key components include weekly dressing changes with film cover, skin antisepsis with 2% chlorhexidine in alcohol, transparent semipermeable membranes, neutral displacement needle-free connectors, regular device checks, and flushing with normal saline daily.
Data Collection and Outcomes
An initial data collection process was conducted around catheter insertion, which included general demographic characteristics (i.e., age, gender, body mass index [BMI]), diagnosis, blood examination to determine coagulation, and questions related to PICCs insertion (i.e., the insertion arm and vein, the attempts at insertion, and whether there was pain or discomfort during placement). Hypercoagulation was identified whether the patient met any two of the following blood laboratory examinations: platelet >300 × 109/L, prothrombin time < 9.6 s, activated partial thromboplastin time < 24.8 s, fibrinogen > 4.0 g/L, and D-dimer > 0.50 mg/L (Chen et al., 2021). Follow-up assessments were conducted with care providers if the participants were still hospitalized or by telephone if discharged approximately every month up to catheter removal except the first week.
The primary observed outcome was the incidence of complications: (1) Infections were defined according to Infectious Diseases Society of America criteria (Mermel et al., 2009); (2) CRT was confirmed by ultrasound or CT examination showing the presence of a thrombus in the vein with a catheter (Taxbro et al., 2019); (3) catheter malposition was defined as exposed length prolapsed ≥5 cm; (4) medical adhesive-related skin injury was defined as the skin around medical adhesive persistently occurring itching, erythema, bulla, or tearing (Kim, Jang, Kim, et al., 2019); (5) phlebitis is characterized by pain, swelling, erythema, induration, purulence, and palpable venous cords around the venipuncture site (Gorski et al., 2021); and (6) leakages from lymphatics, catheter damage, and fibrin sheath formation are all considered.
The secondary outcomes were collected following the procedure, including the amount of bleeding, catheter insertion time, self-reported pain, and one-puncture success rate. The amount of bleeding was observed by subtracting the weight of the blood gauze during the placement and then converting it into volume (ml) = blood weight (g)/1.05. Catheter insertion time was calculated from the first attempt at puncturing the PICCs until complete fixation. The pain was graded according to the 0 to 10 Numeric Rating Scale (Ferreira-Valente et al., 2011). One-puncture success rate refers to the first puncture successfully performed in the vessel.
Statistical Methods
Statistical analysis was conducted using R 4.2 software (R Core Team, Vienna, Austria). The descriptive analyses were expressed as means and standard deviations (SDs), medians and interquartile ranges (IQRs), or percentages. Independent sample t-test, chi-square test, or Fisher’s exact test was used to compare the two study groups. Statistical significance was set at .05 for the final analysis, which excluded missing data from all randomly assigned patients.
Ethical Considerations
On May 13, 2021, this study’s project leader passed the Institution Review Board review at the Shandong University of Qilu Hospital. At the end of the month, two sub-centers completed their ethical reviews. The reference number was 2021042.
Before the trial, all researchers were trained on ZIMTM theory, the subcutaneous tunnel construction process, complications observation and follow-up, adverse event handling, and reporting. Afterward, we registered the clinical trial, and on August 1, all three centers began collecting patients simultaneously. We fully informed the participants about the study’s purpose, procedures, and previous clinical trial results. We assure the patients that they may withdraw from the trial at any time and that their medical treatment will not be adversely affected. Lastly, we will sign an informed consent form if the patient agrees to participate in our study. We have also informed all participants that they may consult with us during the catheterization period if they have any questions or concerns.
Results
Patient Characteristics
A total of 694 patients were recruited and randomly assigned to the study, of whom 256 were included in the leading center and 232 and 206 in the other two. The flowchart of participants in the trial is shown in Figure 1, and it shows that 676 (97.41%) completed the follow-up. Nine participants in each group were excluded from the analysis because these subjects did not receive an allocation, failed to follow up, or actively withdrew trials due to illness worsening. The average catheter dwell time was 115.83 ± 49.28 days in the tunneled group compared to 117.27 ± 57.69 days in the control group (p = .729). Patients’ baseline and PICC characteristics were balanced in both groups, including age, gender, BMI, diagnosis, blood hypercoagulation, insertion arm, insertion vein, and tip position. Detailed information on patients’ demographic and PICC characteristics for each group is provided in Table 1. Because of the tunneled technique, the CVR of the tunneled group at the venipuncture site was lower than that in the control group (0.29 [0.26, 0.32] vs. 0.31 [0.28, 0.35], p < .001).
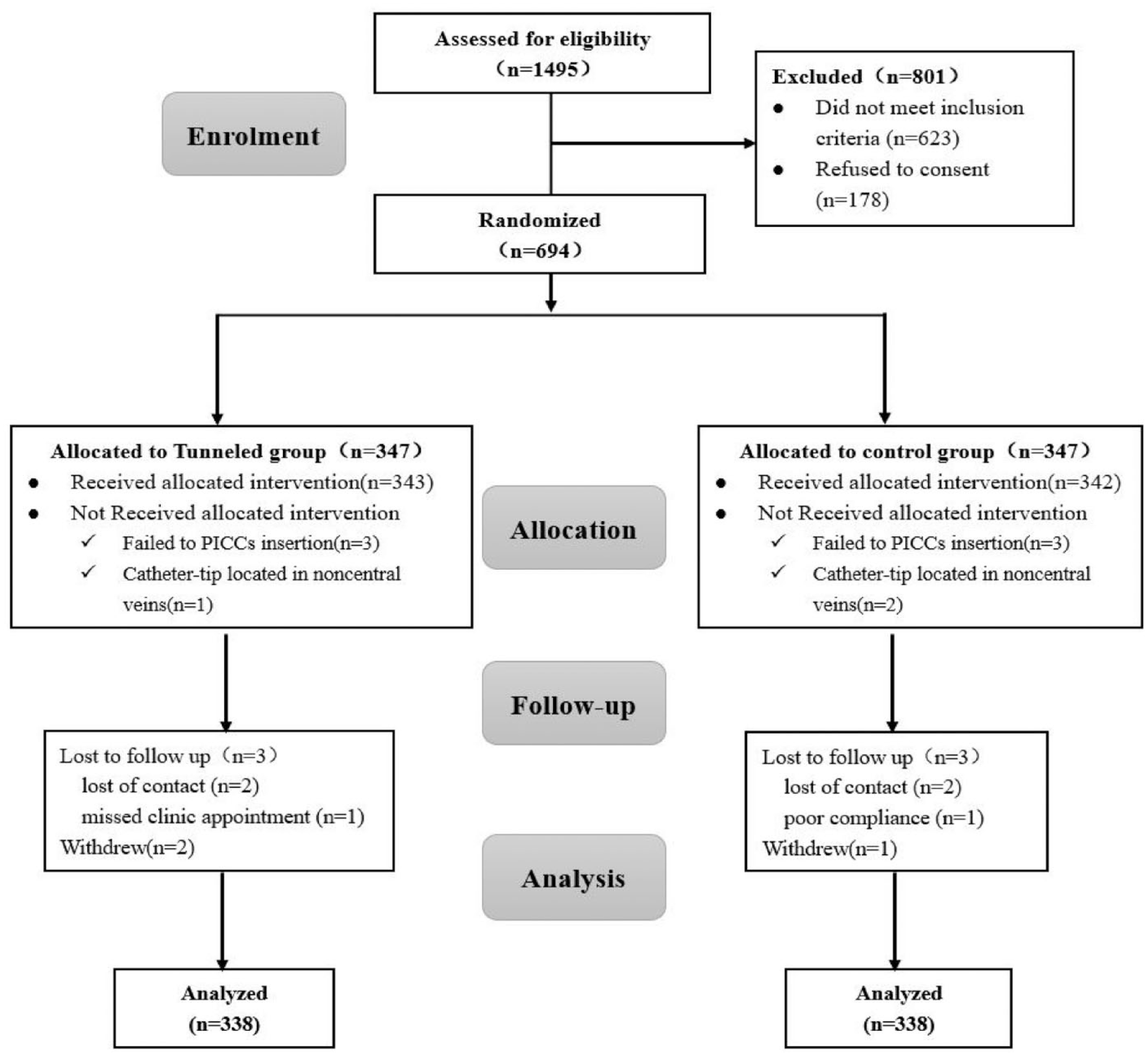
Consolidated Standards of Reporting Trials (CONSORT) flow diagram.
Demographic and PICCs Characteristics at Baseline (n = 672).
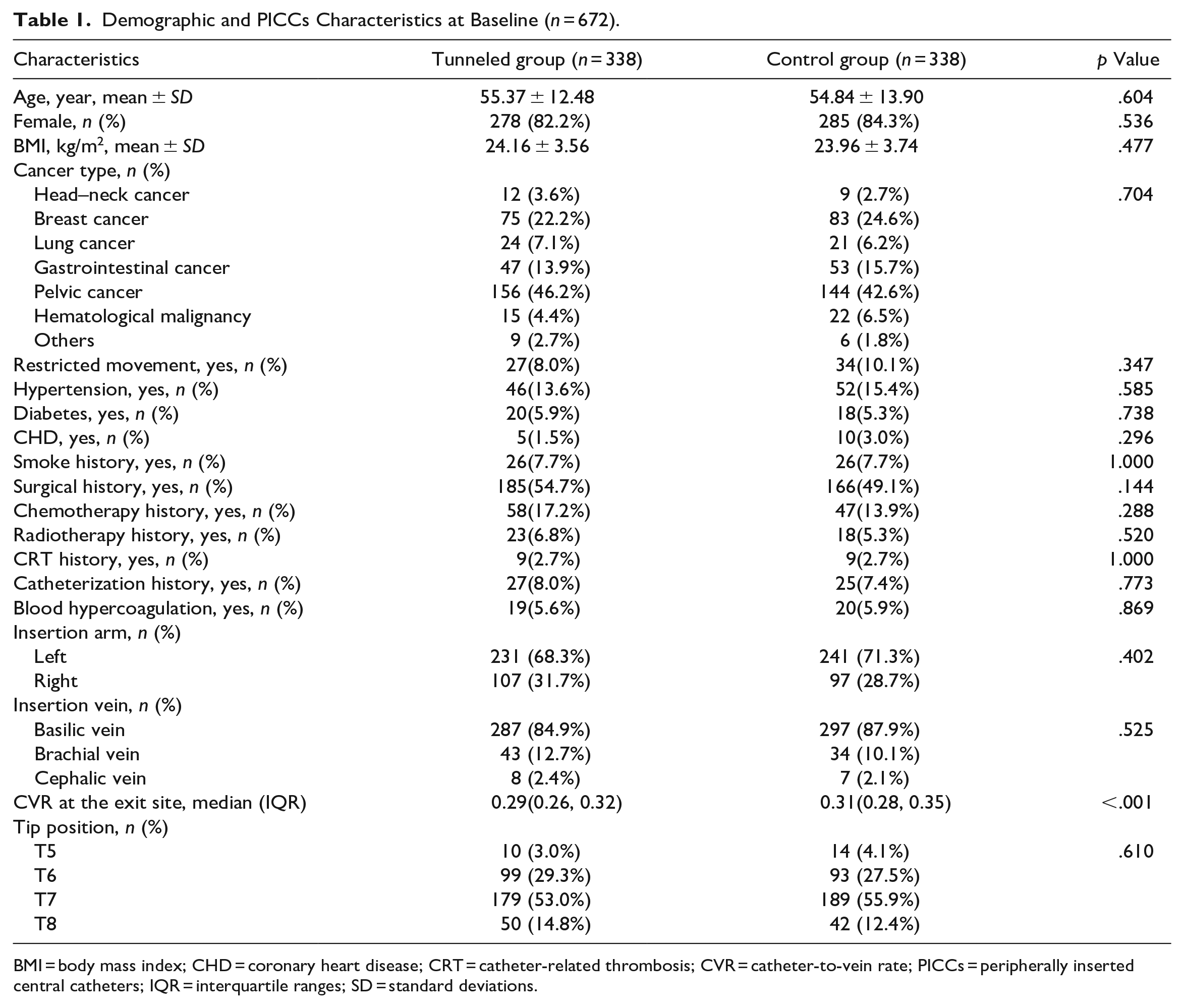
BMI = body mass index; CHD = coronary heart disease; CRT = catheter-related thrombosis; CVR = catheter-to-vein rate; PICCs = peripherally inserted central catheters; IQR = interquartile ranges; SD = standard deviations.
Outcomes and Estimation
Following catheter placement, 161 participants (23.82%) were extubated within 3 months, 86.24% within 6 months, and all follow-ups were completed within 10 months. In each group, no harm or unintended effects were reported.
Table 2 analyzes the cumulative frequency of complications. The tunneled PICCs significantly decreased the frequency of total complications compared to the control group (18.6% vs. 30.5%, p < .001). Among the complications, infection occurred in 10 (3.0%) patients in the tunneled group and 24 (7.1%) patients in the control group, with a significant difference (p = .021). There were 11 (3.3%) cases of CRT detected in the tunneled group compared to 28 (8.3%) cases in the control group (p = .008). Other complications after placement were not significantly different between the two groups (p > .05).
The Primary Outcomes of the Participants.
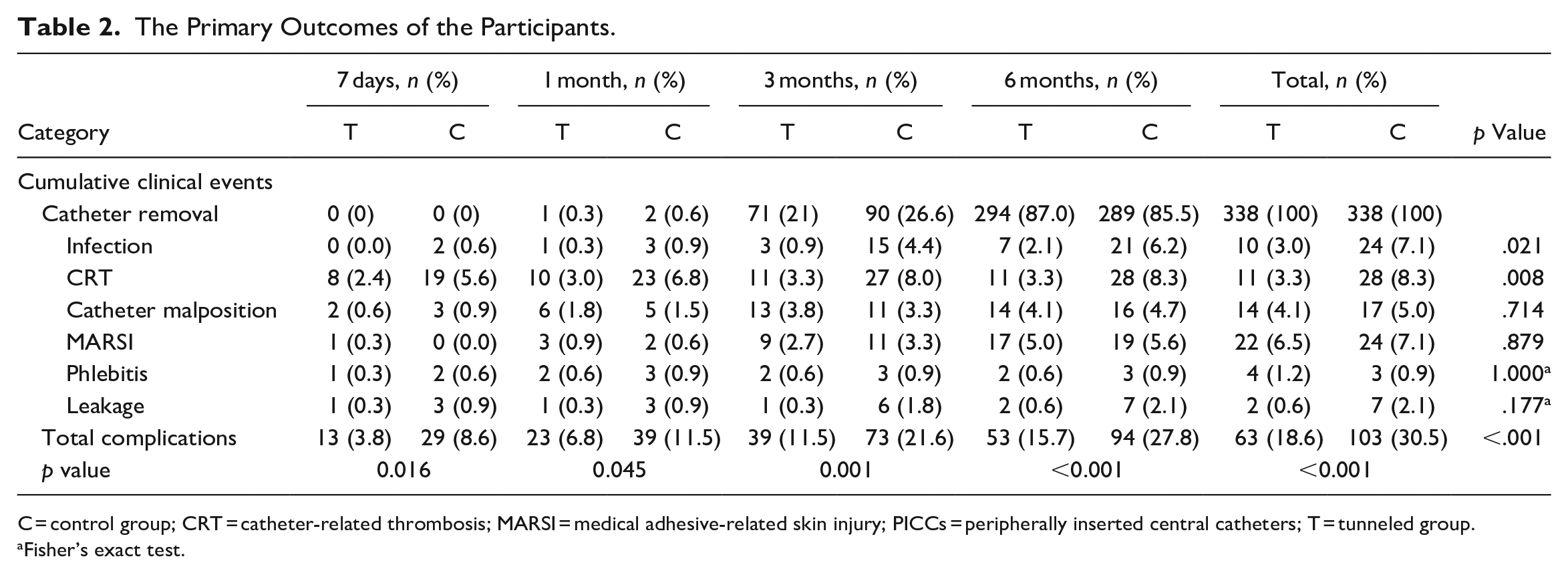
C = control group; CRT = catheter-related thrombosis; MARSI = medical adhesive-related skin injury; PICCs = peripherally inserted central catheters; T = tunneled group.
Fisher’s exact test.
Table 3 analyzed the secondary outcome of the participants between the two groups. The amount of bleeding in the tunneled group was 2.21 (1.23, 3.40) ml, which was significantly higher than that in the control group (1.67 [0.76, 4.30] ml, p =. 013). During the insertion procedure, the time for the control group was less than that for the tunneled groups (20.15 ± 5.58 vs. 16.49 ± 7.66, p < .001). The one-puncture success rate (92.0%) in the tunneled group was higher than that in the control group (89.6%) with no significance (p = .351). The mean scores for self-reported pain (1.71 ± 1.02 vs. 1.76 ± 1.06, p = .484) did not differ between the two groups.
Secondary Outcomes of the Participants.

IQR = interquartile ranges; SD = standard deviations.
Discussion
To our knowledge, this is the largest randomized study assessing the overall risk of catheter-related complications between tunneled PICCs and traditional PICCs in cancer patients. The cumulative data showed that the tunneled group significantly decreased the frequency of total complications, especially in infection and CRT.
In our study, there was a 7.1% infection rate of traditional PICC placement, which corresponded to other studies of 6.25% to 11% (Lacostena-Pérez et al., 2019; Moss et al., 2021). Following a multicenter retrospective study that found tunneled PICCs protected patients from CLABSI (Kim, Shim, Lee, et al., 2019), we first demonstrated that subcutaneous tunneling significantly reduces the risk of infection; while two Chinese scholars (Dai et al., 2020; Xiao et al., 2021) detected the effectiveness, the comparisons failed due to insufficient sample sizes. Preventing infection usually requires multiple strategies, but maintaining sterility and reducing microbial colonization are the primary mechanisms (Bell & O’Grady, 2017). The subcutaneous tunnel makes it more difficult for microbes to travel retrograde along the catheter, thus reducing the incidence.
The incidence of CRT was significantly lower in the experimental group than in the control group (3.6% vs. 12.5%, p < .001). A larger vein diameter in the higher puncture site with a more successful puncture rate in the tunneled group may be contributed to the lower incidence of thrombosis, which is consistent with previous studies, although our findings were higher (Dai et al., 2020; Giustivi et al., 2022; Xiao et al., 2021). In our opinion, the higher incidence might be explained by two reasons: First, CRT can occur very early post-insertion in cancer patients (Trezza et al., 2021; Wang, Li, Wu, et al., 2020). Instead of screening for CRT before catheter removal, we advanced screening to the patient’s first return, which may have allowed us to detect more CRT. Second, the study populations and intervention methods may also have contributed to the differences between the results.
Despite its shortcomings, subcutaneous tunneling is an effective technology. In 2019, Dai et al. (2020) reported that the subcutaneous tunneling technique increased bleeding, whereas the Xiao et al. (2021) studies showed no significant difference between the two groups. The bleeding amount increased by 0.5 ml as a tunnel was created in our research. Still, we believe that the difference in tunneling instruments and intervention methods played an important role. In our procedure, the tunneling takes place after the catheter placement, which avoids the extra pain, skin damage, and time waste associated with successful tunnel placement but failed venipuncture; however, this technology did not work during catheter placement. Taking a tunnel took an average of 3.5 min, with a tunnel needle costing roughly $2.5 (Dai et al., 2020); however, there were fewer complications and a better dwell experience.
There are some limitations to the present study, the first of which is that the current study included only cancer patients from three large regional hospitals. However, this finding will enable researchers to conduct further research in more hospitals and involve patients undergoing PICC placement for different reasons. Second, the intervention cannot be blinded to interventionists and participants, meaning that there is an unavoidable risk of bias, although aseptic drapes were used to screen the procedure and interventionists were not involved in data collection after catheter placement.
PICC-related complications are the leading cause of catheter malfunction. Our study demonstrated that subcutaneous tunneling could significantly decrease CRT and infection frequency. Our responsibility is to maximize patient comfort and safety, making promoting subcutaneous tunneling necessary. Nevertheless, subcutaneous tunneling did not substantially improve catheter malposition, Medical Adhesive Skin Injuries (MRBSI), phlebitis, or leakage; additional tools, such as subcutaneous securement systems and semipermeable transparent dressings, were also required. In addition, ZIMTM requires simple theoretical training before popularization and the optimal tunnel length needs to be determined in specific patient cohorts.
In conclusion, subcutaneous tunneling is a crucial technique to minimize catheter-related complications, particularly in infection and CRT, and it should be recommended as a standard procedure for cancer patients to increase vessel health and preservation.
Supplemental Material
sj-doc-1-cnr-10.1177_10547738231194099 – Supplemental material for Implementation of Tunneled Peripherally Inserted Central Catheters Placement in Cancer Patients: A Randomized Multicenter Study
Supplemental material, sj-doc-1-cnr-10.1177_10547738231194099 for Implementation of Tunneled Peripherally Inserted Central Catheters Placement in Cancer Patients: A Randomized Multicenter Study by Yuan Sheng, Li-Hong Yang, Yan Wu, Wei Gao and Sheng-Yi Dongye in Clinical Nursing Research
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Kashgar Science and Technology Project (No. KS2022013).
Ethical Approval
This study was approved by the Shandong University of Qilu Hospital Ethics Committee (Approval No. 2021042).
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
